Abstract
Spontaneous atherosclerosis is common in psittaciformes, and clinical signs associated with flow-limiting stenosis are encountered in pet birds. Nevertheless, a psittacine model of atherosclerosis has not been developed for research investigations. Sixteen captive-bred Quaker parrots (Myiopsitta monachus) were used in this study. While 4 control birds were fed a maintenance diet, 12 other birds were fed an atherogenic diet composed of 1% cholesterol controlling for a calorie-to-protein ratio for periods ranging from 2 to 8 months. The birds were euthanized at the end of their respective food trial period. Histopathology, transmission electron microscopy, and cholesterol measurement were performed on the ascending aorta and brachiocephalic and pulmonary arteries. Plasma lipoproteins, cholesterol, and triglycerides were also measured on a monthly basis. Significant atherosclerotic lesions were induced within 2 months and advanced atherosclerotic lesions within 4 to 6 months. The advanced lesions were histologically similar to naturally occurring lesions identified in the same parrot species with a lipid core and a fibrous cap. Ultrastructurally, there were extracellular lipid, foam cell, and endothelial changes. Arterial cholesterol content increased linearly over time. Plasma cholesterol and low-density lipoprotein (LDL) significantly increased over time by an average of 5- and 15-fold, respectively, with a shift from high-density lipoprotein to LDL as the main plasma lipoprotein. Quaker parrots also exhibited high plasma cholesteryl ester transfer protein activity that increased, although not significantly, over time. This experiment demonstrates that in Quaker parrots fed 1% cholesterol, advanced atherosclerosis can be induced relatively quickly, and lesions resemble those found in other avian models and humans.
Keywords
Similar to humans, birds have a high susceptibility to atherosclerosis and probably show the highest prevalence and severity of atherosclerotic lesions among vertebrate animals under standard captive conditions. 4,6 Nonpsittacine avian models have been extensively used for the experimental investigations of atherosclerotic lesions and their associated physiologic complications in species such as the White Carneau pigeon (Columba livia domestica), Japanese quail (Coturnix japonica), and chicken (Gallus gallus domesticus). 38,40,41,47
Birds of the order Psittaciformes (eg, parrots, cockatoos) are long-lived species and seem especially prone to atherosclerosis, with a high prevalence of lesions in the captive population. 4,6,8,13 In psittacines, atherosclerotic lesions occur spontaneously and on a cholesterol-free diet but, unlike in pigeons, which develop them in the thoracic aorta, they are predominantly located in the main elastic arteries at the base of the heart and in the carotid arteries. 4,6,8,13 Although identified less often in psittacine species than in humans, coronary and other peripheral arterial lesions are also diagnosed on a regular basis through histopathologic examination. In addition, parrots show clinical manifestations of advanced atherosclerotic lesions caused mainly by arterial luminal stenosis (eg, ischemia, claudication, stroke) with the histologic characteristics of the lesions being similar to those associated with human disease. 7,10,14,26,31,37,39,46 However, atherothrombosis, plaque rupture, and myocardial infarction are considered infrequent in psittaciformes. 4,6 A recent large multicenter case-control study on more than 7600 captive psittacine patients has shown that age, female sex, some genera, and concurrent reproductive diseases were potential risk factors for the development of advanced atherosclerotic lesions of types IV to VI of the American Heart Association (AHA) classification adapted to psittacine atherosclerotic lesions. 6,8 Furthermore, the prevalence of atherosclerotic lesions by age groups was comparable to humans. The pathophysiology and risk factors of psittacine atherosclerosis are still poorly understood. Also, the antemortem diagnosis is challenging, and the correlation between clinical signs and the degree of histologic changes is unclear with current scientific knowledge. 6,9 Therefore, a psittacine model is highly needed to investigate diagnostic tests, explore pathogenesis and risk factors, and test potential treatments of this common disease of parrots. One report published in 1961 documented significant atherosclerosis and hypercholesterolemia in budgerigars (Melopsittacus undulatus) fed a diet composed of 2% cholesterol and 15% lard, 12 but since that study, an experimental psittacine model of atherosclerosis has not been established and used for atherosclerosis research investigation.
