Abstract
The WTC-deafness Kyoto (dfk) rat is a new mutant characterized by deafness and abnormal, imbalanced behavior. WTC-dfk rats carry an intragenic deletion at the Kcnq1 gene; KCNQ1 plays an important role in K+ homeostasis, and the mutation of Kcnq1 causes a cardiac long QT syndrome in humans. Here, we studied stomach lesions in these WTC-dfk rats. The most characteristic pathologic feature in the stomach was the appearance of hypertrophic gastric glands in the stomach body. The hypertrophic cells had many eosinophilic granules in their cytoplasm, and these granules were stained red with Azan stain; stained positively for trypsinogen, amylase, and chymotrypsin; and did not stain positively for pepsinogen when using immunohistochemical analysis. These staining results suggested a metaplasia toward a pancreatic acinar cells. Extensive fibrosis was found in the bottom part of the mucosa of 34–week-old WTC-dfk rats, suggesting a progression of stomach lesions with aging. Although cells that were positive for proliferating cell nuclear antigen were restricted in the area of the glandular neck in WTC control rats, positive cells in WTC-dfk rats were scattered throughout the mucosa. The parietal cells in WTC-dfk rats were negative for KCNQ1 immunohistochemical analysis. These findings indicate that a deficiency in rat Kcnq1 provokes an abnormal proliferation and differentiation of gastric glandular cells.
Introduction
KCNQ1 is a pore-forming (α) subunit of the voltage-gated potassium channel and is expressed in several tissues throughout the body. 1, 15, 21 KCNQ1 plays a crucial role in terminating the cardiac action potential and in controlling water and salt homeostasis in several epithelial tissues. 15 Mutation of the human KCNQ1 gene manifests the cardiac long QT syndrome and deafness, which is known as Jervell and Lange-Nielsen syndrome. 14, 18 KCNQ1 knockout mice also have deafness and imbalance, which are associated with morphologic abnormalities in the inner ear. 3, 17
Rats with abnormal behaviors such as head tossing, drawing back, stepping back, and circling were discovered in the N12F10 generation of a WTC.ZI-Atrnzi congenic strain 16 at the Institute of Laboratory Animals, Graduate School of Medicine, Kyoto University, and the mutation was named deafness Kyoto (dfk). 10 These abnormal behaviors were inherited as an autosomal recessive trait. Auditory brainstem response revealed complete deafness. Histopathologic study of the inner ear of the WTC-dfk rats showed collapsed endolymph; their abnormal behavior is therefore considered to be due to an imbalance associated with the inner ear lesions. 10 Positional cloning of the WTC-dfk rat revealed an intragenic deletion including exon 7 of the Kcnq1 gene. 10 The WTC-dfk rat has a marked reduction of endolymph and a prolonged Q-T interval, as shown on electrocardiograms, which is a similar pathology to that of the Jervell and Lange-Nielsen syndrome in humans. 14, 18 WTC-dfk rats may therefore serve as powerful tools for studying the physiologic functions of KCNQ1.
In the stomach, the KCNQ1 potassium channel is expressed in the apical membrane of parietal cells and luminal K+ channels are required for gastric H+ secretion. 13 Inhibition of KCNQ1 diminishes H+ secretion in vivo and in vitro, 11 and Kcnq1 knockout mice show a large increase in stomach pH and hypertrophy of gastric mucosa. 17 The pH of the stomach fluids in the WTC-dfk rats is almost neutral, suggesting a gastric achlorhydria. 10 Histopathologic study has revealed a hypertrophic eosinophilic gastric gland. Here, we immunohistochemically and electron microscopically analyzed the stomach lesions of WTC-dfk rats. Hypertrophic eosinophilic cells in WTC-dfk rats were positively stained with enzymatic proteins of pancreatic exocrine cells. Thus, mutation of the Kcnq1 gene in rats was found to provoke achlorhydria and metaplasia of pancreatic exocrine cells, resulting from the disturbed proliferation and differentiation of gastric progenitor cells.
Materials and Methods
Animals
WTC-dfk homozygous rats were examined in this study, and age-matched wild-type WTC rats were used as controls. The WTC-dfk strain was provided by the National Bio Resource Project for the Rat in Japan. Rats were handled according to the guidelines for animal experimentation from Osaka Prefecture University and Kyoto University.
