Abstract
We occasionally encounter feline cervical or mesenteric lesions diagnosed histopathologically as abscess or inflammatory granulation tissue with eosinophil infiltration. Gram-positive cocci accompany the lesions. In the present study, such lesions obtained from 27 cats were examined to evaluate the histopathologic features and the nature of the causative bacteria. The average age was 7.3 ± 3.5 years. No sex predilection was observed. Most frequent locations of the lesions included the abdominal cavity with/without mesenteric lymph nodes (11/ 27, 41%) and subcutaneous tissue or lymph nodes of the neck (9/27, 33%). Common clinical presentation was a localized mass. Grossly, the lesions contained abscesses in the center and were surrounded by fibrous tissue. Microscopically, the necrotic zone contained bacterial colonies. Large numbers of eosinophils and macrophages infiltrated the area surrounding the necrotic tissue. The surrounding connective fiber-rich granulation tissue demarcated the eosinophilic abscess. The bacteria were Gram-positive cocci in 23 of the 27 cats and were positive for anti-staphylococcus antiserum in 19 of the 23 cats. In 15 out of 17 lesions, the colonies expressed immunoreactivity to penicillin-binding protein 2′, which is a drug-resistance gene product of methicillin-resistant Staphylococcus (MRS) species. These findings suggest strongly that MRS causes this type of infectious lesion.
Introduction
Abscesses caused by infection with pyogenic microorganisms is common in cats. Histologically, the lesions consist of neutrophil accumulation with bacterial colony formation. 2 , 9 At a late stage of the suppurative inflammatory response, fibrosing granulation tissue encapsulates the abscesses. 2 , 9
Recently, we encountered such encapsulated abscesses in the cervical and mesenteric regions of cats. The lesions had a central necrotic zone with numerous Gram-positive coccal colonies and prominent eosinophils surrounded by fibrosing granulation tissue. In the first cat that we studied, methicillin-resistant Staphylococcus was identified by microbial culture from a surgical specimen. However, microbial culture was not performed in most of the other cats because the lesions were removed by veterinary practitioners as surgical biopsy specimens of a tumor mass.
The mechanism of methicillin resistance is well established and is caused by alternation of the bacterial gene mec A that encodes penicillin-binding protein (PBP). Staphylococcal species often become resistant to a wide variety of antibiotics, including methicillin, through production of an altered PBP2, referred to as PBP2′ or PBP2a. 7 In the present study, we used mouse monoclonal antibodies against PBP2′ for immunostaining by using routine histologic sections of surgical specimens.
The objectives of this study were to characterize the clinicopathologic features of these feline abscesses and to determine the methicillin resistance of the bacteria in paraffin sections by using an immunohistochemical approach.
Materials and Methods
Surgical biopsy specimens were obtained from a total of 27 cats. The lesions were sent to the Research Institute of Drug Safety, Setsunan University, and Marupi Lifetech Co. for histopathologic examination during the period 1994–2000. All the specimens were fixed in 10% phosphate-buffered (pH 7.4) or unbuffered formalin, dehydrated in a graded series of ethanol, and embedded in paraffin, after which 4-μm sections were stained with hematoxylin and eosin. Sections also were stained by the Gram, Giemsa, and Luna methods.
For indirect immunoperoxidase staining, anti–Escherichia coli rabbit polyclonal antibody (diluted at 1:100; DAKO, Kyoto, Japan), three types of anti–Staphylococcus aureus rabbit polyclonal antibodies (diluted at 1:2,560; CH-91, Wood-46, and 3-9D, courtesy of Dr. S. Takeuchi, Fukui Prefectural University), 3 and four types of anti-PBP2′ mouse monoclonal antibodies (diluted at 1:50; B345M, A376M, E303M, and A465M, Denka Seiken, Tokyo, Japan) 6 were used as primary antibodies. The reactivity of the anti–S. aureus antibodies with S. aureus 3 and S. intermedius was confirmed by positive reactions with bacteria smears. After dewaxing and dehydration, the slides for immunostaining with PBP2′ were immersed in 10 mM citrate buffer (pH 6.0) and autoclaved at 121 C for 5 minutes for antigen retrieval. Antigen retrieval was essential for reproducible detection of PBP2′. Slides were then rinsed in 10 mM phosphate-buffered saline (pH 7.2) and sequentially incubated with the primary antibody and horseradish peroxidase–labeled secondary antibody against rabbit or mouse immunoglobulins for 30 minutes at room temperature. Diaminobenzidine solution containing 0.01% hydrogen peroxide was used for color development. The slides were lightly counterstained with Mayer's hematoxylin.
