Abstract
A 6-year-old, gelded, Paint horse displayed clinical signs of muscle wasting and limb stiffness for a 6-month period. The horse's clinical signs abated with corticosteroid therapy, but returned upon cessation of treatment. Upon necropsy, severe lesions of aortic thickening and aortic valve rigidity were observed. Histologically, the tunica media of the aorta, coronary arteries, and pulmonary arteries were expanded by foci of elastin fiber calcification and extracellular matrix with lacunae formation. The vascular lesions are comparative to what has been described as medial arterial calcification, seen in humans suffering from chronic renal failure or diabetes mellitus. No exposure to vitamin D-containing plants or feedstuff could be documented at the time of onset or during the period of clinical signs. The current case describes dramatic lesions of arterial medial calcification of the aorta, coronary, and pulmonary arteries of undetermined cause.
A 6-year-old, gelded, Paint horse was presented to the Iowa State University Veterinary Teaching Hospital (VTH; Ames, IA) with a chronic course of limb stiffness and muscle wasting. Six months prior to referral at the VTH, the referring veterinarian treated the horse for suspected immune-mediated myositis. 9 This diagnosis was based on the acute onset of lethargy, fever, anorexia, loss of muscle mass (topline and gluteal musculature), and pigmenturia. No muscle biopsy was performed to confirm the diagnosis at that time. The horse was polyuric, polydipsic, weak, and moved with a stiff gait. One week after the onset of clinical signs, the leukogram showed a leukocytosis (35,000/μl) characterized by a mature neu-trophilia and mild monocytosis. Serum chemistry revealed a mild hyperphosphatemia, moderate increase in blood urea nitrogen (BUN), increased activity of creatine phosphokinase (CK), and aspartate transaminase (AST), with hypoproteinemia and hypoalbuminemia (Table 1). A urinalysis was not performed at this point or at any other time during the course of illness. In the first 3 weeks of illness, the horse was reluctant to move and appeared wobbly when standing. The horse had transient limb edema and excessive hidrosis. Therapy, consisting of corticosteroids, phenylbutazone, and penicillin, was initiated; only steroids provided improvement of clinical signs. The horse received 2 rounds of corticosteroids at approximately 3-month intervals, each consisting of a moderate dose of dexamethasone (200 mg given over a 7-day period). Limb stiffness returned upon cessation of steroid therapy.
Six months after the onset of clinical signs, the horse was euthanized as a result of nonresolution of gait stiffness, exercise intolerance, and weakness. Serum chemistry analysis and a complete blood count (CBC) were performed 1 day prior to euthanasia; this was the second and final time point for this data. The CBC was within normal limits. On heparinized plasma, BUN was mildly elevated, with mild hypophosphatemia; CK and AST were normal (Table 1). Serum magnesium concentration was within normal limits. Cerebrospinal fluid analysis performed the day of euthanasia revealed moderately elevated protein (85.9 mg/dl, reference [ref.] interval: 32–48 mg/dl); all other parameters were unremarkable. Serum collected prior to euthanasia was assayed for ionized calcium (1.75 mmol/l, ref. interval: 1.58-1.90 mmol/l), 25-hydroxyvitamin D (16 mmol/l, no ref. values established), and parathyroid hormone concentration (PTH; 3.10 pmol/l, ref. interval: 2.00-10.00 pmol/l). These findings were interpreted as supportive of normal calcium homeostasis (Michigan State University Endocrinology Laboratory, Diagnostic Center for Population and Animal Health).
Immediately after the horse was euthanized, muscle biopsies were collected from the gluteal, semitendinosus and semimembranosus, and left triceps muscles. The skeletal muscle was grossly normal. A full necropsy commenced within 1 hr of euthanasia.