Histopathology
Rats were sacrificed under ether anesthesia at 4 and 34 weeks of age. Three affected mutants and 3 age-matched control rats were examined at each point. Perfusion fixation through the left ventricle was conducted using 4% paraformaldehyde in 0.1 M phosphate buffer (PB). The stomachs were removed and fixed further in the same fixative solution. Paraffin-embedded sections were cut and stained with HE for histopathologic evaluation. Representative sections were also stained with Azan, periodic acid Schiff (PAS) reaction, and alcian blue (pH 2.5) stainings.
Immunohistochemistry
The avidin-biotin-peroxidase complex (ABC) method (VECTASTAIN ABC Kit, Vector Laboratories, Inc., Burlingame, CA) was applied. Briefly, the deparaffinized sections were treated for 15 minutes in 0.3% hydrogen peroxide, and masking was carried out using 5% skimmed milk in phosphate-buffered saline. The sections were incubated overnight at 4°C using the following primary antibodies: sheep polyclonal antibody against pepsinogen (Biodesign International, Saco, ME); rabbit polyclonal antibodies against amylase (Sigma, St. Louis, MO), KCNQ1 (Santa Cruz Biotechnology, Santa Cruz, CA), and pancreatic duodenal homeobox-1 (PDX-1) 19 ; and mouse monoclonal antibodies against chymotrypsin (Biogenesis, Poole, UK) and proliferating cell nuclear antigen (PCNA; Dako, Glostrup, Denmark). The sections were incubated using biotinylated anti-sheep IgG, rabbit IgG, or anti-mouse IgG (all three from Vector Laboratories) for 1 hour at room temperature. They were then incubated using ABC for 1 hour at room temperature, and the peroxidase activity was visualized by reaction with 0.05% diaminobenzidine tetrachloride plus 0.01% hydrogen peroxide in 0.1 M Tris buffer (pH 7.6). After incubation, the sections were lightly counterstained with hematoxylin and cover slips were applied.
Electron microscopy
