Abstract
A nestling white-faced ibis (
Keywords
Infections by the oomycete
On gross examination of the skin overlying the wings, neck, torso, and legs, there were numerous raised, yellowish-tan, ulcerated nodules up to 1.5 cm in diameter (Fig. 1). The nodules were firm and dry and, on cut section, extended into the superficial dermis. A complete gross and histologic necropsy was performed, including examination of the cardiovascular, gastrointestinal, respiratory, endocrine, and neural (brain, peripheral nerves) systems and organs of immunity (spleen, bursa, bone marrow); other than the presence of the nodules, the bird was unremarkable. On microscopic examination of regions of feathered skin corresponding to the nodules, the surface, spanning a region of up to 10 sequential follicles, was ulcerated and replaced by thick caseonecrotic debris (Fig. 2) and subjacent follicles were surrounded by collars of similar caseonecrotic debris including fragmented and karyorrhectic granulocytes. In most affected feathers, there was necrosis that extended to include the follicular epithelium. Feather shafts were sometimes absent, and infrequently, the remnant pulp cavity was necrotic. Numerous transverse, round-to-oval, and sagittal sinuous negative profiles of hyphae were visible within the necrotic debris and more rarely within the feather pulp. Hyphae ranged from 3 to 7 µm in diameter, rarely branched, were pauciseptate, and were visible using periodic acid Schiff and Gomori methenamine silver (GMS) stains (Fig. 3). Peripheral to the necrotic cuffs, and occasionally extending into the deeper dermis, were dense populations of granulocytes and histiocytes. Because some avian granulocytes (eosinophils and heterophils) are difficult to distinguish in HE sections, a Luna's stain was used to establish the presence of eosinophils, which are demonstrated by red granules and blue nuclei using this method. Eosinophils were the predominant inflammatory cell type within the perifollicular region and the ulcerated surface and were scattered within the subjacent dermis (Fig. 4). Scattered heterophils were present within the dermis subjacent regions of ulceration.
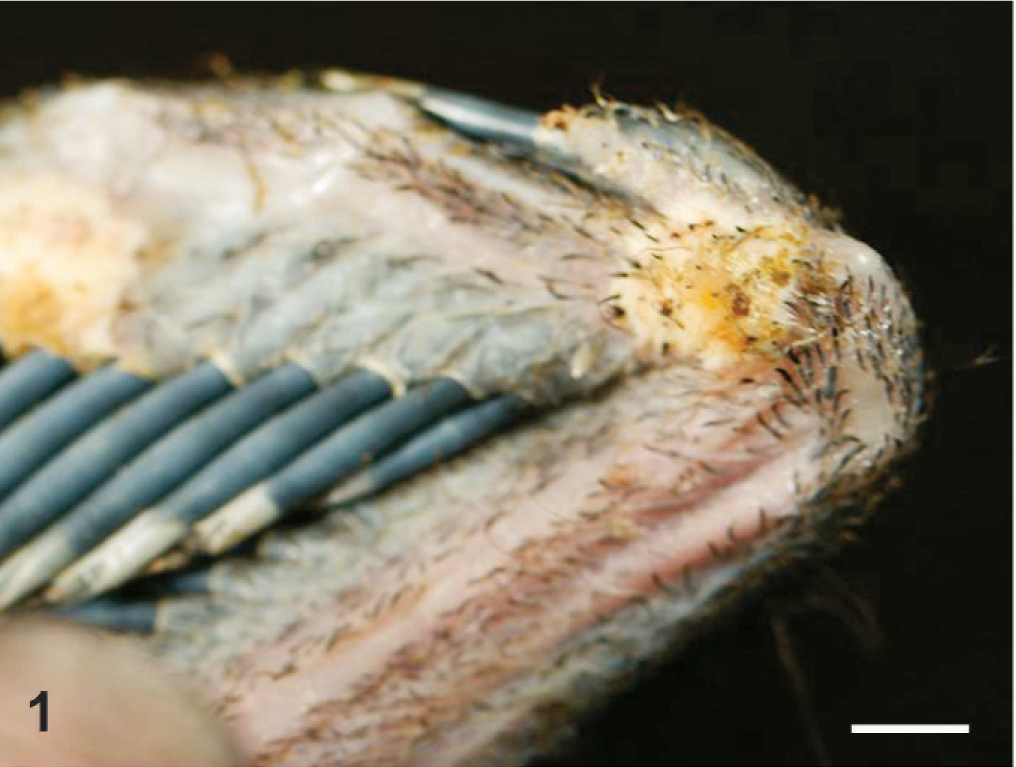
Wing, overlying metacarpal-ulnar joint. A raised, ulcerated, yellowish-tan nodule overlies the lateral surface of the joint. Bar = 0.5 cm.