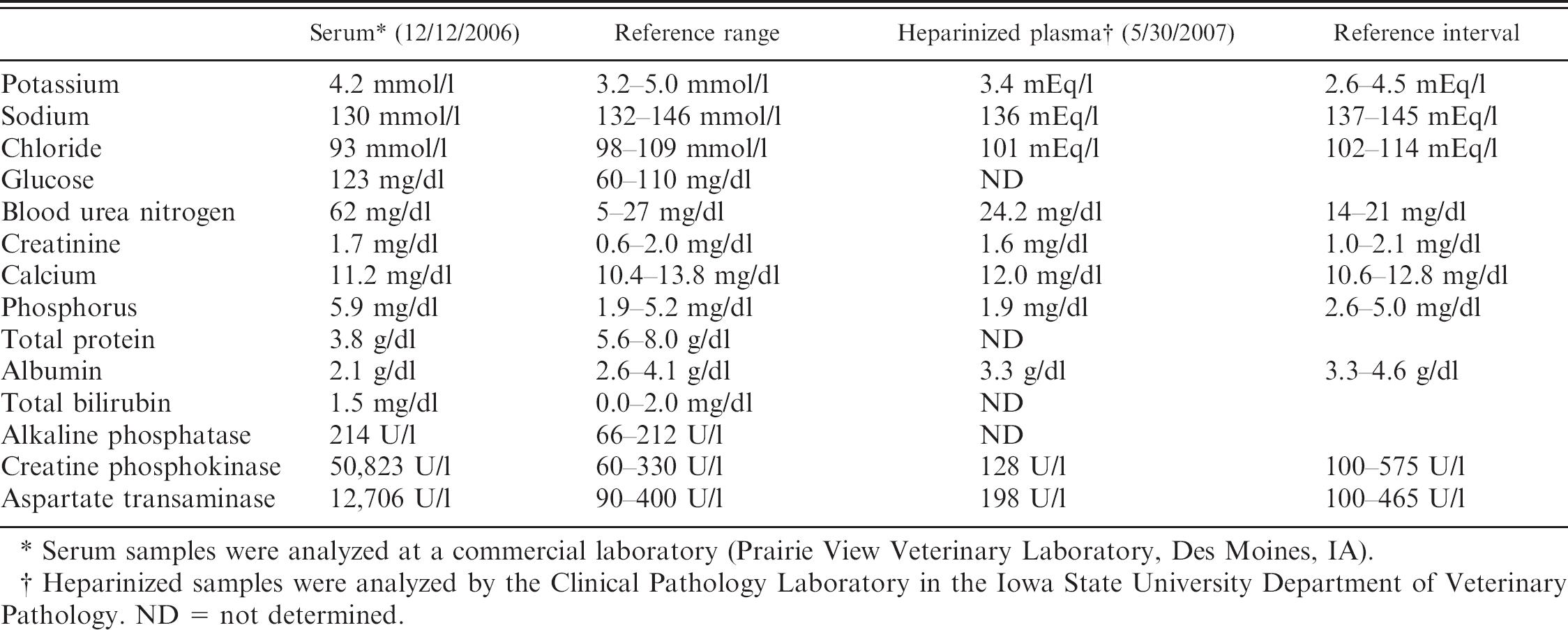
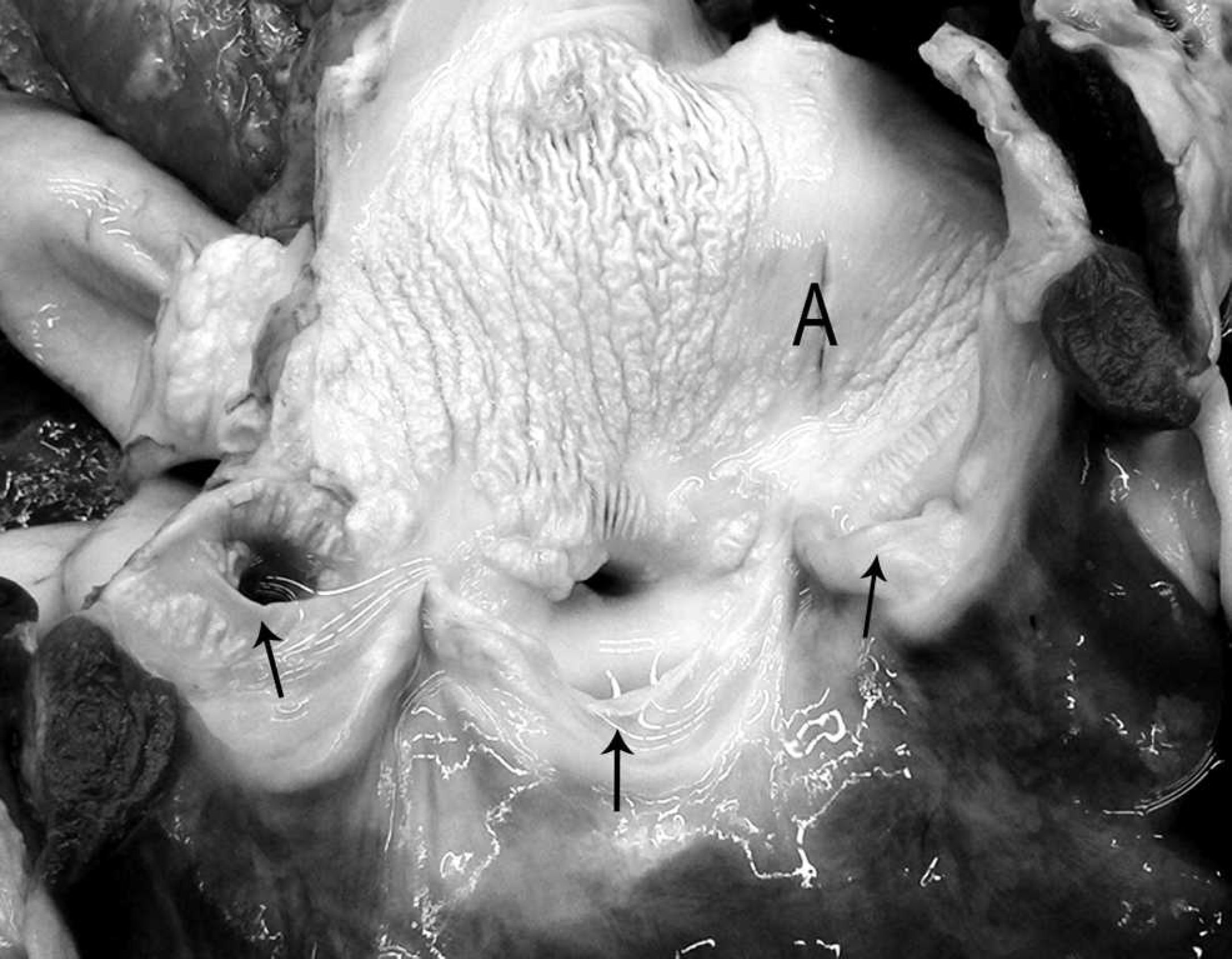
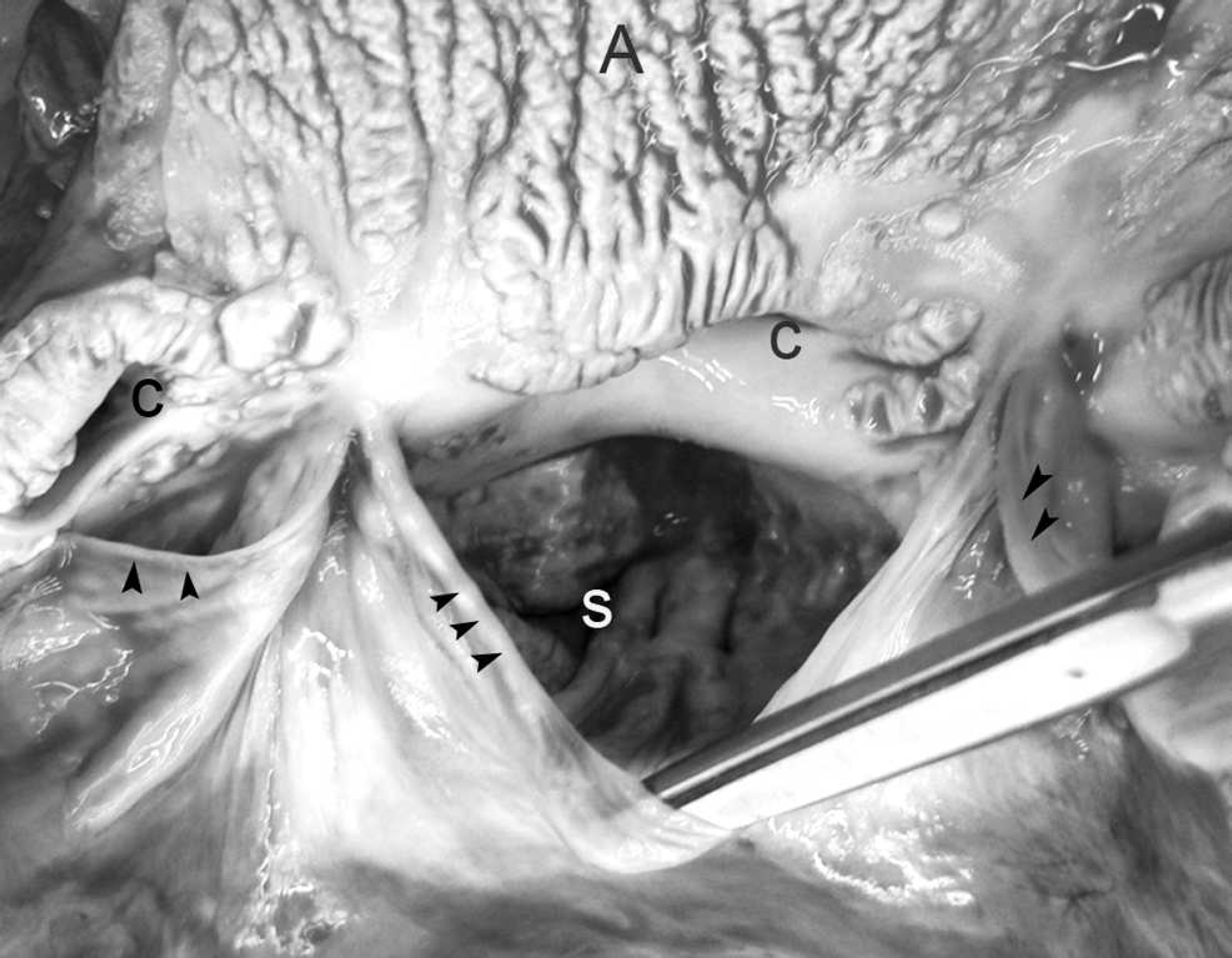
The walls of the aortic arch, descending aorta, subclavian, brachiocephalic, and common carotid arteries were irregularly thick, rigid, and brittle, with severely reduced flexibility. The root of the aorta was ballooned and dilated. Multiple patches of raised, corrugated nodules in the aortic wall extended into the vascular lumen above the aortic valve (Fig. 1). Similar lesions, though of lesser magnitude, expanded from the luminal surface of the right ventricle, pulmonary artery, left atrium, and left ventricle. The aortic valve cusps were ballooned and rigid (Fig. 2). The left and right coronary arteries were similarly affected, with marked reduction in luminal diameter as a result of irregular vascular wall thickening (Fig. 3). On cut section, the thoracic aortic wall was of irregular thickness, with multifocal chalky areas. The thickened arterial wall decreased the luminal diameter of affected arteries as a result of irregularly shaped and sized plaques that extended into the lumen. The pulmonary arterioles within the lung were also affected. The wall thickness of the proximal 60 cm of the thoracic aorta was 8 mm, whereas the thickness of the aorta just proximal to the diaphragm was 2 mm. In contrast, the abdominal aorta was normal, with a uniform thickness of 2 mm to the terminal branches. The entire spinal cord, brain, and complete set of thoracic and abdominal organs were collected in neutral buffered formalin. The thyroid, parathyroid glands, and kidneys were grossly normal. There were no other significant findings at necropsy.
Serum chemistry findings early in disease and 1 day prior to euthanasia.*
Serum samples were analyzed at a commercial laboratory (Prairie View Veterinary Laboratory, Des Moines, IA).
Heparinized samples were analyzed by the Clinical Pathology Laboratory in the Iowa State University Department of Veterinary Pathology. ND = not determined.
