Abstract
Fourteen, 3-month-old calves were intracerebrally inoculated with the agent of chronic wasting disease (CWD) from white-tailed deer (CWDwtd) to compare the clinical signs and neuropathologic findings with those of certain other transmissible spongiform encephalopathies (TSE, prion diseases) that have been shown to be experimentally transmissible to cattle (sheep scrapie, CWD of mule deer [CWDmd], bovine spongiform encephalopathy [BSE], and transmissible mink encephalopathy). Two uninoculated calves served as controls. Within 26 months postinoculation (MPI), 12 inoculated calves had lost considerable weight and eventually became recumbent. Of the 12 inoculated calves, 11 (92%) developed clinical signs. Although spongiform encephalopathy (SE) was not observed, abnormal prion protein (PrPd) was detected by immunohistochemistry (IHC) and Western blot (WB) in central nervous system tissues. The absence of SE with presence of PrPd has also been observed when other TSE agents (scrapie and CWDmd) were similarly inoculated into cattle. The IHC and WB findings suggest that the diagnostic techniques currently used to confirm BSE would detect CWDwtd in cattle, should it occur naturally. Also, the absence of SE and a distinctive IHC pattern of CWDwtd and CWDmd in cattle suggests that it should be possible to distinguish these conditions from other TSEs that have been experimentally transmitted to cattle.
Keywords
Chronic wasting disease (CWD) is a fatal neurodegenerative transmissible spongiform encephalopathy (TSE) that has been identified in captive and free-ranging cervids. 15, 16 Affected animals show accumulation of an abnormal form of prion protein (PrPd) in tissues of the central nervous system (CNS) and lymphoid system. Detection of PrPd in these tissues and characteristic histopathologic changes in the brain are the basis of currently available diagnostic methods for TSEs. 11
Naturally occurring CWD has been documented in three species of North American cervids, namely, mule deer (Odocoileus hemionus hemionus) and black-tailed deer (O. hemionus columbianus), white-tailed deer (O. virginianus), and Rocky Mountain elk (Cervus elaphus nelsoni). 15 Recently, CWD has also been reported in a moose (Alces alces shirasi). 1 CWD of mule deer (CWDmd) has been experimentally transmitted by intracerebral inoculation of affected brain from mule deer into a variety of livestock species, including a goat, 16 sheep, 6 and cattle. 4, 5 In the latter study only 5 of the 13 inoculated animals (38%) showed evidence of PrPd amplification, within 2 to 5 years postinoculation. However, when a second passage was performed, all inoculated animals (n = 6) succumbed to the infection within 17 months postinoculation (MPI). 8
Because cross-species transmission experiments provide valuable information about the biological characteristics of known TSE agents and their potential host range, in this article we describe passage of CWD of white-tailed deer (CWDwtd) into cattle. The primary objective of this study was to compare clinicopathologic observations of CWDmd infection of cattle with those of another source of CWD (white-tailed deer).
Materials and Methods
Inoculum
Material for inoculation was prepared from a pool of 11 CWD-positive (by immunohistochemistry [IHC]) white-tailed deer brains. The pool was positive for PrPd by Western blot (WB), and the inoculum produced CWD in white-tailed deer fawns after intracerebral inoculation (Kunkle et al., unpublished data). The pooled brain tissue was ground in a mechanical grinder, gentamicin was added at 100 μg/ml, and the final concentration of 10% (weight/volume) was made with phosphate-buffered saline as described previously. 3
Animals and experimental design
Sixteen 3-month-old Jersey bull calves were obtained and assigned to inoculated (n = 14) and control (n = 2) groups. Inoculated calves were housed in a Biosafety Level 2 isolation barn at the National Animal Disease Center in Ames, Iowa. Husbandry of these animals has been described. 4, 5 Personnel wore protective clothing while in the isolation facility and showered before leaving.
Calves were inoculated intracerebrally with 1 ml of a 10% CWD brain inoculum as described previously. 2, 3 Briefly, the calves were sedated with xylazine, a midline incision was made in the skin at the junction of the parietal and frontal bones, and a 1-mm hole was drilled through the calvarium. The inoculum was injected into the midbrain via a 22-gauge 9-cm long needle while withdrawing the needle from the brain. The cutaneous incision was closed with a single suture. The 2 control calves were not inoculated. Inoculated animals were examined twice a day and more frequently when showing clinical signs.