Other atherosclerosis animal models involve mainly mammalian species such as mice, rats, hamsters, rabbits, swines, guinea pigs, dogs, and nonhuman primates. 40,47 Although sharing similar lipid physiology and being closer to humans than birds, mammalian models (with the exception of primates) rarely develop spontaneous advanced lesions under normal circumstances and when being fed a standard diet. Therefore, the results obtained from experiments using inbred and genetically modified animals cannot easily be translated to spontaneous-occurring lesions in the same species and humans.
Here, we report the experimental dietary induction of advanced atherosclerotic lesions in Quaker parrots (Myiopsitta monachus), a small psittacine species susceptible to spontaneous atherosclerosis.
Materials and Methods
Experimental Design
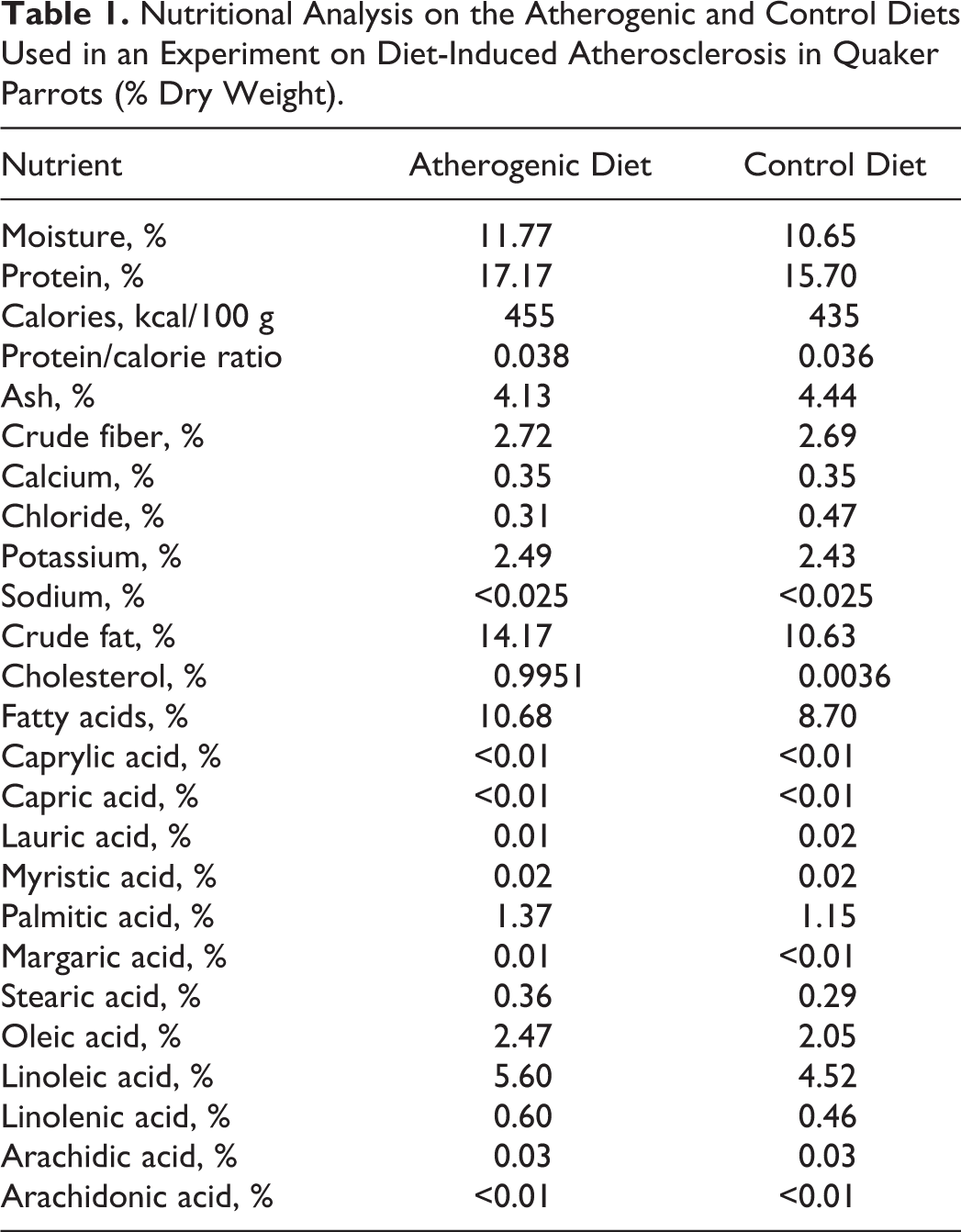
Sixteen captive-bred Quaker parrots (M.monachus) of unknown sex ranging in age from 6 months to 2 years were used in this study and were all purchased from the same private breeder. The birds were housed in pairs and kept in stainless steel cages in a laboratory animal facility. The research protocol was approved by the Louisiana State University–Institutional Animal Care and Use Committee. The parrots were acclimatized for 6 months and maintained on a commercial pelletized psittacine diet (Kaytee Exact Natural; Kaytee, Chilton, WI). The birds were randomized before the acclimatization process using statistical software (R; R development Core Team, Vienna, Austria) to 5 groups: 1 control group of 4 birds and 4 treatment groups of 3 birds each with differing lengths of treatment. A psittacine atherogenic diet was formulated and manufactured by Kaytee from the company’s maintenance parrot pellets formula (Exact Natural) with the addition of 1% cholesterol, keeping a similar calorie and micronutrient to protein ratio with the control diet to obtain similar daily caloric and protein intake among groups. The cholesterol was obtained as 92.5% cholesterol from sheep wool (C8503; Sigma, St Louis, MO). Both the atherogenic and control diets were submitted for nutritional analysis to an independent A2LA-accredited laboratory (Eurofins Scientific, Nutrition Analysis Center, Des Moines, IA) (Table 1). The atherogenic diet was fed for 2, 4, 6, and 8 months with 3 birds allotted to each time period. Each group of birds was euthanized at the end of their specific period by intravenous injection of potassium chloride under isoflurane anesthesia. The control group was fed the control diet. Two birds from the control group were euthanized before the study was initiated to ensure the lack of any detectable atherosclerotic lesions and the 2 remaining birds at the end of the study period at 8 months. Food consumption was monitored daily per pair of birds by providing 18 g of pellets per bird per day and weighing unconsumed food and food waste at the cage bottom before the subsequent feeding. All birds were weighted on a monthly basis until they were euthanized.
