Abstract
Toll-like receptors 2 and 4 (TLR2 and TLR4) are well-characterized cell surface receptors that recognize specific pathogen-associated molecular patterns and play an important role in pathogen recognition and activation of the innate immune system. Variable expression of TLR2 and TLR4 has been described in trophoblasts from normal and diseased placentas; yet, there are limited data regarding trophoblast TLR expression in response to specific placental pathogens, and TLR expression in the guinea pig placenta has not been described. The guinea pig is an effective model for
Toll-like receptors (TLRs) are a well-characterized family of pattern recognition receptors that play a key role in innate immune function, and they have been extensively reviewed. 3,19,30,31 In brief, 13 members of the TLR family have been identified in mice 19 —10 of which (TLRs 1–10) have also been identified in cattle, swine, sheep, and humans—and TLR genes have a high degree of sequence similarity between species. 10 TLRs can be grouped on the basis of the characteristics of their ligands and on the location of cellular expression. TLRs 1, 2, 4, 5, and 6 recognize specific pathogen-associated molecular patterns, such as triacyl lipopeptide (TLR1/TLR2), peptidoglycan (TLR2), lipopolysaccharide (TLR4), flagellin (TLR5), and diacyl lipopeptide (TLR6/TLR2), and they are expressed on the cell surface, whereas TLRs 3, 7, 8, and 9 recognize pathogen-associated nucleic acid patterns and are expressed in intracellular compartments, such as endosomes. 19 TLR2 recognizes the most diverse group of ligands, and this is made possible, at least in part, by the formation of heterodimers with TLR1 and TLR6. 34 Members of the TLR family are expressed by a variety of cell types, including those directly involved in host immunity, such as macrophages, neutrophils, and dendritic cells, as well as nonimmune cells such as fibroblasts, endothelial cells, and various epithelial cells. 19
The maternofetal interface has long been considered an immunological paradox because the endometrium must allow for implantation of the allogenic placenta while maintaining adequate immune surveillance against a diverse spectrum of potential pathogens.
24
The role of pattern recognition receptors in this process—particularly, TLRs—has been an active area of research and the subject of a recent review.
1
Immunohistochemistry (IHC) has been used to characterize the expression of TLR2 and TLR4 in the various trophoblast layers from normal and diseased human placentas;
15,20,22,25
however, there are limited data regarding trophoblast TLR expression associated with specific placental pathogens, and TLR expression in the guinea pig placenta has not been described. We have shown by IHC that
Previous studies have revealed that TLR2 and TLR4, but not TLR5, are capable of binding pathogen-associated molecular patterns of
Materials and Methods
Sample Collection
Blocks of formalin-fixed, paraffin-embedded guinea pig placenta were obtained from animals used in previous experiments
6
in which animals were inoculated orally with
Histologic Evaluation
Hematoxylin and eosin–stained sections of each placenta were evaluated to determine the presence or absence of suppurative placentitis as defined by the presence of infiltrating neutrophils within the junctional zone between the subplacenta and decidua.
Immunohistochemistry
Formalin-fixed, paraffin-embedded guinea pig placental samples were prepared for IHC as previously described. 6 For antigen retrieval, sections were treated with 0.1% protease in a Tris buffer (pH 7.6) at 37°C for 10 minutes, followed by three rinses in phosphate buffered saline solution before placement in an automated cell staining system (BioGenex, San Ramon, CA). Sections were then incubated in 10% neutral goat serum at 22°C for 20 minutes to inhibit nonspecific binding. Primary antibodies consisted of commercially available affinity-purified rabbit antihuman TLR2 and affinity-purified rabbit antihuman TLR4 (Rockland, Gilbertsville, PA) and were used at dilutions of 1:500 and 1:300, respectively. Rabbit antihuman TLR2 and TLR4 primary antibodies have been shown to be cross-reactive in guinea pig tissues. 17 Slides were incubated at 22°C for 60 minutes (TLR2) and 120 minutes (TLR4), followed by rinsing in a bath of phosphate buffered saline solution for 5 minutes. The secondary antibody (BioGenex), horse radish peroxidase–streptavidin conjugate (Invitrogen, Carlsbad, CA), chromogen (Vector, Burlingame, CA), and counterstaining steps were performed as previously described. 6 Positive control samples consisted of guinea pig spleen and lung tissues for evaluation of receptor expression on phagocytes. Negative controls were prepared from the same blocks and consisted of sections in which the primary antibody was omitted and sections in which normal (preimmune) rabbit serum was applied in place of the primary antibody.