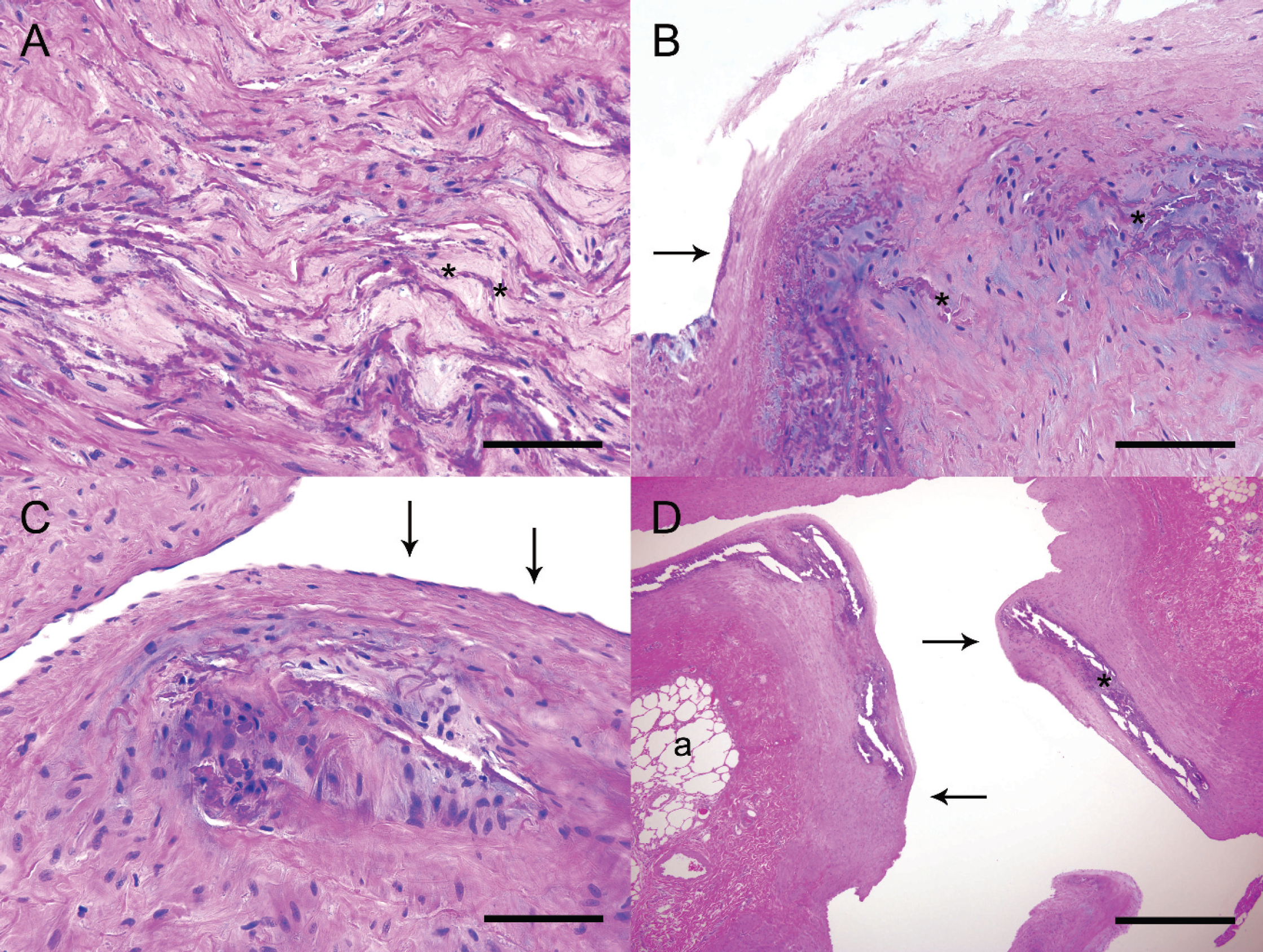
Histologic examination of the aorta and pulmonary arteries demonstrated multifocal expansion of the tunica media due to disarray and mineralization of elastin fibers separated by eosinophilic material (Fig. 4A). Von Kossa stains confirmed that the fibers were calcified. In larger lesions, the calcified elastin fibers were expanded by amphophilic to eosinophilic extracellular matrix (Fig. 4B). In such areas, rare lacunae-like spaces containing chondroblast-like cells were present in the extracellular matrix. This extracellular material stains blue on Alcian blue pH 2.5 stain and purple with Toluidine blue, indicating that this extracellular material contains proteoglycans. In smaller lesions, elastin fibers of the inner third of the tunica media were haphazardly arranged and segmentally calcified (Fig. 4C). Regardless of location, the lesions lacked any appreciable inflammatory cell component or any aggregation of macrophages. The lumina of pulmonary arterioles were severely distorted, as was the surrounding pulmonary architecture (Fig. 4D). In areas of medial mineralization and elastin fiber disorganization, the overlying intima was diffusely undulant and focally expanded by fibroplasia. Endothelial changes ranged from loss of endothelium to endothelial hypertrophy. Vascular calcification was limited primarily to the aorta, coronary arteries, and pulmonary arteries. Additional histologic changes were present in the spinal cord, intestine, pancreas, and kidney.
