Abstract
Reports of thyroid gland neoplasms in guinea pigs (
The guinea pig is a popular companion animal species that is also maintained in many zoological collections. More than 1 million pet guinea pigs (
Materials and Methods
Case Selection
A retrospective search of case files at Northwest ZooPath between 1998 and 2008 was performed. Guinea pig cases with grossly enlarged thyroid glands were included in the study. Patient data included age, sex, radiographic findings, blood thyroxine concentration, and whether kept as part of a zoological collection or as a pet. Concurrent pathological conditions present in the guinea pigs with thyroid neoplasms were also included. Neoplasms were excised during surgery or discovered at necropsy and fixed in 10% neutral-buffered formalin.
Gross Pathology
Enlarged thyroid glands were described by color, greatest dimension, and location. Additional gross pathological findings that were described by the submitting clinician or pathologist were also listed for each case.
Histopathology
Tissues obtained were preserved in 10% neutral-buffered formalin for up to 90 days prior to being processed routinely. Paraffin-embedded tissues were sectioned at 5 μm, mounted on frosted glass slides, stained with hematoxylin and eosin (HE), and examined by light microscopy. Thyroid neoplasms were classified as benign or malignant according to the most current World Health Organization (WHO) classification of thyroid neoplasms of domestic animals. 16 Neoplasms were classified as malignant when neoplastic cells unequivocally penetrated through the full thickness of the affected capsule and/or vessels. Concurrent histologic lesions in other organs were also listed for each case.
Immunohistochemistry
A thyroid immunohistochemistry (IHC) panel that included antibodies against thyroglobulin, thyroid transcription factor 1 (TTF-1), parathyroid hormone, and calcitonin was performed on neoplastic tissue from all cases. Deparaffinization, antigen retrieval, immunohistochemical labeling, and counterstaining were performed on the BOND-MAX Automated Staining System (Vision BioSystems; Leica, Bannockburn, Illinois) using the Bond Polymer Detection System (Vision BioSystems; Leica) or on the Benchmark Automated Staining system (Ventana Medical Systems, Inc, Tucson, Arizona) using an enhanced alkaline phosphatase V-Red Detection System (Ventana Medical Systems, Inc). Primary antibodies and their dilutions, source, host, type of antigen retrieval, and chromogen are listed in Table 1. Thyroid tissue from guinea pigs and dogs was used a positive control tissue. For negative controls, the primary antibodies were replaced with homologous nonimmune sera. Cytoplasmic labeling within neoplastic cells was evaluated as positive for thyroglobulin, parathyroid hormone, and calcitonin; nuclear labeling was evaluated as positive for TTF-1.
Primary antibodies used for immunohistochemistry.
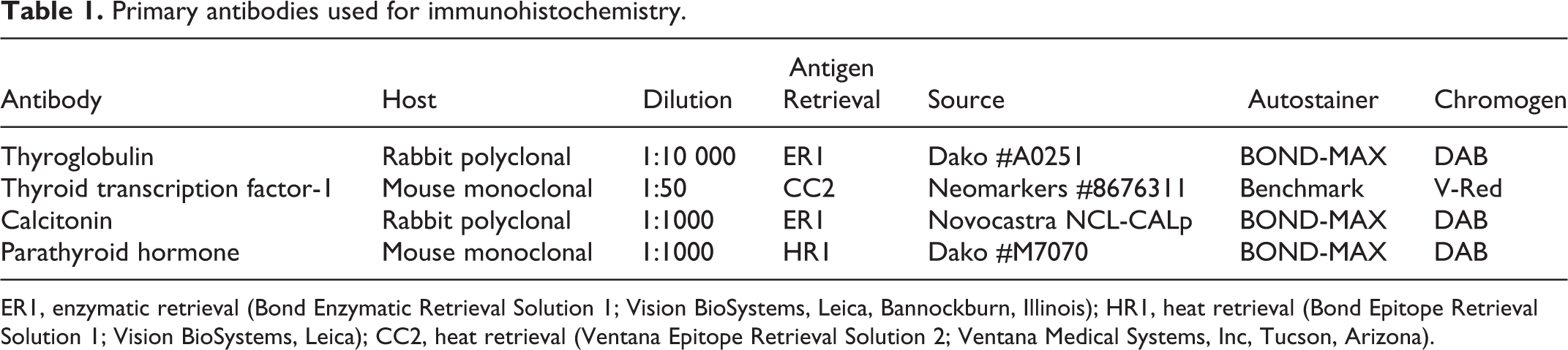
ER1, enzymatic retrieval (Bond Enzymatic Retrieval Solution 1; Vision BioSystems, Leica, Bannockburn, Illinois); HR1, heat retrieval (Bond Epitope Retrieval Solution 1; Vision BioSystems, Leica); CC2, heat retrieval (Ventana Epitope Retrieval Solution 2; Ventana Medical Systems, Inc, Tucson, Arizona).