IHC for
Image Analysis
IHC-stained sections were examined with an Olympus BX60 light microscope (Olympus, Tokyo, Japan) and RGB color digital images were recorded with an Olympus DP70 camera (Olympus) with 2040- × 1536-bit resolution and 24-bit depth. Three magnification images (40×) were obtained for each subplacenta and included one image taken from each lateral aspect of the junctional zone and one from the area just below the central excavation. The recorded microscopic fields were oriented such that subplacental trophoblasts filled as much of the field as possible while excluding any inflammatory exudate present in the junctional zone.
To quantify the level of TLR protein expression in subplacental trophoblasts, all digital images were analyzed with ImageJ image analysis software (National Institutes of Health, Bethesda, MD) and a color deconvolution (stain separation) plug-in implementing methods previously described. 27 Using the plug-in, the red color (positive immunohistochemical staining) was isolated in each image and then converted to a black-and-white image and set to a common threshold. Thresholds of 175 and 200 were selected for TLR2 and TLR4, respectively. The percentage area of each image exhibiting the selected threshold level of staining was then quantified. The mean percentage of the stained area was then determined for the three images from each placenta, and the average staining of all placentas from each individual guinea pig was calculated and compared.
Statistical Analysis
SAS 9.2 was used to perform independent-sample
Results
Histologic Findings in Affected Placentas
Placentas from aborting guinea pigs were typically characterized by one or more of the following changes: congestion of the placental labyrinth, multifocal necrosis of the placental disc, and multifocal to coalescing necrosis of subplacental trophoblasts (Fig. 1
) that was often associated with neutrophilic infiltration. The placental junctional zone in at least one placenta from all 12 aborting guinea pigs was infiltrated with neutrophils; however, not every placenta from each aborting animal was equally affected. This was most notable in guinea pigs with higher fetal numbers, as expected, and placentas from individual fetuses occasionally lacked histologic lesions. In total, 88.6% of placentas from aborting guinea pigs (39 of 44) had evidence of suppurative placentitis. IHC for
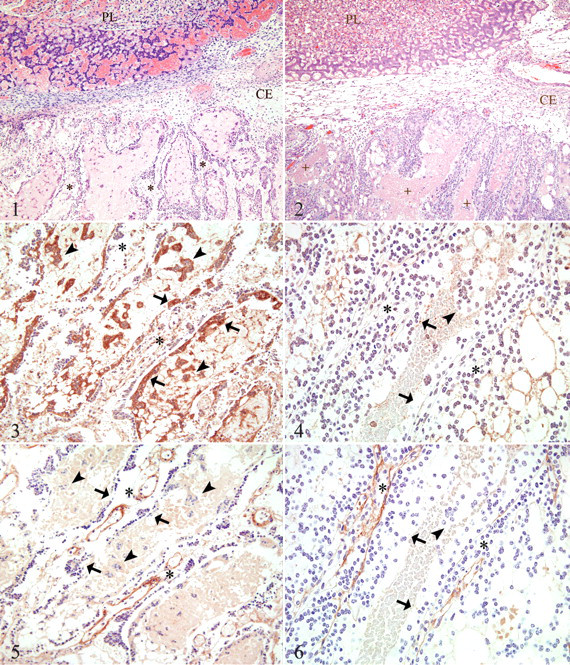
Placentas from sham-inoculated controls lacked significant lesions (Fig. 2 ). Multifocally, there were small areas in which subplacental trophoblasts (1) appeared hypereosinophilic with pyknotic nuclei and (2) often surrounded accumulations of scant to moderate amounts of eosinophilic cellular and karyorrhectic debris (necrosis) admixed with basophilic granular material (mineral); however, these areas were not associated with neutrophilic infiltration and were generally limited to the lateral aspects of the junctional zone.