Removed samples were stored in 2% paraformaldehyde and 2.5% glutaraldehyde in 0.1 M PB, postfixed with 2% osmic acid for 2 hours, and embedded in epoxy resin. Ultrathin sections were double stained with uranyl acetate and lead citrate, and examined using a Hitachi H-7500 electron microscopy (Hitachi, Tokyo, Japan).
Results
Using histologic examination, hypertrophic glands were observed in both 4- and 34-week-old WTC-dfk rats and were distributed in the glandular body to the glandular base (Fig. 1). The nuclei of hypertrophic cells were eccentrically located in the basal cytoplasm. The apical cytoplasm was abundant and deeply eosinophilic, compared with the basal cytoplasm, which was basophilic (1a, b). The eosinophilic cytoplasm of the hypertrophic cells was deep red using Azan stain (Fig. 1c) and negative for PAS or alcian blue stainings. Immunohistochemical analysis for hypertrophic cells was positive for trypsinogen (Fig. 1d), amylase (Fig. 1e), and chymotrypsin, but immunohistochemical analysis for pepsinogen (Fig. 1f) failed to demonstrate positive staining in these cells. Electron microscopy revealed a well-developed rough endoplasmic reticulum (rER), and the rER was densely packed in the basal cytoplasm (Fig. 2). The apical cytoplasm contained many dense granules, which were approximately 1 µm in diameter. These immunohistochemical and electron microscopic findings suggest a metaplasia toward a pancreatic exocrine cell phenotype. 4
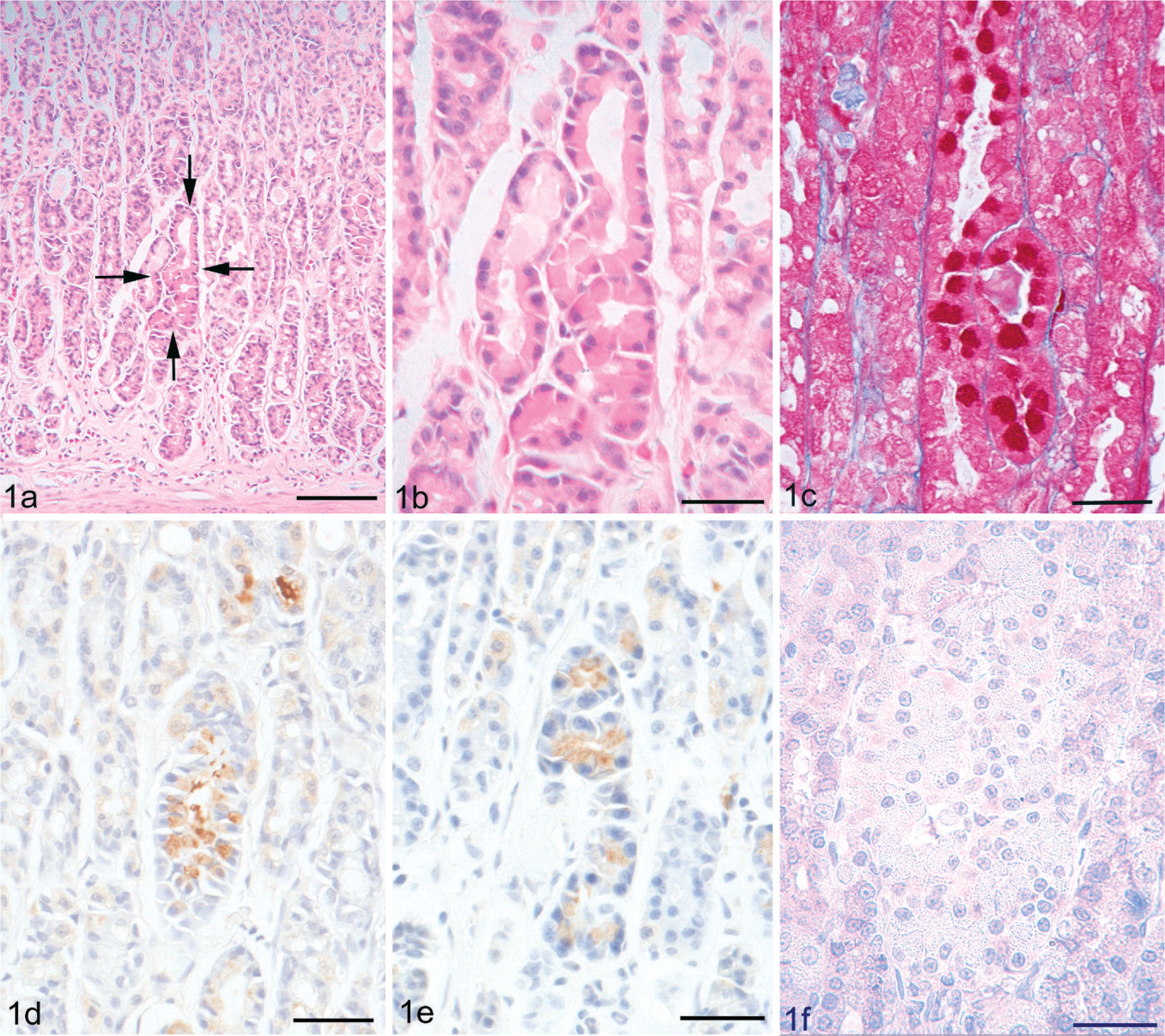
Hypertrophic gastric gland in a 34-week-old WTC-dfk rat. These cells have a deeply eosinophilic cytoplasm when stained with HE (Fig. 1a, b) and are deep red with Azan stain (Fig. 1c). Immunohistochemical analysis using trypsinogen (Fig. 1d) and amylase (Fig. 1e) shows positive staining of hypertrophic cells, but no staining for pepsinogen (Fig. 1f). Bar = 250 µm (Fig. 3a); Bar = 30 µm (Fig. 3b–f).
