Abstract
Objectives
Pyroptosis, a special kind of cell death, is generally believed to aggravate the inflammatory response. It has been found that the acinar cell pyroptosis exits in the course of pancreatitis. Ulinastatin has the function of inhibiting the release of inflammatory mediators, thereby reducing the inflammatory reaction, which suggests that ulinastatin has potential inhibitory effect on pyroptosis. The aim of this study is to investigate the effect of ulinastatin on caspase-1-dependent acinar cell pyroptosis in acute pancreatitis.
Methods
Acute pancreatitis model was established by intraperitoneal injection of L-arginine. Rats in ulinastatin group were injected with ulinastatin intravenously after successful modeling. The pancreatic tissues of rats were subjected to pathological examination and detection of cleaved-caspase-1, GSDMD-N, IL-1β, and IL-18. Serum samples were also collected for measurement of IL-1β, IL-18, amylase, and lipase.
Results
Slight pathological damage of pancreas was observed in the pancreatitis and the ulinastatin group. Compared with the blank control group (p < .05), the protein levels of cleaved-caspase-1, GSDMD-N, IL-1β, and IL-18 were significantly increased in pancreatitis group but decreased in ulinastatin group (p < .05). The positive reaction of caspase-1 in pancreatitis group was significantly higher than that in the blank control group (p < .05). After administration of ulinastatin, the positive reaction of caspase-1 was significantly lower than that in pancreatitis group (p < .05). As for the levels of serum IL-1β and IL-18, they were significantly higher in pancreatitis group than those in the blank control group (p < .05). Administration of ulinastatin significantly decreased the serum levels of IL-1β and IL-18 (p < .05). And the serum amylase and lipase levels elevated in pancreatitis group, while significantly inhibited by ulinastatin (p < .05).
Conclusions
The results showed that the onset of acute pancreatitis was accompanied by obvious acinar cell pyroptosis. Ulinastatin could inhibit caspase-1-dependent acinar cell pyroptosis in acute pancreatitis.
Introduction
Pyroptosis is a mechanistically unique form of programmed cell death. Cell death is a key pathophysiological feature in acute pancreatitis, which directly affects the clinical prognosis of patients. Different modes of cell death such as apoptosis or pyroptosis have different effects on the progression of pancreatitis.1–3 It is generally accepted that pyroptosis can promote inflammatory reaction, trigger “waterfall inflammatory reaction,” cause cell damage, and eventually aggravate the progress of the disease, showing a vicious circle of performance.4–7 A large number of studies have shown that ulinastatin plays an important role in reducing the tissue injury caused by acute pancreatitis.8,9 Ulinastatin, as a glycoprotein hydrolase inhibitor, can inhibit the release of inflammatory mediators and reduce the inflammatory reaction. From this point of view, ulinastatin has potential inhibitory effect on acinar cell pyroptosis. But its specific role is not clear. The aim of this study is to investigate the effect of ulinastatin on caspase-1-dependent acinar cell pyroptosis in acute pancreatitis, and to provide new ideas for clinical treatment of acute pancreatitis.
Materials and methods
Animals
Male Wistar rats, weighing 160–180 g (10–12 weeks), were used. Rats were raised under standard conditions (12-h light/dark rhythm; 35 ± 5% humidity at 25°C) and maintained on food and water ad libitum. Animal procedures were in accordance with the Institutional Animal Care and Use Committee of Shanghai CQ (IACUC) Guide for Care and Use of Laboratory Animals and under the principles of replacement, refinement, and reduction.