Results
Epidemiological and Clinical Findings
Of the 526 guinea pig cases submitted to Northwest ZooPath from 1998 to 2008, 236 cases had neoplasms and 19 (3.6% of guinea pig cases) had thyroid neoplasms. Only lipomas, trichoepitheliomas, lymphoid neoplasms, and mammary gland neoplasms were more common than thyroid neoplasms. An additional enlarged thyroid gland-like mass was submitted but not included in this report because it was too autolyzed for classification. The median age of guinea pigs with thyroid neoplasms was 4.3 years; ages ranged from 2.5 to 6 years (Table 2). Seven of these were male and 10 were female; sex was not reported for 2 cases. Seven cases came from 3 different zoological collections (case Nos. 1, 3, 9, 11, 13, 16, and 18), and the other 12 were companion animals from 9 different veterinary hospitals located throughout the United States. Blood thyroxine (T4) concentration was reported by the submitting clinician to be normal 3 months before death in 1 case (case No. 10; T4 = 1.9 μg/dL, no reference range reported) and elevated shortly before death in another (case No. 17; T4 = 5.9 μg/dl vs T4 = 2.0 μg/dl in a concurrently tested sample from a young unaffected conspecific). Seven of the thyroid neoplasms were collected as an excisional biopsy during surgery (case Nos. 2, 5, 7, 12, 15, 18, and 19), and the other 12 were collected at necropsy. One of the necropsy cases was submitted to Northwest ZooPath as a fresh, whole carcass (case No. 8), and the other necropsies were performed by clinicians who submitted selected tissues. Thyroid neoplasm was listed as the clinical diagnosis in 2 cases (case Nos. 7 and 12). The most common clinical findings reported by submitting clinicians included palpable subcutaneous mass(es) on the ventral neck (case