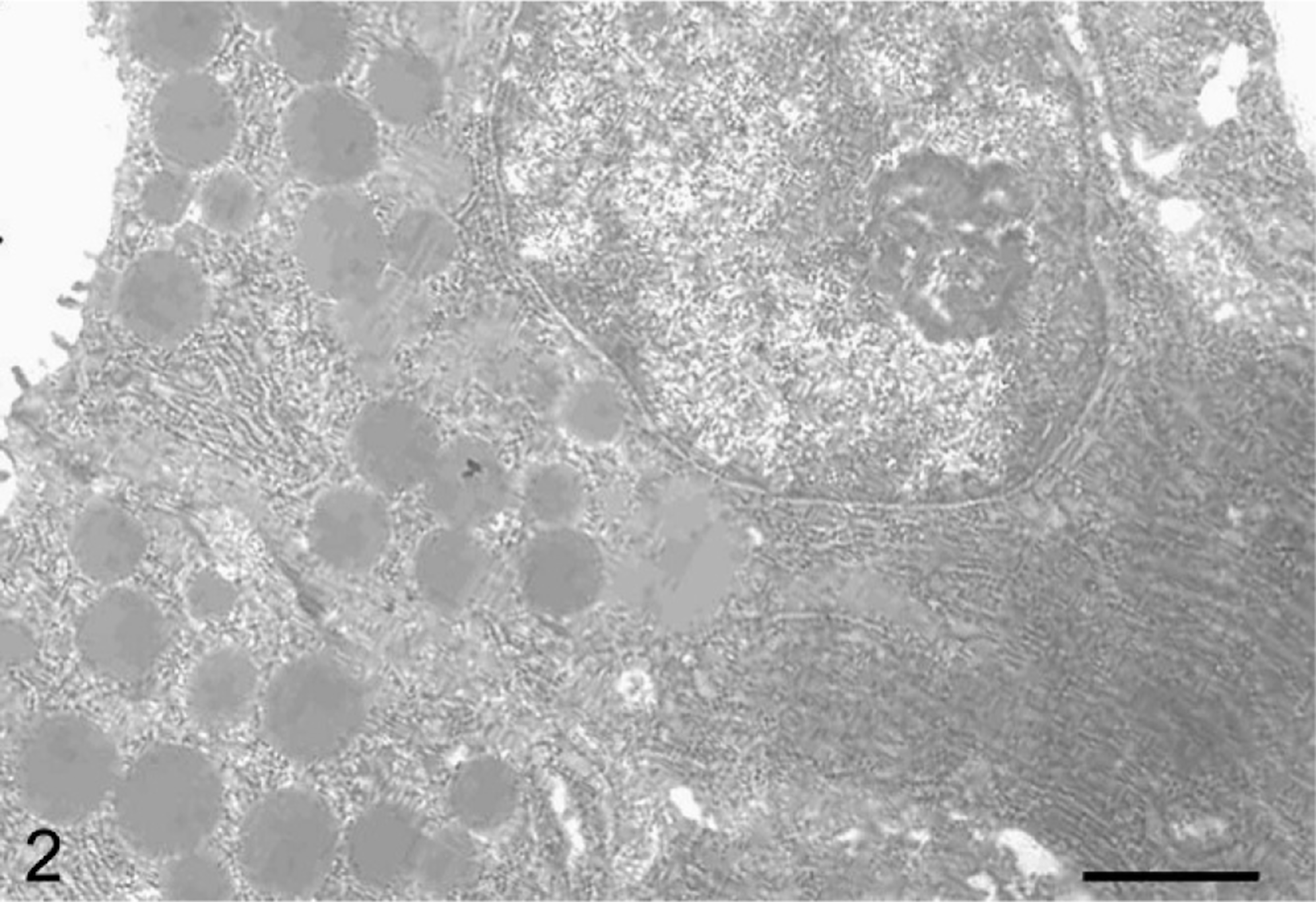
Electron micrograph of a hypertrophic gland from a 34-week-old WTC-dfk rat. Well-developed rER is present in the cytoplasm. The apical cytoplasm contains many dense granules, which are approximately 1 µm in diameter. Bar = 1.6 µm.
Immunohistochemical examination of PCNA was conducted to study the proliferating activity of gastric cells in WTC-dfk rats. A characteristic PCNA-positive distribution pattern restricted to the proliferating neck zone was found in the wild-type control rats (Fig. 3a). However, positive cells were scattered throughout the mucosa in the WTC-dfk rats (Fig. 3b). Disrupted distribution of PCNA-positive cells was observed in both 4- and 34-week-old WTC-dfk rats.
