Abstract
Porcine circovirus type 2 (PCV2) infection is the cause of postweaning multisystemic wasting syndrome (PMWS). It has been speculated whether cell types permissive of replication are found in the primary lymphoid organs and whether infection of these tissues has an important role in the pathogenesis of PMWS. The aim of this study was to determine if primary lymphoid organ cells support viral replication during PCV2 infection. This was done by histopathological examination of thymus and bone marrow from pigs experimentally inoculated with PCV2 (n = 24), mock-infected pigs (n = 12), pigs naturally affected by PMWS (n = 33), and age-matched healthy control animals (n = 29). In situ hybridization (ISH) techniques were used to detect PCV2 nucleic acid irrespective of replicative status (complementary probe, CP) or to detect only the replicative form of the virus (replicative form probe, RFP). PCV2 was not detected in the experimentally PCV2-inoculated pigs or the control animals. Among the PMWS-affected pigs, 19 of 20 (95%) thymuses were positive for PCV2 by CP ISH, and 7 of 19 (37%) of these also supported viral replication. By CP ISH, PCV2 was detected in 16 of 33 (48%) bone marrow samples, and 5 of 16 (31%) of these also supported replication. The 2 ISH probes labeled the same cell types, which were histiocytes in both organs and lymphocytes in thymus. The RFP labeled fewer cells than the CP. Thus, PCV2 nucleic acids and replication were found in bone marrow and thymus of PMWS-affected pigs, but there was no evidence that primary lymphoid organ cells are major supporters of PCV2 replication.
Keywords
Porcine circovirus type 2 (PCV2), a circular single-stranded (ss) DNA virus, is recognized as the causative agent of postweaning multisystemic wasting syndrome (PMWS). PMWS is clinically characterized by increased mortality in pigs of 7 to 15 weeks of age, wasting, dyspnea, diarrhea, and jaundice. 10,15 In the past decades, several circular ssDNA viruses, including PCV2, have been detected in birds, man, and other mammals. These viruses belong to the family Circoviridae (genus Circovirus and Gyrovirus) or Anelloviridae (genus Alphatorquevirus). These viruses have many molecular biological similarities, and the viral infections also have epidemiological, clinical, and histopathological parallels. Shared features of the viruses are the nonenveloped circular ssDNA genome and the replication process, where the virus goes through an intermediary double-stranded (ds) form—that is, the replicative form (RF). Generation of the RF involves de novo DNA synthesis, a process depending on mitosis and nuclear enzymes of the host cell. 26,42 PCV2, Beak and feather disease virus (BFDV), Pigeon circovirus (PiCV), Goose circovirus (GoCV), and Chicken anemia virus (CAV) are widespread and able to cause both clinical and subclinical infections, whereas Porcine circovirus type 1, Torque teno virus (TTV), and Torque teno mini virus have not been directly associated with disease. 20,28,30,37,42 Most of these viruses (CAV, BFDV, PiCV, GoCV, PCV2) affect lymphoid tissues and cause lymphoid atrophy, necrosis, and lymphocyte depletion, leading to immunosuppression and secondary infections. 37,42 Both primary (thymus, bone marrow, bursa of Fab-ricius) and secondary lymphoid tissues (lymph nodes, spleen, tonsil, bronchus-associated lymphoid tissue, gut-associated lymphoid tissue) may be affected, but the predilection site depends on the specific virus. 20,42 In most cases, the viruses can also be isolated from other organs and tissues such as lung, liver, blood, kidney, brain, heart, intestine, trachea, and skin and, for avian species, from crop, claws, beak, and feathers. 20,28,42
An important step in understanding the pathogenesis of viral diseases is the identification of cells permissive of replication. The replication process of the circular viruses is complex, and for most of these viruses, the cells supporting replication are still to be discovered. It has been shown that CAV replicates in bone marrow (hematocytoblasts), thymus (lymphocyte precursors), spleen, and gastrointestinal tract 42 and that TTV replicates in bone marrow (erythroid series and megakaryocytes), peripheral blood mononuclear cells, and liver. 20,22 Much effort has been put into finding cells permissive of PCV2 replication in vivo, but only recently have relevant methods become available. Four different approaches for detecting replication in vivo have been described: Yu et al 45 measured the level of spliced PCV2 capsid messenger RNA (mRNA) by polymerase chain reaction (PCR) in tissue homogenates, Hamberg et al 17 combined in situ hybridization (ISH) for PCV2 and immunohistochemistry (IHC) for ssDNA and dsDNA, Pérez-Martín et al 31 used an ISH probe that hybridized exclusively with the RF of the virus, and finally, Henriksson et al 19 detected replication by the use of padlock probes and rolling circle amplification. A common feature of these studies was that no or relatively few thymus samples were included, and with regard to bone marrow, only 1 study investigated a small number of samples. Since PCV2 is closely related to other circular viruses, which often affect and/or replicate in the primary lymphoid organs, there have been speculations on which role the primary lymphoid organs play in the PCV2 pathogenesis. 10,30
The present study was conducted to determine which cells of the primary lymphoid organs support viral replication during PCV2 infection in swine. Thymus and bone marrow from experimentally infected pigs and from naturally PMWS-affected pigs were examined by histopathology and ISH for the presence of PCV2 nucleic acids and PCV2 replication.
Materials and Methods
Experimental Infection Study
In an experimental infection study, 12,14 36 caesarean-derived, colostrum-deprived (CD/CD) Landrace piglets were obtained from a Spanish farm seropositive for PCV2. At 7 days of age, 24 animals were intranasally and orally inoculated with 105.2 50% tissue culture infectious dose per milliliter (TCID50 ml–1) of a Spanish PCV2 strain (Burgos), whereas 12 control animals were mock inoculated with a sterile cell culture medium. The animals were euthanized at 5 days postinoculation (DPI) (6 PCV2-inoculated and 2 control animals), 8 DPI (7 PCV2-inoculated and 2 control animals), and 29 DPI (11 PCV2-inoculated and 8 control animals), and tissues were sampled (thymus, bone marrow, tonsils, and mesenteric, superficial inguinal, submandibular, and tracheobronchial lymph nodes). Thymus was not obtained from 1 control pig euthanized at 29 DPI. The PCV2-inoculated piglets seroconverted between 14 and 21 DPI; all control animals remained seronegative. No PCV2 infection-related clinical signs or lesions 34 were detected in the control animals. In the PCV2-inoculated group, slight lymphoid depletion, occasional granulomatous inflammation, and presence of multinucleated giant cells were observed in the secondary lymphoid organs at 8 DPI (n = 2) and 29 DPI (n = 10). None of the other PCV2-inoculated animals had any PCV2 infection-related clinical signs or lesions. PCV2 was not detected by ISH in any tissues from the control animals, whereas 1 of 7 and 8 of 11 PCV2-inoculated piglets had low amounts of PCV2 nucleic acids in a few lymph nodes at 8 and 29 DPI, respectively. 12,14
Naturally PMWS-Affected Pigs (Field Study)
From a case-control study in PMWS-affected farms, 16 33 animals with PMWS and 29 age-matched, healthy control animals were selected for the present study. The animals had been euthanized at 12 to 20 weeks of age, and tissues, including thymus and bone marrow, were harvested. However, among the selected animals, thymus was too atrophic to be identified in 13 of the PMWS-affected animals, and in 1 healthy animal, thymus was not excised. The PMWS diagnosis, made by Grau-Roma et al 16 in accordance with accepted standards, 34 was based on clinical signs, presence of characteristic histopathologic findings of lymphoid depletion, granulomatous inflammation, and detection of moderate to massive PCV2 load in secondary lymphoid tissues (tonsils and mesenteric, superficial inguinal, submandibular, and tracheobronchial lymph nodes).
