Abstract
Mortality rate in humans infected with Nipah virus (NiV) has been reported as high as 92%. Humans infected with NiV show a widespread multisystemic vasculitis with most severe clinical and pathologic manifestations in the brain, lungs, and spleen. The purpose of this study was to study pathologic and immunohistochemical findings in guinea pigs infected with NiV. Of 28 animals inoculated intraperitoneally, only 2 survived the infection, and most died between 4 and 8 days postinoculation (dpi). Viral antigen with minimal pathologic changes was first detected 2 dpi in lymph nodes and spleen. More severe changes were noted in these organs 4-8 dpi, where pathologic damage had a vasocentric distribution and viral antigen was abundant in vascular endothelium, tunica media, adventitia, as well as in macrophages lining sinuses. The urinary bladder, uterus, and ovaries were also affected with necrosis and acute inflammation. In these organs, immunohistochemical positive staining was intense in blood vessels, epithelial cells, and ovarian follicles. Approximately 50% of the animals that died or were euthanized in extremis had evidence of viral antigen and histopathologic changes in brain, especially involving meninges and ependymal cells, with lesser changes in the neural parenchyma. A unifying feature of the damage for all affected tissues was necrosis and inflammation of the vasculature, chiefly in arterioles, capillaries, and venules. Inoculation of guinea pigs intraperitoneally with NiV produces a disease with considerable resemblance to the disease in humans, but with reduced pulmonary involvement and marked infection of urinary bladder and the female reproductive tract.
Introduction
Nipah virus (NiV) is the cause of an emerging disease that first surfaced in Malaysia in 1998 with 265 reported cases of illness in humans, including 105 deaths.
4,
5,
15,
18
Almost all cases involved contact with swine, which were also clinically affected, but with a markedly lower mortality rate than that seen in humans.
15,
16,
18
Molecular characterization of this new paramyxovirus revealed striking resemblance to Hendra virus, which was responsible for another emerging disease described in Australia. These 2 viruses are now grouped in a new and distinct genus,
During the initial emergence of NiV, an intense campaign was implemented in Malaysia to control the spread of the virus, including the slaughter of more than a million pigs. 7 Ultimately the origin of NiV, as for Hendra virus, was traced back to pteropid bats (flying foxes), the probable natural reservoir. 6, 19, 20, 28 The disease resurfaced on several occasions in Bangladesh and India with a mortality rate ranging between 40% and 92% in affected humans. 3, 12, 25 In these latter outbreaks, transmission took place without the pig as an intermediary host, and contact with bats or their body fluids has been implicated as the source of infection. Given the high mortality rate, the persistent reservoir in wildlife, and the history of multiple geographic outbreaks, NiV remains a serious and potential threat to human health.
In pigs, considered to be an amplifying host, NiV typically causes a respiratory disease in the young with CNS involvement in older animals. 14, 16 Although the morbidity rate among swine is estimated close to 100%, mortality is low and most affected pigs recover. 16 In domestic cats, which have been infected both naturally and experimentally, the disease is prominently respiratory in nature. 11, 14 In contrast, the infection in humans is a widespread multisystemic vasculitis, with the most severe debilitating clinical and pathologic manifestations in the brain, lungs, and spleen. 11, 27 A more thorough understanding of the pathogenesis would assist in the design of control mechanisms aimed at decreasing viral shedding and interrupting transmission, as well as in the development of vaccines and therapeutic modalities to control and mitigate disease in the event of future outbreaks.
The purpose of this study was to examine pathologic and immunohistochemical changes in archival formalin-fixed paraffin-embedded tissues from a study in which guinea pigs were experimentally infected with NiV. Our goal was to gain more insight into pathogenic mechanisms that might in turn have implications for the disease in humans and other natural hosts.
Materials and Methods
Archived formaldehyde-fixed paraffin-embedded guinea pig tissues were acquired from the Centers for Disease Control and Prevention (CDC) from a study conducted in the CDC BSL4 laboratories for evaluation of the guinea pig as a model for Nipah virus infection (performed in accordance with the CDC IACUC). A medical virologist collected samples and gross findings were not recorded.
This study utilized 30 2–3-month-old female guinea pigs (seronegative for Nipah), of which 28 were intraperitoneally inoculated with 1 ml of Hank's balanced salt solution (HBSS) with 6 × 104 PFU of Nipah virus (CDC; Atlanta, GA; dosage and route identified in pilot study, data not shown), and 2 were inoculated with media only (controls). Two animals were euthanatized at 2 dpi for early examination, and the remaining animals were left to develop clinical disease. Two of the infected animals survived past 8 dpi, recovered, and were euthanatized at 13 dpi (seroconverted to NiV). Of the remaining infected study animals, which died between days 4 and 8 postinfection, only 20 animals had tissues available for evaluation. Tissues included adrenal glands, brain, esophagus, heart, intestines, kidney, liver, lung, mesenteric, and lumbar lymph nodes, ovary, pancreas, spleen, thymus, trachea, urinary bladder, and uterus. For each tissue, 4-µm sections were cut and either routinely stained with hematoxylin and eosin (HE) or used for immunohistochemistry (IHC).
For IHC, slides were deparaffinized in xylene and rehydrated by processing them through graded alcohols. Antigen retrieval was performed by heating slides in a citrate-based solution (AR-10 Solution; Biogenex Laboratories, San Ramon, CA) for 20 minutes in a rice steamer. Normal sheep serum (20%) was used as a blocking agent. The primary antibody (anti-Nipah primary antibody developed as a hyperimmune mouse ascitic fluid [Special Pathogens Branch, CDC, Atlanta GA]) was incubated on the tissue sections at a dilution of 1 : 2,000 for 1 hour at room temperature. Then, a sequential application of biotinylated antibody (Rabbit anti-mouse E0354; DAKO Corporation, Carpinteria, CA), alkaline phosphatase-conjugated streptavidin, and naphthol fast red were applied. Sections were counterstained in Mayer's hematoxylin and mounted using an aqueous mounting medium. Negative antibody controls for each slide included replacing the primary antibody with normal mouse ascitic fluid or with anti-Nipah antibody absorbed with Nipah viral antigens.
