Abstract
Feline spongiform encephalopathy (FSE) is a transmissible spongiform encephalopathy associated with the consumption of feedstuffs contaminated with tissue from bovine spongiform encephalopathy-affected cattle and characterized by the accumulation in the central nervous system of an abnormal isoform of the prion protein (PrPsc). Clinically, it presents as a progressive fatal neurologic syndrome that is not easily distinguished from other feline neurologic conditions. Most cases of FSE have been reported in England, where it was first detected in 1990, but a few cases have been reported from other European countries. To identify possible cases of FSE in Italy, the Italian Ministry of Health funded a 2-year surveillance project during which the brains from 110 domestic cats with neurologic signs were evaluated histologically for spongiform encephalopathy and immunohistochemically to detect PrPsc. Although no cases of FSE were found, the study proved useful in monitoring the Italian cat population for other neurologic diseases: neoplasia (21.8%), toxic-metabolic encephalopathy (18.2%), granulomatous encephalitis (15.5%), suppurative encephalitis (4.6%), trauma (3.6%), circulatory disorders (3.6%), degeneration (2.7%), nonsuppurative encephalitis (2.7%), and neuromuscular diseases (1.8%). No histologic lesions were found in 20% of the brains, and samples from 5.5% of the cats were rejected as unsuitable.
Introduction
Feline spongiform encephalopathy (FSE) is a transmissible spongiform encephalopathy (TSE) caused by conversion of the cellular prion protein (PrPc) into an insoluble protease-resistant isoform (PrPsc). 37 This fatal neurodegenerative disease is associated with the consumption of bovine spongiform encephalopathy (BSE)-contaminated food. Experimental data support a link between FSE and BSE, as mice inoculated with brain homogenates of FSE-affected cats had the incubation period and lesion profile characteristics of the BSE strain. 9
FSE was first identified in the UK in April 1990 during the epizootic of BSE. Ninety cases have been identified in Great Britain, 11 1 in Northern Ireland (Federal Register, 1997), 1 in Norway, 1 1 in Liechtenstein (MAFF [2001] UK statistics on incidence of BSE and related diseases http://www.defra.gov.uk/animalh/bse/othertses/index.html), 1 in Portugal (Silva JF, Correia JJ, Ribeiro J, Carmo S, Orge L: Feline spongiform encephalopathy: first confirmed case reported in Portugal, PRION 2006 Abstract Book, Poster Session-Diagnosis [DIA-45]:392, 2006), and 2 in Switzerland (BVET-OVF-UFV, http://www.bvet.admin.ch/index.html?lang = it). 5 Domestic cats with FSE are generally 4 to 9 years of age (mean, 6.6 years). 38 FSE has also been reported in other felids, 24 including the cheetah (Acinonyx jubatus), 19 ocelot (Felis pardalis), puma (Felis concolor), 36 lion (Felis leo), and tiger (Panthera tigris). 16 The clinical signs and pathologic features of the disease are similar in all susceptible species of Felidae. 13, 32
Clinically, FSE is characterized by progressive neurologic signs such as locomotor dysfunction, abnormal behavior, and altered response to sensorial stimulation. These clinical signs are not easily distinguished from those of other feline neurologic syndromes. 13, 32, 37
FSE, like other TSEs, can be diagnosed only postmortem by histologic, immunohistochemical, or Western blot examination of brain tissue to identify the typical spongiosis and PrPsc deposition. 23 Spongiosis is widespread in the brain and most marked in the medial geniculate nucleus and the basal nuclei. 24, 37 The degree of vacuolation in the neuropil can be marked, progressing to status spongiosis in some cases. Vacuolation is also present in neuronal perikarya, particularly in the raphe nucleus, the dorsal motor nucleus of the vagus nerve, and the red nucleus. There is vacuolation in the white matter mainly in the medulla. 37 Spongiosis is always accompanied by glial responses. PrPsc is typically detectable throughout the brain of cats with FSE as small aggregates, with punctate, particulate, and linear strands of immunoreactivity. 24
In 1998, Zanusso et al. reported the simultaneous occurrence of spongiform encephalopathy in a 60-year-old Italian man and his cat. The prion strain in the brain of the cat was similar to that in the man, but distinct from the UK strain of BSE (new variant Creutzfeldt-Jakob disease and its feline counterpart). 39 Because no BSE-related cases of FSE had been detected in Italy, the Italian Ministry of Health funded a surveillance project to screen adult cats with untreatable neurologic disease for FSE.
Materials and Methods
In this multicenter study, the reference center for Transmissible Spongiform Encephalopathy was the Centro Encefalopatie Animali (CEA) – Istituto Zooprofilattico Sperimentale (IZS) del Piemonte, Liguria e Valle d'Aosta, which collaborated with the Universities of Pisa, Perugia, and Padua, and the IZS of Sicily and Sardinia. The Italian Association of Veterinary Neurology (SINVet) kindly offered technical scientific support. Veterinary practitioners were introduced to the project before its commencement at conferences and congresses, and their voluntary participation was requested. During the 2-year project, 110 cats, older than 1 year with severe neurologic signs and poor prognosis, were submitted after natural death or euthanasia by veterinary practitioners to the CEA and to the participating study centers.