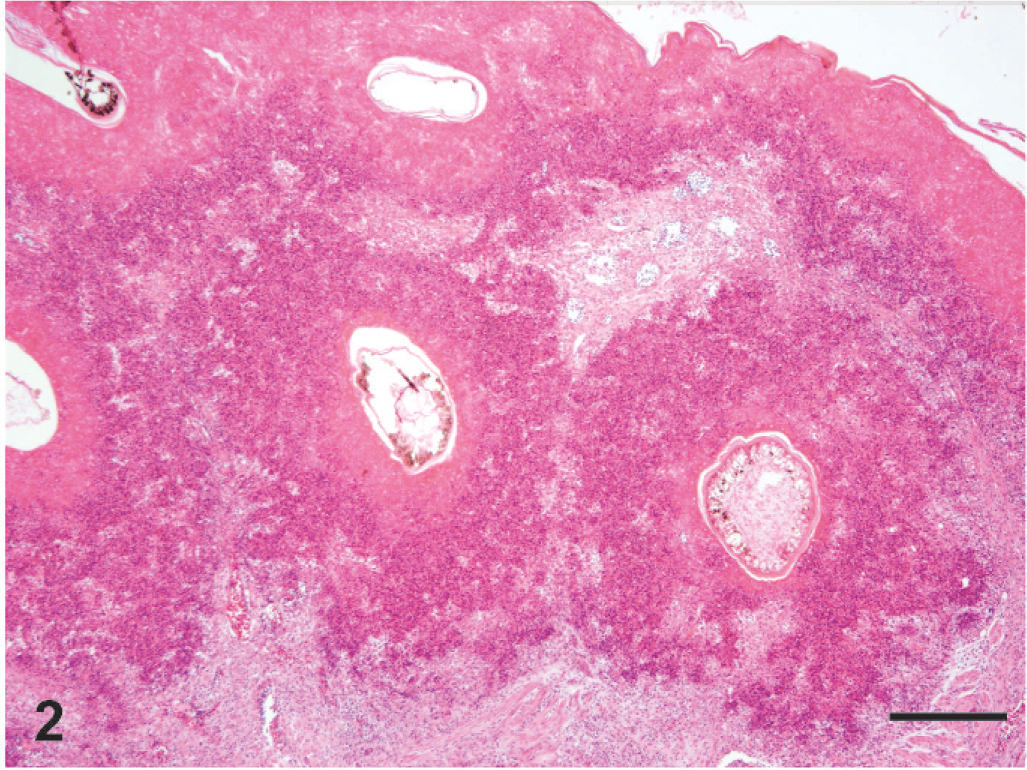
Skin, lateral thorax. The surface and follicular epithelium and dermis surrounding feather follicles are replaced by inflammation and brightly eosinophilic karyorrhectic debris. HE. Bar = 200 µm.