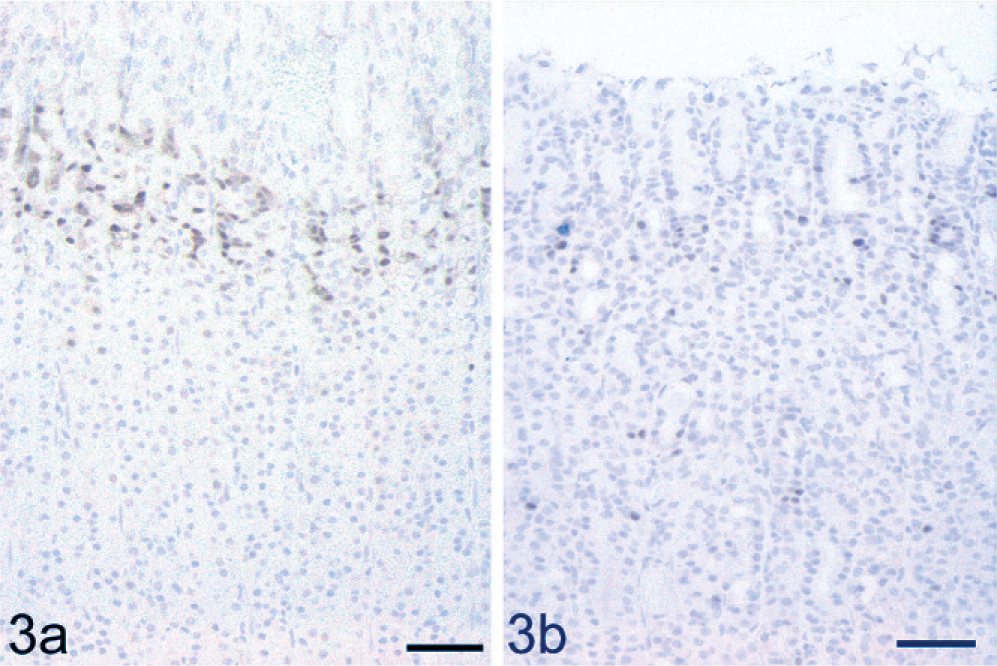
Immunohistochemical analysis for PCNA in a 34-week-old WTC-dfk rat and an age-matched wild-type WTC rat. PCNA positivity is present in the proliferating neck zone in a wild-type control rat (Fig. 3a), while positive cells are scattered throughout the mucosa in a WTC-dfk rat (Fig. 3b). Bar = 50 µm.
To confirm the distribution of the KCNQ1 potassium channel, we performed immunohistochemical analysis for KCNQ1. Many KCNQ1-positive cells were localized in the fundic glands of the wild-type control rats (Fig. 4a), while no immunoreactivity was found in the WTC-dfk rat (Fig. 4b).
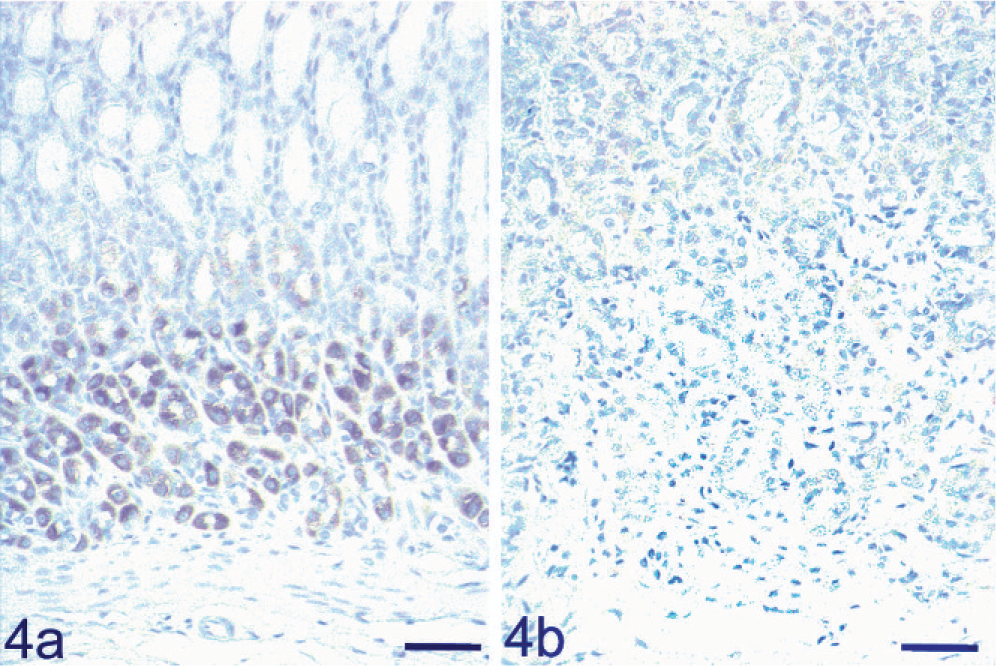
KCNQ1 immunohistochemical analysis in a 34-week-old WTC-dfk rat and an age-matched wild-type WTC rat. KCNQ1 potassium channel immunopositive reaction is present in the fundic glands of a wild-type control rat (Fig. 4a), but not in a WTC-dfk rat (Fig. 4b). Bar = 50 µm.
Expression of PDX-1 has been reported in pancreatic metaplasia of the stomach in humans. 2, 8 No positive reaction was observed in the pancreatic metaplasia of the stomach in the WTC-dfk rats by using immunohistochemical examination for PDX-1.