Histopathology
Within 45 minutes after euthanasia, tissue samples of thymus and bone marrow were fixed by immersion in 10% neutral buffered formalin for 24 hours. After fixation, the bone marrow samples were decalcified in Decalcifier II (Surgipath Europe Ltd, Peterborough, UK) for 1 to 2 hours. Next, all tissue samples were dehydrated, embedded in paraffin wax, cut in 3- to 5-μm-thick sections, and mounted on conventional glass slides, SuperFrost Plus slides (Menzel-Gläser, Göttingen, Germany), or silanized Chemmate capillary gap microscopic slides (DAKO, Glostrup, Denmark) for histochemistry, IHC, or ISH, respectively. All tissue sections were stained with hematoxylin and eosin (HE). 38 For evaluation of hematopoietic precursors and blood cells, all bone marrow sections were stained with periodic acid–Schiff and Giemsa. 7,9 Selected sections of bone marrow and thymus representing the observed histopathological variation were further examined by different histochemical and immunohistochemical staining procedures. Bone marrow was examined for presence of collagen, reticulin, and acid mucopolysaccarides by staining with Masson trichrome (n = 14), 25 Gordon and Sweet reticulin (n = 19), 3 and Alcian blue (pH 3) (n = 25), 7 respectively. Furthermore, IHC staining for CD3 (bone marrow, n = 3) and lysozyme (bone marrow, n = 3; thymus, n = 22) was carried out as previously described. 6,23
In the bone marrow, the following was examined by semiquantitative methods as suggested by the Society of Toxicologic Pathology (STP) Immunotoxicology Working Group: 39 general cellularity, myeloid to erythroid (M:E) ratio, and quantity and morphology of megakaryocytes, erythroid cells, granulocytic cells, and other cells (including lymphocytes, plasma cells, monocytes, histiocytes, osteoclasts, osteoblasts, and reticular stromal cells). 18,41 The general marrow cellularity was presented in grades of hypocellularity as normocellular (0), slightly hypocellular (+), hypocellular (++), or markedly hypocellular/acellular (+++). A mean M:E ratio and standard deviation were calculated for each group of animals. In the field study, 4 bone marrow samples from the control animals and 1 bone marrow from a PMWS-affected animal were acellular, preventing evaluation of the M:E ratio. The examination also included a description of other lesions, when present. A homogeneous amorphous material surrounding the adipocytes was observed in some of the bone marrow stained by HE. This material from hereon is referred to as amorphous fat-associated material (AFM). The amount of AFM was scored semiquantitatively as absent (0), low (+), moderate (++), or high (+++) (Fig. 1a).
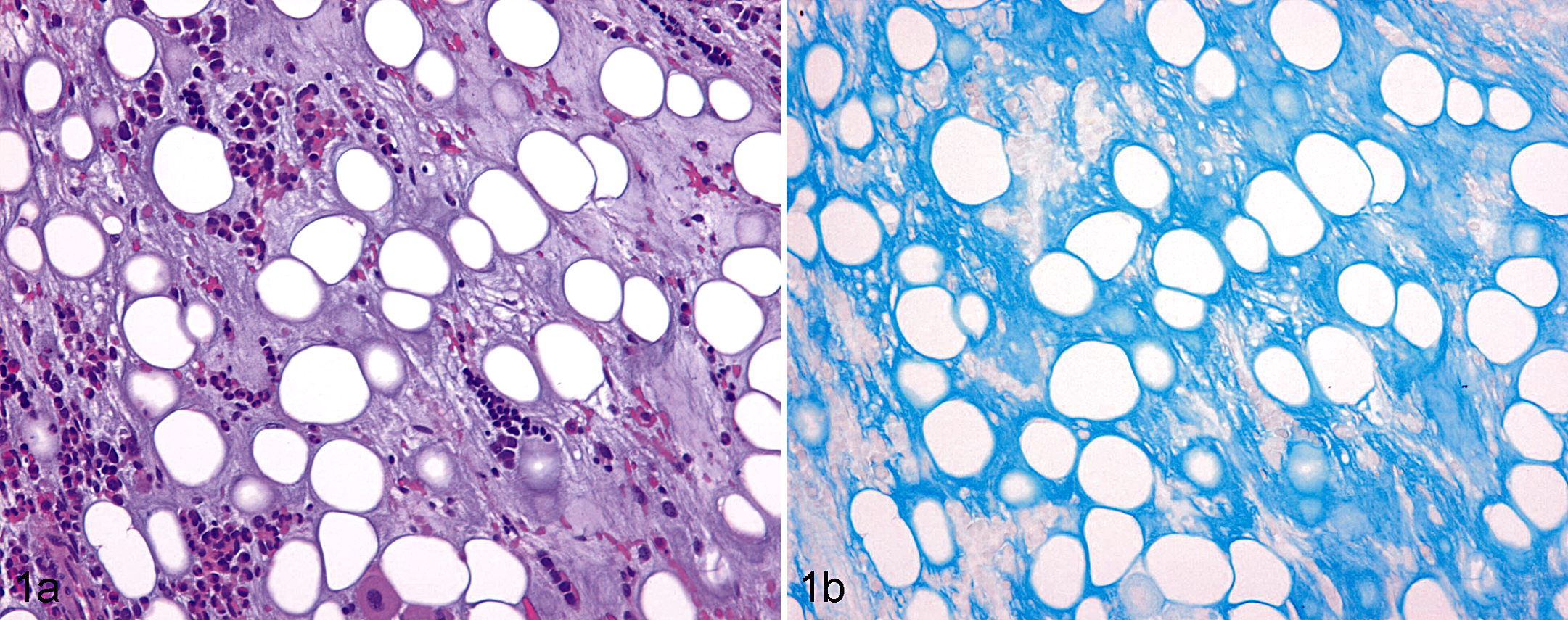
Bone marrow; postweaning multisystemic wasting syndrome–affected pig. (a) Bone marrow with high amount of amorphous fat-associated material (grade +++). Hematoxylin and eosin. (b) Section from same bone marrow stained with Alcian blue.