Sample collection and tests
The affected animals were euthanatized when in extremis (Table 1) with pentobarbital sodium and subjected to a complete necropsy. Representative samples of lung, liver, kidney, spleen, salivary gland, thyroid gland, reticulum, rumen, omasum, abomasum, intestines (ileum, colon), adrenal gland, pancreas, urinary bladder, lymph nodes (retropharyngeal, prescapular, mesenteric, popliteal), tonsils (palatine, pharyngeal), striated muscle (heart, tongue, masseter, diaphragm), eye, sciatic nerve, trigeminal ganglion, pituitary gland and spinal cord (cervical, thoracic, lumbar) were immersion-fixed in 10% neutral-buffered formalin. The brain was cut sagittally; half was fixed in formalin and the remainder was frozen (−20° C). The formalin-fixed brain was cut into coronal sections, 2–4 mm wide. A minimum of 5 brain areas per animal were selected for examination by light microscopy. These were rostral cerebrum, hippocampus, superior colliculus, cerebellum, and medulla (at the level of obex). Two sections of spinal cord at each location (cervical, thoracic, and lumbar) were also selected for microscopic examinations. The tissues were processed for routine histopathologic examination, embedded in paraffin wax, and sectioned at 5 μm. All tissues collected at necropsy were stained with hematoxylin and eosin (HE) and examined by IHC for PrPd, as described previously, 10 with a monoclonal antibody (F99/97.6.1). 13 This antibody recognizes PrP sequences conserved in most mammalian species in which natural TSEs have been reported.
Findings in cattle intracerebrally inoculated with the agent of chronic wasting disease from white-tailed deer (CWDwtd) and in uninoculated control cattle. ∗
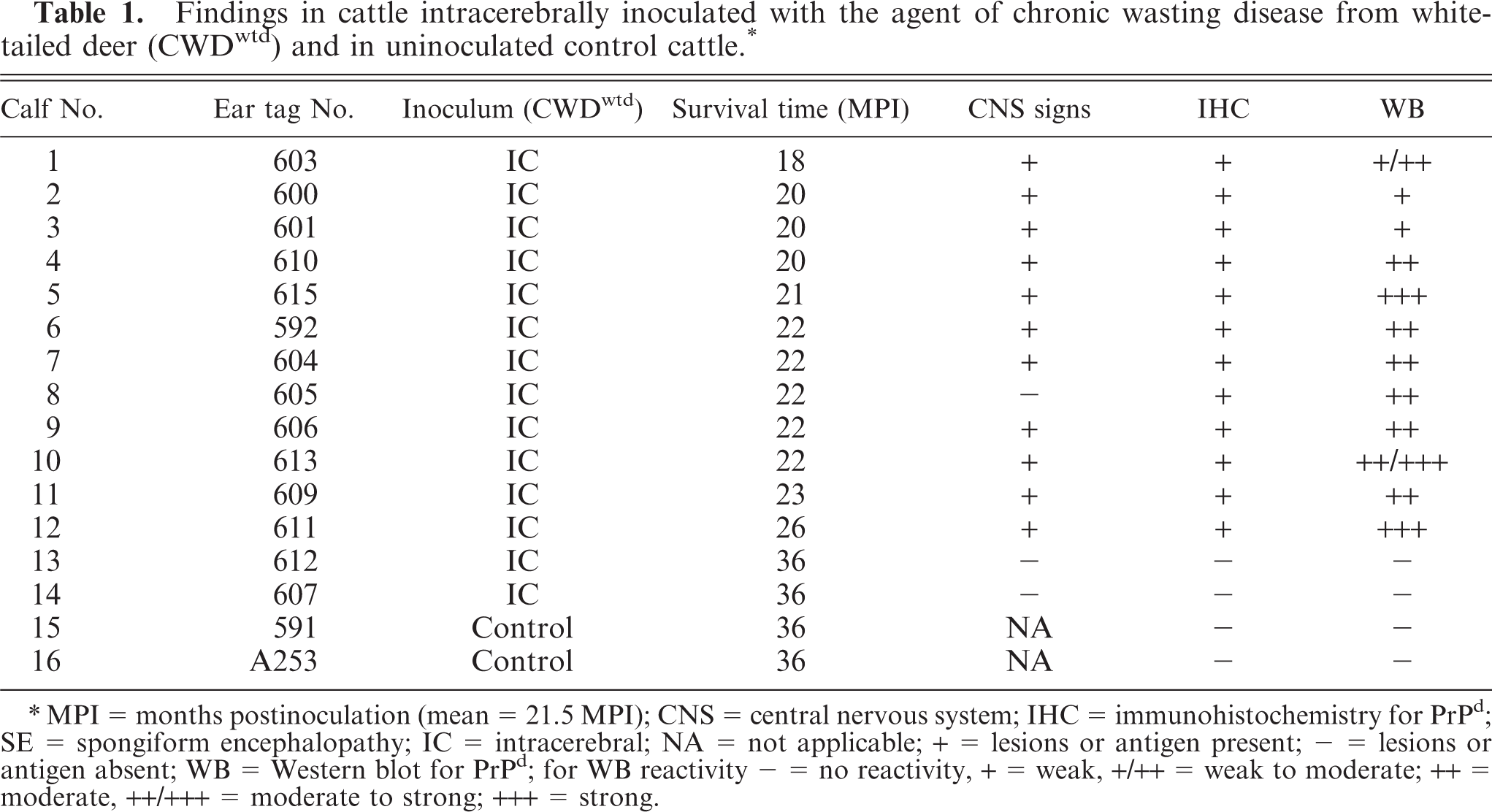
∗MPI = months postinoculation (mean = 21.5 MPI); CNS = central nervous system; IHC = immunohistochemistry for PrPd; SE = spongiform encephalopathy; IC = intracerebral; NA = not applicable; + = lesions or antigen present; − = lesions or antigen absent; WB = Western blot for PrPd; for WB reactivity − = no reactivity, + = weak, +/++ = weak to moderate; ++ = moderate, ++/+++ = moderate to strong; +++ = strong.