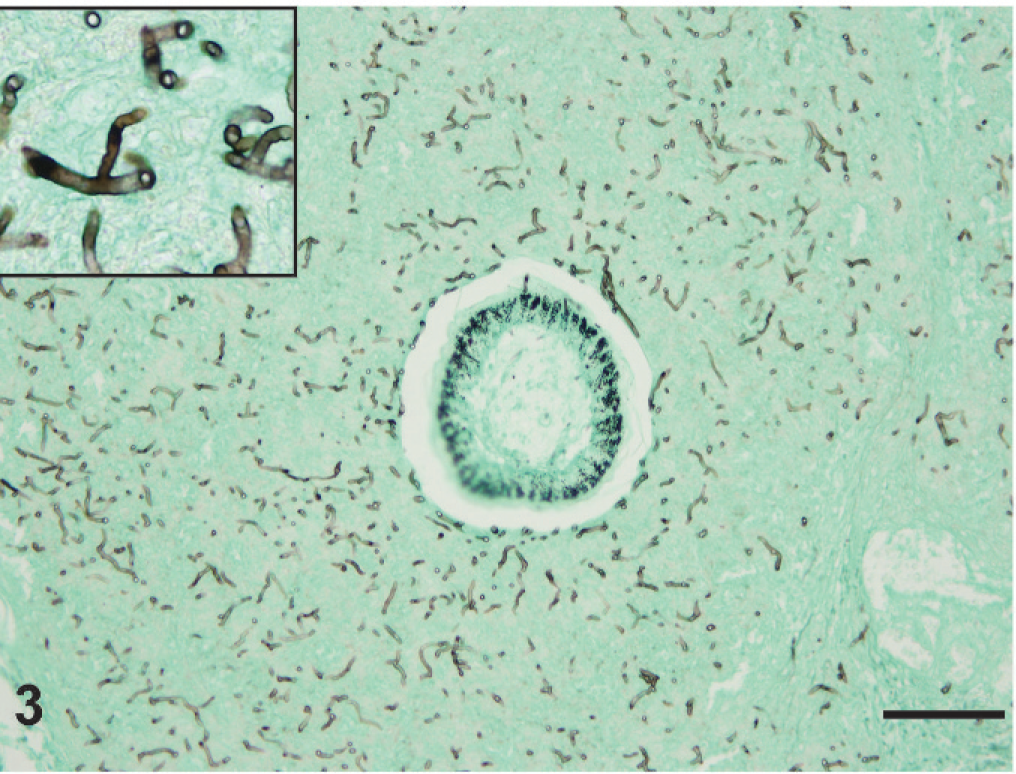
Skin, feather follicle and perifollicular regions, lateral thorax. The bottom image is a single feather follicle. The surrounding dermis contains numerous oomycete hyphae that are blunt to sinuous, pauciseptate, and range from 3 to 7 µm in diameter. GMS stain. Bar = 10 µm.
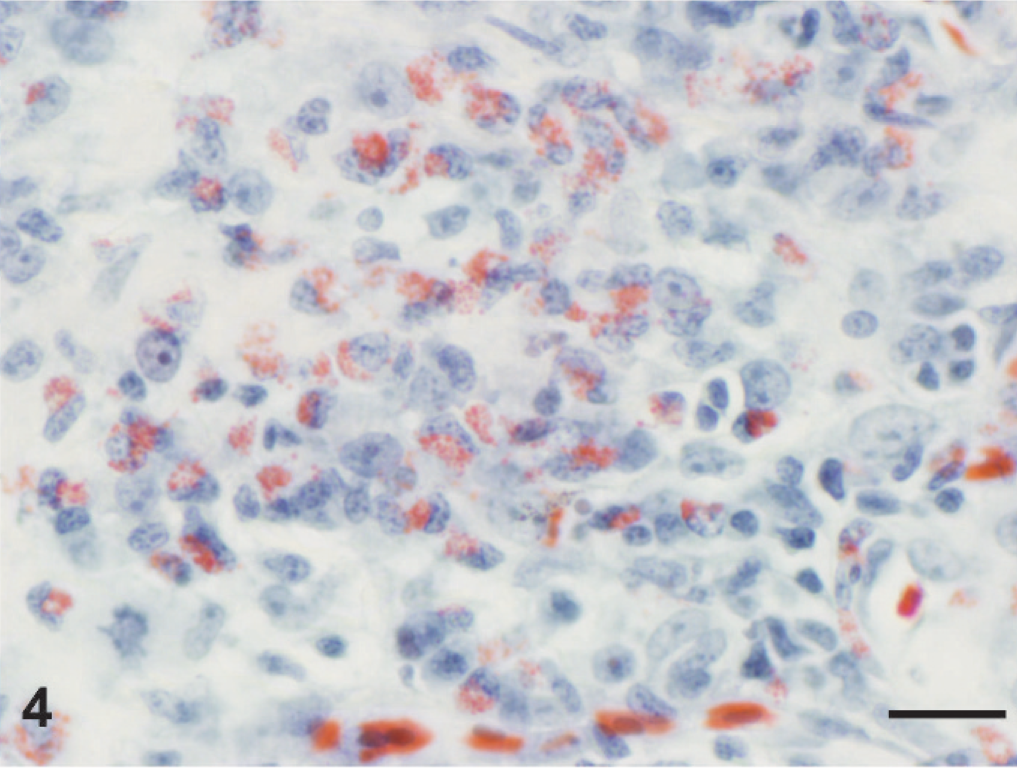
Skin, dermis. The cytoplasmic granules of granulocytes within the inflamed regions are reddish-brown using a Luna's stain, indicating that these are eosinophils. Luna's stain. Bar = 20 µm.
Based on initial gross observations at necropsy, dermatophytosis was considered a possible cause of the skin lesions and nodular material was planted on dermatophyte-specific isolation media (Dermatophyte Test Media, Hardy Diagnostics, Santa Monica, CA) and incubated at 25° and 37°C. After histopathology revealed the presence of large, hyaline, and pauciseptate hyphae, isolation attempts were also conducted using a general mold isolation agar (Inhibitory Mold Agar, Hardy Diagnostics). Incubation was carried out at 25°C. Both the dermatophyte-specific media and general fungal isolation agar plates were incubated for 1 month, but no isolations were made.
Total DNA was isolated from one of the skin nodules using a previously described method.
6
Briefly, tissue was ground using a sterile pestle and mixed in 1 ml of phosphate-buffered saline (PBS). The mixture was centrifuged at 20,800 ×
Amplified product representing the 5′ end of the large subunit rRNA gene was generated using previously described fungal primers TW13 and Ctb6.
16