Nos. 2, 3, 5, 6, 8, 12, 14, 15, 17, and 18) and progressive weight loss (case Nos. 1, 2, 4, 10–13, 16, and 19). Three of the 7 guinea pigs from which neoplasms were removed as an excisional biopsy died within a few days after the surgery (case Nos. 7, 18, and 19), and postoperative information is not available for the other 4 cases. Two cases (case Nos. 2 and 6) had radiographic evidence of small mineral densities within the mass (Fig. 1), and in 1 case (case No. 16), radiographs of the patient were interpreted to be “within normal limits” but were not available for further review.
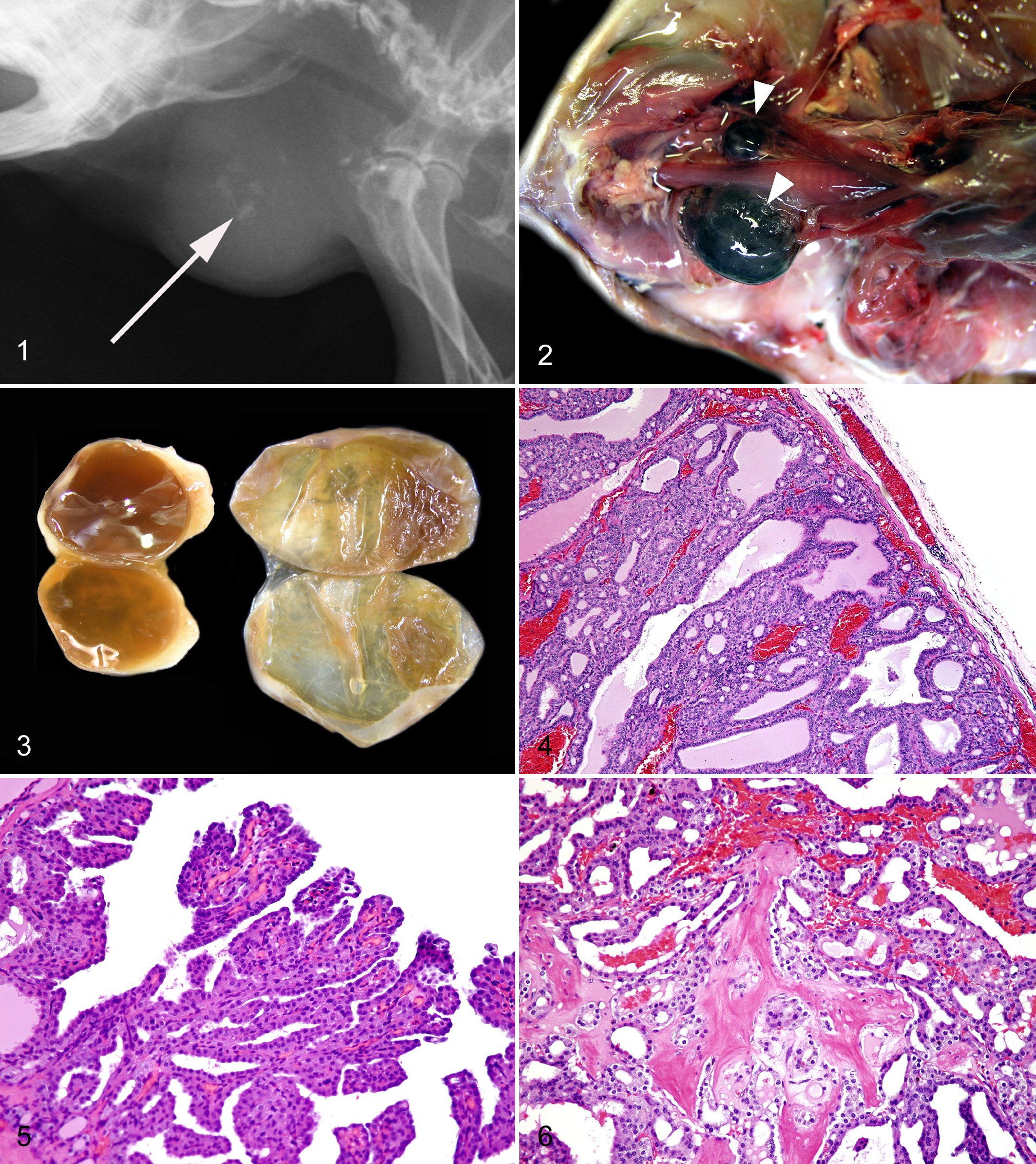
Radiographic projection; guinea pig. Radiographic appearance of follicular-compact thyroid carcinoma; note the small radio-opaque foci of osseous metaplasia (arrow) within the cervical soft tissue mass.
Histologic Classification, Signalment, Presence of Osseous Metaplasia, and Notable Clinical Findings in Guinea Pig Thyroid Neoplasm Cases Submitted to Northwest ZooPath Between 1998 and 2008.
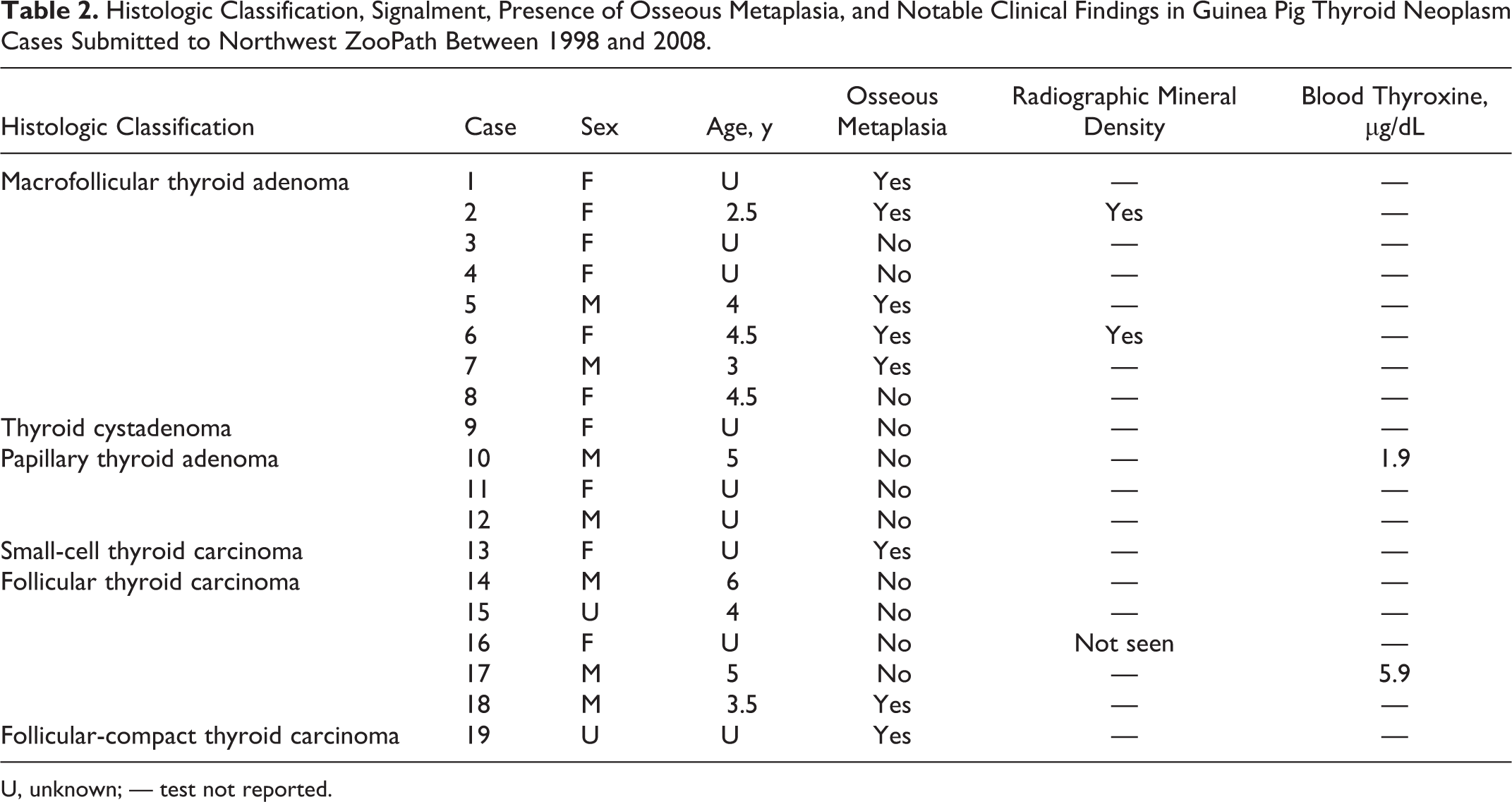
U, unknown; — test not reported.
Gross Pathological Findings
Gross pathological findings in the thyroid were reported for 8 of the 19 cases. The most commonly reported finding was a cystic mass, and all 5 of these were characterized as either black or a dark color (Fig. 2). Size was reported in a few of the cases and ranged from 1 to 2.5 cm in the largest dimension. Enlarged glands were unilateral in all cases except case No. 17 (Figs. 2–3). Gross necropsy findings in addition to an enlarged thyroid gland were reported in 10 of the 19 cases. The most common additional gross pathological finding was subjectively enlarged adrenal glands (case Nos. 3, 10, 11, and 14), although adrenal gland measurements were not reported by any of the submitting clinicians. Liver cysts were reported in 3 cases (case Nos. 1, 10, and 16). Thirty-four other gross postmortem abnormalities were reported, none of which was described in more than 2 cases.