Microbial culture using pus drained from the lesions was performed on two samples.
Results
The clinical, histopathologic, and immunohistochemical findings are summarized in Table 1.
The clinical, histopathologic, and immunohistochemical findings in 27 cats with abscess-forming inflammatory granulation tissue.
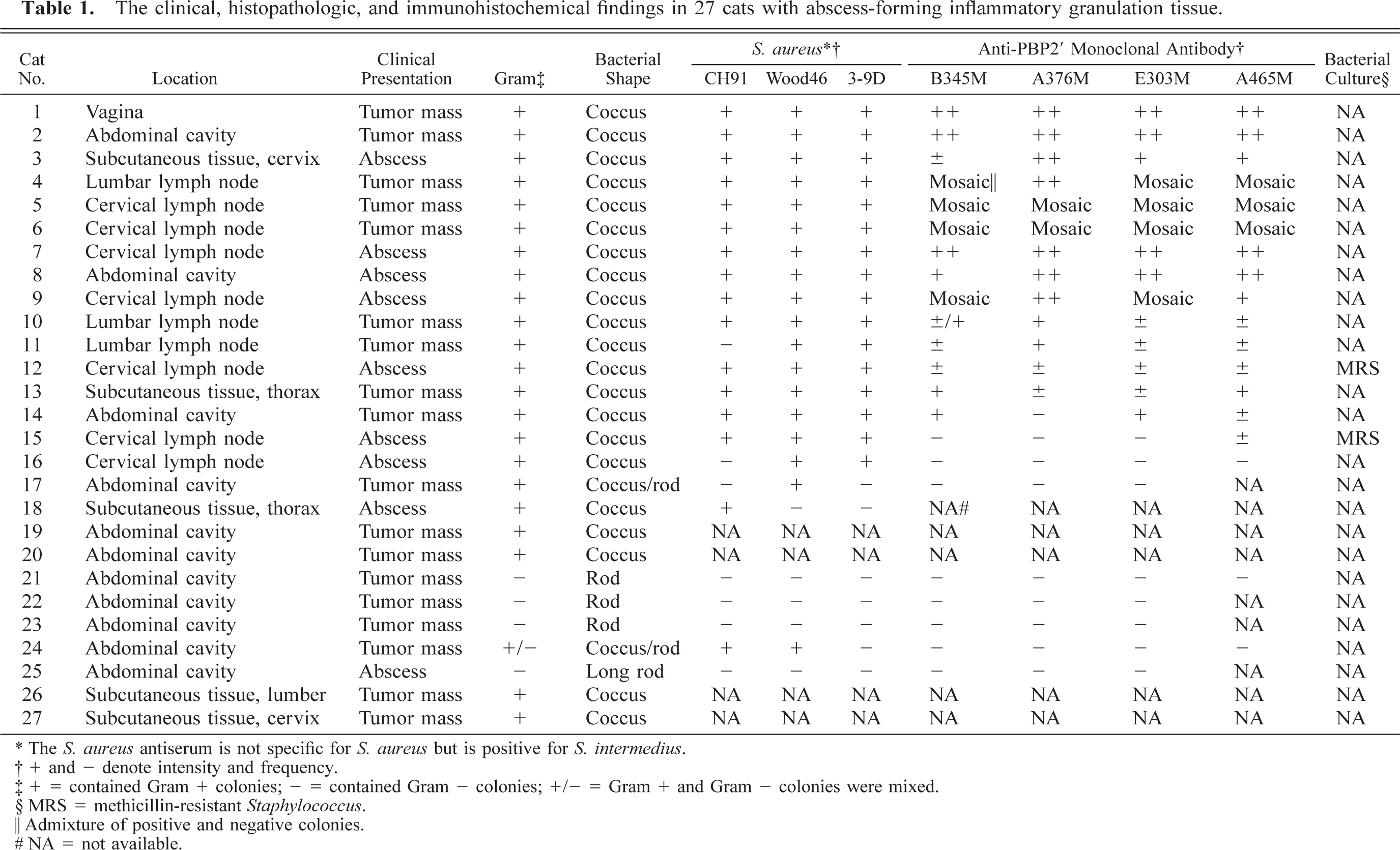
∗ The S. aureus antiserum is not specific for S. aureus but is positive for S. intermedius.