Preparation of acute pancreatitis model and intervention
A total of 24 rats were fasted for 12 h but were allowed to drink water freely before the experiment. Then, the rats were randomly divided into four groups with six animals in each group: blank control group; ulinastatin control group; pancreatitis group; and ulinastatin group. The sample size was determined by power analysis using preliminary data obtained in our laboratory with the following assumptions: α of 0.05 (two-tailed) and power of 90%. Rats from pancreatitis group were intraperitoneally injected with 20% L-arginine solution (2 × 100 mg/100g) at an interval of 1 h. Each rat in ulinastatin group received a bolus injection of ulinastatin (10,000U/kg; Techpool) through tail vein after establishing pancreatitis model. In the blank control group, each rat received a bolus injection of normal saline (1.0 mL × 2). Each rat in ulinastatin control group received a bolus injection of ulinastatin (10,000U/kg) after being intraperitoneally injected a bolus of normal saline (1.0 mL × 2). Further histopathological analysis and index detection were performed in all rats 24 h after the acute pancreatitis model was established.
Collection and treatment of serum samples
Blood samples were collected from the rats via the tail vein 24 hour after the model was established and the supernatant was centrifuged at room temperature and stored at −70°C. The serum levels of IL-1β, IL-18, amylase, and lipase were determined by using ELISA kits (R&D System Europe Ltd, UK) as described by the manufacturer.
Pancreatic tissue sampling
Rats were anesthetized with urethane and then sacrificed by decapitation. Pancreas of all animals was excised and pancreatic heads were obtained. Tissues to be stained with hematoxylin and eosin (H&E) and immunohistochemistry staining (IHC) were soaked in neutral-buffered 10% formalin solution and then embedded in paraffin blocks wax. Pancreatic tissue sections (3 mm) were taken and detected by Western blot analysis.
Histopathological analysis
Paraffin-embedded tissues were sectioned at 3-μm thickness and stained by hematoxylin and eosin for microscopic observation. The morphological changes were evaluated according to the scoring system of Schmidt by two certified pathologists in a blinded manner. 10
Immunohistochemistry
After a preliminary treatment with hydrogen peroxidase (0.3%), dewaxed pancreatic sections were rinsed with blocking buffer for 30 min at room temperature. After antigen thermal repair, the tissue sections were incubated with the primary goat anti-mouse FBP antibody at a dilution of 1:100 (Biyuntian Biotechnology Co., Ltd) at 4°C overnight. Then, they were incubated with the appropriate biotinylated secondary rabbit anti-goat antibody (Biyuntian Biotechnology Co., Ltd), during which diaminobenzine was used for color development and hematoxylin was used for restaining. The sections were observed under microscope and photographs were taken by a digital camera. Quantitative analysis of the caspase-1 content was measured by Medical Image Quantitative Analysis System (MIQAS) (Shanghai Qiuwei Biotechnology Co., Ltd) via calculating the cytoplasmic positive expression intensity (optical density, OD). Three visual fields of view were randomly inspected to count the positive value, and then averaged the results. All steps in negative control were the same as presented above except that the primary antibody was replaced by PBS.
Western blot analysis
Pancreatic sections were lysed and subjected to Western blotting. Protein concentration was assayed by the Bio-Rad method. The equivalent proteins of each sample were subjected by SDS-PAGE through a 10% acrylamide gel and transferred to polyvinylidene difluoride transfer membranes (Millpore, Bedford, MA) which were blocked at room temperature with blocking buffer (5% non-fat milk powder and 0.1% Tween-20 buffer in PBS) for 2 h, and incubated with primary antibodies (cleaved-caspase-1, Santa Cruz Co., Ltd; IL-18: Biyuntian Biotechnology Co., Ltd; GSDMD-N, IL-1β: abcam) overnight at 4°C in hybridization solution. After washing three times for 5 min in TBST buffer, the membranes were subjected by 2-h incubation with secondary antibody (cleaved-caspase-1, Santa Cruz Co., Ltd; IL-18: Biyuntian Biotechnology Co., Ltd; GSDMD-N, IL-1β: abcam) in hybridization solution at room temperature. The signals were detected by using enhanced chemoluminescence detection system (EMD Millipore, USA). Images of the radiographic bands were recorded by using the Quantity One system (Bio-Rad Laboratories, Inc., USA). The optical density ratio of the target protein to β-actin was used to express the values.