Immunohistochemical Findings
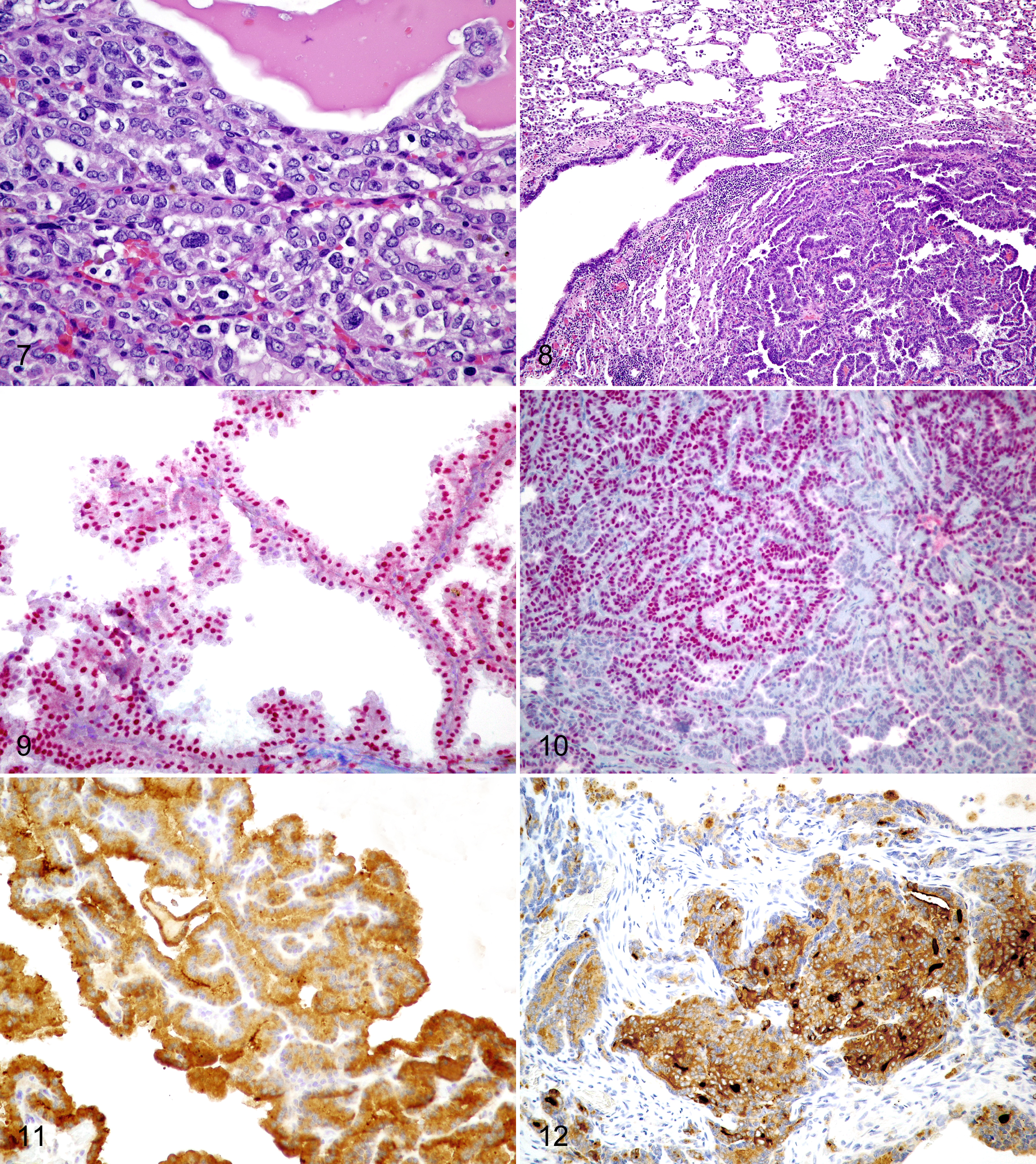
All 19 neoplasms were positive for TTF-1 and thyroglobulin but negative for parathyroid hormone and calcitonin, thereby confirming a thyroid follicular cell origin. All adenomas had diffuse, strong labeling of the nuclei for TTF-1 (Fig. 9) and diffuse, strong labeling of the cytoplasm for thyroglobulin (Fig. 11). Labeling was more variable in thyroid carcinomas, with 50% to 60% of cells expressing strong nuclear labeling for TTF-1. The remaining neoplastic cells had weak TTF-1 expression or negative nuclei (Fig. 10). Commonly, decreased labeling was observed in focal areas, but there was no underlying morphologic difference between TTF-1-positive and TTF-1-negative neoplastic cells. Thyroglobulin was strongly expressed by 90% of the neoplastic cells in thyroid carcinomas. Both TTF-1 and thyroglobulin were detected in the vast majority of neoplastic cells at metastatic sites (Fig. 12).
Histopathological Findings
Seven thyroid follicular neoplasms had microscopic features of malignancy; the other 12 neoplasms were diagnosed as adenomas. Malignant neoplasms were present in 3 of the 7 males and 2 of the 10 females. Histologically, benign neoplasms (case Nos. 1–12; Figs. 4–6, 9, 11) were encapsulated and associated with displacement or compression of surrounding tissues. These thyroid adenomas had follicular, papillary, and cyst patterns lined by 1 or 2 layers of cuboidal epithelial cells that had minimal anisokaryosis and variability in nuclear polarity. Mitoses were rare (approximately 1 per 