Thymus was evaluated semiquantitatively as suggested by the STP Immunotoxicology Working Group. 39 Thus, the quantity and morphology of lymphocytes in cortex and medulla, granulocytes, histiocytes, and multinucleated giant cells and the quantity of Hassall’s corpuscles were assessed. 5 Lymphoid depletion (ie, reduced number/density of lymphocytes) was scored as absent (0), mild (+), moderate (++), or severe (+++). In the thymuses, IHC for lysozyme was used to evaluate the number of histiocytes, which was scored as normal (0; ie, a few individualized histiocytes dispersed in the cortex and medulla), increased (+; ie, many individualized and a few small clusters of histiocytes in the cortex and medulla), or markedly increased (++; ie, many individualized and clusters of histiocytes in the cortex and medulla). The number of multinucleated giant cells was scored as none (0), few (+), some (++), or many (+++). The examination also included a description of other lesions, when present.
In Situ Hybridization
For detection of PCV2 nucleic acid, ISH with 2 complementary digoxigenin (DIG)–labeled oliogonucleotide probes, named complementary probe (CP) and RF probe (RFP), was performed. 31,32 The probes were constructed from the open reading frame (ORF) 1 sequence of PCV2 encoding for the replicase proteins. The CP was complementary to ORF1 32 of the viral genome and hybridized with viral ssDNA, RF, and mRNA and thus detected PCV2 irrespective of replicative status. The RFP was identical to ORF1 31 of the viral genome and could therefore solely hybridize with the RF form, which only is present during viral replication. All thymus and bone marrow samples were investigated with the CP, and the positive samples were further examined with the RFP. The number of positive cells by CP and RFP ISH was scored as low, moderate, or massive, and the cells were morphologically characterized. The ISH process for both probes was identical and was carried out according to Rosell et al. 32 Briefly, sections were deparaffinized, rehydrated, and treated with 0.3% pepsin for 10 minutes at 37°C, followed by heating to 105°C for 8 minutes. At this temperature, the sections were incubated with 100% formamide for 5 minutes and then prehybridized for 20 minutes by incubation with 0.1 or 0.3 nmol/ml of the CP or RFP, respectively. The hybridization process was subsequently carried out at 37°C for 60 minutes, and sections were washed at high stringency with saline sodium citrate. Detection was done by incubation with a 1:500 dilution of anti-DIG antibody (Roche Diagnostics, Barcelona, Spain) at 37°C for 60 minutes, followed by color development by NBT/BCIP stock solution (Roche Diagnostics) at 37°C for 20 minutes. Sections were counterstained by Fast Green (Sigma-Aldrich, Madrid, Spain), dehydrated, and coverslipped. A section of a lymph node or trachea from a PMWS-affected pig was the positive control for validation of the ISH process with the CP or RFP, respectively. As negative control of the assay, the technique was applied on the same tissues without addition of probe.
Statistical Analyses
The chi-square test, Fisher’s exact test, and/or Student’s 2-sample t-test of SAS version 9.1 (SAS Institute, Cary, NC) were used for analyzing the results. Statistically significant level was set at P < .05. The small group sizes of the experimentally infected pigs precluded valid statistical analysis.
Results
Histopathology of Bone Marrow
The main histopathological findings are listed in Table 1 and the M:E ratios are presented in Fig. 2. No association was found when comparing the general cellularity of PMWS cases and controls by the chi-square test, and the M:E ratio was not different between these groups using the Student’s 2-sample t-test and Fisher’s exact test. The quantity and morphology of megakaryocytes and other cells in the bone marrow were normal.
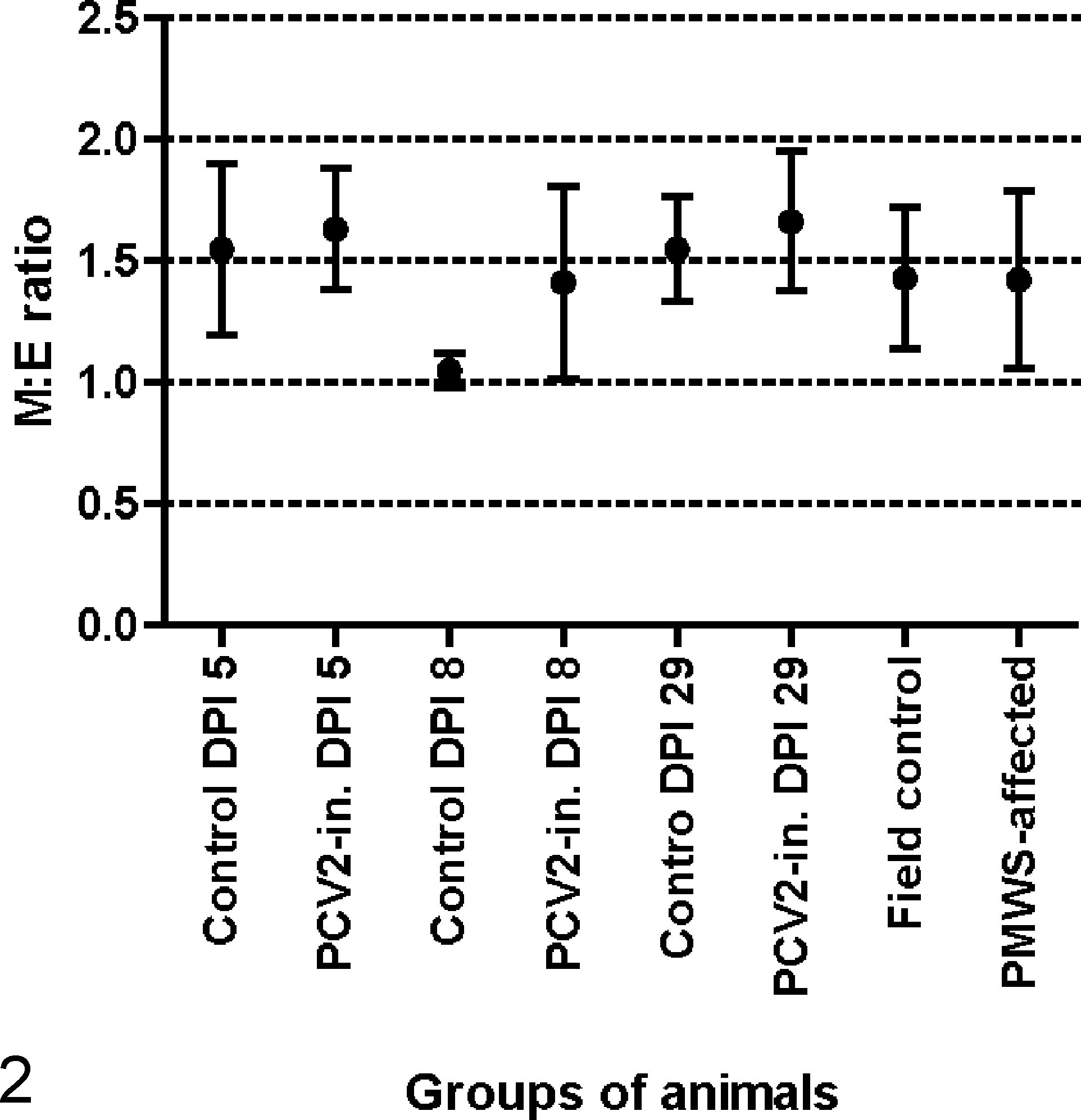
Myeloid to erythroid ratio (M:E ratio) presented as mean and corresponding standard deviation for each group of animals. DPI, days postinoculation; PCV2-in., inoculated with Porcine circovirus type 2; PMWS, postweaning multisystemic wasting syndrome.