Heart, aortic root; horse. The aorta is thickened and the vascular surface is irregularly raised and rugose. Semilunar valves are indicated with arrows. A = aortic lumen.
There was mild, chronic axonal degeneration of the cervical spinal cord. This change was distributed randomly around all funiculi of the cord; no evidence of a traumatic etiology was observed at necropsy. Mild eosinophilic and lymphoplasmacytic enterocolitis was noted in the small and large intestinal mucosa. Lympho-plasmacytic inflammation and edema expanded the periductular stroma of 1 pancreatic duct of the pancreas. The renal lesion consisted of mild, multifocal lympho-plasmacytic perivasculitis and rare, segmental occlusion of distal tubules by pale eosinophilic debris. Parathyroid glands were histologically normal. Von Kossa stains performed on sections of brain, spinal cord, and kidney did not identify calcified soft tissue. Sections of skeletal muscle from multiple sites showed mild, sporadic variation of myofiber diameter, but lacked any evidence of inflammation or fibrosis.
Heart, aortic valve; horse. The valve leaflets (arrowheads) of the aortic valve are of decreased flexibility, and the sinus of Valsalva (S) is dilated. The entrances to the coronary sinuses (C) are partially occluded by the rugose vascular surface. A = aortic lumen.
The vascular lesions in this horse consist exclusively of elastin fiber disorganization and calcification. Histologically, the pattern of mineralization within the tunica media is similar to arterial medial calcification, as described in humans. 2 The lack of cholesterol granulomas, inflammation, and lipid core formation in these lesions distinguishes them from atherosclerotic plaques. The absence of inflammation within the arterial lesions and the restriction of the lesions to the thoracic cavity make parasitic migration less likely. Therefore, the differential diagnoses for the lesions of arterial medial calcification consisted of vitamin D toxicosis (excessive supplementation or ingestion of Cestrum or Solanum spp. plants), chronic renal disease, nutritional secondary hyperparathyroidism, magnesium imbalance, or idiopathic mineralization.
Similar lesions in horses have been reported sporadically. A 1983 review 1 of 1,066 horses from a British slaughter plant showed that 9.28% of the horses had medial mineralization and elastin fiber disarray with a chondroid core in the pulmonary artery. The average age of affected animals was 10.75 years, and racing thoroughbreds were overrepresented at 26.9%. As a result of the increased incidence of this lesion in thoroughbreds (vs. cobs, ponies, and draught horses) in that study, the authors attributed the formation of pulmonary artery medial mineralization to hemodynamic injury incurred by the explosive exertions of a racehorse. 1 Lesions of arterial medial calcification were found in equine cases of accidental and experimental overdose of ergocalciferol (vitamin D2) and cholecalciferol (vitamin D3). 5,6 Clinically those horses exhibited depression, recumbency, weakness, limb stiffness, fever, and polyuria. Arterial medial mineralization of thoracic and abdominal viscera was reported in a foal from a herd of Andalusian brood mares that were fed a diet with an improperly balanced calcium/phosphorus ratio. 3 The mares and foals displayed joint pain, lameness, and stiff gait. The PTH concentration of the mares was increased 10-fold, while the foals' PTH was slightly elevated. A single foal was necropsied from that herd; no adult horses died. Clinical signs resolved once the appropriate calcium/phosphorus ratio of the feed was restored.
Heart, right coronary artery; horse. The lumen of the coronary artery (outlined by arrows) is partially occluded by irregular thickened areas within the arterial wall. e = epicardial fat.
Medial calcification of the aorta can be induced experimentally in animals by means of vitamin D overdose in rats, pigs, and rabbits; vitamin D combined with nicotine in rats and rabbits; vitamin D and magnesium deficiency in pigs; or models of renal failure in rats and rabbits. 2 Aortic mineralization has been associated with infection with Mycobacterium avium subsp. paratuberculosis (Johne's disease) in cattle and goats. In the current study, there was no evidence of mycobacterial disease or other forms of chronic, granulomatous inflammation in the tissues examined.