† + and - denote intensity and frequency.
‡ 1 = contained Gram + colonies; − = contained Gram - colonies; +/− = Gram + and Gram - colonies were mixed.
§ MRS = methicillin-resistant Staphylococcus.
‖ Admixture of positive and negative colonies.
# NA = not available.
Twenty-six cats were of mixed breed, and one cat was of Himalayan breed; all specimens were collected from Japan. The average age was 7.3 ± 3.5 years (range 2–14, median 7.5). There were five intact males (19%), seven intact females (26%), seven neutered males (26%), and eight neutered females (30%). The locations of the masses included the abdominal cavity with or without the involvement of the mesenteric lymph nodes (11 lesions, 41%), the subcutaneous tissue with or without cervical lymph nodes (nine lesions, 33%), the subcutaneous tissue with or without lumbar lymph node involvement (four lesions, 15%), the subcutaneous tissue of the thorax (two lesions, 7%), and the vagina (one lesion, 4%). Clinical diagnoses included tumor mass (18 lesions, 67%) and abscess (nine lesions, 33%). Serum positivity for feline immunodeficiency virus antibody was observed in three of the 12 (25%) cats for which test results were available. None of these 12 cats tested positive for feline leukemia virus antibody.
In gross specimens, variably sized single or multiple abscesses (1–15 mm) were identified within each mass. These foci were commonly surrounded by fibrous tissue of variable thickness. Some abscesses formed fistulae from which pus drained.
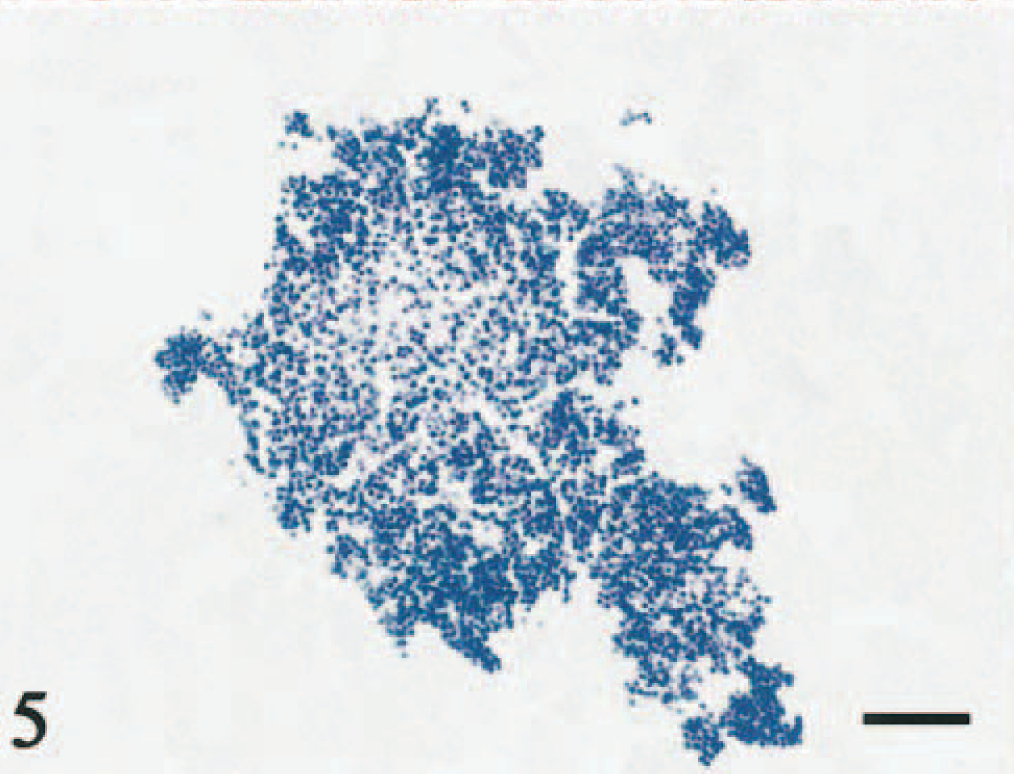
Microscopically, a necrotic zone with numerous bacterial colonies formed the center of each abscess. The necrotic zone was surrounded by fibrous granulation tissue or densely collagenous tissue (Fig. 1). Many eosinophils, neutrophils, and macrophages infiltrated the fibrous connective tissue (Figs. 2, 3), and there was a lymphoplasmacytic cellular infiltrate. The irregularly shaped necrotic zones varied considerably in size, and the large ones tended to be cavitated. In some lesions, there was advanced fibrosis, occasionally hyalinized (Fig. 4). Among the hyalinized collagen bundles, there were often numerous mast cells and eosinophils (Fig. 5).