Major histopathological findings in bone marrow.
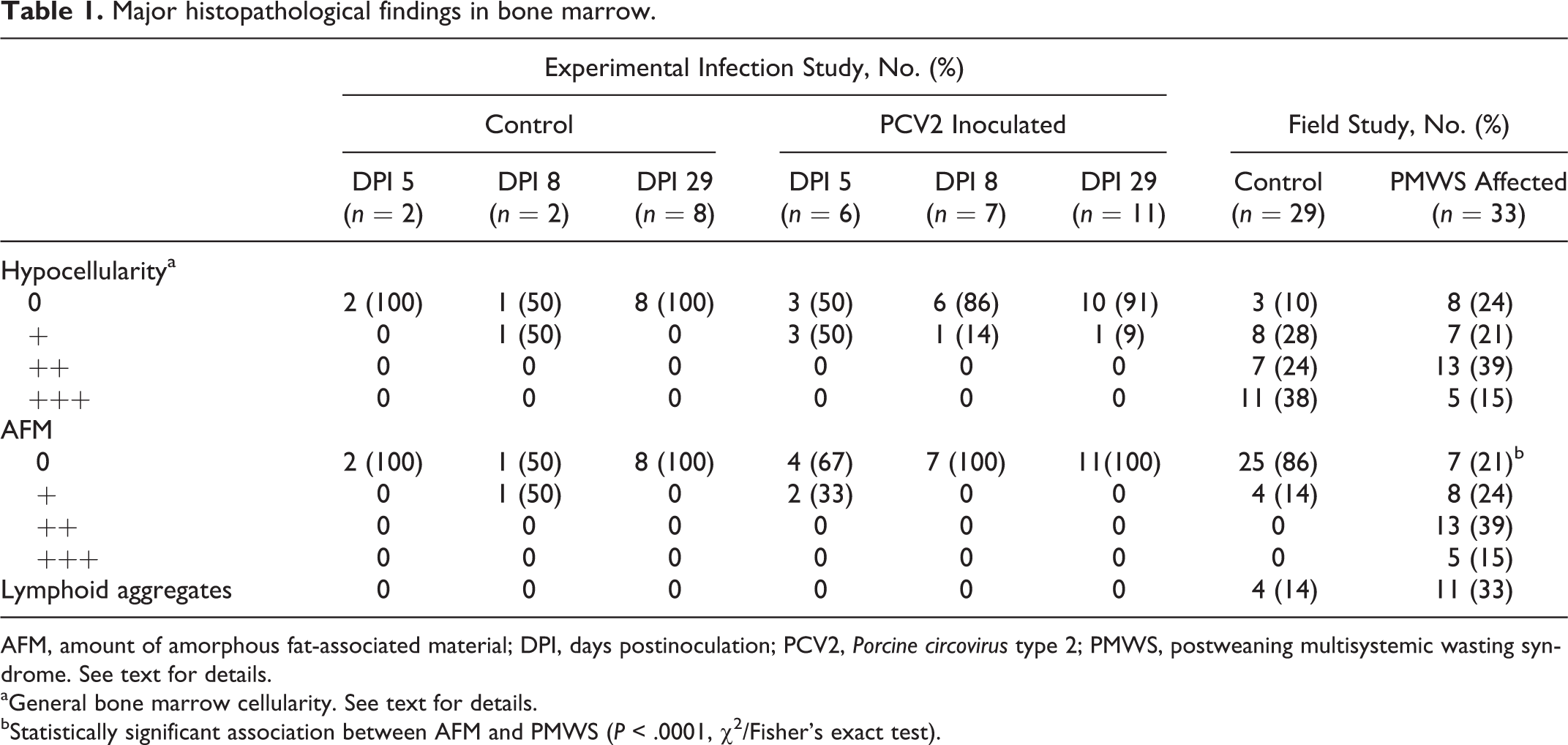
AFM, amount of amorphous fat-associated material; DPI, days postinoculation; PCV2, Porcine circovirus type 2; PMWS, postweaning multisystemic wasting syndrome. See text for details.
aGeneral bone marrow cellularity. See text for details.
bStatistically significant association between AFM and PMWS (P < .0001, χ 2 /Fisher’s exact test).
The distribution of AFM was focal, multifocal, or diffuse. Adipocytes in the affected areas showed varying degrees of atrophy. Alcian blue stained the AFM, identifying the mucopolysaccaride nature of the substance, and the staining intensity seemed to have a positive association with the amount of AFM (Fig. 1b). The AFM was negative by staining for collagen and reticulin. In the study of field cases, AFM was significantly more abundant in PMWS cases compared with controls (P < .0001, Fisher’s exact test).