Differential diagnoses for arterial medial calcification in humans include systemic effects of abnormal regulation of pH, electrolytes, or metabolic disorders. 2 Specific examples include hypervitaminosis D, uremia from chronic renal disease, chronic dysregulation of hyperglycemia from diabetes mellitus, dysregulation of PTH, hyperparathy-roidism, and idiopathic arterial calcification. 2,12,15 An idiopathic form of mineralizing coronary arteriopathy is described for human infants, resulting from a nonsense mutation for ectonucleotide pyrophosphatase phosphodi-esterase-1. 7
Deactivation of local calcification inhibitors within the tunica media of the aorta and coronary arteries has been proposed as a mechanism for elastin fiber calcification. 10 Bone formation observed in these lesions may be due to differentiation of vascular smooth muscle cells into osteoblast-like cells 10 and promoted by hyperphosphatemic conditions. 13 These “transdifferentiated” cells likely play a role in deposition and calcification of extracellular matrix. 12,13 Elastin degradation products and transforming growth factor-β1 may promote osteogenic differentiation of aortic myofibroblasts. 14 Evidence indicates that vascular medial calcification is an active process, similar to endochondral ossification, and may be regulated by paracrine signaling. 2,13 In humans, the alterations in coronary perfusion due to aortic medial sclerosis result in increased cardiovascular mortality 10,15 and increased risk of limb amputation in diabetic patients. 11 Cortico-steroid therapy is a risk factor in human cases of atherosclerosis and vascular mineralization, along with hypertension and hypercholesterolemia. The mechanism by which corticosteroids promote mineralization of extracellular matrices is not fully known, although dexamethasone has been shown to promote differentiation of pericytes (and other cell types) into osteoblast-like cells while down-regulating synthesis of calcification-inhibitory molecules. 8
The inciting cause of mineralizing medial arteriopathy in the current equine case remains undetermined. As serum chemistry values were checked only twice during the progression of disease, the full spectrum of variation within calcium, phosphorus, BUN, and parathyroid concentrations was not documented. One week after onset of signs, the horse was mildly hyperphosphatemic, normocalcemic, polyuric, polydipsic, and moderately azotemic. Six months later, at the time of euthanasia, the horse was normocalcemic and mildly hypophosphatemic, with normal PTH concentrations and presumed normal levels of 25-hydroxyvitamin D. These findings do not support long-term nutritional or renal secondary hyperparathyroidism or recent oversupplementation of vitamin D. However, these data do not exclude the occurrence of transient abnormality in calcium/phosphorus homeostasis resulting in an irreversible lesion. The paradox of normal levels of ionized calcium, vitamin D, and PTH in the face of severe arterial medial calcification of thoracic arteries may be corroborated by a recent report on humans with chronic renal disease, which showed no correlation between vascular calcification and serum markers of mineral metabolism. 16
Chronic renal failure remains as a possible cause of arterial medial calcification, as the horse had increases in BUN in the 2 instances during which serum chemistry was performed. Furthermore, the horse was polyuric and polydipsic at the time of initial clinical signs. A urinalysis was not performed; thus, assessment of renal function remains unknown. Evidence that is less supportive of renal failure includes consistently normal creatinine concentration at the 2 time points checked and minimal histologic changes in the kidneys. There was no clinical history or histologic evidence to support a diagnosis of diabetes mellitus. There is no history available to support exposure to excessive amounts of vitamin D, although the lesions and clinical signs in this case are similar to those described in other equine cases of vitamin D toxicosis. 3,5,6 Though ionized calcium, vitamin D, and PTH concentrations were within normal limits 6 months after onset of clinical signs, these values were not assessed at the onset of clinical signs.