10 high-power fields [hpf]), and capsular invasion was not detected. These benign neoplasms were further subclassified as macrofollicular thyroid adenoma (case Nos. 1–8), thyroid cystadenoma (case No. 9), and papillary thyroid adenoma (case Nos. 10–12) if follicular, cystic, or papillary patterns predominated, respectively (Table 2). Malignant neoplasms (case Nos. 13–19; Figs. 7, 8, 10, 12) generally had a more compact or solid cellular pattern of cuboidal to elongated follicular epithelial cells with moderate to marked anisokaryosis, occasional mitotic figures (1–3 per 10 hpf), and multifocal capsular invasion. These neoplasms were further characterized as small-cell thyroid carcinoma (case No. 13), follicular thyroid carcinoma (case Nos. 14–18), and follicular compact thyroid carcinoma (case No. 19) based on degree of follicular differentiation, respectively. 16 No vascular invasion was noted for any malignant neoplasm, although in 1 case, the neoplasm had metastasized to the lungs (Fig. 8). Foci of osseous metaplasia were noted in 5 benign (case Nos. 1, 2 5, 6, and 7) and 3 malignant neoplasms (case Nos. 13, 18, and 19; Figs. 8–12; Table 2). For benign and malignant neoplasms, foci of osseous metaplasia within the neoplasm were well differentiated, and for 1 malignant neoplasm, the metaplasic focus also contained medullary hematopoietic precursors representing all cell lines and with orderly maturation, as well as predominance of myeloid cells (case No. 18). Osseous metaplasia was more prominent in the benign neoplasms than in the malignant neoplasms. All neoplasms produced colloid-like proteinaceous fluid within cystic or follicular lumina, although generally, follicles were less well defined and fluid production was less prominent in the malignant neoplasms.