Results
Histopathology and IHC results are presented in Table 1. Uninfected control animals had no histologic lesions considered typical of Nipah virus infection and were negative for NiV antigen by IHC. Similarly, all IHC results using normal mouse ascites fluid or anti-Nipah antibody absorbed with NiV antigen were negative. Histopathologic changes in the animals that were euthanized at 2 dpi were minimal and only observed in the lumbar lymph node of 1 of the 2 animals (guinea pig No. 1). Changes in this lymph node consisted of focal coagulation necrosis that contained syncytial cells and a single follicle with lymphoid depletion and germinal center necrosis. Nipah virus antigen was identified by IHC within these syncytial cells. While lymph nodes were not available for evaluation in the second 2 dpi animal, NiV antigen was evident in scattered lymphocytes and macrophages surrounding splenic periarteriolar lymphoid sheaths (PALS).
Severity of histopathologic changes (HE) and antigen distribution (IHC) in extraneural organs of guinea pigs inoculated IP with 6 × 104 PFU of NiV.∗
DPI = days postinoculation; GI tract = stomach and intestines.
HE = hematoxylin and eosin; - = no abnormal findings; + = minimal changes consisting of focal inflammation with some necrosis and rare syncytial cells; ++ = moderate changes consisting of necrosis that can be focally extensive with or without inflammation, syncytial cells are usually prominent; +++ = severe inflammation, extensive necrosis, hemorrhage, and numerous syncytial cells.
IHC = immunohistochemistry; - = negative; + = less than 2 positive cells/high-power field (hpf); ++ = 2-10 positive cells/hpf; +++ = more than 10 positive cells/hpf; NA = tissues not available for evaluation.
Histopathologic findings in animals that died or were euthanatized between 4 and 8 dpi were similar to each other. All tissues evaluated had evidence of endothelial and/or epithelial syncytial cells as well as mural lymphohistiocytic vasculitis with fibrinoid vascular change. The most notably affected organs were the spleen, lymph nodes, urinary bladder, ovary, uterus, and brain.
Splenic and lymph node changes were most dramatic histologically. Of the 20 4–8 dpi animals, spleen was available from all, and lymph nodes were available from 17 animals. In addition to multifocal to widespread transmural vasculitis with necrosis of vascular walls and fibrinoid change, the spleen and lymph nodes had moderate lymphoid depletion and necrosis (Figs. 1–4). Splenic changes ranged from moderate focal lymphoid depletion and scattered necrosis to severe multifocal to coalescing necrosis of lymphoid areas with obliteration of normal architecture and numerous and prominent syncytial cells. Splenic parenchymal necrosis was prominent in PALS and occurred circumferentially around the central arterioles, appearing as zones of coagulation necrosis with loss of normal architecture (Fig. 1A). Syncytial cells were frequently evident both within the areas of necrosis as well as in the surrounding red pulp. Similar findings were present in the lymph node as in the spleen, ranging from normal or mild focal depletion and necrosis of lymphoid tissue with scattered syncytial cells (Fig. 3) to marked multifocal depletion and necrosis of lymphoid areas with numerous syncytial cells scattered throughout. These syncytial cells were also frequent in lymphoid follicles within lymph nodes. Erythrophagocytosis was frequently observed in the lymph nodes.
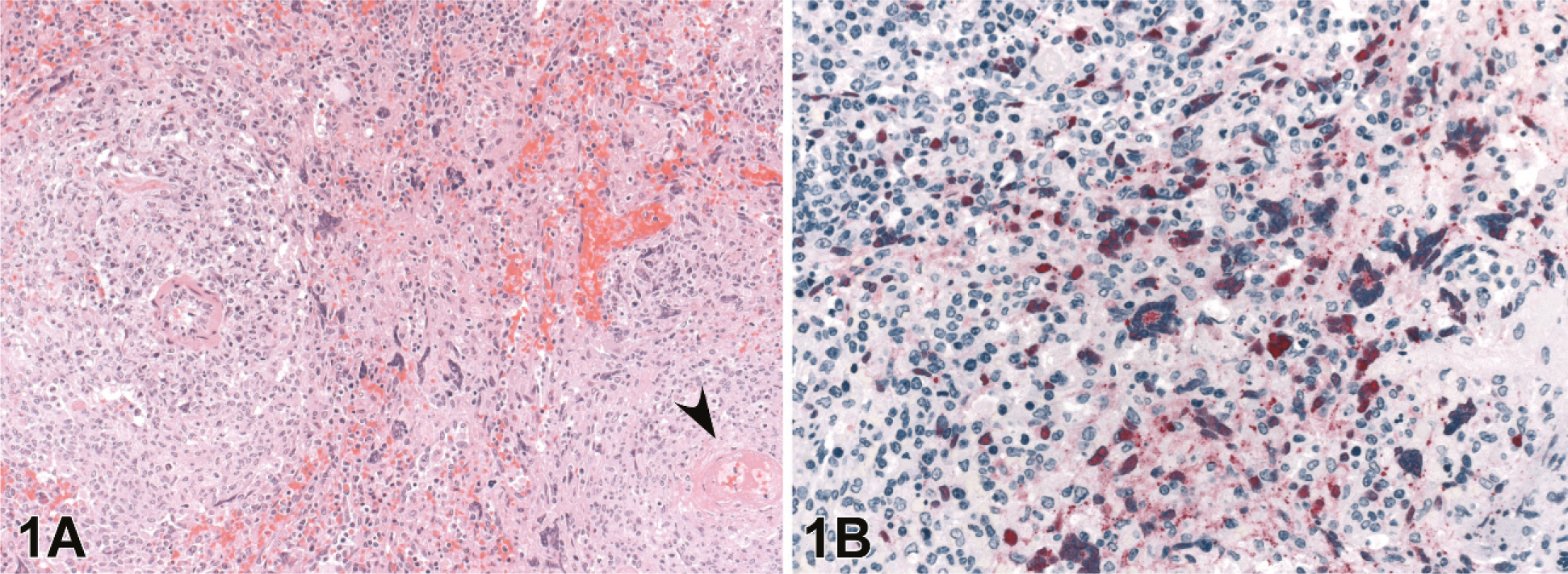
Spleen; guinea pig 6 dpi (guinea pig No. 9).