Statistical analysis
All statistical analyses were showed as mean ± standard deviation and were carried out by using SPSS 19.0 statistical software package. (IBM Corp,USA). One-way analysis of variance (ANOVA) was performed for the comparison of qualitative variables. A two-tailed p value < .05 was considered statistically significant.
Results
Validation of the acute pancreatitis model in rats
Histopathological investigation was performed and representative photomicrographs of the pancreas were taken (Figure 1). No obvious abnormalities were found in the pancreas of rats in the blank control group and the ulinastatin control groups (Figure 1(a)). Pathological changes were characterized by interstitial edema, cavitation formation, and inflammatory cell infiltration of the pancreas in pancreatitis and ulinastatin groups (Figure 1(a)). No obvious acinar cell necrosis was observed in both groups. The differences of histological score between pancreatitis and ulinastatin groups were found not to be statistically significant (Figure 1(b)). Histological findings of rat pancreas in each group. Section A: Hematoxylin and eosin staining of rat pancreas in each group (magnification × 200). A. Blank control group; B. Ulinastatin control group; C. Pancreatitis group; D. Ulinastatin group (Scale bar=100 μm). Figure A and B show normal morphology of pancreas. Figure C and D show interstitial edema, cavitation formation and inflammatory cell infiltration of the pancreas. No obvious acinar cell necrosis was observed. Section B: Histological score of pancreatic tissue. The scores of Pancreatitis and Ulinastatin group significantly increased as compared with blank control group. *p<0.05 vs blank control group. #p>0.05 vs Pancreatitis group. &p>0.05 vs blank control group.
Verification of the inhibition effect of ulinastatin on pyroptosis in the acinar cells of acute pancreatitis
Western blot technique was applied to detect the protein expression of cleaved-caspase-1, GSDMD-N, IL-1β, and IL-18 in the acinar cells of acute pancreatitis. All rats of pancreatitis groups demonstrated a significant increase in the protein levels of active caspase-1, GSDMD, IL-1β, and IL-18, compared with those in the blank control group (p < .05; Figure 2). Administration of ulinastatin significantly inhibited the increase of active caspase-1, GSDMD, IL-1β, and IL-18 protein expression. Therefore, the protein levels of active caspase-1, GSDMD, IL-1β, and IL-18 in the ulinastatin group significantly decreased compared with those in the pancreatitis group (p < .05; Figure 2). Expression of pro-caspase-1, cleaved-caspase-1, GSDMD-FL, GSDMD-N, IL-1β, and IL-18 in rats pancreatic tissue measured by Western blot. A: Expose of the electrophoresis gel film; B: Quantified expression of cleaved-caspase-1, GSDMD-N, IL-1β, and IL-18 in each group. Expression of cleaved-caspase-1, GSDMD-N, IL-1β, and IL-18 elevated in Pancreatitis group, while significantly inhibited by ulinastatin. *p < 0.05 vs blank control group. # p <0.05 vs Pancreatitis group. &p > 0.05 vs blank control group.