Neurologic signs were assessed using a standard clinical examination protocol. Details on age, sex, breed, history, clinical signs, serum biochemistry, ancillary investigations, and differential diagnosis were recorded on a standardized form. At postmortem examination, brains and major organs were sampled, fixed in 10% buffered formalin, and routinely processed for histopathologic examination. Sections of brain from representative areas (medulla oblongata, pons, cerebellum, mesencephalon, diencephalon, and telencephalon), and from different spinal cord segments were stained with HE, periodic acid–Schiff (PAS), and Luxol fast blue. When necessary, immunohistochemistry was performed on selected central nervous system sections to establish an etiologic diagnosis. Feline coronavirus antigen was detected by immunohistochemistry with monoclonal antibody FCV3-70 (Custom Monoclonals International, West Sacramento, CA). In case of neuromuscular disease, peripheral nerve samples were fixed in 2.5% glutaraldehyde and processed for electron microscopy. Ultrathin sections were stained with uranyl acetate and lead citrate and examined with electron microscope. In 1 case, 15-µm-thick paraffin sections of cerebellum were used for DNA extraction with DNeasy Tissue Kit (QIAGEN, GmhH, Hilden, Germany). 25 A polymerase chain reaction (PCR) test was performed to amplify the entire open reading frame 2 of canine and feline parvovirus (CPV-2/FPV), using the set of primers P33/34 (V. Martella, unpublished results). In some cases, the diagnostic workup included computed tomography or magnetic resonance imaging. In suspected cases serologic tests for feline leukemia virus (FeLV) and feline immunodeficiency virus (FIV) were carried out.
After morphologic or etiologic diagnosis, the study centers sent the medulla oblongata (obex) samples and any other brain regions with vacuolar change to the CEA laboratories for histologic and immunohistochemical detection of prion disease. For immunohistochemical detection of PrPsc accumulation, deparaffinized and rehydrated sections were treated with 98% formic acid for 20 minutes at room temperature, then autoclaved in buffered citrate (pH 6.1) at 121°C for 30 minutes. After rinsing, the sections were incubated overnight at 4°C with anti-PrP monoclonal antibody L42 (R-biopharm RIDA mbL42, Italia s.r.l. Cerro al Lambro, Milan, Italy) diluted 1 : 250. Subsequent antibody detection was carried out using biotinylated goat anti-mouse secondary antibody diluted 1 : 200 for 20 minutes (Vector Laboratories, Burlingame, CA) at room temperature, followed by the avidin–biotin–peroxidase complex (Vectastain ABC kit, Vector Laboratories). Immunoreactivity was visualized using 3,3′-diaminobenzidine as chromogen; the sections were counterstained with Mayer's hematoxylin counterstain. Paraffin sections of medulla oblongata of a cat with FSE (kind gift of A. Oevermann, University of Berne, Switzerland) were used as the positive control.
Results
The most commonly recorded clinical signs were ataxia, altered grooming, hypersalivation, tremors, hyperesthesia and incoordination. The age of 93/110 cats was recorded. Mean age was 7.5 years; 67 cats were 1 to 10 years of age; the remaining 26 were 10 to 20 years of age. Breed was recorded for 94 cats. Most cats were of mixed breed (n = 81); 9 were Persian; 2 were Siamese; 1 was Soriano; 1 was Certosino. Gender was recorded in all but 7 cats: 32 intact females, 14 spayed females, 45 intact males, and 12 castrated males.
No histologic lesions of FSE were found in any cat; spongiform changes were present at the level of the obex in only 2 cases, which were diagnosed respectively as hepatic and renal encephalopathy. All immunohistochemical examinations for PrPsc were negative. Nevertheless, histologic lesions were present in 74.5% of the brain samples (Fig. 1). The lesions were ascribed to a spectrum of neurologic diseases that were classified in descending order of frequency as: neoplasia (21.8%), toxic-metabolic encephalopathy (18.2%), granulomatous encephalitis (15.5%), suppurative encephalitis (4.6%), traumatic lesions (3.6%), circulatory disorders (3.6%), degenerative lesions (2.7%), nonsuppurative encephalitis (2.7%), and neuromuscular disease (1.8%). No histologic lesions were detected in 20% of the samples; 5.5% were rejected as unsuitable.
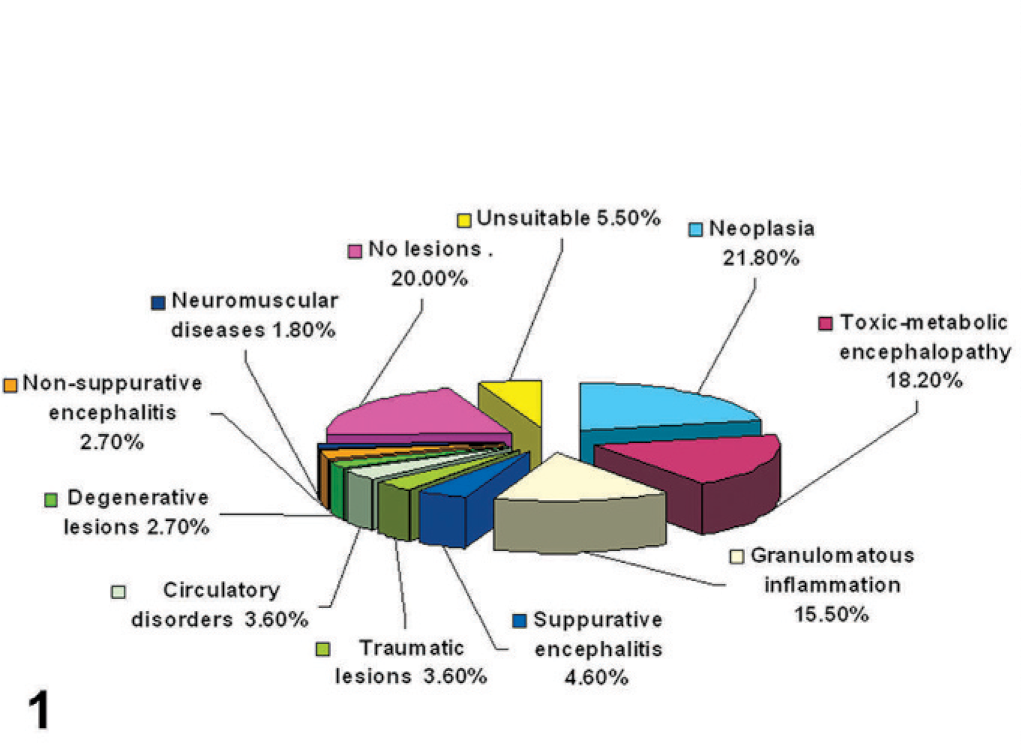
Diagram. Graphic representation of the diagnoses from examination of brain samples of 110 cats with neurologic signs.