Localized aggregates of CD3-positive lymphocytes and lysozyme-positive histiocytes were detected in the bone marrow of 11 of 33 (33%) PMWS-affected animals and 4 of 29 (14%) controls from the field study (see Suppl. Fig. S1a,b). However, this difference was not significant (P = .07, chi-square test). The lymphoid aggregates were not seen in the bone marrow of the experimentally infected animals.
No inclusion bodies were found in the bone marrow sections of animals from the experimental or field studies.
Histopathology of Thymus
The main histopathological findings are summarized in Table 2. The lymphoid depletion primarily affected the cortical lymphocytes. In the severely depleted thymuses, only a few Hassall’s corpuscles were detected. Multinucleated giant cells were mainly located in the medullary area in the thymuses without lymphoid depletion but were present in both cortex and medulla of the depleted thymuses. There was a strong association between PMWS and degree of lymphoid depletion (P < .0001, Fisher’s exact test), number of histiocytes (P < .0001, Fisher’s exact test), and multinucleated giant cells (P = .0002, Fisher’s exact test).
Major histopathological findings in thymus.
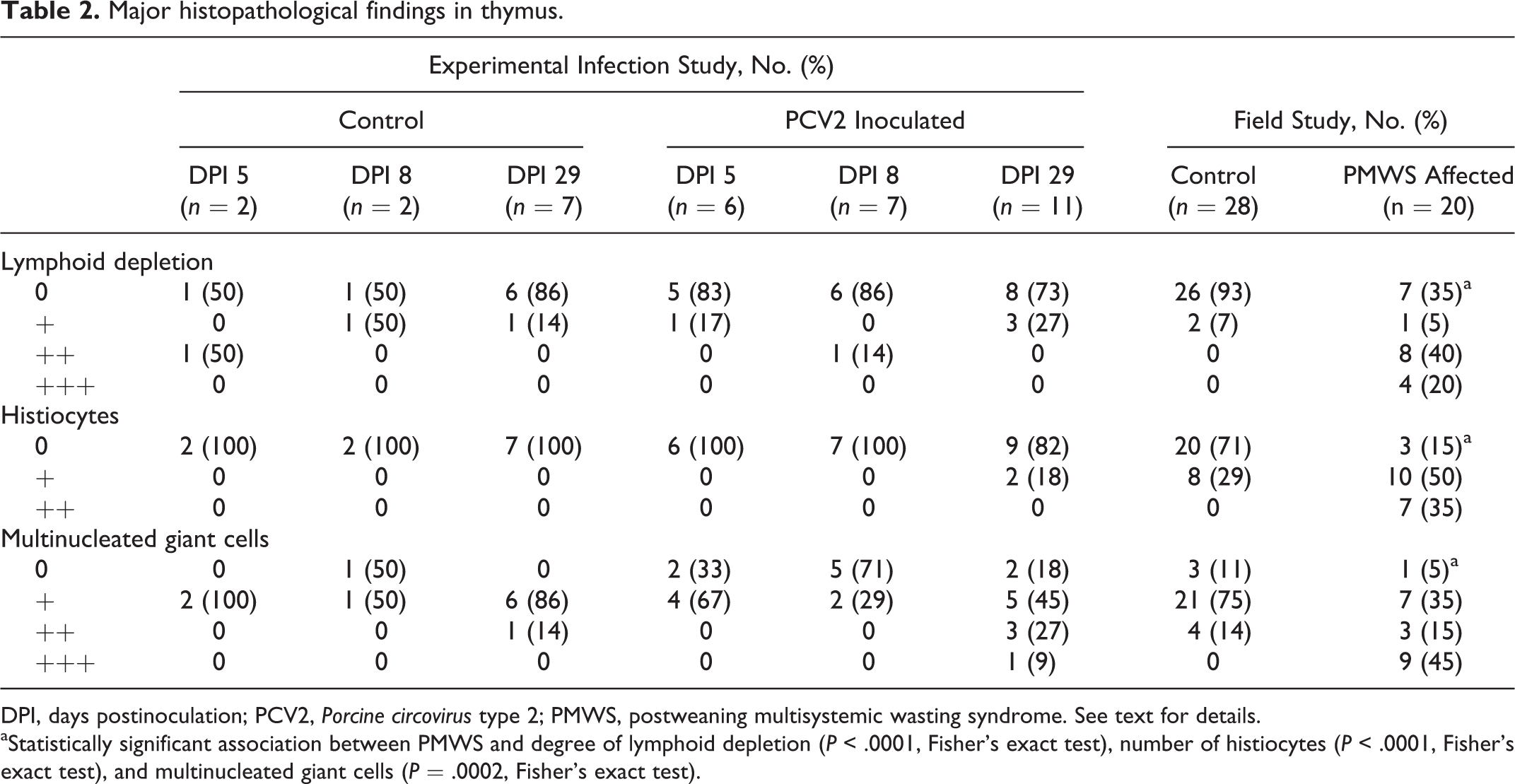
DPI, days postinoculation; PCV2, Porcine circovirus type 2; PMWS, postweaning multisystemic wasting syndrome. See text for details.
aStatistically significant association between PMWS and degree of lymphoid depletion (P < .0001, Fisher’s exact test), number of histiocytes (P < .0001, Fisher’s exact test), and multinucleated giant cells (P = .0002, Fisher’s exact test).
Inclusion bodies were not found in any of the thymuses.
All sections of thymus contained variable numbers of eosinophilic mononuclear cells, which contained multiple small uniform, intensely eosinophilic cytoplasmic granules (see Suppl. Fig. S2a). The majority of these cells had abundant cytoplasm and contained 1 large, round to oval and often eccentrically located nucleus, and mitotic figures could be seen. Accumulations of these cells were mainly seen in the connective tissue of trabeculae and vessels, but individualized cells were also located at the corticomedullary junction and diffusely spread in the medullary and cortical tissues. Mature eosinophilic granulocytes could also be seen adjacent to the mononuclear eosinophilic cells, and both cell types stained positive by Luna staining (see Suppl. Fig. S2b). The proportion of eosinophilic mononuclear cells to mature eosinophilic granulocytes differed from pig to pig.