To investigate the caspase-1 expression patterns in pancreatic tissues, immunohistochemical analysis was further performed on formalin-fixed and paraffin-embedded tissue specimens. And the results were the same as those yielded by Western blot. As showed in Figure 3, compared with the control group, the positive reaction of caspase-1 in the pancreatitis group was significantly higher (p < .05). In ulinastatin group, the positive reaction of caspase-1 was substantially lower than that in the pancreatitis group (p < .05). Expression of caspase-1 in rats pancreatic tissue measured by immunohistochemistry. Section A: Caspase-1 immunoreactive cells in pancreatic tissue in each group (× 200) A. Blank control group; B. Ulinastatin control group; C. Pancreatitis group; D. Ulinastatin group (Scale bar = 100 μm). The arrows in the figure show immunoreactive cells. Immunopositive cells were stained brown-yellow color in the cytoplasm. Figure A and B show small amount of caspase-1 immunoreactive cells. Figure C shows the amount of caspase-1 immunoreactive cells increased significantly. While in Figure D, the amount of caspase-1 immunoreactive cells decreased as compared with Pancreatitis group. Section B: The comparison of the expression of caspase-1 measured by immunohistochemistry in each group. Expression of caspase-1 elevated in Pancreatitis group, while significantly inhibited by ulinastatin. *p < 0.05 vs blank control group. #p < 0.05 vs Pancreatitis group. &p > 0.05 vs blank control group.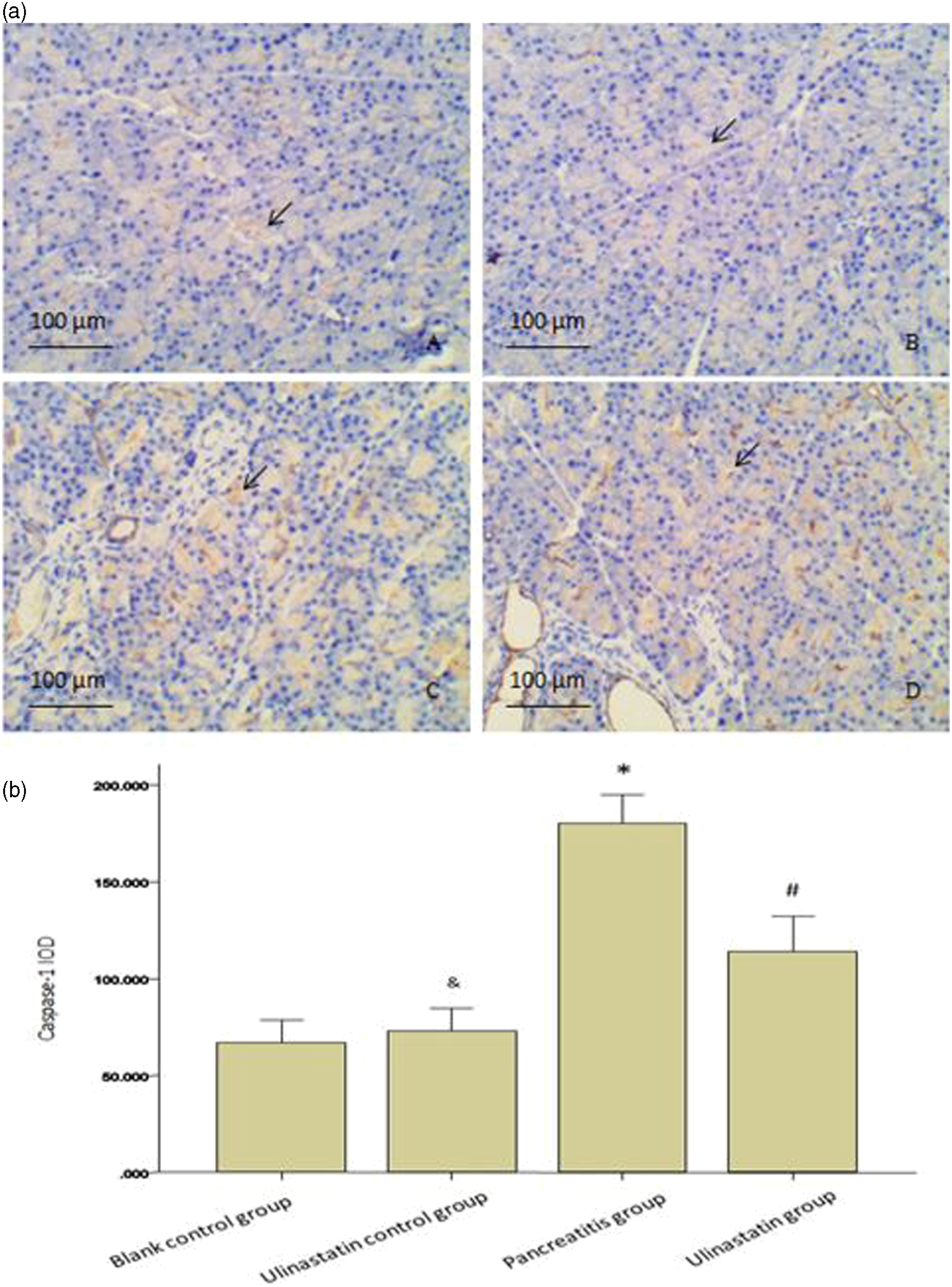
Comparison of serum levels of IL-1β and IL-18 in rats in each group (

ap > 0.05 vs blank control group.