Neoplasia
The 24 cases of neoplasia included 11 metastatic tumors, 8 lymphomas, 4 meningiomas, and 1 pituitary adenoma. Of the 11 metastatic tumors, 3 were pulmonary carcinomas, 2 ceruminous carcinomas, 2 mammary carcinoma, 1 lingual squamous cell carcinoma, 1 mast cell tumor, 1 cutaneous squamous cell carcinoma, and 1 renal lymphoma. One lymphoma formed a discrete mass that was limited to the brain and composed of angiocentric neoplastic cells mainly in white matter. Tumor cells were large with a round nucleus and a prominent central nucleolus. The other 7 lymphomas had similar cytologic features and formed part of multicentric lymphoma (Fig. 2). All 8 cats with lymphoma tested negative for FIV antibodies and FeLV antigens.
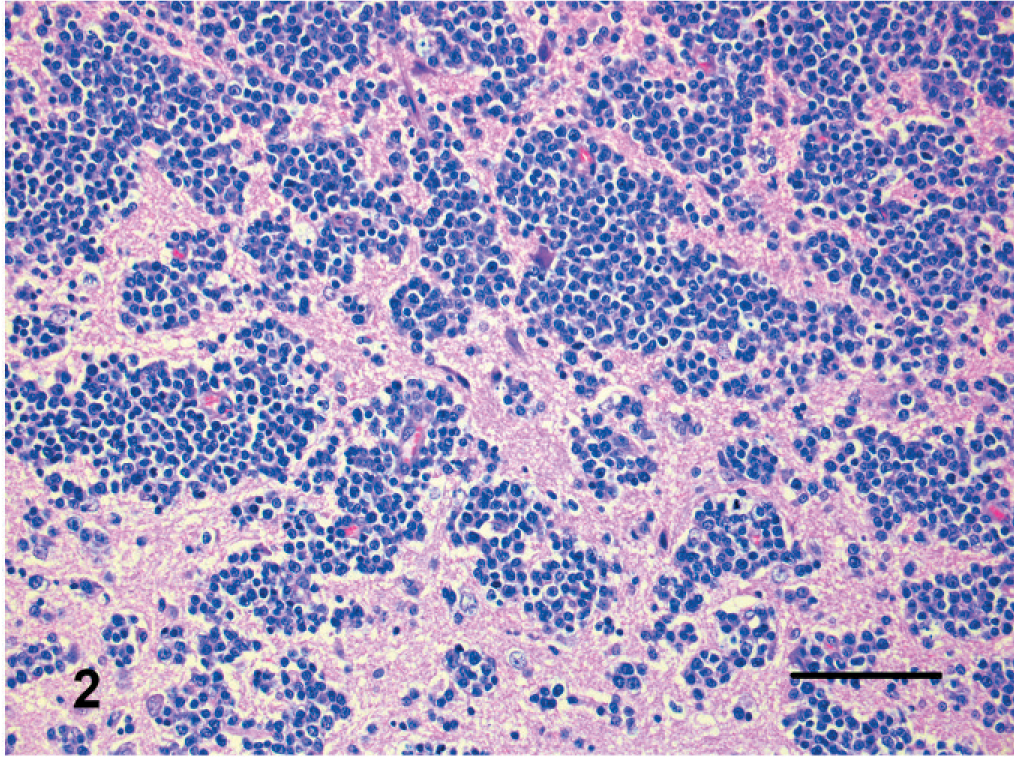
Brain; cat. Neoplastic lymphocytes have perivascular orientation with neuroparenchymal infiltration. HE. Bar = 150 µm.
Two meningiomas were classified as transitional; 2 were psammomatous. Transitional meningiomas (Fig. 3) had features of both meningiotheliomatous and fibroblastic meningioma. In psammomatous meningiomas (Fig. 4), neoplastic cells were arranged in whorls around small vessels or hyalinized structures with degenerated and mineralized wall (psammoma bodies).
