Abstract
Amyloid precursor protein (APP) immunocytochemistry was used to study axonal and neuronal changes in guinea pig brains exposed to tunicamycin. Substantial axonal injury was found in ischemic-hypoxic foci and more generally, but this injury was not readily appreciable in conventionally stained sections. Neuronal perikaryal APP expression was also widely distributed, possibly as an acute phase response to this neurotoxin.
Keywords
Corynetoxins are responsible for a severe neurologic disorder of grazing livestock in southern Australia. The corynetoxin-producing
The fundamental morphologic change in brains exposed to tunicamycin appears to be microvascular damage with capillary obstruction, leading to impaired local perfusion. The resultant ischemia causes a spectrum of injury ranging from selective neuronal damage to focal parenchymal necrosis. 3 4 We used sensitive amyloid precursor protein (APP) immunocytochemistry to more fully investigate the range and distribution of axonal and neuronal changes in tunicamycin-treated brains.
Studies were conducted on 8–10-week-old guinea pigs because tunicamycin produces cerebral lesions in this species that closely resemble those found in sheep. 3 Tunicamycin-treated guinea pigs were euthanatized at 48 hours (6 animals) and 72 hours (6 animals) postinjection. Six control guinea pigs were injected at time 0 with a similar volume of physiologic saline, and 3 of these control animals were euthanatized at each time interval. Tunicamycin (Sigma Chemical Co.) was prepared for injection according to a published method, 5 and a single dose of 400 µg/kg in physiologic saline was administered by subcutaneous injection within 1 hour of preparation. 3 This dose was shown in a previous study to be lethal 3–4 days postinjection, with focal necrosis only becoming apparent 48 hours after tunicamycin administration. 3
Brains were fixed by a perfusion technique. Guinea pigs were anesthetized by intramuscular injection with ketamine (100 mg/kg) and xylazine (4 mg/kg), the thorax was opened rapidly, the right auricle was incised, and heparinized saline (0.9% NaCl containing 0.02% heparin) was injected by gentle manual pressure into the left ventricle through a needle in the apex of the heart. When clear fluid issued from the auricle, the injection of 50 ml of 4% paraformaldehyde was commenced in a similar manner. After remaining in situ for 2 hours, the brain was removed and immediately immersed in 10% buffered formalin for 4 days. Coronal sections were taken from six levels along the brian and from the cervical spinal cord, and the levels were selected to ensure that a wide range of neuroanatomic structures was examined. The levels were (from rostral to caudal) through the cruciate sulcus down to a point midway along the olfactory tract, centrally at a level through the mammillary body, slightly caudal to the previous cut to include the lateral geniculate body and pulvinar, through the anterior colliculus near the roots of the oculomotor nerves, through the posterior colliculus at the level of the pons, and through the medulla oblongata to include the cerebellum. Coronal slices were embedded in paraffin, sectioned at 6 µm, and stained with hematoxylin and eosin (HE).
The axonal and neuronal reaction in these brains was detected using sensitive APP immunohistochemistry. Brain sections were incubated overnight with a monoclonal antibody to APP (clone 22C11, gift from C. Masters, University of Melbourne) at a dilution of 1:3,000, stained with 3,3-diaminobenzidine tetrahydrochloride (Sigma) using avidin–biotin peroxidase (Vector Laboratories), and counterstained with hematoxylin.
APP is a membrane-spanning glycoprotein that is synthesized in normal neurons and has putative neuroprotective, neurotrophic, synaptic, and adhesive functions. 1 6 It is transported anterogradely by fast axoplasmic transport, and APP immunocytochemistry is the earliest and most reliable marker of axonal injury (AI). 9 APP probably accumulates after disruption of the axonal cytoskeleton, causing interruption of axoplasmic flow, and only labels injured axons while all others remain unstained. 1 9 AI can be detected <60 minutes after injury in contrast to more traditional silver impregnation techniques, which only identify axonal swellings with certainty about 15 hours after injury, and there is a high level of background staining with silver. 1 APP-positive axons have been described after traumatic brain injury, around infarcts, abscesses, and tumour metastases, and in neurodegenerative disorders. 9 Neuronal APP is widely upregulated as early as 30 minutes after various traumatic and ischemic insults to the brain, before other signs of cellular injury are evident, 10 and could play an important role in the response of the brain to injury. 6
Axonal and neuronal reaction was quantified microscopically using a grid system. A transparent graticule comprised of 4-mm grid squares, each with a unique reference number, was placed over the section. The graticule had reference marks so the correct alignment could be made with the underlying slide and independent evalution of brain sections could be conducted. The detection of any APP immunostaining of axons in a grid square or APP-reactive granules occupying ≥50% of the neuronal cytoplasm resulted in a positive score. The number of positive grids (from an average total of 140 for the seven coronal sections) was then summed, and the percentage of APP-positive grids for axons and neuronal cell bodies was calculated.