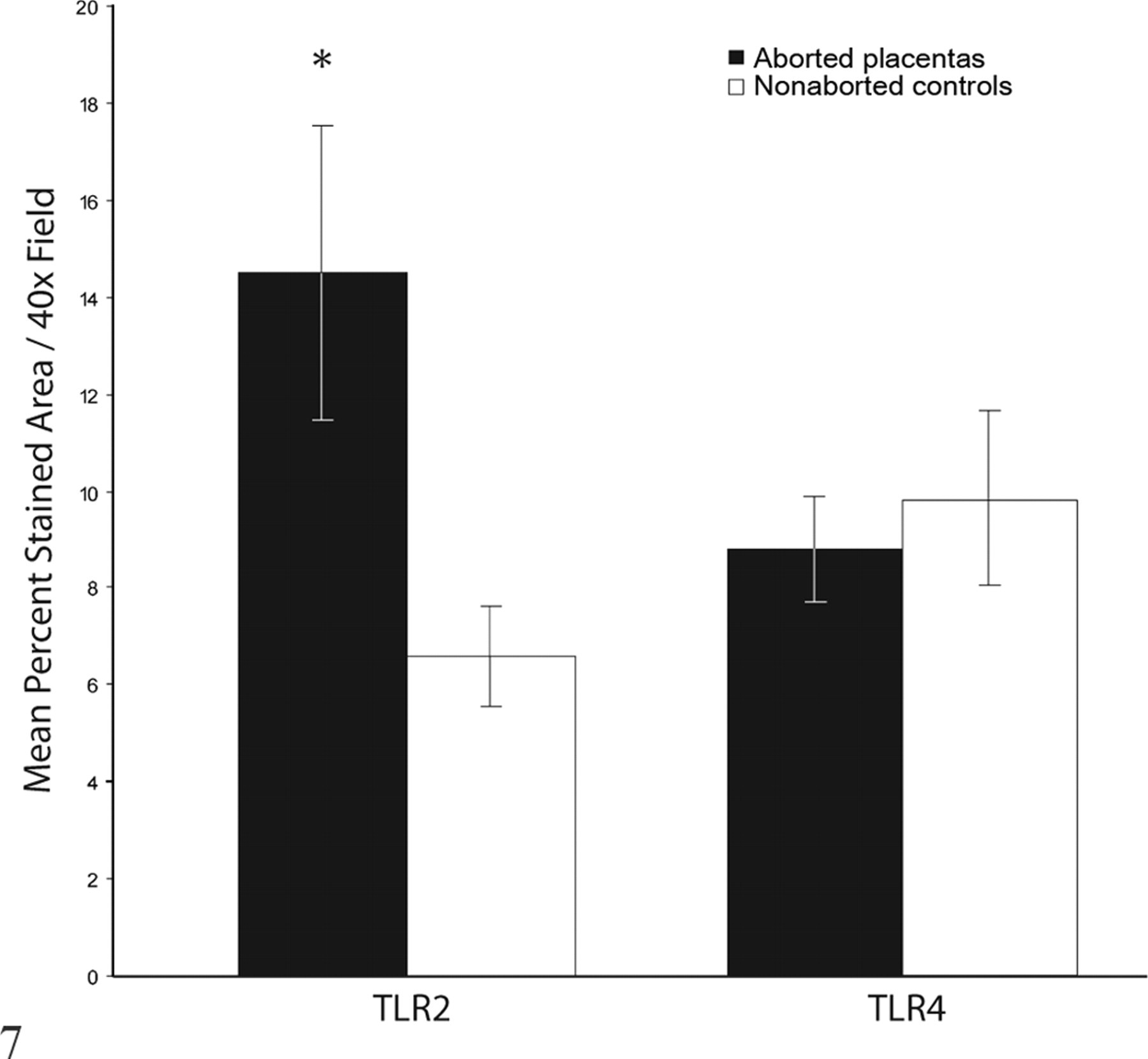
Percentage area of subplacental photomicrographs corresponding to positive staining for Toll-like receptors 2 and 4 (TLR2 and TLR4) per 40× field (mean ± SEM). Values represent mean immunoreactivity for all placentas from 12 aborting guinea pigs (44 placentas) and 9 nonaborted controls (28 placentas). Asterisk indicates that values differ significantly (
Expression of TLR2
IHC of aborted placentas, as compared with controls, revealed a moderate increase in TLR2 immunoreactivity within trophoblasts of the main placenta; however, strong positive immunoreactivity was a consistent feature of subplacental syncytiotrophoblast cells and syncytial islands within lacunae, whereas the cytotrophoblast layer lacked significant staining (Fig. 3 ). In sham-inoculated control tissues, a low level of diffuse cytoplasmic immunoreactivity for TLR2 was evident in trophoblasts of the interlobium of the main placenta, whereas the syncytiotrophoblast and cytotrophoblast cells of the subplacenta were generally unstained (Fig. 4 ). Positive immunoreactivity was also observed in neutrophils and macrophages within control tissues, whereas negative controls were nonreactive (not shown).