For detecting PrPd in the brainstem by WB, a commercial kit (Bio-Rad, Marnes-La-Coquette, France) with antibody 6H4 (0.1 μg/ml of a mouse IgG1 monoclonal antibody antiserum raised against human residues 144–152 of the PrP peptide; Prionics AG, Switzerland) was used.
Results
Approximately 6 to 8 weeks before euthanasia, 12 of the 14 inoculated cattle gradually started to lose appetite and weight. Eleven of these showed intermittent abnormal neurologic signs (Table 1) that included various combinations of circling, bruxism, head pressing, depression, and nonresponsiveness to external stimuli. Between 18 and 26 MPI, 12 of the 14 calves became recumbent and were unable to rise without assistance; at this stage they were euthanatized. The 2 CWD-inoculated cattle without clinical signs and the 2 control calves were euthanatized 36 months after the study was initiated (Table 1).
Although the affected cattle had lost considerable weight, gross postmortem examination did not reveal obvious signs of emaciation, such as serous atrophy of fat.
Microscopic examination of HE-stained sections of brain and spinal cord (cervical, thoracic, and lumbar) failed to reveal characteristic lesions of spongiform encephalopathy (SE). In one animal (No. 5) a focal area of vacuolation in the neuropil was seen in one section of the brain (rostral colliculus) and a few isolated neurons with single, clear vacuoles of variable size were seen in the red nucleus of one control and four inoculated animals. Nonneuronal lesions consisted of the presence of a few sarcocysts in striated muscles of 12 of 16 cattle.
Of the 14 inoculated cattle, 12 had evidence of PrPd in CNS tissue, as determined by IHC (Table 1). However, the intensity of immunoreactivity varied between individual animals and at different anatomic locations in the brain and the spinal cord (Table 2). In 2 cattle (Nos. 3 and 9), the immunoreactivity was limited in both amount and distribution within the brain (primarily colliculi and hippocampus) and was absent in the obex of No. 3. In the other 10 cattle there was moderate to extensive labeling in almost all brain sections, except for the cerebellum, where the amount of labeling was usually minimal and in 5 calves was absent. In the 7 cattle that had IHC reactivity in cerebellum, labeled areas were present primarily in white matter, and only sparse foci appeared in the granular layer. Similarly, the IHC labeling in sections of hippocampus and cerebrum was predominantly in white matter. In spinal cord, almost all cervical and lumbar samples had reactivity (restricted to grey matter), but several thoracic samples were negative. It should be noted that in the 2 cattle that had limited labeling in brain sections (Nos. 3 and 9), all spinal cord sections were IHC negative.
Immunohistochemical (PrPd) findings in cattle intracerebrally inoculated with the chronic wasting disease from white-tailed deer (CWDwtd) and in uninoculated control cattle.