Microscopically, well-circumscribed foci of parenchymal injury were most common in the cerebellum, particularly in the granular layer (Fig. 1). Necrotic foci were also randomly distributed throughout the brain at 48 and 72 hours after injection of tunicamycin, but especially in the thalamus, corpus striatum, cerebral cortex, and corpus callosum. Lesions were less commonly found in the vestibular nuclei, colliculi, paraventricular area, medulla, red nucleus, substantia nigra, hippocampus, pons, and gray matter of the cervical spinal cord. These focal areas had a similar appearance at both times but were more numerous at 72 hours. The neuropil was often markedly vacuolated and this spongy change sometimes progressed to necrosis. Neurons within and adjacent to these foci often showed ischemic change with shrunken, angular, and hypereosinophilic cytoplasm and often pyknotic nuclei; this neuronal change was not observed in perfusion-fixed control brains. Moreover, small blood vessels in areas of neural damage were frequently plugged with erythrocytes (Fig. 2), whereas vessels in the surrounding unaffected parenchyma were normally perfused (Fig. 3), suggesting local vascular stasis. Focal necrosis was also unattended by an acute inflammatory reaction, attributed to vascular occlusion and inadequate reperfusion at the time intervals examined. 8
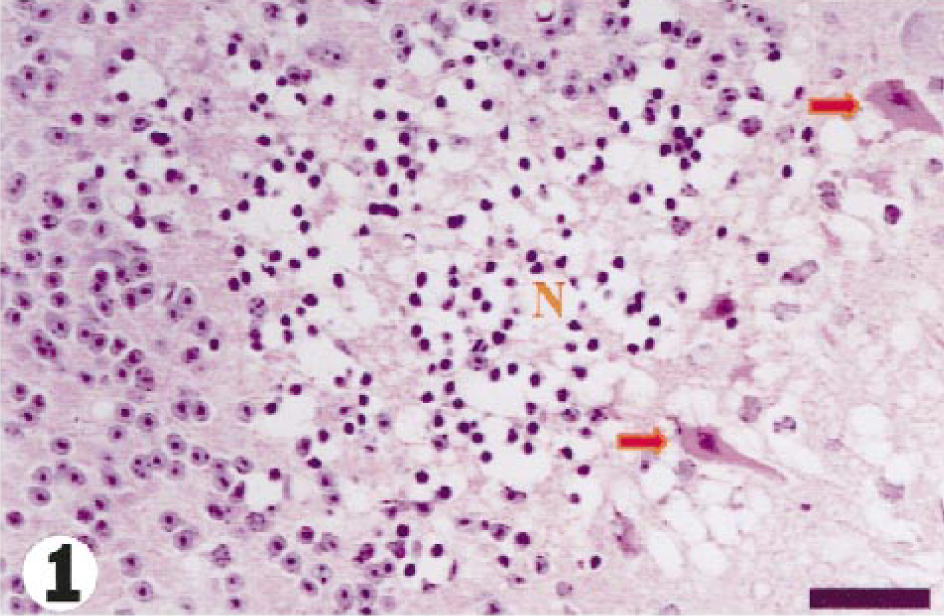
Cerebellum; guinea pig No. 4/99. Focal granular layer necrosis (N) with ischemic change in adjacent Purkinje cells (arrows), 48 hours after injection of tunicamycin. HE. Bar = 65 µm.