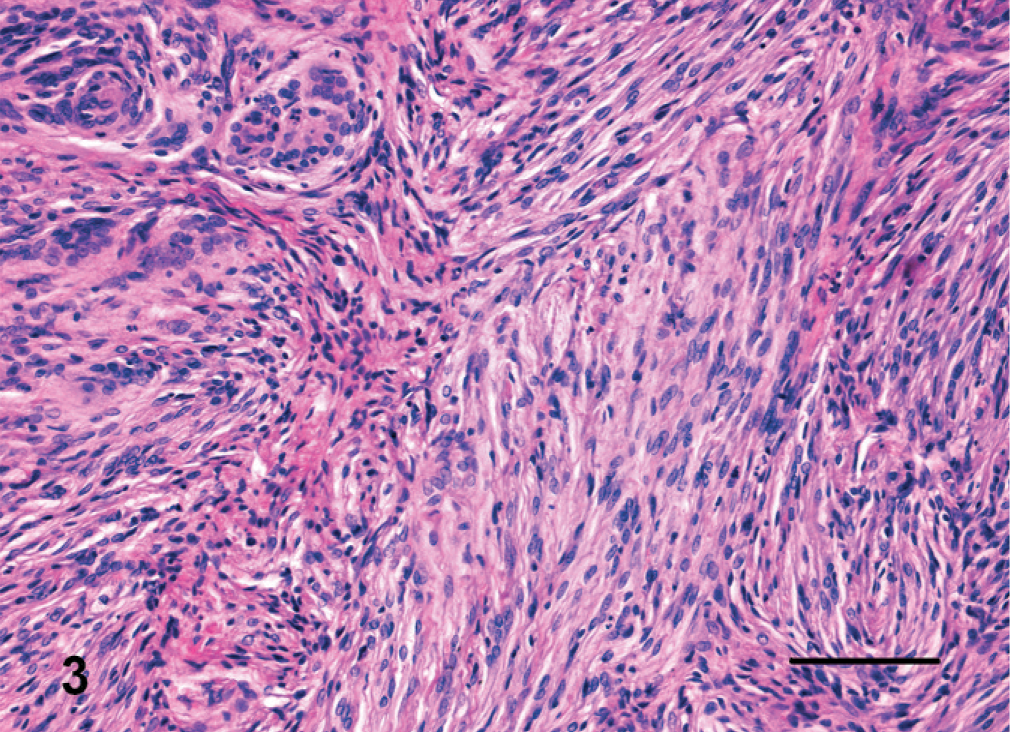
Brain; cat. Transitional meningioma has whorls of neoplastic meningothelial cells separated by streams of spindloid cells. HE. Bar = 150 µm.
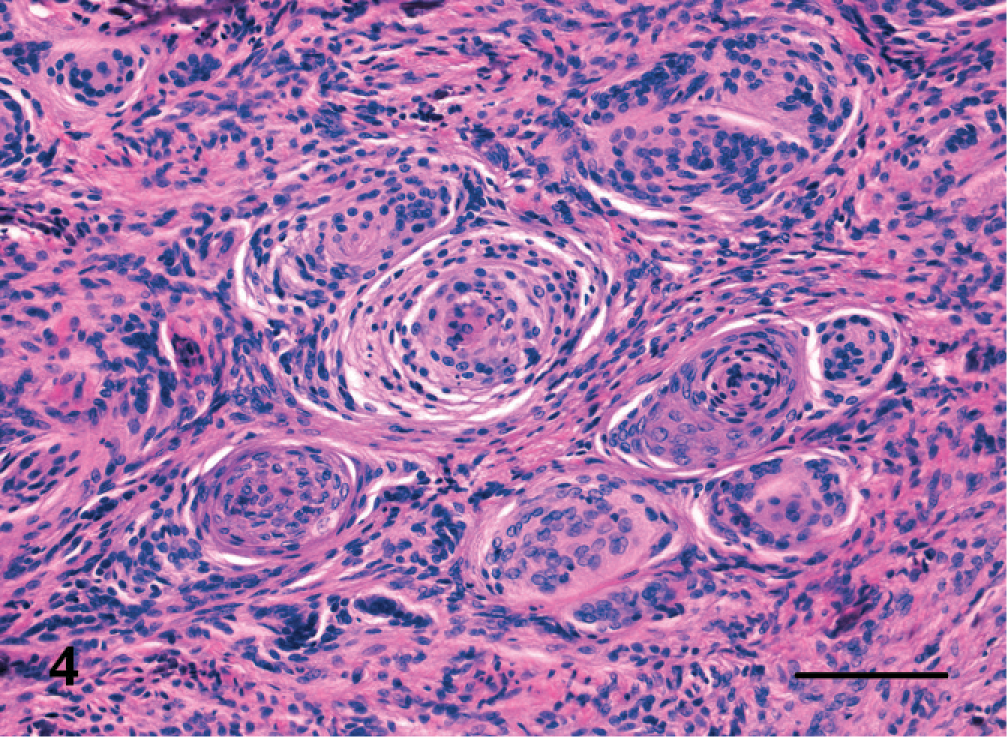
Brain; cat. Psammomatous meningioma characterized by small whorls of neoplastic cells and vessels, sometimes encrusted with calcium salt forming psammomatous bodies. HE. Bar = 150 µm.
The pituitary tumor consisted of sheets and trabeculae of basophilic cells with a moderate amount of cytoplasm and large round oval nuclei with a prominent nucleolus.
Toxic-metabolic disorders
The heterogeneous lesions in this category were ascribed to hepatic or renal encephalopathy (n = 15) (Fig. 5), thiamine (vitamin B1) deficiency (n = 1) (Fig. 6) and hippocampal necrosis (n = 4) (Fig. 7). Hepatic encephalopathy was diagnosed on the basis of degenerative changes in the brain associated with severe hepatic degeneration and necrosis (n = 7). In the cerebral hemispheres, microcavitation of the white matter at the junction with the gray matter, as well as bilateral and symmetrical vacuolation of the internal capsule, cerebellar peduncles and caudal brain stem were the most common lesions. Alzheimer type II cells (astrocytes with clear nuclei and marginated chromatin) were observed in some cases.