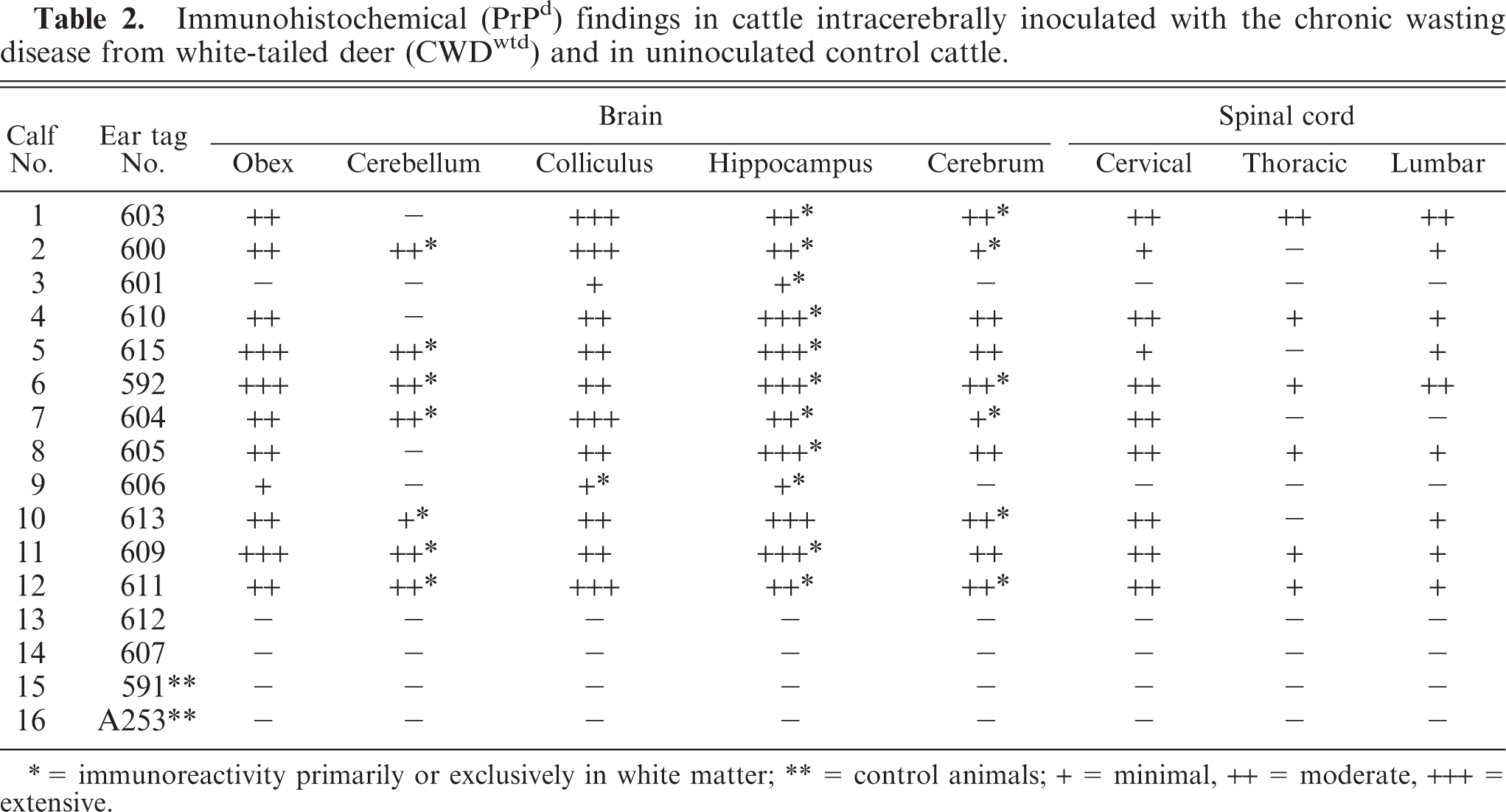
∗ = immunoreactivity primarily or exclusively in white matter; ∗∗ = control animals; + = minimal, ++ = moderate, +++ = extensive.
In most immunolabeled sections, the pattern of labeling was multifocal (Fig. 1), although in some areas of grey matter the reactivity was so extensive that it was essentially diffuse. Most of the labeling appeared as discrete particles or particulate, often concentrated in or around glial cells, but small aggregates and plaques were sometimes present. Notably, intraneuronal, perineuronal (Fig. 2), and perivascular immunolabeling were not observed. Other than brain and spinal cord, PrPd was not found in any of the other tissues examined (including multiple lymphoid tissues, retina, and pituitary gland).
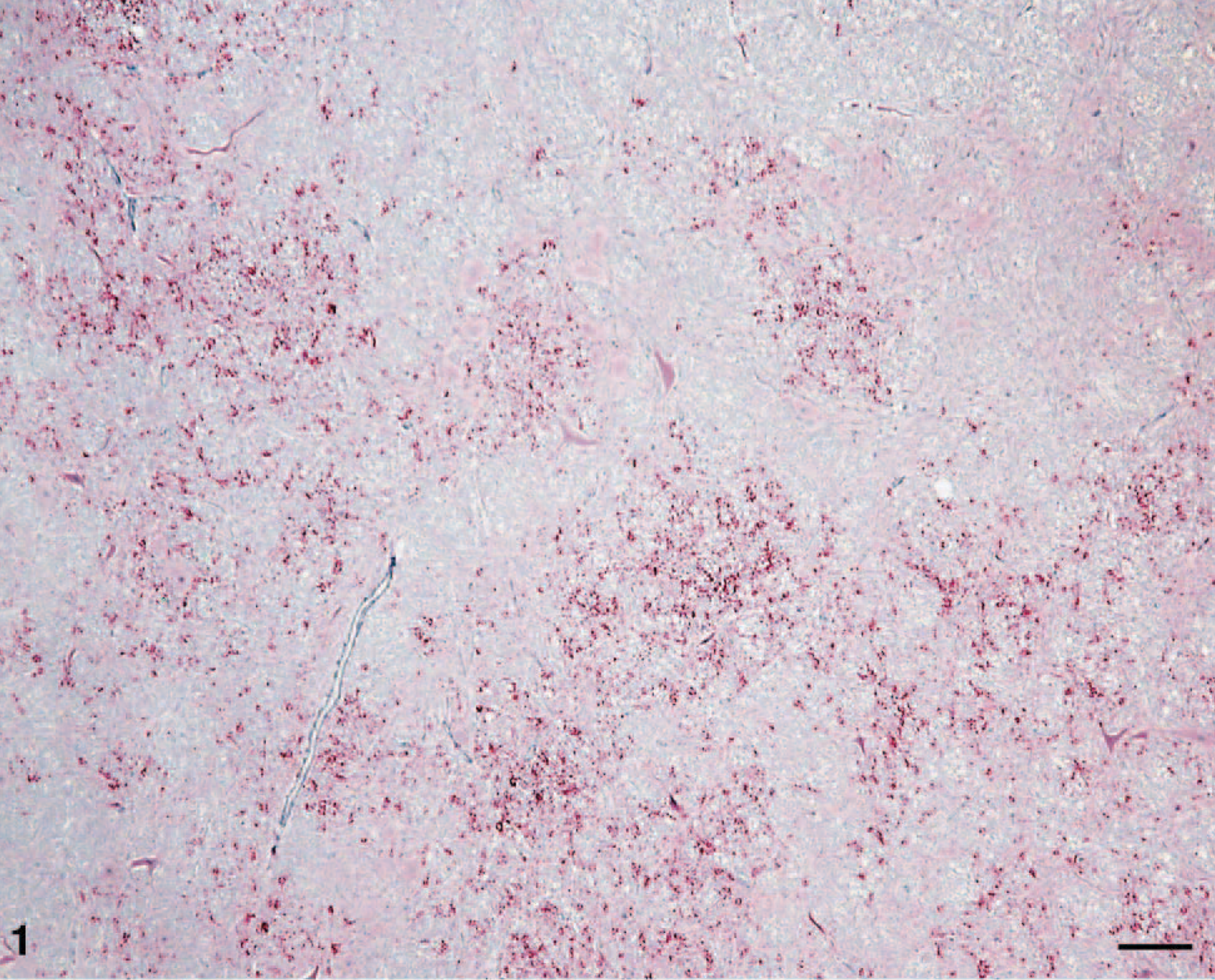
Brain; brainstem (obex) of calf No. 1. Note the multifocally distributed concentrations of particulate labeling of PrPd (red) in the section. Labeled for PrPd by IHC. Bar = 130 μm.
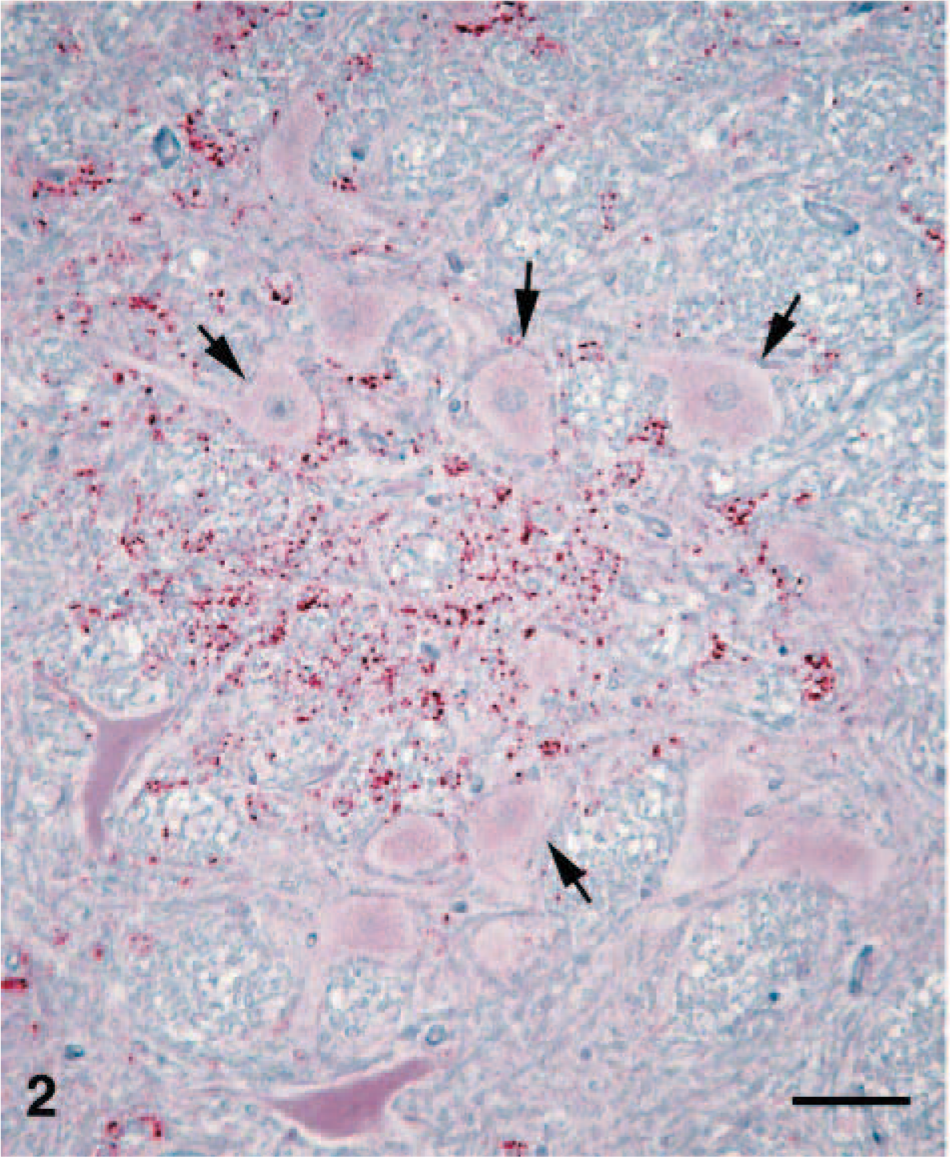
Brain; brainstem (obex) of calf No. 1. Higher magnification of a focal area of PrPd labeling. Particulate labeling is scattered in the neuropil with very little labeling associated with neurons (arrows). Labeled for PrPd by IHC. Bar = 90 μm.
WB analyses of brainstem samples from these animals revealed that all cattle inoculated with CWDwtd and showing clinical signs of TSE disease (Table 1) were positive for the presence of PrPd as detected by monoclonal antibody 6H4 (Fig. 3). In addition, calf No. 8 did not exhibit CNS