The amplification mixture contained 100 to 300 ng of DNA template, 1× polymerase chain reaction (PCR) buffer (II/Gold Buffer, Applied Biosystems, Foster City, CA), 1.5 mM MgCl2, 150 µM of each deoxynucleotide (Applied Biosystems), and 1 U of polymerase (AmpliTaq Gold, Applied Biosystems) in a reaction volume of 50 µl. Amplification parameters were heating at 95°C for 10 minutes followed by 50 cycles of 94°C for 30 seconds, 50°C for 1 minute, 72°C for 2 minutes, and a final extension period at 72°C for 7 minutes (GeneAmp PCR System 9700, Applied Biosystems). A 5 µl aliquot from the PCR reaction was electrophoresed on a 3% agarose gel to check for an amplicon of expected size range. The amplicon was purified (Montage PCR cleanup kit, Millipore, Bedford, MA) and sequenced (ABI 3730 DNA sequencer, Davis Sequencing Inc., Davis, CA). Primers used for amplification were also used for sequencing forward and reverse fragments. A consensus sequence was generated from individual sequenced strands and a basic local alignment search tool (BLAST) search was performed (VectorNTI Advance 10.3, Invitrogen Corporation, Carlsbad, CA). Search results for the consensus sequence showed 100% homology (503/503 base pairs) with
Infections caused by the oomycete
Infection sites in the case of cutaneous forms of pythiosis in most animals are typically concentrated on the distal limbs, which most likely involve the skin regions in contact with water. The lesions in this ibis were more generalized, and there was a distinct surface and perifollicular distribution of the hyphae. Microscopic features of both the fungus and inflammation are otherwise similar to those described in affected mammals. It is possible that ibis nestling behavior supports this expanded distribution or that there was a contributory or predisposing cause in this bird that was not detectable by histopathology.