Thyroid; guinea pig. Follicular thyroid carcinoma; note cellular pleomorphism. Hematoxylin and eosin (HE).
Sixty-five histopathological diagnoses were found in addition to thyroid neoplasia in the 13 cases in which multiple tissues were available. Systems affected by co-conditions included hepatobiliary (case Nos. 1, 3, 4, 6–8, 10, 11, 13, 14, 16, and 18), respiratory (case Nos. 3, 4, 6–8, 11, 13, 14, 17, and 18), urinary (case Nos. 1, 3, 4, 8, 11, 13, 16, 17, and 18), cardiovascular (case Nos. 1, 3, 4, 6, 7, 11, 13, and 18), hematopoietic (case Nos. 4, 10, 11, 14, and 15), reproductive (case Nos. 8, 13, and 16), gastrointestinal (case Nos. 11, 16, and 17), and endocrine (case Nos. 11, 13, and 16). The most common co-condition was pulmonary congestion (case Nos. 3, 6, 8, 11, 13, 14, and 18), followed by myocardial degeneration (case Nos. 1, 3, 7, 11, and 13), nephrosclerosis (case Nos. 4, 8, 13, 16, and 17), and atrophy of fat (case Nos. 9, 11, and 17–19). Eight cases (case Nos. 1, 3, 6, 8, 11, 13, 14, and 18) had postmortem lesions consistent with congestive heart disease (ie, pulmonary, hepatic, and other tissue congestion). Nine cases (case Nos. 1, 4, 8, 10, 11, 14, 15, 17, and 18) had an array of inflammatory diseases. Concurrent neoplastic diseases included pulmonary adenoma (case Nos. 6 and 8), uterine leiomyoma (case No. 16), transitional cell carcinoma in situ (case No. 1), pulmonary bronchoalveolar adenocarcinoma (case No. 14), malignant pancreatic islet cell neoplasm (case No. 11), and duodenal leiomyosarcoma (case No. 16).
Discussion
Thyroid neoplasms were more common in the guinea pig cases submitted to Northwest ZooPath between 1998 and 2008 (19 cases) than would be expected by the number of previously reported cases (5 cases).17,18,26,38 The relative prevalence of thyroid neoplasms in the cases reported here could be in part because the sample group is composed of zoological specimens and privately owned pets rather than laboratory animals. This relative prevalence was not reflected in a report of 20 spontaneous neoplasms in pet guinea pigs at a university pathology service in the Czech Republic, which did not include a single thyroid neoplasm. 13 It is possible that thyroid neoplasms are present in that population, but the number of guinea pig submissions could have been too small to detect neoplasms with a prevalence that is much less than the neoplasms that were included. It is also possible that the true prevalence of thyroid neoplasms is greater in North American companion and zoo guinea pigs than it is in those of the Czech Republic.