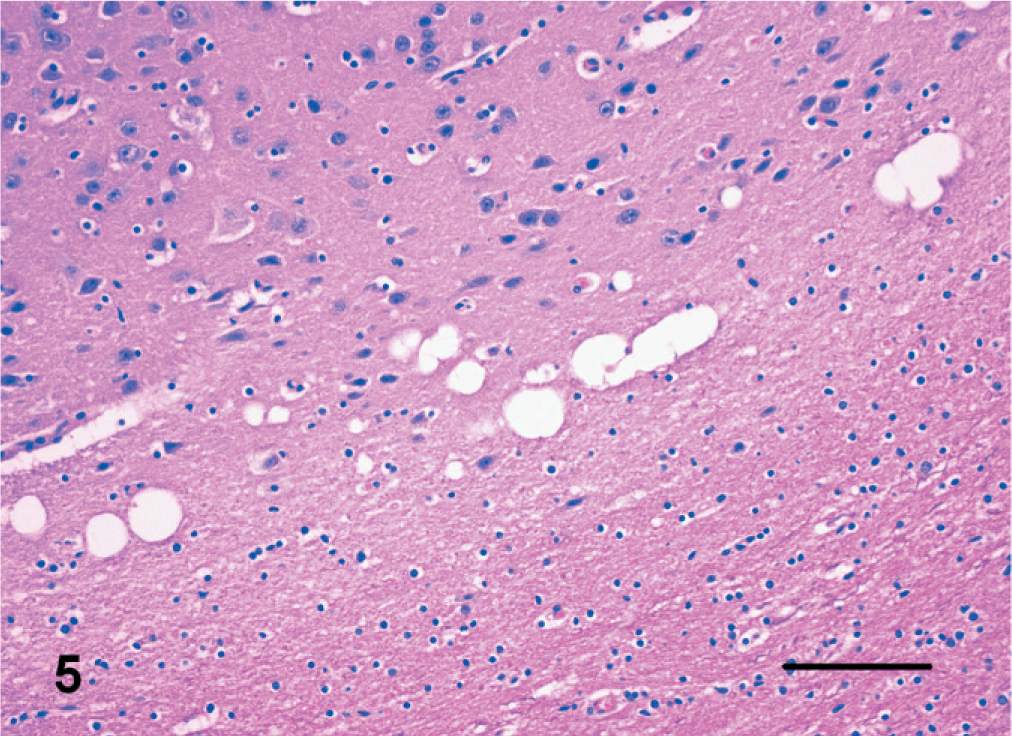
Brain; cat. Hepatic encephalopathy with microcavitation of white matter at the junction with cerebral cortex. HE. Bar = 150 µm.
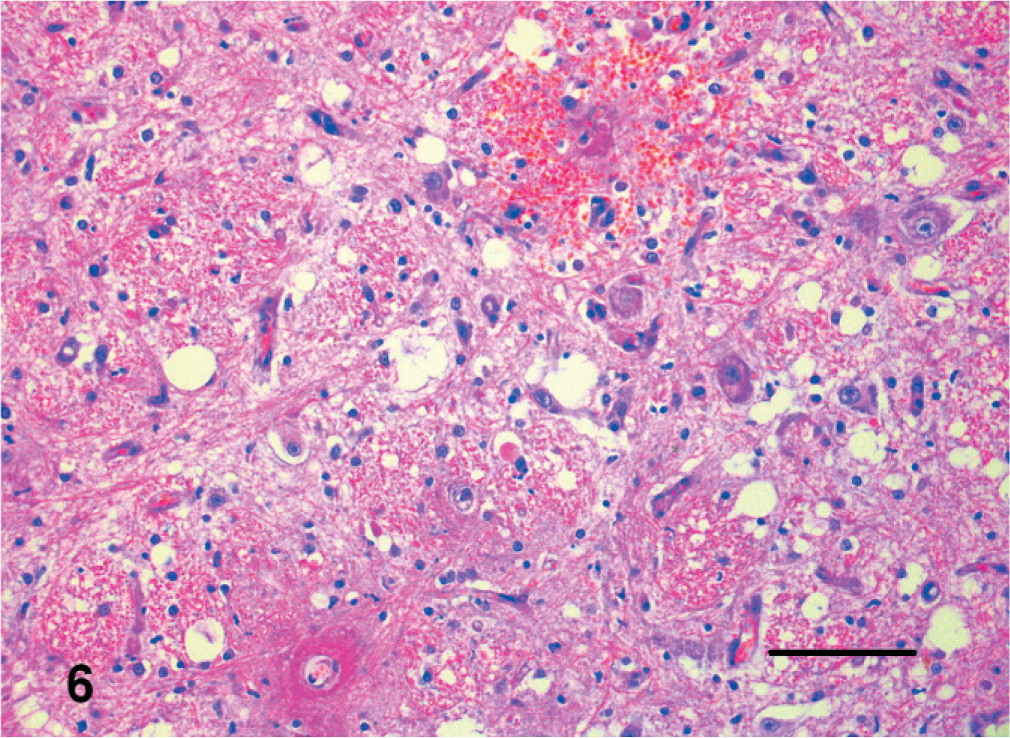
Brain; cat. Lesions of thiamine deficiency include focal hemorrhage, astrocytosis, and vacuolation. HE. Bar = 150 µm.