signs (Table 1) and was also positive for PrPd. In contrast, Nos. 13 and 14, which were inoculated with CWDwtd and sacrificed 36 MPI (Table 1) as well as Nos. 15 and 16, the noninoculated control animals, were negative for PrPd (Fig. 3). Both of the positive control samples, from a bovine infected with CWDmd and a white-tailed deer infected with CWDwtd, had a strong positive WB reaction (Fig. 3).
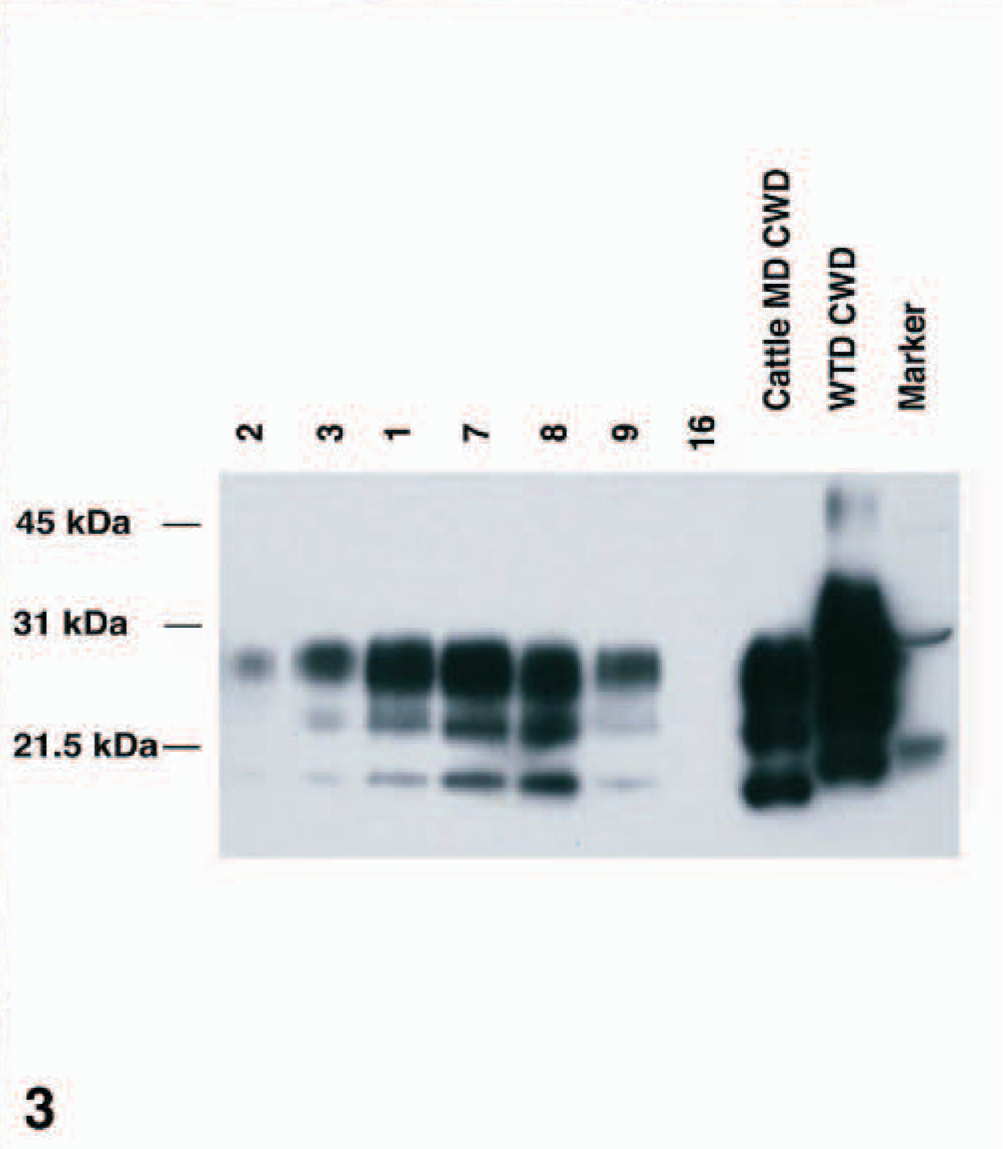
Western blot analysis of brain homogenates of cattle infected with CWDwtd. M = protein weight marker; calf Nos. 2, 3, 1, 7, 8, 9: brain homogenates from cattle infected with CWDwtd. No. 16 = brain homogenate from control cattle; Cattle MD CWD = brain homogenate from cattle infected with CWDmd; WTD CWD = brain homogenate from white-tailed deer infected with CWDwtd. Positive samples show a clear reaction with the 3 protease resistant polypeptide isoforms (di-, mono-, and unglycosylated) of PrPd; no reaction is found with the negative sample No. 16. Monoclonal antibody 6H4 (Prionics AG, Switzerland) was used for immunodetection of PrPd.
Discussion
This study used the intracerebral route of inoculation to provide the most efficient means of testing the absolute susceptibility of cattle to the agent of CWD from white-tailed deer and to provide a comparison with similarly designed previous studies. 2– 5, 7 Such studies do not inform on susceptibility by natural routes of transmission but, if negative, can provide evidence supporting the existence of a considerable species barrier. In instances of successful transmission they also provide an understanding of the descriptive pathology that might characterize the natural disease in the recipient species.