The age of the guinea pigs in this study may also explain in part the large number of thyroid neoplasms. The companion and zoo population in North America could have a greater average life span than the populations described in previous reports of guinea pig spontaneous neoplasm relative prevalence.11,13,15,31 Expected life span of guinea pigs is not clearly defined and probably differs with breed and environmental conditions. Some authors report a 4- to 5-year expected life span with maximum of 8 years, whereas others report a 5- to 6-year expected life span and a maximum life span of 12 years.8,12,24,30,32 The median age of guinea pigs with thyroid neoplasms in this study was 4.3 years, and the maximum age was 6 years. This is consistent with previous reports that neoplasia occurs after 1 year but usually not before.13,27,36
Degenerative diseases are common in aged guinea pigs, 27 and thyroid disease may or may not contribute to the progression of these diseases. In dogs, nonfunctional thyroid follicular carcinomas are the most commonly diagnosed thyroid neoplasm, and the concurrent degenerative, neoplastic, and inflammatory diseases such as those found in this study do not usually occur. Conversely, functional nodular hyperplasia is the most common lesion in cats, 2 and feline hyperthyroidism is associated with hypertension, heart disease, hepatic disease, neoplasia, inflammatory bowel disease, inflammatory pulmonary disease, diabetes mellitus, central nervous system disease (including seizures), renal polyuria/polydipsia, muscle weakness, and predisposition to arterial thromboembolism.1,9,19,25,28,29,33,35 The pathophysiology of hyperthyroidism-induced disease may involve changes in tissue oxygen and glucose consumption, altered microvascular blood flow, and thyroid hormone interference with neurotransmitters. 25
Eight of the 19 guinea pig cases described in this study had histopathologic abnormalities of the myocardium, and 8 had lesions consistent with congestive heart disease (ie, pulmonary, hepatic, and other tissue congestion). By comparison, 70 of the 257 cases submitted to Northwest ZooPath with ≥4 tissues during this 10-year period had similar heart disease (unpublished data). Hyperthyroidism can induce heart disease in cats and could contribute to the formation of cardiac lesions in guinea pigs. The pathophysiology of hyperthyroidism-associated heart disease in cats includes both direct and indirect thyroid hormone effects. Hyperthyroidism is associated with increases in myocardial contractility, mitochondrial protein synthesis, sarcoplasmic reticulum calcium cycling, diastolic depolarization rate, and decreases in sinoatrial nodal cell action potential duration. 1 These effects combine to enhance systolic performance, improve diastolic function, and increase heart rate. Indirect cardiovascular effects of thyroid hormone include an increased number of β1-adrenergic receptors and increased basal metabolic rate, which lead to reduced systemic vascular resistance and increased cardiovascular volume. 1 The hypertension that occurs with hyperthyroidism in cats is somewhat paradoxical. The decrease in systemic vascular resistance is countered by increased cardiac output, activation of the renin-angiotensin-aldosterone system, and increased sympathetic tone, which results in hypertension. 1 The net effect of hyperthyroidism-associated changes is an increase in cardiac oxygen requirements with decreased myocardial oxygen delivery, which leads to progressive myocardial cell death, fibrosis, and failure. 1 The clinical manifestations of hyperthyroidism-associated cardiovascular disease in cats include bounding femoral pulses; strong, apical cardiac impulse; heart murmur (high-flow or mitral valve regurgitation); gallop heart beats; tachyarrhythmias; systemic hypertension; diastolic and systolic dysfunction; and congestive heart failure manifested by pleural effusion and/or pulmonary edema. 1 Hyperthyroidism also induces an increase in the number of β-adrenergic receptors in the hearts of cats, which enhances the cardiac response to catecholamines. 29 The potential for these clinical signs should be assessed in clinical cases of suspected thyroid disease in guinea pigs.
Seven of the 19 guinea pig cases described in this study had histopathologic evidence of renal disease compared with 25 of the 257 cases submitted to Northwest ZooPath with ≥4 tissues during the same 10-year period (unpublished data). Hyperthyroidism occurs concurrently with chronic renal failure in cats, although it is unclear whether this is because both diseases are prevalent or because hyperthyroidism contributes to the pathogenesis of renal disease. Renal failure does become evident in many cats that have been treated for hyperthyroidism. 10 The proposed pathogenesis for thyroid hormone–induced renal disease is primarily based on the cardiovascular effects of thyroid hormone that are described above. Increased cardiac output and hypertension could lead to renal damage, and glomerular filtration rate (GFR) can increase due to the loss of renal autoregulation that occurs with thyrotoxicosis. 10 It is interesting that this increase in GFR can lead to decreased serum creatinine together with an increase in blood urea nitrogen that results from thyrotoxic muscle catabolism. 10 A hyperthyroid-associated increase in GFR can mask underlying renal insufficiency, and treatment of feline hyperthyroidism will unmask the condition. 10 Renal disease could be associated with thyroid disease in guinea pigs, so those with clinical signs of hyperthyroidism should be evaluated for renal insufficiency, particularly if treated for hyperthyroidism.
Information about blood thyroxine (T4) concentration was provided by the submitting clinicians in 2 of the 19 submitted cases. The submitting clinician in case No. 10 reported that T4 was normal in blood collected 3 months before death, and the concentration (1.9 μg/dl) was less than the published reference range (2.3–3.5 μg/dl) available at that time. 3 Case no. 17 had a T4 concentration (5.9 μg/dl) that was greater than 2 times the blood level of an unaffected young guinea pig submitted for testing at the same time (2.0 μg/dl). Case no. 10 also had progressive weight loss and a liver cyst, and case No. 17 had nephrosclerosis and atrophy of fat. It is not possible to determine whether the neoplasms in this report were functional without more thorough testing of thyroid hormones.