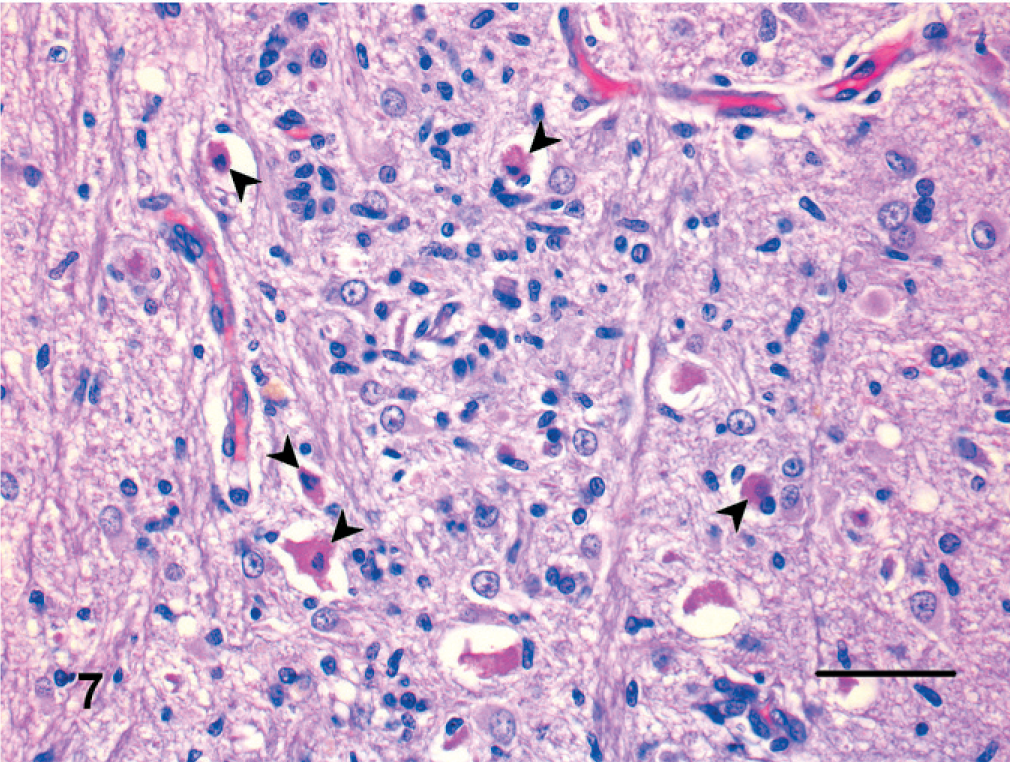
Brain; cat. Hippocampal necrosis. Many pyramidal neurons are shrunken with pyknosis (arrowheads). Reactive astrocytes are also evident. HE. Bar = 75 µm.
Multifocal vacuolation of the white matter was also seen in the brain of 8 cats with severe chronic interstitial nephritis and a clinical history of renal failure. In these cases, a diagnosis of renal encephalopathy was made.
In the cases of thiamine deficiency, bilaterally symmetric malacic areas in the caudal colliculi were characterized histologically by necrosis with macrophage infiltration, astrogliosis, and vascular proliferation.
In the 4 cases of hippocampal necrosis, lesions were mostly restricted to the hippocampus and pyriform lobe. There were neuronal loss and necrosis in the pyramidal cell layer and at the level of the small neurons of the pyriform lobe. Lesions were characterized also by the presence of hypertrophic astrocytes (gemistocytes) and fibrillary astrocytes. Most neurons were shrunken and had pyknotic or lytic nuclei surrounded by brightly eosinophilic cytoplasm. In addition, capillary proliferation and swelling of the endothelial nuclei were prominent.
Granulomatous encephalitis
All 17 cats (mean age, 4 years) with granulomatous inflammation were affected by feline infectious peritonitis (FIP) caused by feline coronavirus (Fig. 8). Diagnosis was based on the presence of characteristic lesions of FIP, including pyogranulomatous ependymochoroiditis and meningitis with vasculitis. Immunohistochemistry was performed in inconclusive cases, and the presence of viral antigen, mainly within the cytoplasm of macrophages, confirmed the diagnosis.
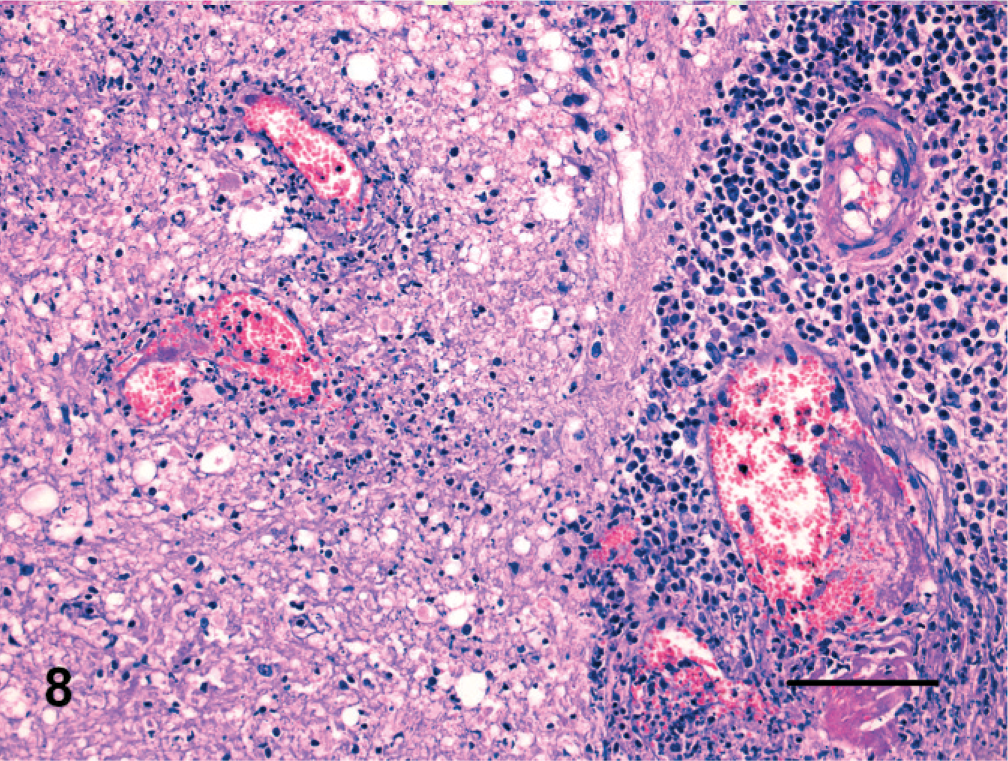
Brain; cat. Feline infectious peritonitis characterized by pyogranulomatous meningitis with infiltration of plasma cells, neutrophils, lymphocytes, macrophages, and vessel wall necrosis. Degenerative changes of subpial neuroparenchyma are also evident. HE. Bar = 150 µm.