CWD, like all other TSEs, is characterized by a long incubation period, which in deer is seldom less than 18 months. 15, 16 In an experimental study of cattle inoculated intracerebrally with CWD from mule deer (first passage; CWDmd), PrPd was demonstrated in only 5 of 13 cattle (38%), after incubation periods that ranged from 23 to 63 MPI. 4, 5 In contrast, 12 of 14 inoculated cattle in the present study were positive for PrPd (86% attack rate) within 26 MPI (21.5 ± 0.5 MPI). The increased incidence and shorter incubation periods indicate that the CWDwtd inoculum was more pathogenic in cattle than the CWDmd inoculum on first passage. 4, 5 However, there may be several reasons for these differences, such as genetic variability of the recipient hosts, the differences in PrP genotype of pooled brain material in the inoculum, and the infectivity titer of the inocula used in the CWD transmission studies of cattle. 4, 5 Regarding the latter, in a separate study of intracerebral CWD inoculation of white-tailed deer, we found both sources of CWD (from mule deer and white-tailed deer) successfully transmitted to each of two groups of white-tailed deer within two years, and there was no significant difference in incubation times and lesions between the groups (Kunkle et al., unpublished data).
In cervids, clinical CWD is characterized by emaciation, changes in behavior, and excessive salivation. 15, 16 Although the latter was not observed in the CWDwtd-inoculated cattle, 12 of 14 inoculated calves showed anorexia and considerable weight loss, and the majority of these cattle also showed intermittent abnormal CNS signs. However, despite the advanced clinical signs in the affected cattle, none of the animals had histopathologic changes of SE, but all were positive for PrPd by IHC and WB. The absence of morphologic lesions in cattle with cross-species transmitted TSEs has been documented previously with cattle inoculated intracerebrally with the sheep scrapie agent. 3 In that study, 100% of cattle succumbed to the disease between 14 and 18 months after inoculation with a US strain of the scrapie agent derived from sheep. None of the cattle showed microscopic lesions, and all were positive for PrPd. A similar lack of morphologic changes was observed in cattle inoculated with CWDmd, even after the second passage. 4, 5, 8
The predominant characteristics of IHC reactivity reported previously for cattle inoculated with mule deer CWD 4, 5, 7 were also observed in the diseased cattle that died after inoculation with CWD from white tailed-deer. The PrPd accumulations were primarily multifocal and presumably glial cell–associated, with no evidence of intraneuronal or perineuronal labeling. The only difference noted between calves inoculated with CWDmd and CWDwtd was that the latter seemed to show more extensive labeling, both in amount and anatomic distribution within the CNS. In addition, labeling of white matter was more prominent in the CWDwtd-inoculated calves.