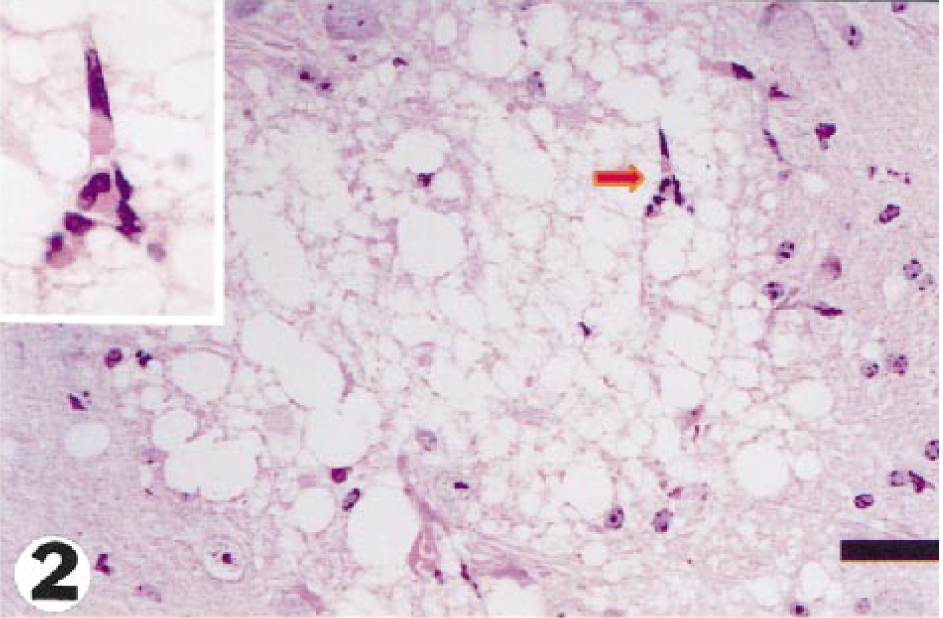
Pons; guinea pig No. 3/99. Focal spongy change of the neuropil with an occluded capillary (arrow) enlarged in the inset, 48 hours after injection of tunicamycin. HE. Bar = 52 µm.
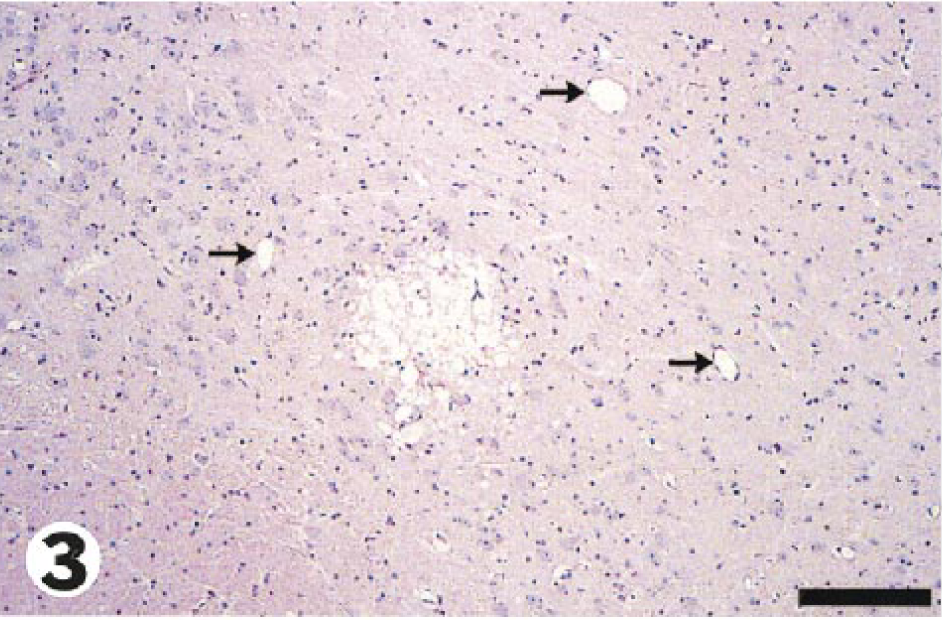
Medulla; guinea pig No. 4/99. Lower magnification of Fig. 2 showing normally perfused blood vessels (arrows) in unaffected parenchyma surrounding the area of focal damage containing the occluded capillary. Bar = 46 µm.