Antemortem diagnosis of hyperthyroidism is often not possible with a single measurement of blood T4 concentration. The relationship between blood concentrations of thyroid hormones and thyroid disease is not always clear. Laboratory methods for measurement of thyroid hormones in dogs and cats require rigorous protocols for sampling, sample handling, and testing, so differences among values reported by different laboratories could be a result of different protocols. 14 Reference ranges for T4, free T4, triiodothyronine (T3), and free T3 have been reported for guinea pigs, and the range of concentrations found in healthy guinea pigs differs between reports.3,23 In addition to interlaboratory variability, circulating blood thyroid hormone concentrations are affected by nonthyroidal factors, including medications and chronic illness (ie, euthyroid sick syndrome). Many of the animals reported here had chronic illness, so blood thyroid hormone concentrations could have been falsely depressed, which would have added to the difficulty in making an antemortem diagnosis of hyperthyroidism. Free T4 (by equilibrium dialysis) concentration is measured in dogs and cats with suspected thyroid disease because it is less affected by nonthyroidal factors than T4 and T3. A T3 suppression test or thyrotropin-releasing hormone test may be used to confirm a diagnosis of suspected thyroid disease in dog and cat cases with normal T4 and T3. 14 Nuclear scintigraphy, which is also used to help diagnose thyroid disease in dogs and cats, was used to confirm a diagnosis of suspected hyperthyroidism in a guinea pig with clinical signs of hyperthyroidism, T4 within reference ranges, and mildly elevated free T4.20,22
Osseous metaplasia was present in 8 of the 19 thyroid neoplasms, including the 2 cases in which the submitting clinician reported radiographic mineral densities (Table 2). This suggests the possibility that plain radiography could be useful to help determine whether a ventral cervical mass is a thyroid neoplasm or other mass (eg, abscess). Radiography, however, is probably not useful to differentiate malignant from benign neoplasms because osseous metaplasia was present in both malignant and benign neoplasms in this study. This differs somewhat from thyroid neoplasm mineralization in humans, where ultrasonagraphic evidence of thyroid neoplasm mineralization has been associated with greater risk of metastasis to regional lymph nodes. 7
Even though palpable masses were identified in the ventral neck of 11 of the 19 cases, thyroid disease was listed as the working diagnosis in only 2 cases. Veterinarians who work with guinea pigs may not suspect thyroid disease because it is newly described in veterinary literature that is written in the English language, even though it is reported to be relatively common in Germany.5,20–22 It follows, therefore, that the clinicians who submitted the cases in this study did not report that any treatment for thyroid disease had been attempted prior to death or thyroidectomy.
Treatment could include surgical thyroidectomy, percutaneous ethanol injection, radioactive iodine (I-131) therapy, external beam radiation therapy, and methimazole therapy.2,22 Some authors have recommended surgical thyroidectomy for the treatment of hyperthyroidism in guinea pigs, but this may be particularly risky because at least 3 of the 7 excisional biopsy patients in this study died within a few days after surgery. These deaths could have been a result of hemorrhage because the blood supply to the thyroid glands of guinea pigs is highly variable or because removal of functional neoplasms might unmask underlying disease.4,17,37 Surgical procedures were unilateral, and no parathyroid gland tissue was found in the samples that were submitted for histopathology, so it is unlikely that death resulted from complications associated with parathyroid gland removal. To reduce the risk of perioperative complications, patients with hyperthyroidism should be evaluated for concurrent illness, including heart disease and renal disease. Treatment with oral methimazole is recommended for cats and may be useful to determine whether underlying renal insufficiency will be unmasked when hyperthyroidism is controlled in guinea pigs. 22
In summary, thyroid neoplasms were found in 19 of the zoo and pet guinea pig cases submitted to Northwest ZooPath between 1998 and 2008. Most of the thyroid neoplasms were identified as palpable masses on the ventral neck, one-third were malignant, and many contained osseous metaplasia that may be evident radiographically. The neoplasms could be functional, and an array of degenerative diseases, including cardiomyopathies and nephropathies, occur concurrently in guinea pigs with thyroid neoplasms. Research is needed to improve antemortem case management and to determine whether thyroid disease plays a role in the pathogenesis of chronic degenerative diseases in guinea pigs.
Footnotes
Acknowledgements
We thank Dr Jörg Mayer for his assistance interpreting literature written in German, Dr Richard Nye for the radiographic image, Dr Adolf Maas for providing additional clinical data, and Roy Brown of Histology Consultation Services for excellent slide preparation. We also thank the staff of Northwest ZooPath, including Jamie Kinion and Sue Hutton for data retrieval and Christie Buie for photo editing and plate layout.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