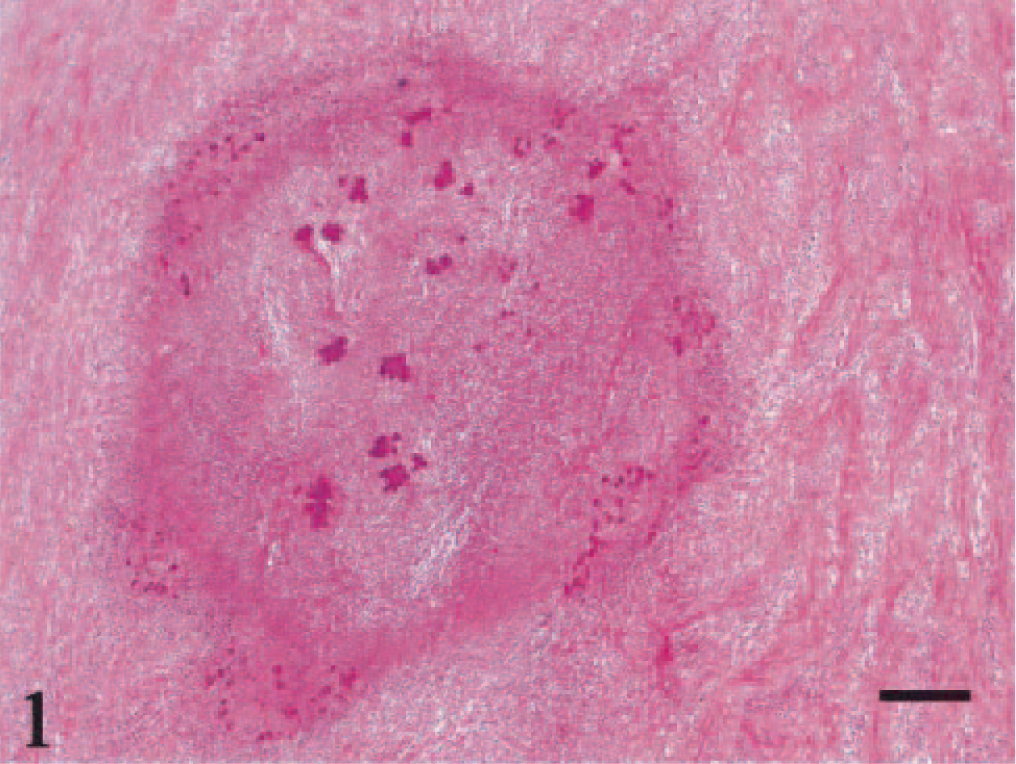
Abdominal mass; cat No. 961206. A necrotic zone is surrounded by fibrosing granulation tissue. HE. Bar = 200 μm.