Suppurative encephalitis
This group included 1 cat with brain abscessation, 3 with other purulent inflammations, and 1 fungal infection. The mycotic infection was associated with suppurative meningoencephalitis with multiple cystic cavities in which there was central necrosis, and PAS+ encapsulated yeasts of about 20-µm diameter consistent with Cryptococcus. The yeasts were associated with vascular necrosis; adjacent parenchyma was infiltrated by lymphocytes, plasma cells, and epithelioid macrophages. No bacterial cultures were done on these brains, since they were submitted to our study as formalin-fixed tissues.
Circulatory disorders
Circulatory disorders were seen in 4 cats. Lesions were characterized by a massive spongy state attributed to edema and variably associated with ischemic neuronal necrosis.
Traumatic lesions
Hemorrhage was present in the brains of the 4 cats with head trauma. Some hemorrhagic foci were infiltrated by macrophages and fibrillary astrocytes.
Degenerative lesions
This group consisted of 2 cases of cerebellar abiotrophy and 1 of motor neuron disease. The 2 cats with cerebellar abiotrophy had a history, since 1 year of age, of difficulty in walking and neurologic signs attributable to cerebellar dysfunction. Marked multifocal degeneration and loss of cerebellar Purkinje cells, with an increase in the number of Bergmann's glia, were consistent with cerebellar abiotrophy. Diffuse myelin vacuolation and axonal swellings (spheroids) were observed in the cerebellar white matter. Medial vestibular and cerebello-vestibular nuclei also had apparent decrease in the number of neurons with gliosis and axonal spheroids. No remarkable changes were found in the cerebrum and the PCR test for feline panleukopenia virus on brain tissue was negative.
The cat with motor neuron disease had severe neuronal loss in ventral horns of the spinal cord with reactive gliosis. The white matter of descending tracts had swollen axons and formation of digestion chambers with phagocytosis of axonal and myelin debris. Motor nerve roots had loss of myelin and presence of gitter cells. Ascending tracts were largely spared. In the brain, marked gliosis affected the medulla oblongata, pons, colliculi, mesencephalic tegmentum and thalamus. Axonal degeneration and secondary demyelination were observed focally.
Nonsuppurative inflammation
Three cats from different premises not adjacent to pig farms died within approximately 48 hours after the onset of aggressive behavior, postural defects, abnormal and continuous vocalization, excessive salivation, and severe dehydration with progression to polypnea and respiratory failure. All 3 cats had neuronal degeneration and necrosis, localized to the medulla and pons, with thin perivascular cuffs of lymphocytes and plasma cells and reactive microglial cells. Although intranuclear eosinophilic inclusions were not found in neurons, based on clinical and histologic features, nonsuppurative encephalitis was attributed to pseudorabies virus infection. 33 No further diagnostic tests were performed.
Neuromuscular diseases
This group included 1 case of diabetic polyneuropathy and 1 of ischemic neuromyopathy; neither case had brain lesions. Diabetic polyneuropathy was identified by the presence of splitting and ballooning of the myelin sheath. The ballooned, intramyelinic space was often spanned by strands of still-compacted myelin lamellae or vesicular debris. Aside from myelin defects, reactive, degenerative, and proliferative changes of Schwann cells were also evident. Ischemic neuromyopathy was characterized by severe axonal degeneration in the peroneal nerves, with numerous myelin ovoids and loss of nerve fibers, mainly involving the central portion of the fascicles in the proximal aspect of the nerve. The distal aspect of the nerve had a more random, multifocal pattern of axonal degeneration.
No detectable lesions
Twenty-two of the 110 cats had no significant lesions in brain or any other examined tissue. In an additional 6 cases, autolysis precluded complete pathologic assessment of the central nervous system.
Discussion
The aim of this project, as in a previous study, 15 was to search for possible FSE cases in the Italian feline population. However, in our sample of 110 adult cats with neurologic signs, no cases of FSE were found. Even so, this is the first detailed diagnostic evaluation of a large case series of neurologic disease in the Italian cat population. Some of the clinical signs in cats in this study have been described in FSE, 37 which emphasizes the importance of screening cats with neurologic disease for possible cases of FSE.