In Situ Hybridization
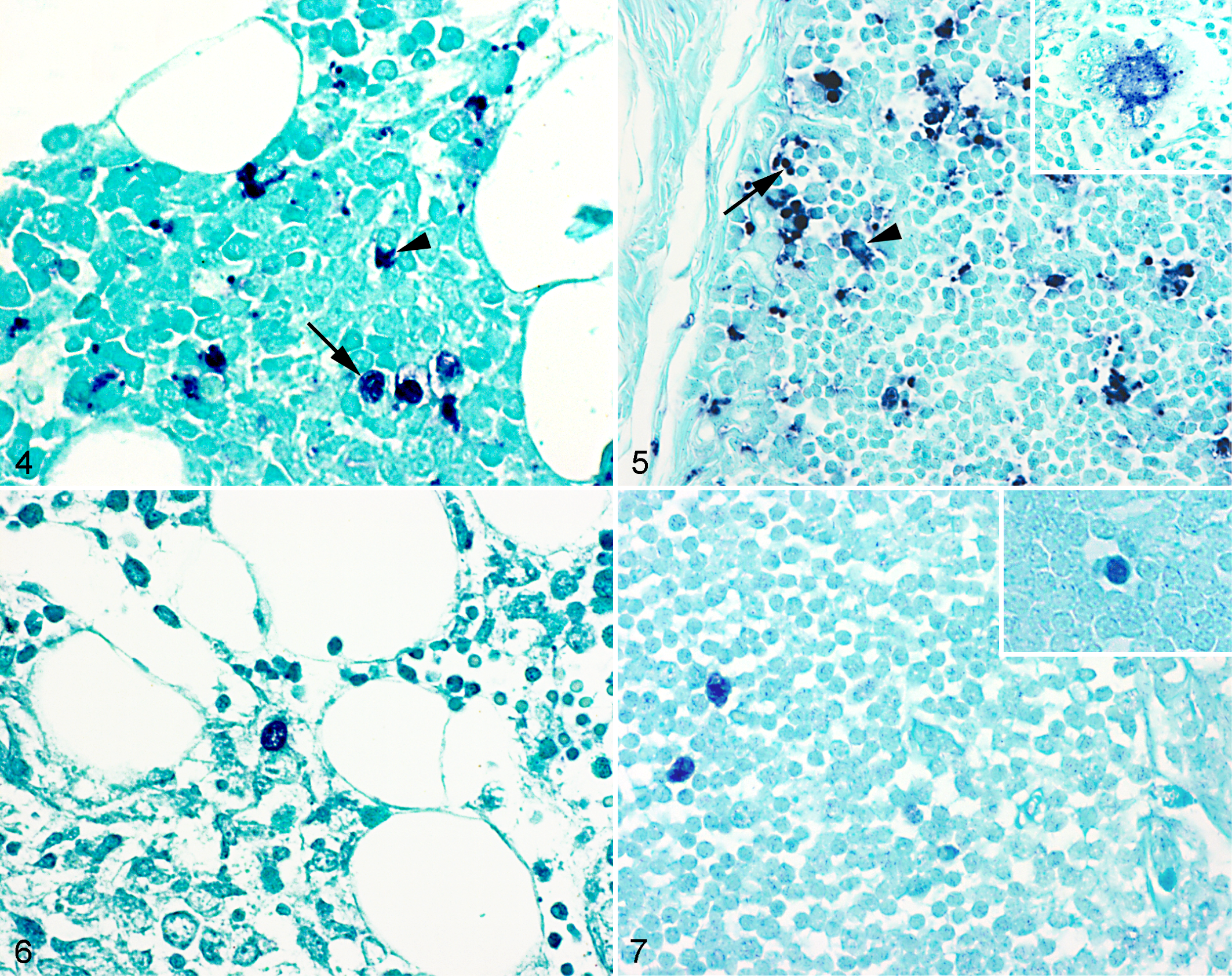
The results of CP ISH are displayed in Fig. 3. All sections of bone marrow and thymus from the controls and PCV2-inoculated pigs of the experimental infection study were negative for PCV2 by CP ISH, and this was also the case for the healthy animals from the field study. In the PMWS-affected animals, 19 of 20 (95%) available thymuses had a positive reaction of varying intensity with the CP ISH, whereas the CP ISH only gave a low-grade reaction in the bone marrow (16/33, 48%) (Figs. 4 and 5). Detection of PCV2 in thymus and bone marrow was associated with systemic moderate or massive PCV2 loads, detected by CP ISH in the secondary lymphoid organs (Table 3).
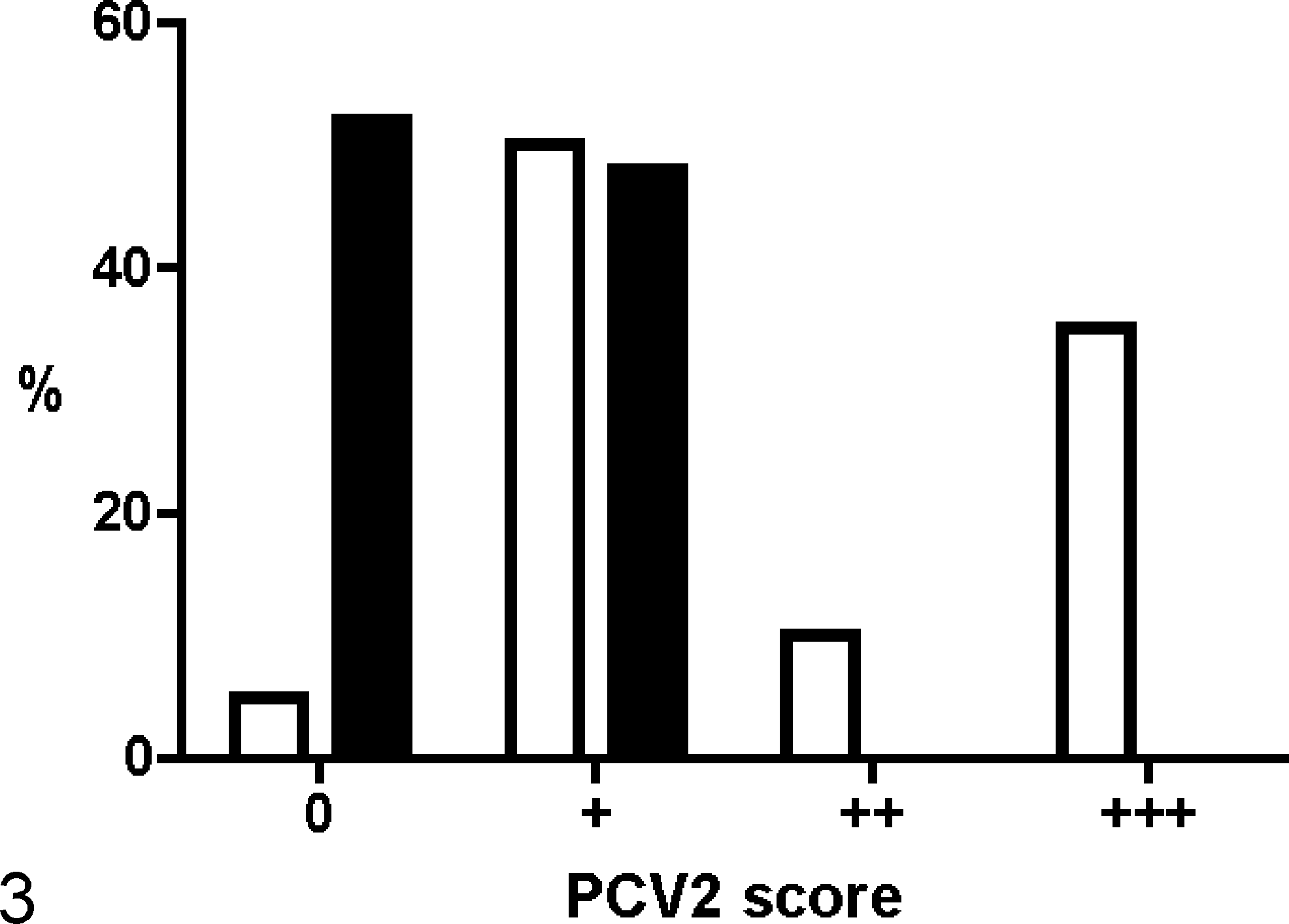
Scoring and detection of Porcine circovirus type 2 (PCV2) nucleic acids by complementary probe in situ hybridization in thymus and bone marrow from naturally infected pigs. PMWS, postweaning multisystemic wasting syndrome; %, percentage of pigs; □, thymus, PMWS affected (n = 20); ▪, bone marrow, PMWS affected (n = 33).