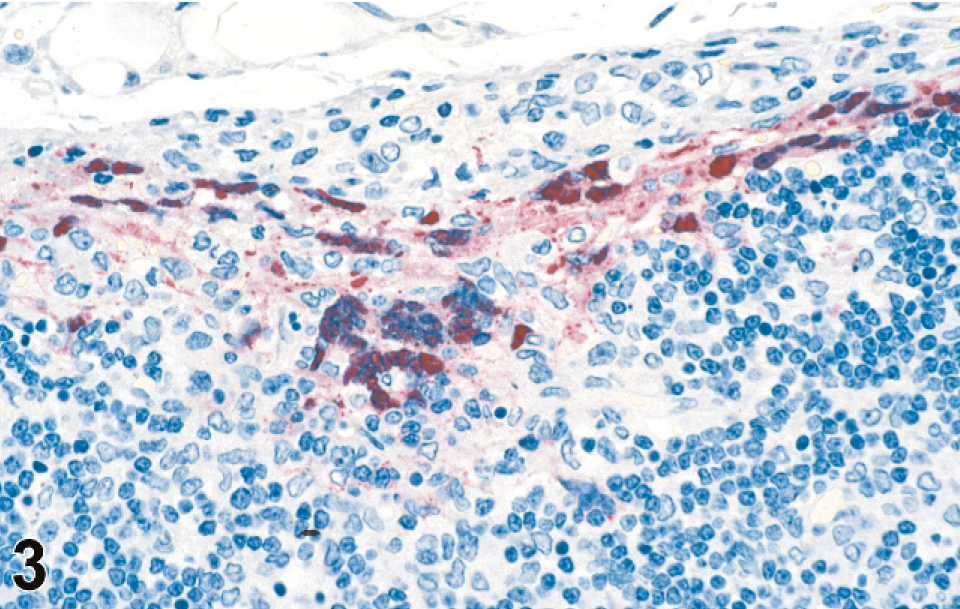
Lymph node; guinea pig 7 dpi (guinea pig No. 14). IHC. Nipah virus antigen present in lymph node subcapsular sinus and syncytia. Fast Red substrate and Mayer's hematoxylin counterstain.
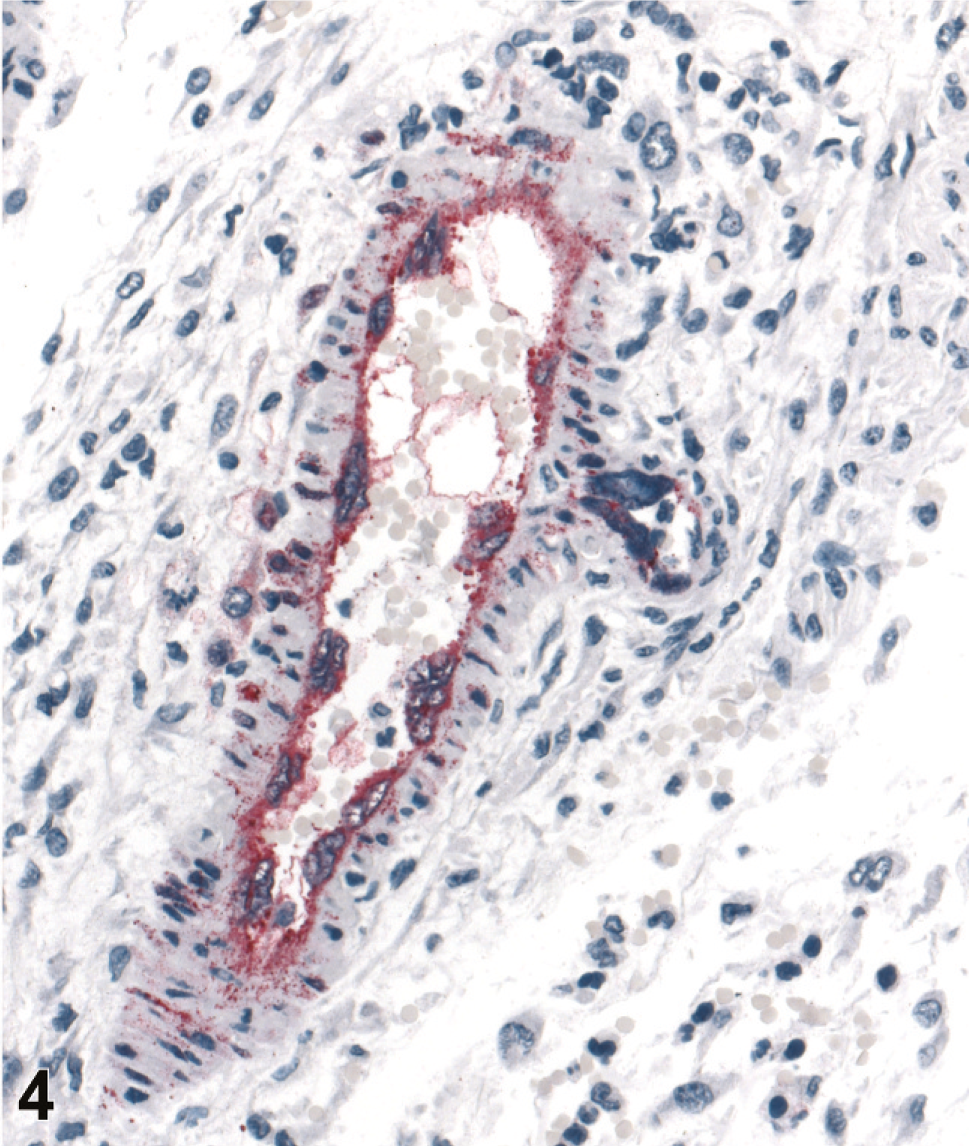
Affected blood vessel in lymph node peri-capsular adipose tissue; guinea pig 4 dpi (guinea pig No. 5). Prominent endothelial syncytia with abundant viral antigen scattered in cytoplasm as well as throughout the vessel wall. IHC. Mayer's hematoxylin counterstain.
Urinary bladders from 15 of the 20 4–8 dpi animals were available for evaluation. Changes were seen in 10 of these 15 animals and were characterized by submucosal edema and submucosal lymphohistiocytic vasculitis with frequent necrosis and ulceration of the overlying transitional epithelium. Lesions ranged from focal inflammation with scattered necrosis and rare syncytial cells to severe lymphohistiocytic and heterophilic inflammation, extensive necrosis, scattered hemorrhage and numerous syncytial cells. Within the transitional epithelium of the mucosa, syncytial cells were frequently observed at the luminal edge of the bladder epithelium and also appeared as sloughed cells within the bladder lumen (Fig. 5A).