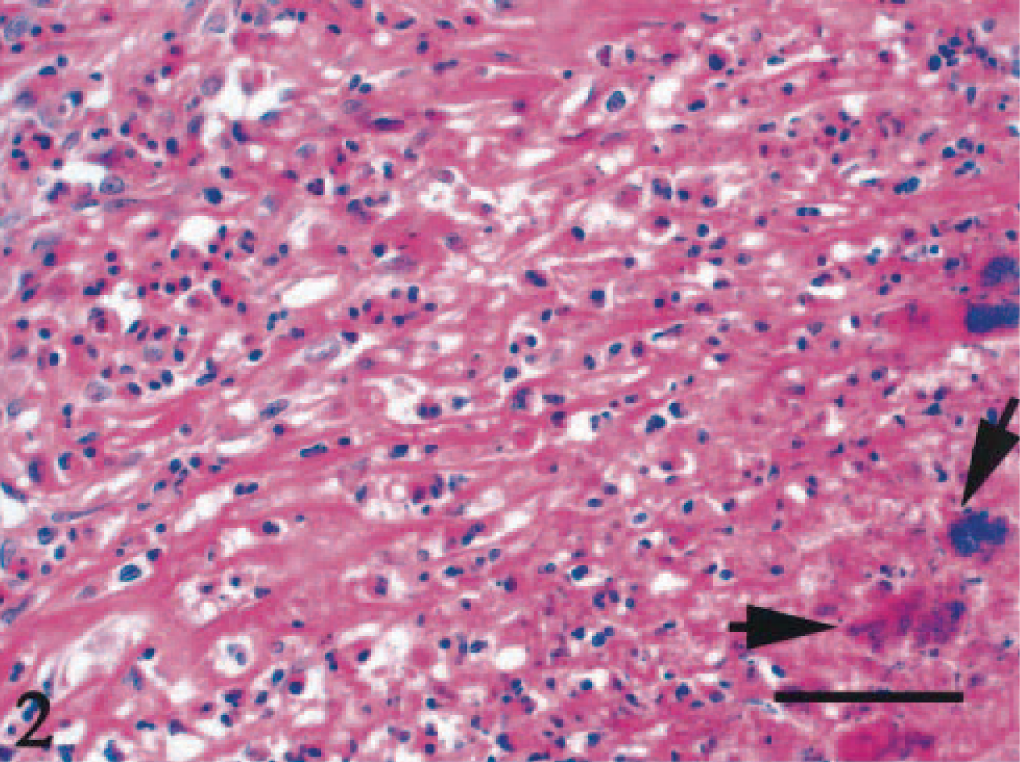
Abdominal mass; cat No. 961206. Numbers of coccal colonies (arrows) are distributed within the necrotic zone, surrounded by fibrous tissue with infiltration of eosinophils, neutrophils, and macrophages. HE. Bar = 50 μm.
Abdominal mass; cat No. 961206. Eosinophils in the lesion are stained using the Luna method. Luna. Bar = 50 μm.
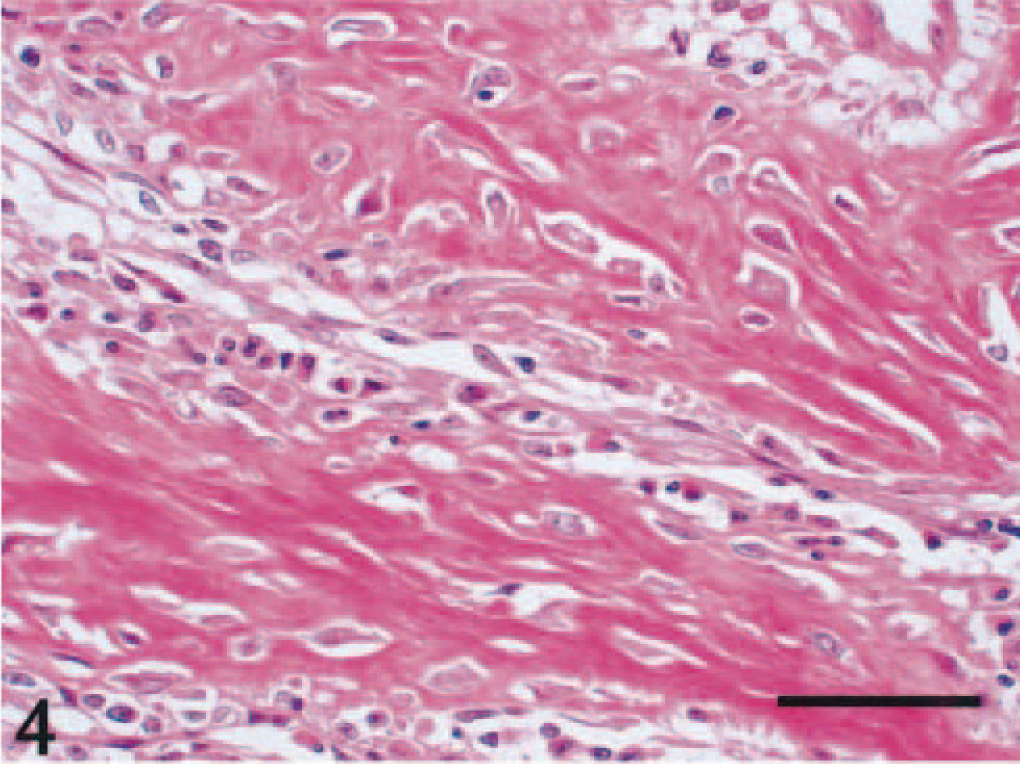
Abdominal mass; cat No. 961206. Fibrosing granulation tissue is characteristic of the mass lesion. Thick collagen bundles and intervening fibroblasts are observed. HE. Bar = 50 μm.