Nutritional Analysis on the Atherogenic and Control Diets Used in an Experiment on Diet-Induced Atherosclerosis in Quaker Parrots (% Dry Weight).
Samples Collection and Analysis
A 1-ml blood sample was obtained monthly from each bird from the right jugular vein and placed in heparinized tubes (Microtainer; BD, Franklin Lakes, NJ). For biochemical analysis, blood was centrifuged for 10 minutes at 1000 × g, and the plasma was harvested and stored at –80°C until analysis. The samples were used to measure packed cell volume, avian plasma biochemistry profile (Avian/Reptile rotor, Abaxis VetScan; Abaxis, Union City, CA), triglycerides, total cholesterol, and high-density lipoprotein (HDL) (Ortho Vitros 250 Chemistry Analyzer, Rochester, NY). Very low-density lipoprotein (VLDL) and low-density lipoprotein (LDL) concentrations were obtained using the Friedewald formula. 13 A larger blood sample (3 ml) was obtained under anesthesia prior to euthanasia. Plasma cholesteryl ester transfer protein (CETP) activity was measured using a commercial assay according to the manufacturer instructions (ab65383; Abcam, Cambridge, UK) and a complete blood count performed using the Leukopette technique. 11
The ascending aorta and brachiocephalic and pulmonary arteries were collected within 15 minutes of euthanasia, and sections were fixed in 10% neutral buffered formalin for histopathology and in 2% paraformaldehyde and 1.25% glutaraldehyde in a 0.1 M cacodylate buffer for transmission electron microscopy (TEM). The heart and liver were also collected and fixed in 10% neutral buffered formalin. Additional fresh sections of arteries and liver were stored at –80°C for cholesterol measurement. Sex was determined at necropsy. Formalin-fixed tissues were routinely processed into paraffin. Five-micrometer sections were cut and stained with hematoxylin and eosin, von Kossa for calcium salts, and Masson’s trichrome for collagen. Frozen formalin-fixed sections of arteries were also stained with oil red O (ORO) for lipid. Arterial tissues fixed for electron microscopy were processed, sectioned, and stained using standard techniques previously described for psittacine arterial samples. 8 In addition, 0.5-μm-thick sections obtained from the blocks used for TEM sections were stained with toluidine blue. TEM sections were mounted on collodion-coated copper grids, stained with Reynolds lead citrate, and imaged with a JEM-1011 transmission electron microscope (JEOL, Tokyo, Japan).