bp < 0.05 vs blank control group.
cp < 0.05 vs pancreatitis group.
Comparison of serum levels of amylase and lipase in each group (

ap > 0.05 vs blank control group.
bp < 0.05 vs blank control group.
cp < 0.05 vs pancreatitis group.
These data revealed that ulinastatin significantly prevented the protein levels of caspase-1, GSDMD, IL-1β, and IL-18 in pancreatic tissues. Considering the characteristic and representative role of caspase-1 and GSDMD in pyroptosis, we think these results indicated that the caspase-1-dependent pyroptosis of acinar cells was considerably inhibited by ulinastatin.
Discussion
According to the data of louhimo et al., about 40% of cell death in acute pancreatitis is not due to necroptosis or apoptosis. 11 Scholars believe that there should be another way of cell death. With the development of pancreatitis research, pyroptosis gradually emerged from the study of apoptosis. In 2001, Cookson first defined this mode of cell death as pyroptosis. 12 The team of Shen Feng and other scholars has found that there exists pyroptosis of acinar cells in the development of pancreatitis. 13 Pyroptosis is a kind of inflammatory cell death accompanied by the synthesis of inflammasome and the activation of IL-1β and IL-18, which leads to a waterfall of inflammatory response and cell damage. 5 When the activation pathway of inflammasome is inhibited, the mortality of pancreatic cells decreases significantly, which indicates that pyroptosis plays an important role in the development of pancreatitis. 14 Most scholars believe that pyroptosis can aggravate inflammatory reaction. For example, Mazzolini’s study on liver inflammation found that pyroptosis aggravated the inflammatory response. 15 Labbé shared the same idea of pyroptosis. 6 However, the specific role of pyroptosis in pancreatitis remains unclear.
There are a series of special pathological changes in pyroptosis. At the beginning, many tiny pores formed on the cell membrane, then the ionic gradient on both sides of the cell membrane disappeared and the extracellular water influx across the membrane, eventually resulting in the serious swelling of the cells until they ruptured and died. 16 In addition, nucleus condensation and chromosome degradation in pyroptosis were found similar to those in apoptosis. 17 Therefore, the pyroptosis cells present some characteristics of apoptosis and necrosis at the same time, which explains why it is particularly important to distinguish pyroptosis from apoptosis. Obviously, pyroptosis is lack of characteristics in HE staining and is difficult to be observed, as shown in our study. In particular, the model in this study is not severe pancreatitis, so it is more difficult to distinguish pyroptosis through the pathological changes showed by HE staining. Related studies have shown that caspase-1 is advantageous to deal with this problem. Caspase is an important group of protein family with high homology and similar structure. Research studies have shown that caspase and its substrate related to apoptosis do not participate in pyroptosis, and caspase-1, on which pyroptosis depends, is not involved in the apoptotic pathway. Therefore, caspase-1 detecting can be used to distinguish pyrotosis from apoptosis.17,18 In the present study, caspase-1 activation has been found in acinar cells of acute pancreatitis by immunohistochemistry, which is a favorable evidence of the occurrence of pyroptosis in these cells.