Abdominal mass; cat No. 961206. The cocci in the center of the abscess are shown to be Gram-positive. Gram. Bar = 1 μm.
The bacteria were identified as Gram-positive cocci (21 cats), Gram-negative rods (3 cats), Gram-negative long bacilli (1 cat), and mixed infections of Gram-positive cocci and Gram-negative rods (2 cats). Gram-positive cocci were identified as Staphylococcus, based on the morphologic features and immunoreactivity (Fig. 6, Table 1). Gram-negative rods and long bacilli were not identified. From two lesions, in which Gram-positive cocci were demonstrated, methicillin-resistant, coagulase-positive Staphylococcus was identified by microbial culture.
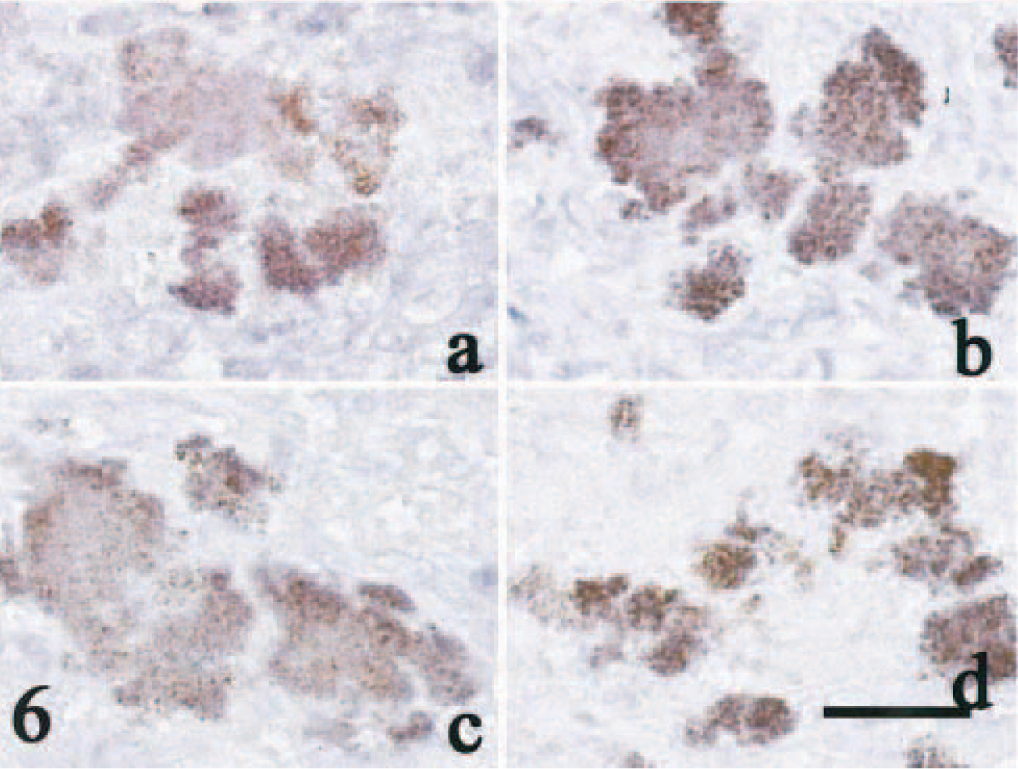
Seventeen lesions with numerous colonies of Gram-positive cocci and five lesions with Gram-negative bacilli were examined immunohistochemically for expression of PBP2′. Fifteen lesions with Gram-positive cocci were immunohistochemically positive for PBP2′ with at least two of the four monoclonal antibodies (Fig. 6); Gram-negative bacilli did not stain with any of these antibodies. In 13 lesions, all four monoclonal antibodies reacted with the cocci (Table 1). The two lesions from which methicillin-resistant coagulase-positive Staphylococcus were cultured had positive immunohistochemical reactions.