The horse in the current study was originally presented for a 6-month history of gait stiffness, rather than for cardiovascular dysfunction. Immune-mediated myositis and Lyme myositis were considered as clinical differential diagnoses for gait stiffness and muscle atrophy. However, there was no support for either of these diagnoses histologically. Similarities exist between the histologic appearance of the arterial lesions in this case and the lesions recently described for Equine Systemic Proteoglycan Accumulation (ESPA), 4 also known as degenerative suspensory ligament desmitis when limited to the suspensory ligaments. Alcian blue pH 2.5 and Toluidine blue stains confirm that some or all of the amphophilic material separating elastin and collagen fibers within arteriolar and aortic lesions in the present case are proteoglycans. However, fragmentation and mineralization of elastin fibers is not described as a feature of ESPA. Proteoglycan deposition and multifocal cartilage metaplasia could indicate overlapping patterns of primary disease, or they could reflect a common response of degenerative, weakened mesenchymal tissue. The mesenchymal tissues in the present case were not examined by electron microscopy, nor were the types of proteoglycans identified; thus, ESPA cannot be ruled out as a contributing factor to lesion development in this case. However, renal function, calcium-phosphorus homeostasis, and other indicators of systemic function have not been evaluated as potential cofactors in the development of ESPA lesions 4 ; thus, it remains to be seen whether ESPA is a unique, primary lesion of collagenous tissues or a side effect from other influences.
Alterations in thoracic arterial wall elasticity could be expected to have systemic consequences. Physical exam of the horse at rest revealed no obvious signs of cardiovascular dysfunction. A cardiac work-up was not performed in the present case; thus, there are no data regarding blood pressure or cardiac output. In humans, arteriolar mineralization may result in “stiffening of arterial tone, increased systolic blood pressure, and impairment of endothelium-dependent relaxation.” 2 This is supported by recent findings that pulse wave velocity and vascular calcification are correlated in humans with chronic renal disease. 16 In horses, arterial medial mineralization has been associated with clinical signs of lethargy, painful joints, musculoskeletal weakness, and stiff gait. 3,5,6 Furthermore, in humans, femoral bone mineral density is inversely correlated with superficial femoral artery vascular calcification. 16 The cause of elevated CK and AST noted 1 week after the onset of clinical signs remains undetermined; recumbency and decreased perfusion of skeletal muscle are possible causes. Histologically, there was mild to moderate variation in myofiber diameter (myofiber atrophy) in the sections of skeletal muscle taken 6 months after onset of signs. Weight loss (as much as 25% of body weight) has been described in experimental and clinical cases of vitamin D toxicosis in horses, though the mechanism for reduction in body mass was not described. 5,6
The current study documents an unusual and unexpected pathologic finding of arterial medial calcification in an adult horse. There was no documented exposure to plants or substances causing alterations in calcium homeostasis. Serum chemistry was performed at 2 different times in the progression of clinical signs, once at the onset of signs and again 6 months later, prior to euthanasia. In the 6-month duration of clinical signs, the horse was initially hyperpho-sphatemic, then hypophosphatemic, while the horse was normocalcemic at the 2 times during which serum chemistry analysis was performed. Serum magnesium concentration was not evaluated at the onset of clinical signs; thus, hypomagnesemia is not excluded. The serum chemistry and urinalysis data are incomplete; thus, renal failure is not ruled out as a cause for altered calcium-phosphorus homeostasis. Evidence to support chronic renal failure in the present case consists of a mild elevation in BUN and mild histopathologic changes of the kidney. Corticosteroid therapy could conceivably have played a role in the progression of vascular wall mineralization, although the dosage of dexamethasone was moderate, and windows of treatment were separated by 3 months. Regardless of the role played by corticosteroids in lesion progression, it is likely the dexamethasone was administered after the initial development of vascular lesions.
In conclusion, the current case describes the presence of dramatic mineralization of the tunica media of the aorta and arteries of the heart and lungs. The cause of arterial medial calcification remains undetermined. Though arterial medial calcification is an infrequently encountered lesion in horses, it is a consideration for equine cases involving clinical signs of prolonged lethargy, gait stiffness, and exercise intolerance.
Acknowledgements. Dr. Claire Andreasen is gratefully acknowledged for her assistance with interpretation of clinical pathology data. Collection of data for this case was assisted through funds from the Equine Sections of the Iowa State University (ISU) Veterinary Teaching Hospital. Dr. Phil Edler of Hudson Veterinary Clinic is acknowledged for the referral of this case to the ISU Veterinary Teaching Hospital. Ms. Ann Staniger is acknowledged for her assistance with preparing the photomicrographs for publication.