In APP-immunostained sections from tunicamycin-treated animals, APP expression in neurons (Fig. 4) was widely distributed throughout the brain, with a mean percentage of positive grids of 34% and 56% at 48 and 72 hours, respectively. In control brains (Fig. 5), the mean percentage of neuronal positivity was 4%. Many APP-immunopositive neurons showed no other morphologic changes (Fig. 4), but some had progressed to necrosis (Fig. 6). The mean percentage of positive grids for axons was 9% and 12% at 48 and 72 hours, respectively, and no AI was found in control brains. APP-positive axonal swellings were common in foci of spongy change (Fig. 7), and the axonal reaction in these sites was generally not appreciable in HE-stained sections. In addition, APP-reactive axons were found, to a more limited degree, in other areas of brain where the neuropil was otherwise unaffected (Fig. 8).
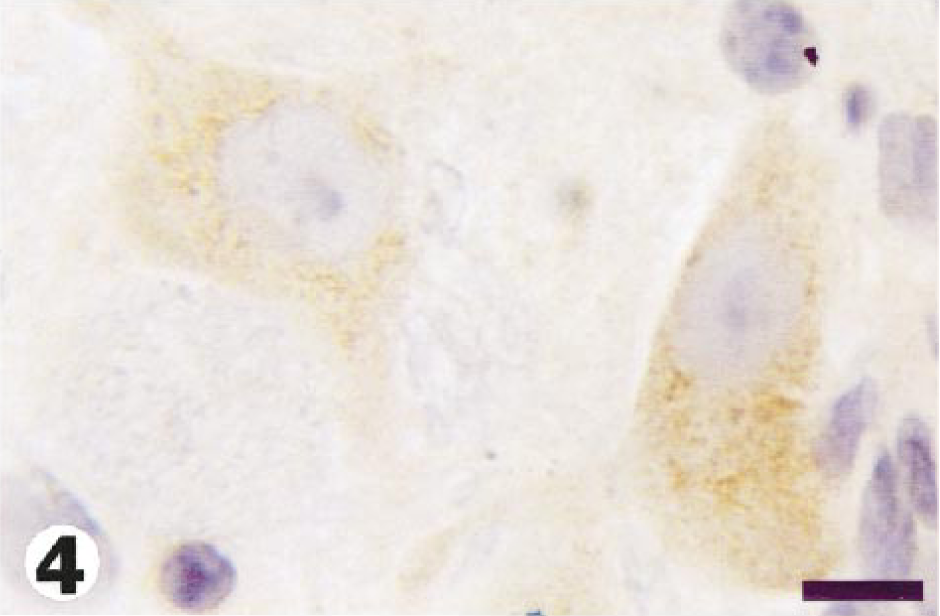
Medulla; guinea pig No. 4/99. Numerous APP-positive cytoplasmic granules in all neuronal cell bodies, 48 hours after injection of tunicamycin. Avidin–biotin peroxidase method, hematoxylin counterstain. Bar = 15 µm.
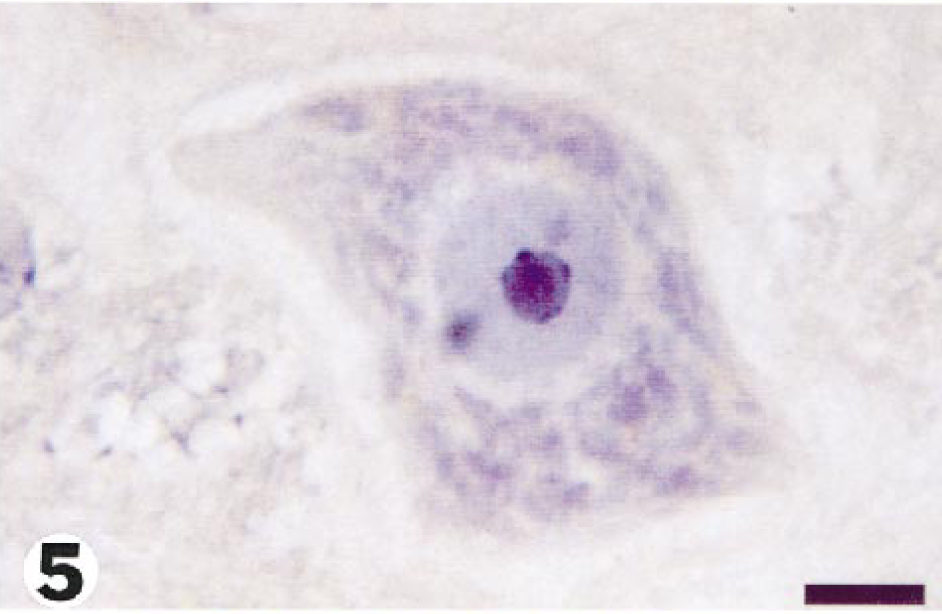
Medulla; guinea pig No. 7/99. Neuron from the same area shown in Fig. 3 in a control guinea pig. Bar = 10 µm.