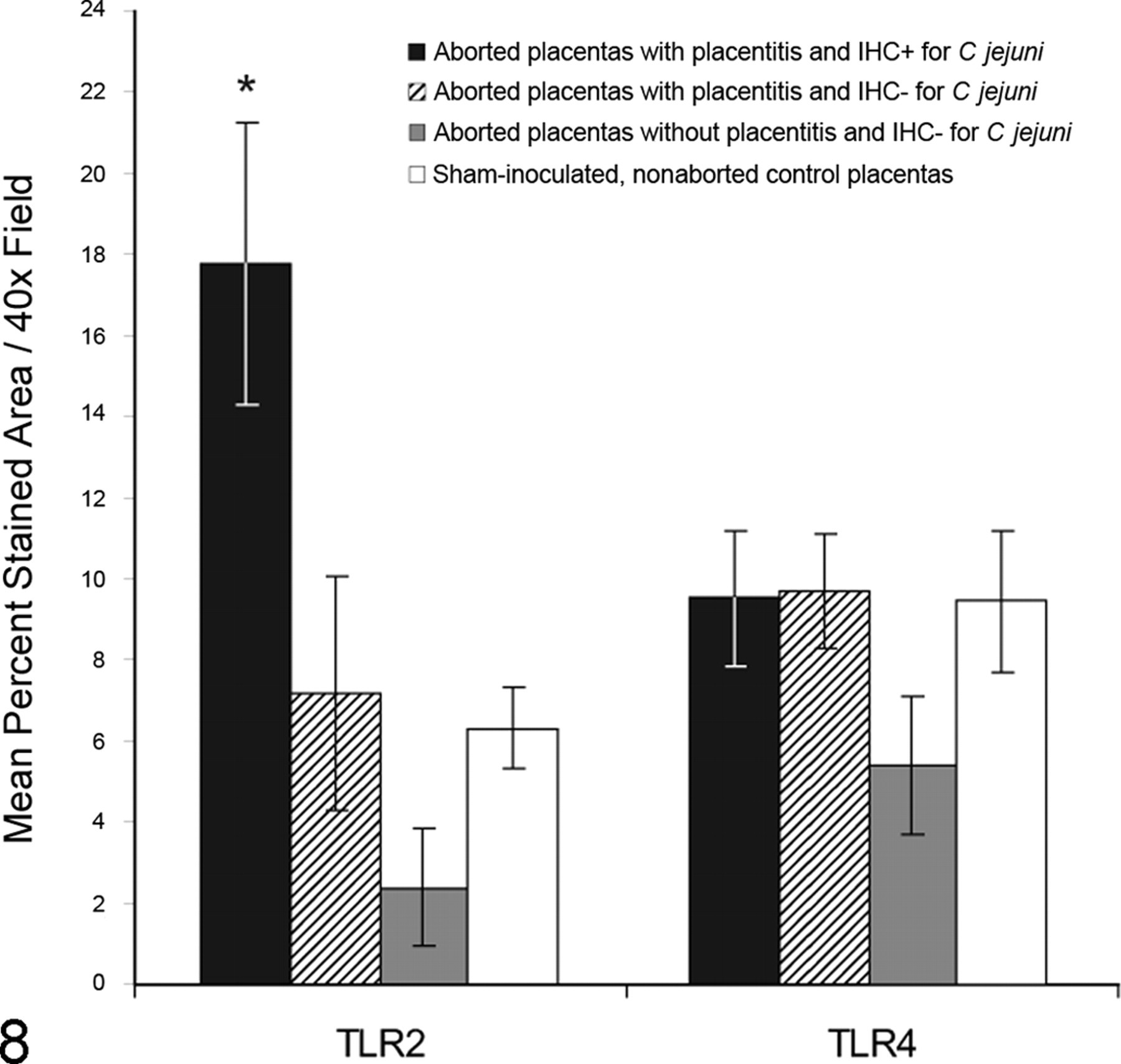
Percentage area of subplacental photomicrographs corresponding to positive staining for Toll-like receptors 2 and 4 (TLR2 and TLR4) per 40× field (mean ± SEM). Values represent mean immunoreactivity for all aborted placentas immunohistochemistry positive (IHC+) for
Expression of TLR4
IHC of aborted placentas revealed moderate TLR4 immunoreactivity within trophoblasts of the main placenta and in the mesenchyme and surrounding vascular spaces of subplacental lamellae. A low level of positive immunoreactivity was an inconsistent feature of subplacental syncytiotrophoblast cells, whereas the adjacent cytotrophoblast layer lacked significant staining (Fig. 5 ). IHC of sham-inoculated control tissues revealed scant cytoplasmic immunoreactivity for TLR4 in trophoblasts of the interlobium, moderate immunoreactivity within the mesenchyme and surrounding vascular spaces in subplacental lamellae, and minimal to no immunoreactivity within subplacental syncytiotrophoblast and cytotrophoblast cells (Fig. 6 ). Strong positive immunoreactivity was also observed in neutrophils and macrophages within control tissues, whereas negative controls were nonreactive (not shown).
Image Analysis of Subplacental Immunoreactivity
TLR2 immunoreactivity was significantly higher in subplacental trophoblasts from aborted placentas compared with nonaborted, sham-inoculated controls (
TLR4 immunoreactivity did not differ significantly between aborted placentas and controls (
Discussion
Campylobacteriosis is a well-recognized cause of small ruminant abortion worldwide; however, the molecular mechanisms involved in this process are not known. We recently showed using IHC that, following oral inoculation,
The guinea pig placenta consists of a disclike, labyrinthine chorioallantoic main placenta with separate subplacenta and yolk sac placenta.
16
The subplacenta is unique to hystricognath rodents, and its histological, histochemical, and ultrastructural properties have been thoroughly described elsewhere.
11,23,26
In brief, the subplacenta is separated from the main placenta by a thin layer of fetal mesenchyme,