Porcine circovirus type 2 (PCV2) load in the secondary lymphoid organs vs PCV2 load in thymus and bone marrow detected by in situ hybridization (ISH) with the complementary probe (CP) and the replicative form probe (RFP).
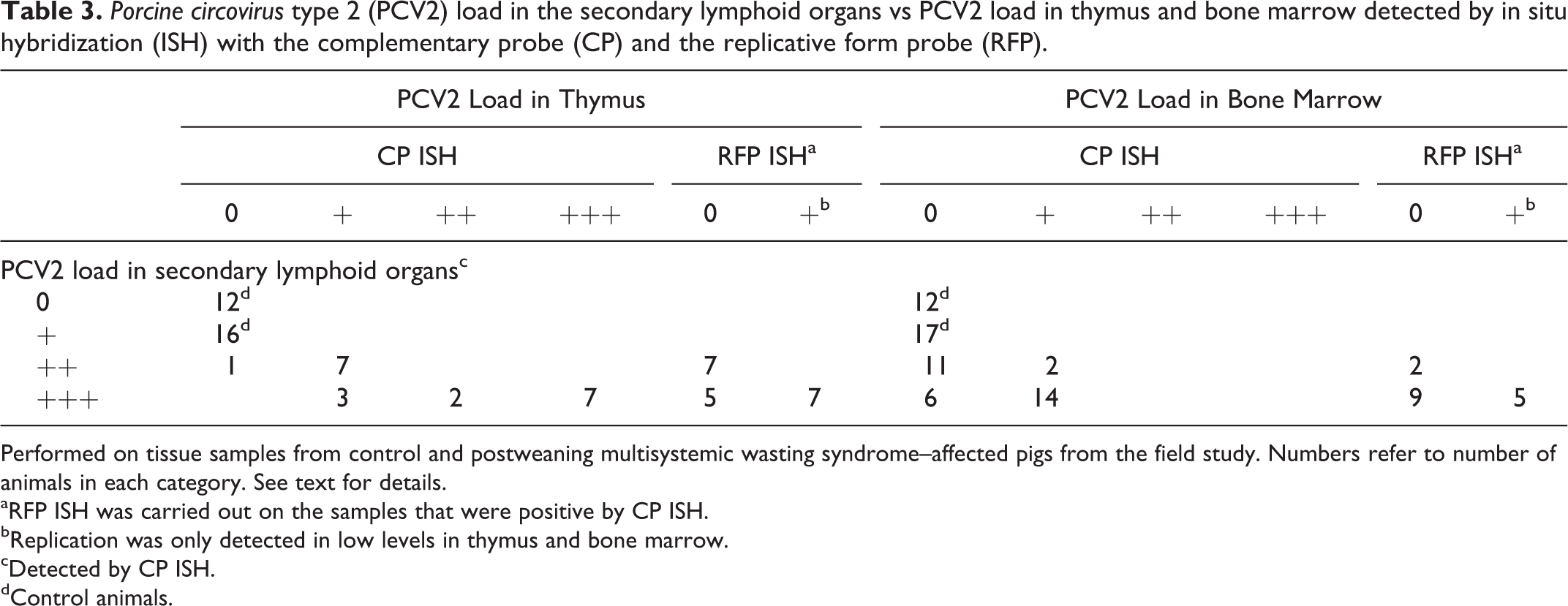
Performed on tissue samples from control and postweaning multisystemic wasting syndrome–affected pigs from the field study. Numbers refer to number of animals in each category. See text for details.
aRFP ISH was carried out on the samples that were positive by CP ISH.
bReplication was only detected in low levels in thymus and bone marrow.
cDetected by CP ISH.
dControl animals.
By RFP ISH, a few replication-positive cells were detected in 7 of 19 (37%) and 5 of 16 (31%) CP ISH-positive thymus and bone marrow samples, respectively (Table 3). RFP ISH-positive reaction was seen exclusively in animals with massive PCV2 load by CP ISH (Figs. 6 and 7). The CP gave a positive reaction in both bone marrow and thymus for 9 of the 20 animals (45%) where both organs were available. Four of these 9 animals (44%) had a positive reaction in both bone marrow and thymus with the RFP, 3 (33%) animals were only positive in the thymus, and the remaining 2 (22%) animals were negative in both organs.
The CP ISH labeled the cytoplasm and sometimes the nucleus of the positive cells in both thymus and bone marrow, whereas the RFP ISH only stained the nucleus of the positive cells. The CP- and RFP-positive cells in the bone marrow had the morphology of histiocytes (ie, large cells with a large oval nucleus and abundant cytoplasm), which were primarily located in the lymphoid aggregates and perivascular tissues (see Suppl. Fig. S1b). In thymus, the CP mainly labeled the cytoplasm of histiocyte-like cells in the cortex and medulla, but labeling of the nucleus of cortical lymphocytes (which were small- to medium-sized round cells with a round darkly stained nucleus and sparse cytoplasm) could also be detected in some thymuses (Fig. 5). The RFP-positive cells were seen primarily in the cortex of the thymus, and most of them were histiocyte-like, but positive cortical lymphocyte-like cells were also observed in some of the thymuses (Fig. 7).