Atherosclerotic lesion types were categorized into types I to V according to a published classification system in parrots that mirrors the AHA classification. 8 The heart and liver were evaluated on histopathologic sections, and about 15 to 20 intramyocardial coronary arteries per heart of approximately 40 to 250 μm in diameter were also evaluated. Total cholesterol was measured in fresh arterial samples using a commercial assay according to the manufacturer instructions (ab65359; Abcam). Lesions were also qualitatively compared with spontaneous lesions in 16 pet Quaker parrots (age range, 2–16.5 years; sex: 6 males, 3 females, 7 unknown) obtained from a veterinary pathology laboratory (Zoo/Exotic Pathology Service, Sacramento, CA).
Statistical Analysis
Hepatic cholesterol content and CETP plasma activity between groups were compared using a Kruskal-Wallis test. Repeated measures over time were analyzed using linear mixed models with parrots as the random variable and treatment, time, and polynomial terms as fixed variables when appropriate. Arterial cholesterol content was similarly analyzed but with time and artery as fixed effects. Variance heterogeneity and dependence among observations were modeled using variance functions and serial correlation structures when appropriate. Competing models were evaluated based on the Akaike information criterion, and model assumptions were evaluated on residual and quantile plots. Parametric and nonparametric post hoc comparisons were performed with a Tukey adjustment. Correlations between lesion types in aorta and brachiocephalic arteries and plasma cholesterol at euthanasia or arterial cholesterol content were assessed using a Kendall τ correlation coefficient. Correlations between plasma cholesterol at euthanasia and arterial and liver cholesterol concentrations were assessed using a Spearman correlation coefficient. A P < .05 was used for statistical significance. R (R Development Core Team) was used for statistical analysis and the R package “nlme” for mixed modeling. 32
Results
Four of 16 Quaker parrots were females, and 1 female was in each group, except for the 8-month group. The Quaker parrots consumed an average of 9.9 g (95% confidence interval [CI], 9.4–10.3) of pellets daily (intercept-only mixed models). There was a significant time × treatment interaction effect on the daily food consumption (t = 15.6, P < .001). The control parrots had a food consumption that increased with time (approximately 20% in 8 months, t = 17.5, P < .001) and was, overall, greater than the parrots in the treatment groups (approximately 15%; t = 2.55, P = .025). Parrots in the treatment groups had a stable food consumption over time of 9.6 g (95% CI, 9.5–9.6) daily, corresponding to 84.3 mg (788.6 mg/kg) of cholesterol daily. The mean (SD) weight of the parrots was 106.9 (10.1) g, which did not change over time (t = –0.6, P = .55) or with treatments (t = –0.8, P = .86). No bird displayed any abnormal clinical signs during the study. Overall, there was a high interindividual variability for all measurements that frequently accounted for close to 70% of the total variance of the mixed models.