Caspase-1 belongs to proteolytic enzymes family, which exists as its preemzyme at the initially formative stage. Once the cells are stimulated by pathogens or danger signals, the inflammasome will be assembled inside the cells, which can cleave the inactive caspase-1 into the active caspase-1, also known as the specific executive protein of pyroptosis.19,20 Caspase-1 gene deletion can reduce the severity of inflammation induced by caerulein in rats of acute pancreatitis.4,5 Some studies have also found that caspase-1-specific blocker can inhibit the formation of membrane micropores of pyroptotic cells, which leads to a series of pyroptotic changes, such as imbalance of electrolyte between the inner and the outer of cell membrane, cell swelling, membrane rupture, and release of inflammatory mediators.16,18 Clearly, caspase-1 activation is the key step of pyroptosis. Furthermore, the downstream signaling transduction pathways after caspase-1 activation become clearer with the development of research, and further progress has been made in the study of Gasdermin D protein, which is now believed to be an executive protein of pyroptosis. Activated caspase-1 cleaves the junction of N-terminal and C-terminal domains of GSDMD protein and releases the N-terminal domain with punching activity on the phospholipid membrane, thus disrupting the cell membrane and triggering pyroptosis.21,22 This is one of the important ways of pyroptosis. In our study, the activation of caspase-1 and GSDMD in pancreatic acinar cells was simultaneously detected, which provided strong evidence for the existence of caspase-1-dependent pyroptosis. Moreover, the inhibition of caspase-1 and GSDMD activation was also detected in rats injected with ulinastatin, which showed the inhibitory effect of ulinastatin on pyroptosis. Ulinastatin, also known as urinastatin, is a refined glycoprotein extracted from human urine. Previous studies have shown that ulinastatin can reduce apoptosis by inhibiting caspase-3 activation. 23 Recent studies have also shown that ulinastatin can inhibit the inflammatory activation induced by lipopolysaccharide. 24 These results also suggest the possible intervention pathway of ulinastatin on pyroptosis. It remains elusive how caspase-1 activates and specifically recognizes and precisely cleaves GSDMD; further research studies are needed on the specific mechanism of ulinastatin in pyroptosis.
It has been known that caspase-1 can also convert pro-IL-1β and pro-IL-18 into IL-1β and IL-18 with pro-inflammatory activity, which can be secreted out of the cells. Then, IL-1β and IL-18 can recruit and activate other immune cells and induce the synthesis of inflammatory factors, adhesion factors, and chemokines, thus amplifying local and systemic inflammatory response.19,25 In the present study, the results show that the activation of caspase-1 was accompanied by the activation of IL-1β and IL-18, indicating the presence of caspase-1-dependent pyroptosis in the course of acute pancreatitis, which is also consistent with previous studies. In rats injected with ulinastatin, the levels of IL-1β and IL-18 in pancreatic tissue and serum were significantly decreased, suggesting that ulinastatin may play a role in inhibiting the activation of caspase-1. Previous studies have shown that ulinastatin can inhibit the level of IL-1β in acinar cells of acute pancreatitis in a dose-dependent manner.26,27 Therefore, whether ulinastatin has other mechanisms to inhibit the activation of IL-1 needs further studies.
Conclusion
In summary, the present findings demonstrate that there exists obvious acinar cells pyroptosis in the course of acute pancreatitis and ulinastatin can inhibit caspase-1-dependent pyroptosis of these cells. The present study provides novel insights into the role of ulinastatin in acute pancreatitis, but it has certain limitations. The interaction between pyroptosis and other cell death modes in acute pancreatitis has not been observed. What role ulinastatin plays in this process has not been deeply studied. Whether the inhibitory effect of ulinastatin on pyroptosis can affect the mortality needs to be discussed in the following study. In addition, this study is only aimed at mild acute pancreatitis. The effect of ulinastatin on pyroptosis in severe pancreatitis deserves further investigation. In brief, the exact mechanism via which ulinastatin inhibits caspase-1-dependent pyroptosis remains to be studied to provide a scientific basis for the clinical diagnosis and effective treatment of acute pancreatitis.
Footnotes
Declaration of conflicting interests
The author(s) declared no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Xiamen Municipal Bureau of Science and Technology project (3502Z20184030).