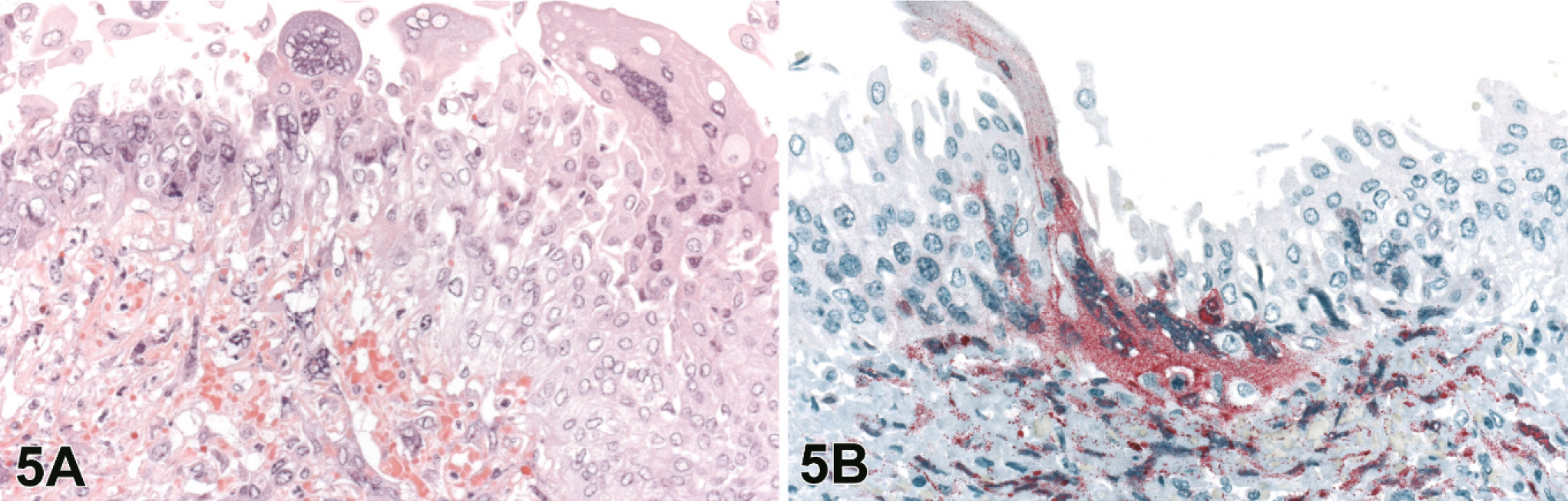
Urinary bladder; guinea pig 6 dpi (guinea pig No. 9).
The ovaries and uterus were evaluated in 8 and 10 of the 20 4–8 dpi animals, respectively. Lesions in the ovaries were generally minimal with focal regions of inflammation and scattered necrosis with rare syncytial cells, although some had severe lymphohistiocytic inflammation with necrosis, hemorrhage, and syncytial cells. Lesions in the ovaries were generally localized to the maturing follicles and corpora lutea, and regions of the ovary with primordial and primary follicles were unaffected (Fig. 6A). Uterine changes consisted of varying degrees of multifocal necrosis of the myo- and endometrium associated with fibrinoid vascular change, endothelial and epithelial syncytial cells, and lymphohistiocytic heterophilic inflammation (Fig. 7A).
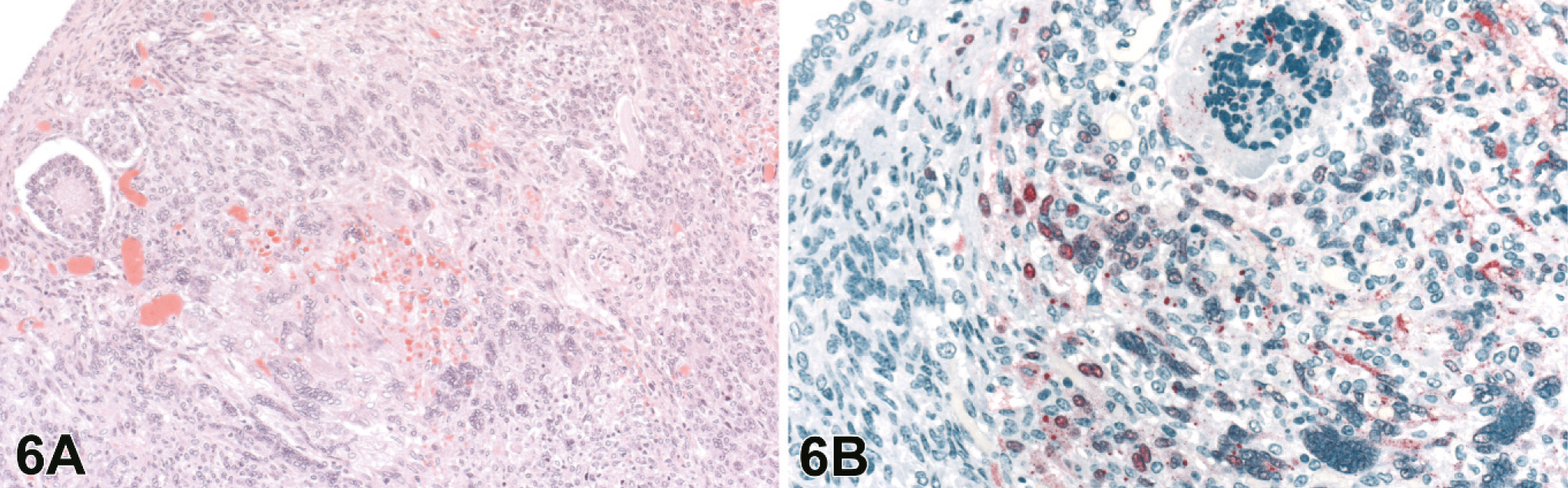
Ovary; guinea pig 7 dpi (guinea pig No. 17).