Discussion
In the present study, PCV2 nucleic acids and viral replication were detected in bone marrow and thymus of PMWS-affected pigs, but there was no evidence that primary lymphoid organ cells are major supporters of PCV2 replication.
Like others, 31,45 we have considered if the viral replication takes place early in the PCV2 infection, even before clinical signs develop, and therefore materials from experimentally PCV2-inoculated animals were examined. In the original experimental infection study by Fernandes et al, 12 all the PCV2-infected pigs seroconverted, and lymph nodes were found PCV2 positive by CP ISH, but in the present study, no PCV2 DNA could be detected in thymus or bone marrow from these animals. This is in contrast with previous experimental infection studies, where PCV2 was detected by IHC in thymus (bone marrow not examined) from specific pathogen-free piglets 27 and conventional pigs. 45 In the present study, the piglets were inoculated with a PCV2a genotype, 12,36 which induced low-grade disease. A review found that the highest potential of reproducing PMWS follows an experimental protocol that includes inoculation with high doses of PCV2b together with another porcine pathogen, 43 which might partially explain why PCV2 was not detected in the thymus and bone marrow in the present study.
In general, the finding of PCV2 (by CP ISH) in thymus and bone marrow of the PMWS-affected pigs was associated with high amounts of PCV2 nucleic acids in the secondary lymphoid organs. This could indicate that the virus has a preference for the secondary lymphoid organs over the primary lymphoid organs. Among the PMWS-affected pigs, most of the thymuses (95%) were positive for PCV2 by CP ISH, whereas PCV2 could only be detected in the bone marrow samples of half of the pigs. Furthermore, the virus load in the thymus was in general higher compared with that of the bone marrow. These findings indicate that thymus is more susceptible to the PCV2 infection or that bone marrow is more resistant. The percentage of RFP ISH-positive thymuses (37%) and bone marrow samples (31%) was almost identical, indicating that the ratio of replication was similar in the 2 organs.
In the bone marrow and thymus, PCV2 nucleic acids were primarily found in the cytoplasm of histiocytes, which supports the general accepted idea that histiocytes are the main cell type responsible for passive accumulation and systemic spread of PCV2. 10,30 Most of the RFP-positive cells in the bone marrow and thymus were also histiocytes, but some cortical lymphocytes also supported PCV2 replication, although it was not possible to tell whether the cortical lymphocytes were thymocytes or lymphocytes migrating through thymus. PCV2 replication was not found in hemopoietic or reticular cells of the bone marrow; furthermore, the M:E ratios were within the normal range, 41 indicating that the bone marrow is not a specific target organ with regard to PCV2 infection. These results are in agreement with previous reports by Pérez-Martín et al, 31 Yu et al, 45 and Hamberg et al, 17 who could not link PCV2 replication to a specific cell type but found that replication was supported by different cell types, including histiocytes and lymphocytes. In the search for other organs that may support viral replication, it could be interesting to look into the ileal Peyer’s patch (IPP), since it has become known that it functions as a primary lymphoid organ in juvenile mammals. 4,44 Furthermore, the IPP is a mammalian homologue to the avian bursa of Fabricius, which is a major supplier of B lymphocytes, 44 and it is affected by the avian circoviruses. 37,42 In PMWS, the associated lymphopenia and lymphoid depletion affect B lymphocytes before T lymphocytes, 29,33 and the B lymphocytes also seem to be more susceptible to PCV2 infection. 24,45 Therefore, the function of the IPP and its influence on the total number of B lymphocytes in vivo should be examined further in pigs with PMWS. Ideally, a longitudinal study permitting temporal tracing of events in individual pigs should be carried out; however, this requires a solid PMWS experimental model, which is not available at present.
Serous fat atrophy of bone marrow is characterized macroscopically by the presence of a gelatinous substance. Histological findings are atrophic adipocytes, marrow hypoplasia, and deposition of acid mucopolysaccarides, which are visible on HE stainings as an amorphous ground substance that stains positive by Alcian blue. 2,8 Based on this, the observed AFM likely represents serous fat atrophy, although no macroscopic findings were observed during necropsy. Serous fat atrophy is seen in many different conditions. 2 This can explain the finding of AFM in 1 of 12 control animals from the experimental study and 4 of 24 control animals from the field study, indicating that these animals may have had another disease. However, the AFM score was higher (P < .0001) in the PMWS-affected animals compared with the control animals, which is consistent with the fact that reduced weight gain and wasting are some of the general clinical signs of PMWS. 15 To our knowledge, serous fat atrophy of bone marrow has not been described before in relation to PMWS.
In the bone marrow samples, the CP- and RFP-positive cells were often detected in lymphoid nodules, 40 which are clusters of histiocytes, B lymphocytes, and T lymphocytes, and can be seen in relation to chronic inflammation and viral infections. 11,40 Although not significant (P = .07), this finding in the bone marrow of the PMWS-affected animals correlates well with the chronic nature of PMWS.
In the thymus, the significant findings of lymphoid depletion (P < .0001) and infiltration of histiocytes (P < .0001) and multinucleated giant cells (P = .0002) in cases of PMWS, compared with the control animals, correlate with previous reports. 35 Moreover, all thymuses contained varying numbers of mononuclear cells with multiple uniform eosinophilic granules. Thymus eosinophilia has been described in humans, 21 mice, 21 and pigs. 1,5 Based on the morphology, positive Luna staining, and descriptions from the human thymus, 13 we believe that the eosinophilic cells in the porcine thymus were eosinophilic precursors. Since the exact function of these thymic mature and immature eosinophilic granulocytes in humans is still obscure, 13,21 it might be beneficial to use the pig as a model to elucidate the role of the thymic eosinophilic granulocytes. The eosinophilic precursors seemed not to be associated with PMWS, since the quantity and distribution were the same in PMWS cases and controls.
In conclusion, the primary lymphoid organs are definitively affected by the PCV2 infection, but in the present study, there was no evidence for thymus or bone marrow being of importance to support PCV2 replication in cases of PMWS.
Footnotes
Acknowledgements
We acknowledge Betina Andersen, Lisbeth Kioerboe, Hanne H. Moeller, Dennis Brok, Annie Ravn Pedersen, Mónica Pérez, and Rosa Valle for excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