In this and in the earlier study of CWDmd in cattle, 4, 5 IHC labeling differed from that seen in cattle with bovine spongiform encephalopathy (BSE) or experimental transmissible mink encephalopathy (TME). 6 In both (BSE and TME) the IHC labeling was similar and was characterized by widespread diffuse (usually perineuronal) labeling of grey matter neuropil, with labeled particles that were not cell-associated except occasionally at neuronal cell membranes. 5, 14 The IHC pattern in bovine CWD also contrasts markedly with that seen in scrapie-inoculated cattle, in which intracytoplasmic labeling of neurons was a prominent feature. 2, 3
Compared with experimental TME in cattle, 7 the experimental bovine CWD in this study was associated with less extensive IHC labeling in neural tissues other than brain and spinal cord. Whereas the retina was positive in all cattle inoculated with TME, none of the CWD-infected cattle in this experiment had any retinal labeling. Similarly, in the present study there was no labeling in the pituitary gland, a tissue that in some TME-infected cattle is positive. 7 The different affinities of tissues may reflect different abilities of these agents to amplify in these tissues. However, given sample sizes and uncontrolled experimental variables, other features, including host genetics, titers of inocula, and stage of disease cannot be excluded.
IHC labeling for PrPd was not observed in striated muscles (heart, tongue, masseter, diaphragm) of the experimental calves. This observation is in accordance with our previous findings 9 in which striated muscle from 20 animals (cattle, sheep, elk, and raccoons) was examined for PrPd by IHC. In these animals, all of which had developed a TSE after experimental inoculation, PrPd was found by IHC examination in the CNS, but not in striated muscle. However, recent investigations with an enriched WB technique 12 have enabled us to detect PrPd in the tongues of some sheep and elk that were experimentally inoculated with scrapie and CWD, respectively (Bessen et al., unpublished data).
When brainstems of CWD-infected cattle were analyzed by WB for the presence of PrPd, 12 of 14 samples were found to be positive for PrPd (Table 1; Fig. 3), showing a clear reaction with the three protease resistant polypeptide isoforms (di-, mono-, and unglycosylated). Interestingly, No. 3, which was found by IHC to be negative in the obex area, but positive in midbrain region (Table 2), was found positive by WB (Fig. 3). This might indicate that the WB technique used for these studies could be more sensitive than the IHC method applied. However, this difference could also be attributable to the sampling of different portions of obex used for IHC and WB analyses. The WB molecular mass was similar in all cattle inoculated with CWDwtd and similar to that of cattle-passaged CWDmd (Fig. 3). However, PrPd isoforms from deer infected with CWDwtd had a higher molecular mass (Fig. 3).
This study indicates a high susceptibility of cattle to the CWDwtd agent by the most direct route of inoculation of the CNS, but it remains to be shown if natural routes of exposure would result in infection and disease and whether differences in attack rates and incubation periods between cattle inoculated with CWDmd and CWDwtd agents reflect true differences in susceptibility or are attributable to differences in experimental factors.
Although susceptibility of cattle to intracerebral inoculation of CWDwtd was demonstrated, it should be noted that this is an unnatural route, and it suggests only a potential for cattle to become infected under natural conditions of exposure. It is likely that transmission of CWD to cattle by a more natural route, such as per os, would require a much larger dose of inoculum and might even be difficult to accomplish within the normal life span of the animal. Because the present study resulted in a higher attack rate and shorter incubation periods than similar inoculation with CWDmd, the CWDwtd agent would be the initial choice for experimental oral inoculation of cattle.
Further transmission studies of CWD isolates (e.g., CWD of elk or moose in cattle) are required to explore possible differences in clinicopathologic features that might indicate different disease phenotypes, which may in turn, reflect different CWD agent strains.
Finally, the findings of this study suggest that diagnostic techniques presently used for confirming BSE (IHC and WB) would also detect CWDmd and CWDwtd in cattle should it occur naturally.
Footnotes
Acknowledgements
We thank Dr. Katherine I. O'Rourke for providing the antibody for the IHC procedure; Drs. L. Nusz and J. Laufer for clinical assistance; Dr. Marcus Kehrli Jr. for constructive comments and for his enthusiastic support; and James Fosse for the photomicrographs. Martha Church, Kevin Hassall, Joe Lesan, Dennis Orcutt, Sharla Van Roekel, Ginny Montgomery, and animal caretakers at the National Animal Disease Center provided expert technical assistance.
This study was carried out under the guidelines of the institutional Animal Care and Use committee at the National Animal Disease Center. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the United States Department of Agriculture.