In this study, the most common neurologic lesions were categorized as neoplasia, toxic-metabolic encephalopathy, and granulomatous inflammation. Lymphoma was the most common brain tumor, but has been second to meningioma in most studies. 17, 20, 21, 30 Cats with lymphoma are variably seropositive for FeLV or FIV; 7, 31 however, all samples from the 8 cats with lymphoma in this study were negative for FIV antibodies and FeLV antigens. In 5 cases, lymphoma involved mainly the meninges of the brain and in 1 case was confined in the dorsal motor nucleus of the vagus of the obex. Similar localizations of this tumor were described in another study. 2
Thiamine deficiency encephalopathy in cats usually results from a fish diet rich in thiaminase; thiamine deficiency also may develop after consumption of meat preserved with sulfur dioxide. 28 The lesions in our cats were similar to those reported in other cases of thiamine deficiency. 4
As described in a previous report, 8 cats with hippocampal necrosis had bilateral degenerative lesions confined to the hippocampus and the pyriform lobe. The lesions resembled those of feline ischemic encephalopathy (FIE), but FIE was not found in this series. Feline ischemic encephalopathy is characterized by asymmetric cerebral signs and lesions that are attributed to ischemia; in some cases, a parasitic cause has been demonstrated. 34 The pathogenesis of hippocampal necrosis is still unknown. 9
FIP, which comprised the category of granulomatous inflammation, was diagnosed in 15.5% of the samples, but its prevalence in the Italian cat population as a whole was probably underestimated because FIP mainly affects cats less than 1 year old, an age range excluded by the study protocol.
All 3 cases of nonsuppurative encephalitis were diagnosed as pseudorabies. This herpesvirus infection occurs in cats after ingestion of contaminated pork; however, in these cases, no consumption of infected meat was reported by the cat owner. The clinical signs in our cases were similar to those in a previous report, 12 except for the lack of pruritus, which has also been absent in some natural and experimental cases. 10 The pathologic findings in the brain were the same as described elsewhere. 6, 28 Eosinophilic intranuclear inclusions may be found in neurons in pseudorabies but were absent in our cats.
Cerebellar abiotrophy has been described in most species, including cats, dogs, cattle, sheep, horses, and swine. A hereditary basis (usually consistent with autosomal recessive transmission) for the disease has been hypothesized. The clinical signs of cerebellar hypoplasia, caused by in utero exposure to the panleukopenia virus, 14 differ from those of cerebellar abiotrophy in that the former are present at birth or in the neonatal period and are nonprogressive. Our cats, in contrast, were normal at birth and acquired signs at around 1 year of age. Histopathologically, the lesions of cerebellar abiotrophy differed from the classic panleukopenia virus lesions, which include failure of normal development of the granular cell layer and disorganization and loss of Purkinje cells. 14, 29, 35
Motor neuron disease (MND) is a group of disorders whose principal clinical and pathologic features are related to degeneration and loss of motor neurons. Most MND syndromes predominantly affect the lower motor neurons of the spinal cord and brain stem; however, amyotrophic lateral sclerosis (ALS), the most notable MND in humans, may affect the upper motor neurons of the cerebral cortex. 33 To our knowledge, lesions of MND in animals have been restricted to the ventral horns of the spinal cord and the brain stem. However, the lesions observed in the 1 case of MND resembled those in human beings with progressive muscular atrophy, a form of ALS. The prevalence and pathogenesis of MND in cats are unknown. 26
In the cases of diabetic polyneuropathy and ischemic neuromyopathy, lesions were limited to the peripheral nervous system. Diabetic polyneuropathy, which commonly affects diabetic dogs and cats, is caused in part by prolonged hyperglycemia and results in dysfunction of one or both tibial nerves and a plantigrade stance. Clinical signs may resolve with treatment to regulate blood glucose. The pathologic features of this condition closely resemble those of human diabetic neuropathy. 22 Multiple factors, such as vascular, mechanical, and biochemical disturbances, have been proffered as likely causes of axon and Schwann cell derangements in dogs and cats. 3
Ischemic neuromyopathy occurs in cats with cardiomyopathy and is caused by occlusion of the aortic trifurcation with thromboembolic obstruction of the internal and external iliac arteries and the median sacral artery. Ischemia of muscle and peripheral nerve is exacerbated by vasoconstriction of the collateral circulation, induced by platelet substances such as serotonin and thromboxane A2. Histopathologic studies have disclosed that peripheral nerve changes develop at the level of the middle to lower thigh. Proximal to the level of ischemic degeneration of axons, nerve fibers commonly undergo paranodal and segmental demyelination. 27 The lesions in our case were identical to those described in a previous report. 18
Only 2 of the 110 brains had spongiform changes with vacuolation at the obex; however, no evidence of PrPsc deposition was detected by immunohistochemistry, and one fifth (20%) of the samples had no neurologic lesions or pathologic alterations elsewhere. An in-depth neurologic examination in these cases would have facilitated correlation of clinical signs with the neuropathologic changes. However, because this part of our data was incomplete, no such correlation could be established. Nevertheless, this study demonstrated that concerted cooperation between pathologists and clinicians enhances diagnostic accuracy in cases of neurologic disease. Within the framework of the project, the participating study centers set up an archive of feline neurologic diseases.
Footnotes
Acknowledgements
The authors wish to thank C. L. Florio, M. Corsi, P. Gazzuola, and SINVet. We are indebted to A. Oevermann for kindly providing the control slide sections of an FSE-positive cat. This work was supported by a grant from the Italian Ministry of Health (IZSPLV 004/2002).