The mean plasma total cholesterol concentration in control birds was constant over time, with a mean (SD) of 6.5 (1.6) mmol/L, and overall was lower than the treatment group parrots (P < .001). The mean plasma cholesterol concentration as well as the variance in treatment groups increased significantly over time. It was best modeled using a cubic polynomial (cholesterol [mmol/L] = 3.3 + 27.3 × time-5.9 × time 2 + 0.4 × time3; P < .001, P < .001, P = .03, and P = .19, respectively) with a power of variance covariate for the variance structure (Var = σ2(time)2×0.87). Even if the cubic term was not significant, it brought substantial smoothing of the curve and an overall better fit to the data. The mean cholesterol concentration reached a plateau at about 3 to 4 months (Fig. 1).
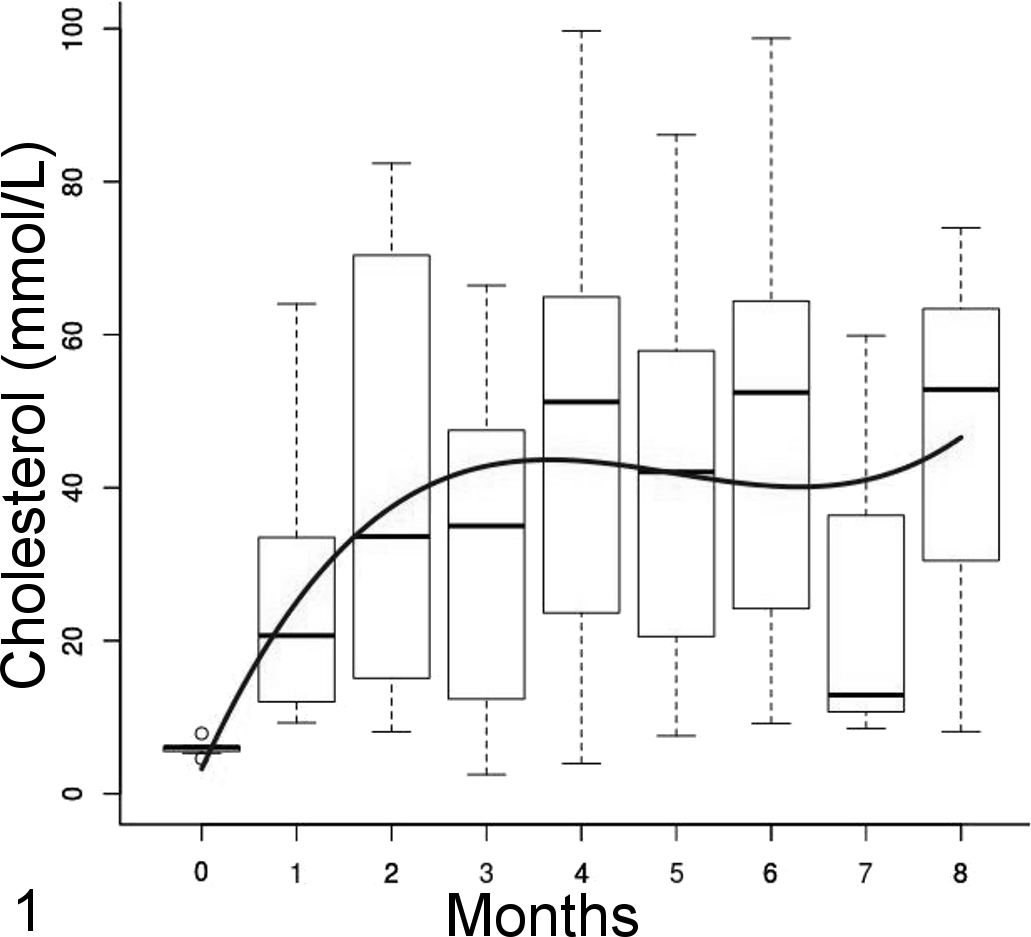
Plasma cholesterol concentration over time represented by side-by-side boxplots with a polynomial regression line. Mean concentration increased over time and reached a plateau at 3 to 4 months.