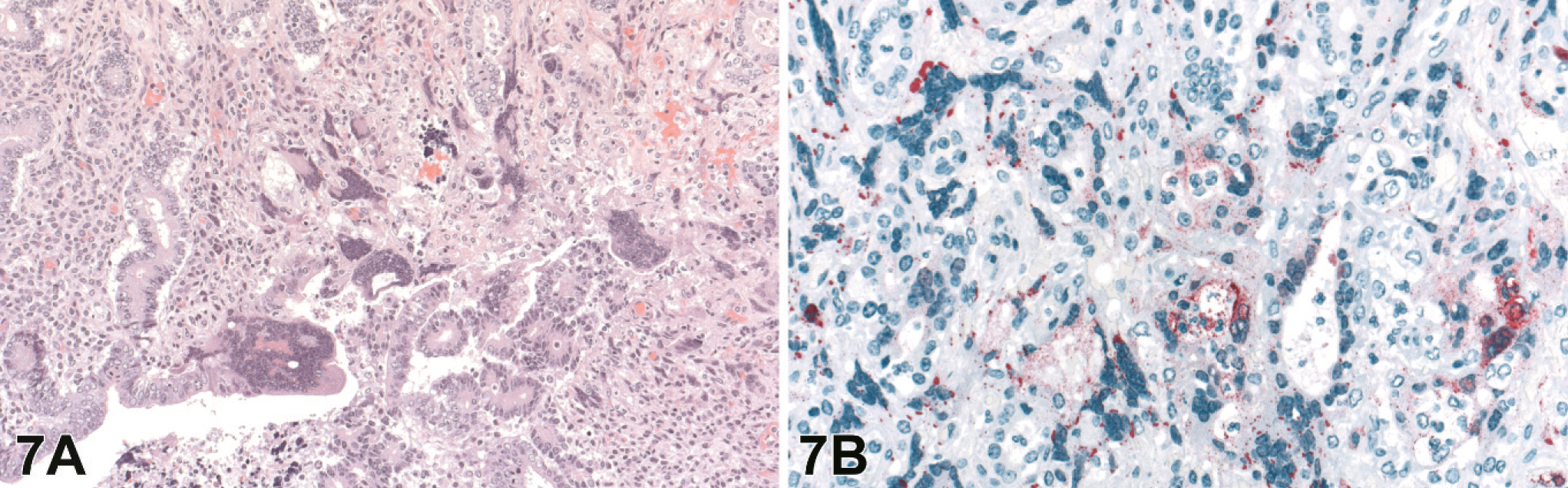
Uterus; guinea pig 7 dpi (guinea pig No. 17).
Brains were evaluated for all 20 of the 4–8 dpi infected animals, and lesions were present in 10. Histopathology and IHC findings are presented in Table 2. There was mild to severe histiocytic and lymphocytic meningitis (Fig. 8) and meningeal vasculitis in all of these and lymphohistiocytic meningoencephalitis with rarefaction of parenchyma associated with the inflammation in some. Animals harvested at the later time points (6–8 dpi) had more severe and widespread lesions than those taken at 4 dpi. In those animals with an encephalitic component, there were scattered intracytoplasmic and intranuclear eosinophilic inclusions within neurons and microglia that were rarely associated with neuronophagia (Figs. 9, 10). These lesions were seen predominantly in the brainstem and rarely in the cortex.
Histopathologic changes (HE) and antigen distribution (IHC) in the brain of animals with CNS involvement (IP inoculation of 1 × 104 PFU NiV).∗

DPI = days postinoculation; Np = neuroparenchyma.
HE = hematoxylin and eosin; - = no abnormal findings; + = mild pathologic changes characterized by mixed mononuclear inflammation; ++ = moderate pathologic changes characterized by mixed mononuclear inflammation; +++ = severe pathologic changes characterized by mixed mononuclear inflammation in addition to neuronal death, rarefaction of neuropil, inclusion bodies, or neuronophagia as pertinent to tissue type.
IHC = immunohistochemistry; - = negative; + = less than 2 positive cells/high-power field (hpf); ++ = 2-10 positive cells/hpf; +++ = greater than 10 positive cells/hpf.
Vasculitis and correlating immunostaining were prominent features in meningitis.
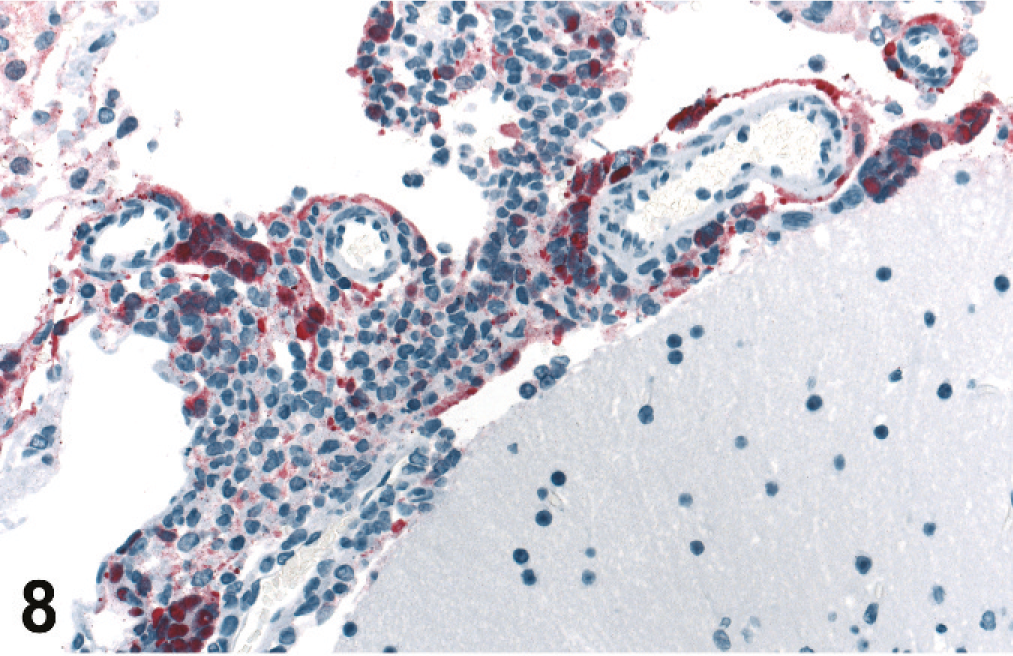
Brain; guinea pig 7 dpi (guinea pig No. 18). Meningitis with prominent viral antigen in inflammatory cells, syncytia, and adventitia of vessels. IHC. Fast Red substrate and Mayer's hematoxylin counterstain.