9
and the distal segments of the uteroplacental arteries enter the main placenta through the periphery of this junction.
11
It is in this location that we consistently observe
In the study reported here, we observed markedly increased immunoreactivity for TLR2 protein in the syncytiotrophoblast layer of the subplacenta from guinea pigs aborting following infection with
In contrast to our findings with TLR2, TLR4 protein expression in subplacental trophoblasts did not differ significantly between controls and placentas from aborting animals overall or from any of the subdivided groups. These results suggest that altered TLR4 expression is not associated with
Limitations of this study include its retrospective nature and the lack of a specific time-matched control placenta for each aborted placenta. The presence of noninflamed placentas within the pool of aborted tissues did, however, serve as an internal control. Furthermore, the immunoreactivity of these tissues was similar to sham-inoculated control tissues; as such, the constitutive expression of TLR2 and TLR4 in the guinea pig subplacenta did not significantly vary throughout the period studied. In addition, the relevance of these findings to ovine campylobacteriosis is uncertain; however, a recent study of obesity-induced placental inflammation in ewes revealed elevated mRNA expression for TLR2 and TLR4 with a concurrent increase in proinflammatory cytokines, 36 and these results support a role for TLRs in the response of ovine placenta to inflammatory stimuli.
In summary, whereas TLR4 immunoreactivity was essentially unchanged, we observed increased TLR2 immunoreactivity in subplacental syncytiotrophoblast cells from aborting guinea pigs following oral inoculation with
Footnotes
Acknowledgements
We would like to thank Deborah Moore for her assistance in developing the immunohistochemistry procedures used in this work.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This study was supported by the Iowa Livestock Health Advisory Council (grant No. 109-05-66) and the Iowa State University CVM Summer Scholars Research Program.