Discussion
Prominent histopathologic features of these infectious lesions included a central abscess that contained numerous bacterial colonies and was surrounded by collagenous granulation tissue. Infiltration of eosinophils, neutrophils, and mast cells was common, and hyalinization of the collagen fibers occurred occasionally. These lesions are encapsulated abscesses. 2 , 9
Formation of inflammatory granulation tissue with prominent eosinophil infiltration in cats occurs as part of the feline eosinophilic granuloma complex or with inflammation in response to parasites. 10 , 11 Lesions of the eosinophilic granuloma complex are usually formed in the skin and oral cavity and are characterized by nodular or diffuse granulomatous dermatitis with multifocal “flame figure” formation. The flame figures are variably sized foci of degenerate collagen bundles. Pathogens are usually absent from these lesions, except in ulcerated areas. In our series these features were not observed in lesions.
Subcutaneous abscess formation is common in adult male cats. 9 The predilection in male cats is related to the fact that the lesions are frequently caused by bite wounds. Therefore, castration, which results in less fighting and roaming, can prevent this type of lesion. 9 Lesions in the present series were associated with bacterial colonies, but it is unlikely that the lesions were caused by bite wounds because of the absence of sex predilection and the frequent occurrence of lesions in the abdominal cavity. Clinically, two-thirds of the lesions were surgically removed in the belief that they were tumor masses. The incorrect clinical diagnosis probably resulted from the presence of dense fibrosis. The reason the collagen fiber bundles became so dense in the lesion was unclear, but infiltration of mast cells among the collagen bundles may have influenced the fibrotic reaction. 1
A high rate of infection with Gram-positive cocci, Staphylococcus, was characteristic in our series; moreover, most of the bacteria were considered to be methicillin resistant based on the immunohistochemical findings. Staphylococcus is reported from about 14% of feline abscesses. 2 This frequency is much lower than that observed in the present series. Using microbial culture, methicillin-resistant Staphylococcus has been isolated from the skin surface of the cat, but such findings are infrequent. 9
When bacterial culture is not available, methicillin resistance can be determined in formalin-fixed histopathologic sections by using immunohistochemical techniques, as shown in the present study. In our series, bacterial culture was performed in only two cats, and it was found to be coagulase-positive Staphylococcus. However, this could not identify the species of Staphylococcus because S. intermedius, a species frequently cultured from cat tissue, 5 cannot be distinguished from S. aureus. Both species are commonly coagulase-positive and biochemically similar in nature. 4 S. intermedius is not infectious for humans, whereas methicillin-resistant S. aureus (MRSA) is an important and serious human pathogen, particularly in hospital-acquired infection. 8 It may be worth noting that two cats with Gram-positive cocci were kept by nurses who may have brought back MRSA from their hospital.
It is known that S. aureus does not readily infect the cat, although it has been isolated from the cat and dog occasionally. 5 Therefore, if the pathogen in the cat is true MRSA, the infected cat may become a source of MRSA infection for human family members. Even if the bacterium is a different kind of MRS, such as methicillin-resistant S. intermedius, which does not infect humans, it may represent a serious problem in veterinary health, as the lesions in the present study contained numerous bacterial colonies and often formed a fistulous tract to the overlying skin surface or into the digestive tract. Thus, it is important to investigate the pathogenesis of abscess-forming fibrosing lymphadenitis in domestic cats and to identify conclusively the bacteria associated with these lesions.
Footnotes
Acknowledgements
We thank Akira Shimizu, DVM, PhD, Kobe University, for providing the bacterial colony of S. intermedius and Hiroshi Takagi, DVM, Marupi Lifetech Co., Ltd., for technical assistance.