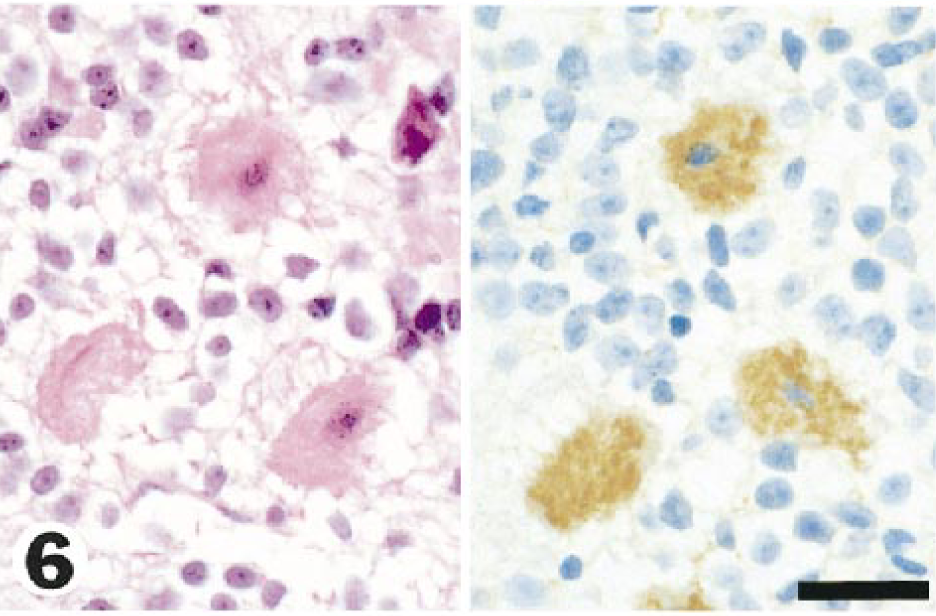
Cerebellum; guinea pig No. 4/99. Purkinje cells showing ischemic cell change (at left) and APP-positive neurons from the same cerebellum that also showed ischemic change with HE (right). Bar = 24 µm.
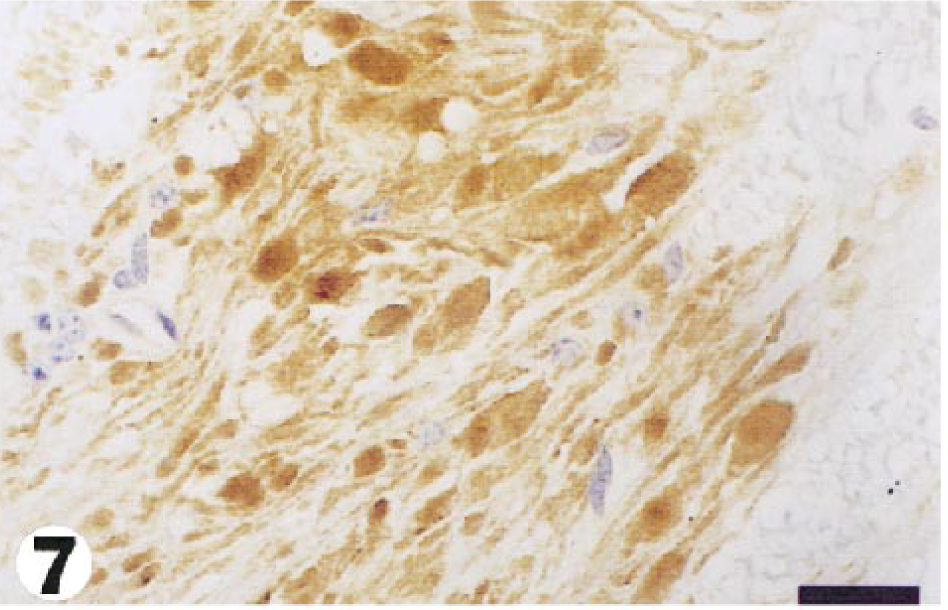
Medulla; guinea pig No. 11/99. Numerous axonal swellings showing strong APP immunoreactivity in an area of focal ischemic injury, 72 hours after injection of tunicamycin. Bar = 80 µm.