The mean plasma LDL cholesterol concentration in control birds was constant over time, with a mean (SD) of 0.5 (0.4) mmol/L, and overall was lower than in parrots in treatment groups (P < .001). The rising total cholesterol concentrations in treatment groups was explained by the LDL cholesterol, and thus it followed a similar trend (LDL [mmol/L] = –0.6 + 22.6 × time-4.0 × time 2 + 0.2 × time3; P = .3, P < .001, P = .04, and P = .34, respectively; variance structure: Var = σ2(time)2×0.89). Likewise, the mean LDL concentration reached a plateau at about 3 to 4 months (Fig. 2).
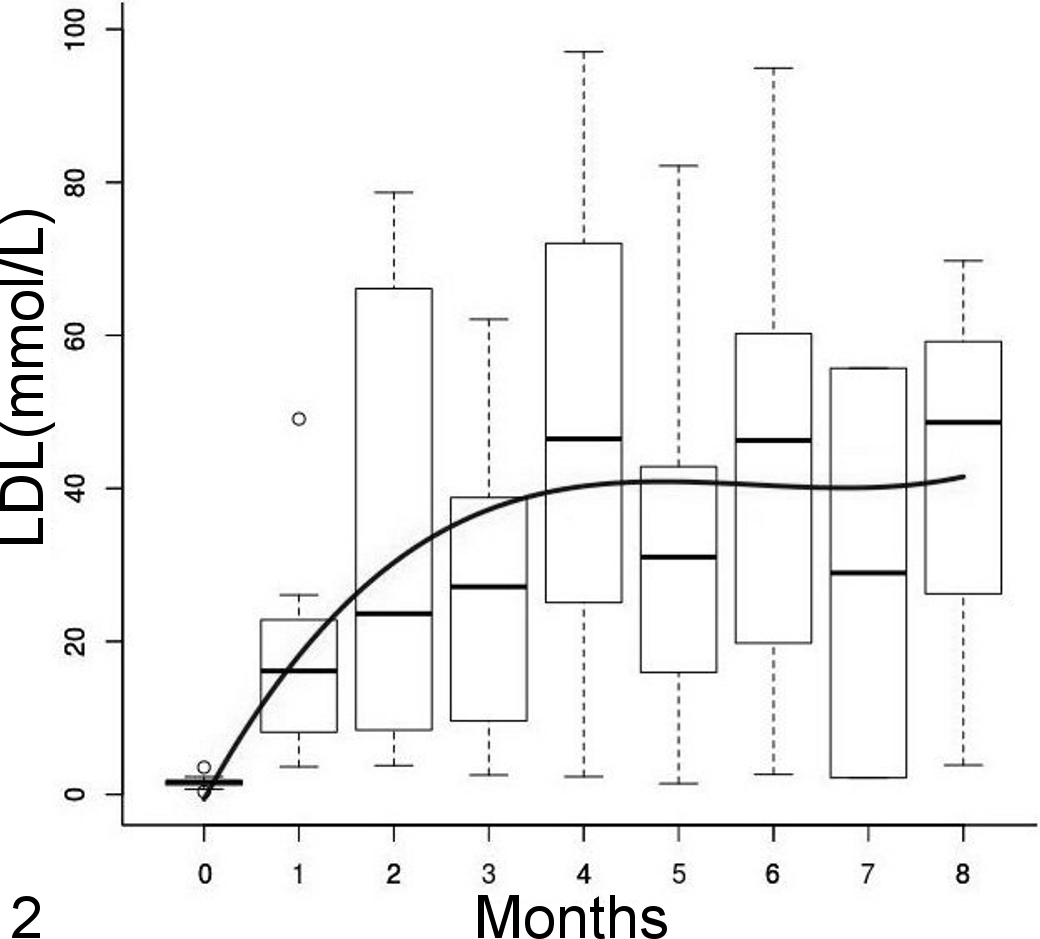
Low-density lipoprotein (LDL) cholesterol concentration over time represented by side-by-side boxplots with a polynomial regression line. Mean concentration increased over time and reached a plateau at 3 to 4 months.