In 34-week-old WTC-dfk rats, fundic glands were often dilated and contained proteinous fluids. Dilated glands were lined by flattened and shrunken epithelial cells. Fibrosis was advanced in the mucosa of these rats, especially in the area of glandular crypts (Fig. 1a). These fibrotic lesions were clearly demonstrated by bluish connective tissue when stained with Azan stain. Fibrotic lesions were not found in the 4-week-old WTC-dfk rats and in age-matched wild-type control rats.
Discussion
Using immunohistochemical analysis, hypertrophic cells with deeply eosinophilic cytoplasm in the HE-stained sections in the WTC-dfk rats were observed to show a positive reaction to several enzymes for acinar cells of the pancreas, and electron microscopic findings were also consistent with the characteristics of pancreatic acinar cells. Although the chief cell in the gastric gland has a similar ultrastructure, hypertrophic cells were negative for pepsinogen, which is the main component of chief cell granules. Hypertrophic cells are therefore considered to be a metaplasia to the pancreatic acinar cells.
In humans, pancreatic (acinar) metaplasia is frequently found and is associated with chronic gastritis. 4, 7, 23 The so-called hyperplastic endocrine nodules can be found in severe atrophic corpus gastritis, and these nodules may be preneoplastic lesions for neuroendocrine tumors. 22 Exocrine pancreatic metaplasia of the stomach is considered to be associated with endocrine cell hyperplasia. 8 The homeobox transcription factor PDX-1 plays an important role as a switch for glandular exocrine and endocrine differentiation in the pancreas. 2 PDX-1 expression has been demonstrated in pancreatic metaplasia in chronic gastritis in humans, and it may therefore be a candidate switch factor for glandular exocrine and endocrine differentiation. 2 We failed to demonstrate PDX-1 immunoreactivity in pancreatic metaplasia of the stomach in the WTC-dfk rat. Further study is therefore needed to clarify the pathogenesis of pancreatic metaplasia in the WTC-dfk rat.
Kcnq1-deficient mice also show achlorhydria and increased weight of the stomach resulting from mucous neck cell hyperplasia; however, neither pancreatic metaplasia nor fibrosis were reported in the mice models. 3, 6, 17 In addition, no hyperplastic change was found in the WTC-dfk rats. Similar hypertrophic cells with eosinophilic cytoplasm have been described in rats administered lansoprazole, which has an antisecretory effect on the proton pump inhibitor. 5, 9 Loxtidine-induced eosinophilic cells were found to be negative for pepsinogen and were not likely to be derived from the chief cells. 9, 20 The same pepsinogen-negative result was obtained in the metaplastic cells in WTC-dfk rats. Thus, hypertrophic cells might be a nonspecific reaction to the achlorhydria rather than being specific to dysfunction of the KCNQ1 potassium channel.
All gastric cells originate from the proliferating progenitor cells located in the neck zone. 12 Daughters from the progenitor cells migrate either upward toward the lumen, forming the surface mucous cells, or downward toward the base, forming chief cells, parietal cells, and enteroendocrine cells. 12 Ki-67–positive cells were expanded in both the antrum and fundus of the Kcnq1-deficient mice. 17 PCNA-positive cells were also scattered throughout the mucosa in the WTC-dfk rats. Although the thickness of the mucosa does not differ between the WTC-dfk rats and wild-type control rats, this disrupted distribution of proliferating cells suggests an impaired differentiation of gastric progenitor cells. These data raise the possibility that hypertrophic cells of the WTC-dfk rats are related to impairment in the differentiation of gastric progenitor cells. Fibrosis found in 34-week-old WTC-dfk rats suggests the presence of progressive and degenerative gastric lesions in Kcnq1-deficient rats.
In conclusion, a deficiency of the Kcnq1 potassium channel in rats provokes achlorhydria, apparently by reduction of secretion from parietal cells. Pancreatic exocrine cell metaplasia of stomach glands is present in the WTC-dfk rats. This metaplasia may be related to the dislocated and impaired differentiation of proliferating progenitor cells. The WTC-dfk rat may provide a powerful model to clarify the pathophysiology of Kcnq1-related stomach disorders.
Footnotes
Acknowledgements
We thank Dr. H. Nagai (Development Research Center, Takeda Pharmaceutical Company Limited) for his valuable advice and Dr. Chris Wright (Department of Cell and Developmental Biology, Vanderbilt University) for generously providing us with the PDX-1 antibody. This work was supported by a Grant-in-Aid for Scientific Research from Japan Society for the Promotion of Science (JSPS; No. 00081589 and 20244668, to M. Kuwamura). We thank the National Bio Resource Project for the Rat in Japan for providing the WTC-dfk rat strain.