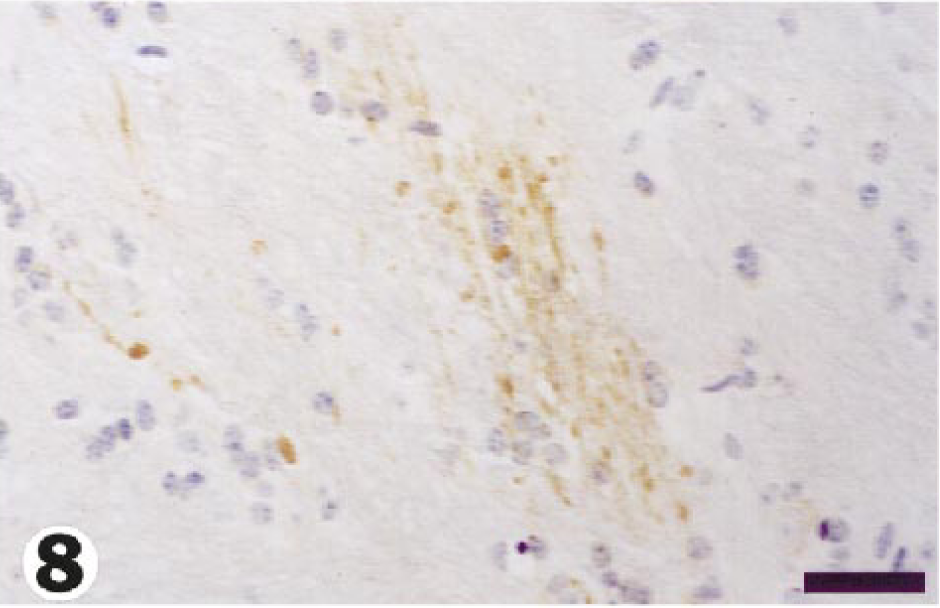
Pons; guinea pig No. 6/99. Many APP-immunoreactive axons in otherwise unaffected neuropil, 48 hours after injection of tunicamycin. Bar = 40 µm.
The widespread expression of APP in neuronal perikarya probably represented an acute phase response to trauma, 6 because it is unlikely that all these immunoreactive neurons were irreversibly damaged. Some APP-positive neurons in this and other 7 studies showed ischemic cell change, but many of these neurons were morphologically unaltered. Furthermore, although some APP-positive axons were obviously swollen, others were not, and the concentration of this protein may reach a detectable level before signs of axoplasmic swelling appear because of its delivery by fast axoplasmic transport. 9
Tunicamycin-induced changes in axons and neurons were found to be more widely distributed in guinea pig brains when using APP immunocytochemistry than when using traditional, less sensitive staining methods. Furthermore, many of these structural alterations occurred distant to foci of ischemic-hypoxic injury, suggesting that tunicamycin may have a direct toxic effect on axons and neurons and on endothelial cells.
Footnotes
Acknowledgements
We thank Mr. F. Madaras (IMVS Veterinary Services) and the Plant Toxins Unit, CSIRO Australian Animal Health Laboratory, Geelong, Victoria, for preparing the commercial tunicamycin used in this study.