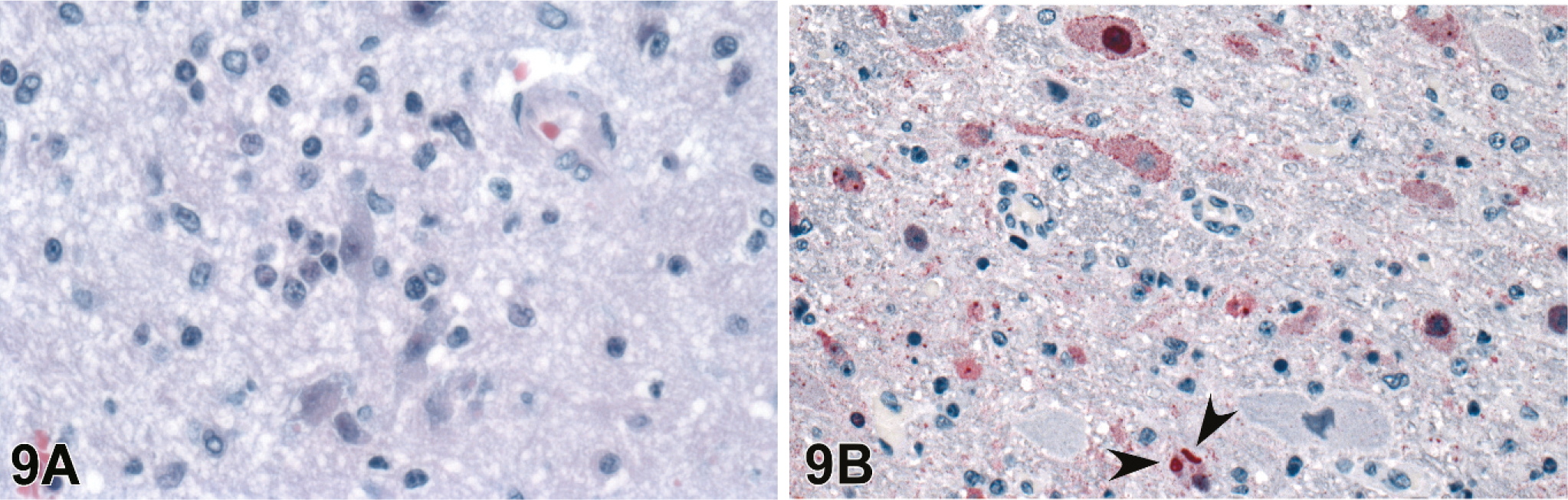
Brain; guinea pig 6 dpi (guinea pig No. 12).
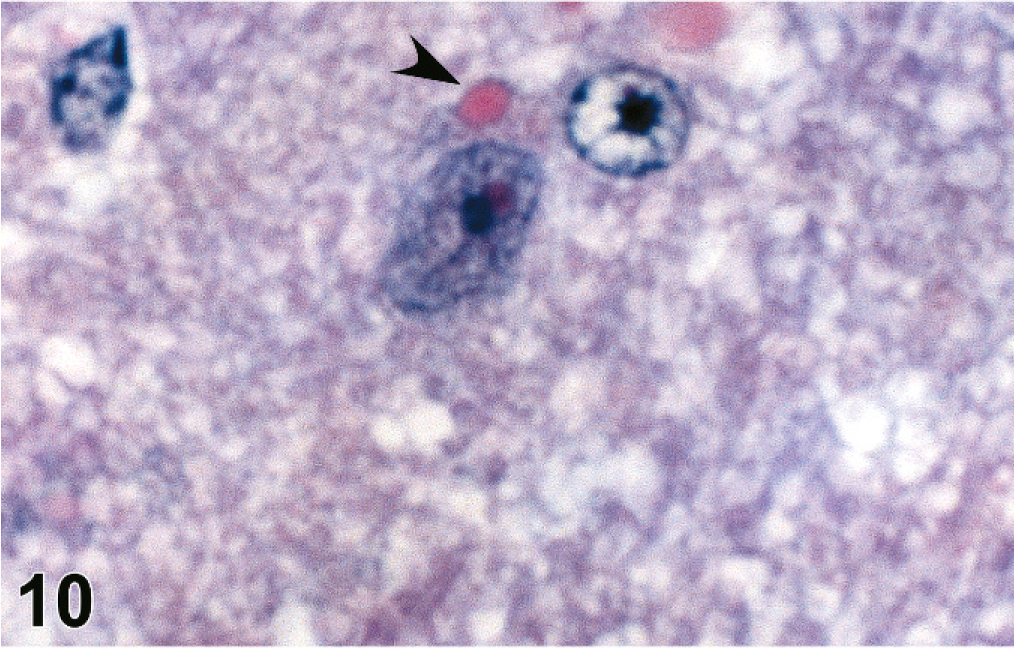
Brain; guinea pig 6 dpi (guinea pig No. 12). Intracytoplasmic eosinophilic viral inclusion (arrowhead). HE.
Histopathologic changes in other organs examined were subtle or minimal. The kidneys had sporadic lymphohistiocytic vasculitis and syncytial cells within the glomerular tufts (Fig. 11A). Lesions in the lungs were primarily a mild diffuse interstitial pneumonia characterized type II pneumocyte hypertrophy and hyperplasia. Only one animal had a small subpleural focus of necrosis. Vasculitis associated with parenchymal necrosis was occasionally seen in the adrenal glands, thymus, thyroid, intestinal mesentery, and small intestine.
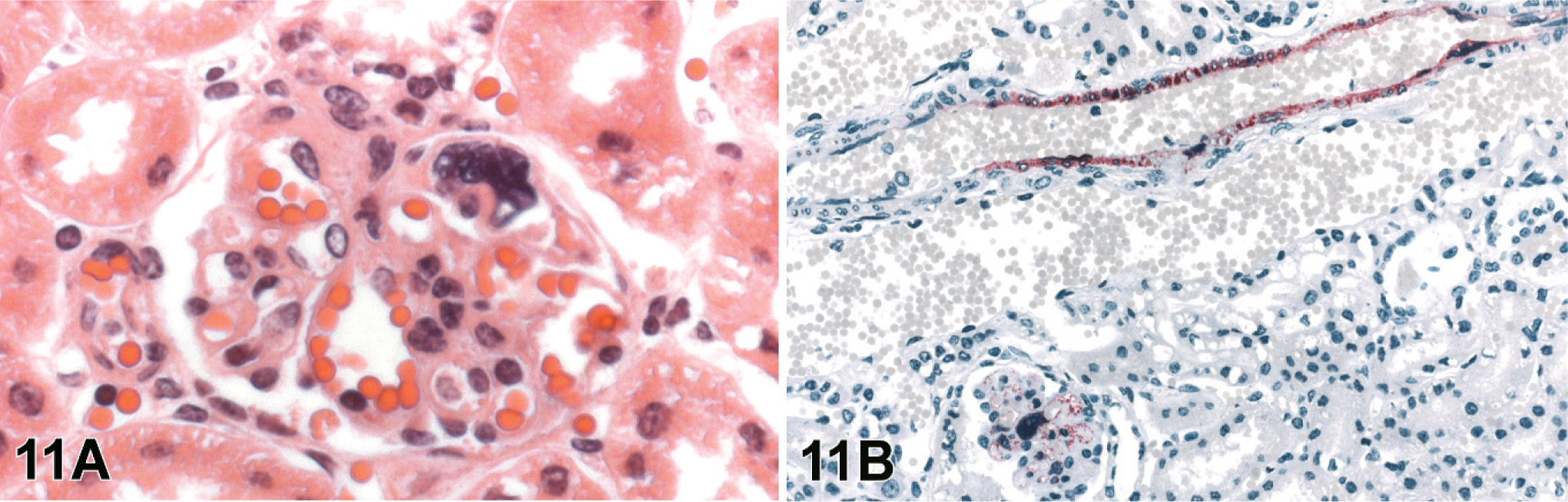
Renal cortex; guinea pig 4 dpi (guinea pig No. 3).
Viral antigen as detected by IHC was present in multiple organs and usually corresponded with degree of histopathologic changes, with greater amounts of staining in foci with more extensive lesions. It was particularly prominent in blood vessels (Fig. 4), both transmurally and within endothelial cells, especially endothelial syncytia. This vascular staining was most notable in lymphoid tissues, uterus, and urinary bladder. Immunopositive mononuclear cells, morphologically consistent with macrophages, were seen occasionally in viable PALS and lymphoid follicles (Figs. 1B, 2). Syncytia in the glomerular tuft (Fig. 11B), endometrial glandular epithelium (Fig. 7B), and urinary bladder transitional epithelium (Fig. 5B) were strongly immunopositive. In the brain, viral antigen was detected in the meninges, neurons, microglial, and scattered ependymal cells (Figs. 8, 9B). Often IHC correlated with neuronal inclusion bodies noted on HE (Fig. 9B). The discrete focus of subpleural necrosis was also positive by IHC, represented by poorly defined and degenerating inflammatory cells, as well as sporadic mesothelial cells in most animals. Immunostaining was rare in gastrointestinal serosal mesothelial cells and parenchyma of the adrenal glands and thymus.
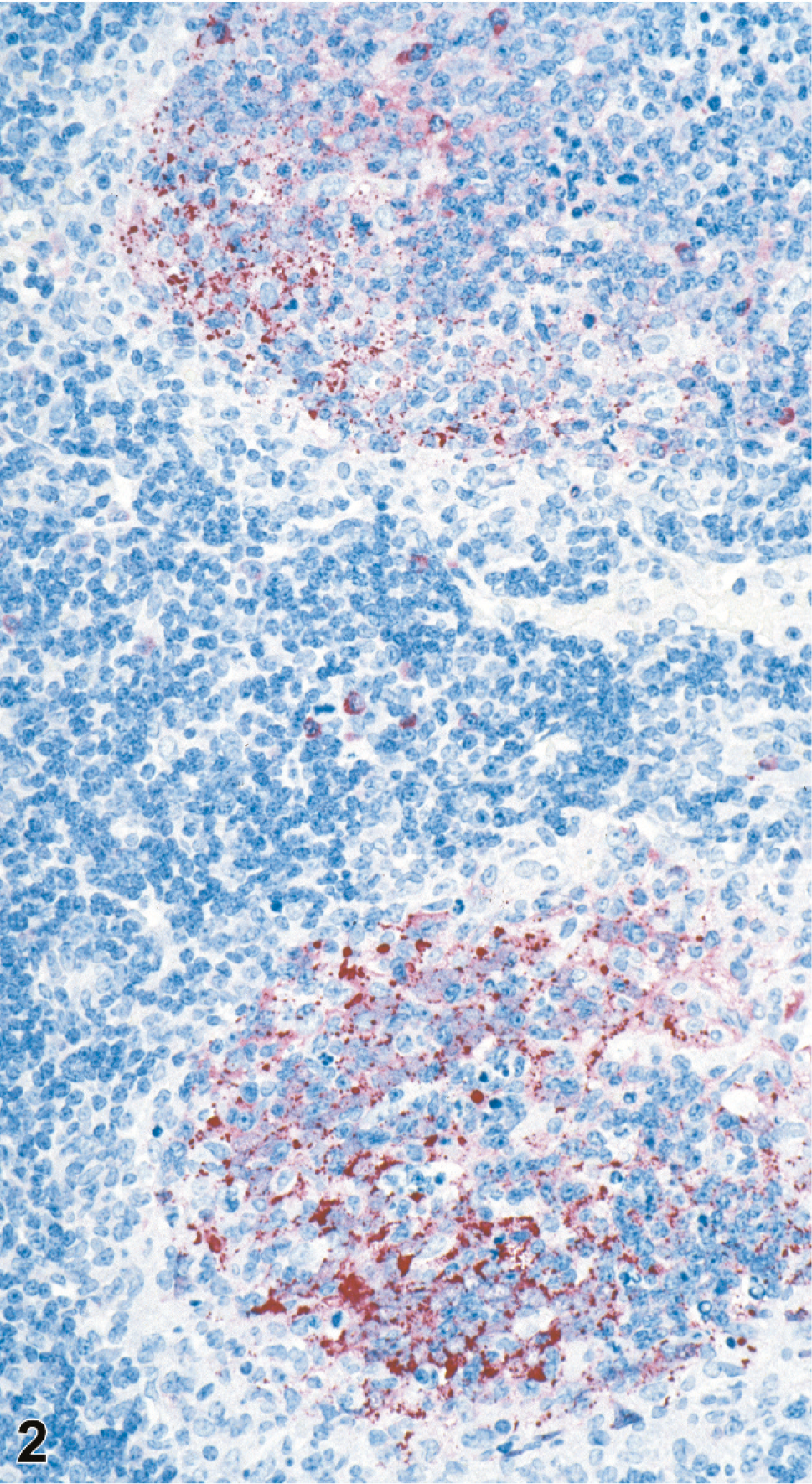
Lymph node; guinea pig 7 dpi (guinea pig No. 14). IHC. Nipah virus antigen present in moderately depleted and necrotic lymphoid follicles. Fast Red substrate and Mayer's hematoxylin counterstain.
Discussion
In this study we examined tissues collected from guinea pigs experimentally infected with NiV. The inoculation of 6 × 104 PFU of NiV given IP produced severe clinical disease, with most animals dying between 4 and 8 dpi. At 2 dpi, there were minimal histologic changes, identified only in an abdominal lymph node (lumbar) that was associated with viral antigen, as detected by IHC staining. Between 4 and 8 dpi, damage was predominantly in the lymph nodes, spleen, urinary bladder, uterus, ovary, and brain. In all tissues, pathologic changes were most severe in blood vessels and were associated with parenchymal coagulative necrosis. There were varying degrees of lymphohistiocytic vasculitis, fibrinoid vascular change, and endothelial syncytia formation. Viral antigen was extensively distributed in a wide variety of cell types and tissues, but blood vessels were consistently immunopositive. Viral inclusions were more prominent in the brain but easily identify with IHC in other affected tissues.
Other attempts to establish experimental animal infections with NiV have included pigs, domestic cats, pigs, small rodents, and bats. In pigs and domestic cats inoculated subcutaneously and oronasally, respectively, the disease was predominantly respiratory in nature. 11, 14 Cats had extensive lymphoid necrosis and prominent syncytial cell formation in vascular endothelium and lymphoid organs and marked epithelial necrosis and inflammation throughout the lung, with numerous syncytial cells. 11, 14 In the pigs, there was a bronchointerstitial pneumonia with syncytial cells in both epithelium and endothelium. In a few of the inoculated cats and pigs, there was neurologic involvement as well, consisting almost exclusively of meningitis, never with any neuroparenchymal involvement in the cats. However, in a subsequent study in which each pig was simultaneously inoculated intranasally, orally, and ocularly, viral antigen was detected in neurons and glial cells. 22 In a paper that describes NiV inoculation in several rodent species, intranasal inoculation of 105 PFU in 2 guinea pigs was unsuccessful in establishing infection. 26 In the same study, Swiss mice did not appear to be susceptible to NiV infection, but hamsters experimentally inoculated IP or intranasally demonstrated systemic disease with predominant involvement of blood vessels, although endothelial syncytia were rare. In these hamsters, which were given a very high dose of virus (107 PFU), damage was most severe in the brain, with relative sparing of spleen and lymph nodes, and mild scattered focal parenchymal pulmonary inflammation. The liver, an organ not affected in other species, had evidence of vasculitis. 26
Gray-headed fruit bats (
In all domestic species studied, NiV has a fairly broad cellular tropism and infects multiple organ systems. Autopsy findings in humans (which by definition are all end-stage disease, and usually after multiple intervention therapies), describe a predominantly neurologic disease, with necrotic plaques in the neural parenchyma being the outcome of vasculitis with mural necrosis and thrombosis. In addition, viral antigen has been detected by IHC in neurons and glial cells of humans succumbing to the infection. 27 In our study, many of the infected guinea pigs had meningoencephalitis and necrosis, which consistently colocalized with NiV antigen by IHC. It is presumed that virus entered the brain through infected vessels, as neuroparenchymal involvement was more extensive in areas adjacent to foci of vascular damage and immunopositivity. Neurons, ependymal cells, and microglia to a lesser extent, all had evidence of viral infection, as determined by IHC. Most extensive and severe neurologic involvement occurred in those guinea pigs surviving the longest, indicating that this is a later phase of disease progression, similar to the situation in humans. Occasionally there were foci of NiV by IHC in the brains of guinea pigs that were unassociated with histologic lesions; this may indicate that the virus causes neuronal cell death directly, rather than through vascular compromise. This may be in contrast to the situation in humans, where brain damage is thought to largely result from extensive vascular damage and avascular necrosis. However, brain histology in humans has been in terminal cases, and there are no reports on the effects of NiV infections effects on the human brain early in the disease.
In humans, infection is thought to take place via the oronasal route. The vascular lesions and pathologic changes are most severe in the brain, with spleen and lung (vasculitis, fibrinoid necrosis, and syncytial cells in airways) also prominently affected. 27 A possible explanation for this NiV tropism can be postulated based on in vitro studies of NiV receptor affinity, which highlight the EphrinB2 receptor as a key factor in NiV binding and cell entry. 2, 17 EphrinB2 receptor is widely expressed in humans, particularly in endothelial cells, smooth muscle cells of vessels, bronchial epithelium, and neurons; consequently, general vascular distribution and prominent involvement of lungs and brain are expected. 9 The EphrinB2 receptor is highly conserved in mammals; therefore, lesion distribution in susceptible animal species might be anticipated to follow the same pattern as in humans. In swine and cats, the disease is predominantly respiratory, with some involvement of brain. In hamsters, blood vessels, and brain were most affected, with some pulmonary involvement. In the guinea pigs in this study, spleen and lymph nodes were the most severely affected organs, brain less so, and lung only minimally, which follows the Ephrin B2 receptor predictions to some degree as well. Relative sparing of lung in the guinea pig may be a result of inoculation route. There was a focus of pulmonary necrosis in 1 guinea pig and sporadic immunopositive mesothelial cells in the pleura of most animals. However, this viral uptake by pleural mesothelium could be attributed to virus inoculum (which was delivered IP) spreading from adjacent diaphragmatic and intercostal lymphatics associated with the peritoneal cavity drainage system. 1, 8
We did, however, find a surprising departure in the guinea pigs in this study compared with findings in other domestic animals and humans, in the extent of viral immunostaining and damage in the urogenital system. The urinary bladder and uterus were both severely affected, with extensive inflammation and ulceration of the mucosa, usually with inflamed and virus-laden blood vessels in the adjacent submucosa. IHC positivity was intense in the epithelial cells, where many had formed syncytia containing abundant viral antigen. There was often evidence of infected endometrium and urothelium sloughing off into luminal cavity, a potential mechanism for viral shedding. Comparable antigen detection in uterus and bladder has been previously reported in a study using a small number of guinea pigs as NiV infection controls, and in experimental infections of guinea pigs with the closely related Hendra virus. 13, 23, 24 Bat urine or placental fluids are presumed to be a vehicle for natural transmission of Henipaviruses, although the exact cellular source in these species has not been elucidated for NiV. Replication in bladder and uterus has implications for transmission in nature, and our findings in guinea pigs provide the tools to further explore possible mechanism of viral shedding into the environment. Also, in the guinea pigs in this study, the ovaries were affected dramatically, with growing follicles and corpora lutea extensively damaged and relative sparing of primordial follicles. Studies with the related Hendra virus have shown strong affinity of the virus for not only the uterus but also the ovaries of guinea pigs. 24 The possible role that infected ovarian tissue might play in transmission is obscure, and the ovaries may be infected because of some hormonally dependent effect that also predisposes the uterine epithelium to infection; or it may simply be an artefactual effect as a result of the extensive vascularity of the ovary.
In summary, NiV infection can be reliably established in guinea pigs by IP inoculation, and the pathogenesis appears to involve early replication in the mononuclear phagocytic system followed by dissemination through affected and infected vasculature. Organs with the most damage include spleen, urinary bladder, female reproductive tract, and brain. There are many aspects that share similarities with the disease in humans and the tropism for urinary bladder and uterine epithelium could provide the means for further exploration of natural transmission.
Footnotes
Acknowledgements
We gratefully acknowledge Mr. Mitesh Patel for the outstanding graphics technical assistance in this manuscript. This study was funded in part by NIH K08 grant (No. AI060629-02).