The HDL cholesterol plasma concentrations did not change significantly over time (t = –0.5, P = .61) or between treatment and control groups (t = 0.19, P = .85), with a median (interquartile range) of 3.8 (2.7) mmol/L (Fig. 3). Mean triglycerides and VLDL cholesterol plasma concentrations did not change significantly between treatment and control groups (t = –4.8, P = .39) but slightly decreased over time (triglycerides [mmol/L] = 1.58 – 0.05 × time, all P < .001) (Fig. 4). HDL was the predominant cholesterol type in plasma before starting the feed trial, but LDL became preponderant on the atherogenic diet.
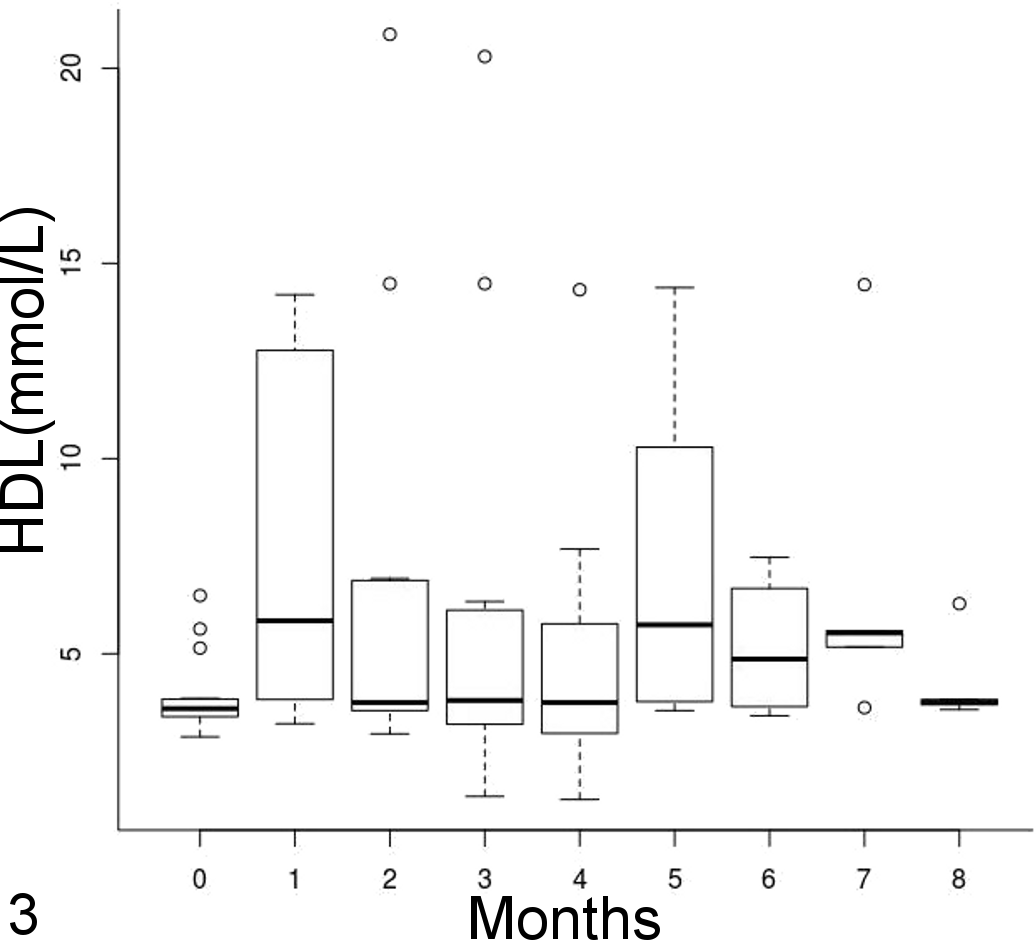
High-density lipoprotein (HDL) cholesterol concentration over time represented by side-by-side boxplots. No significant trend over time was identified.
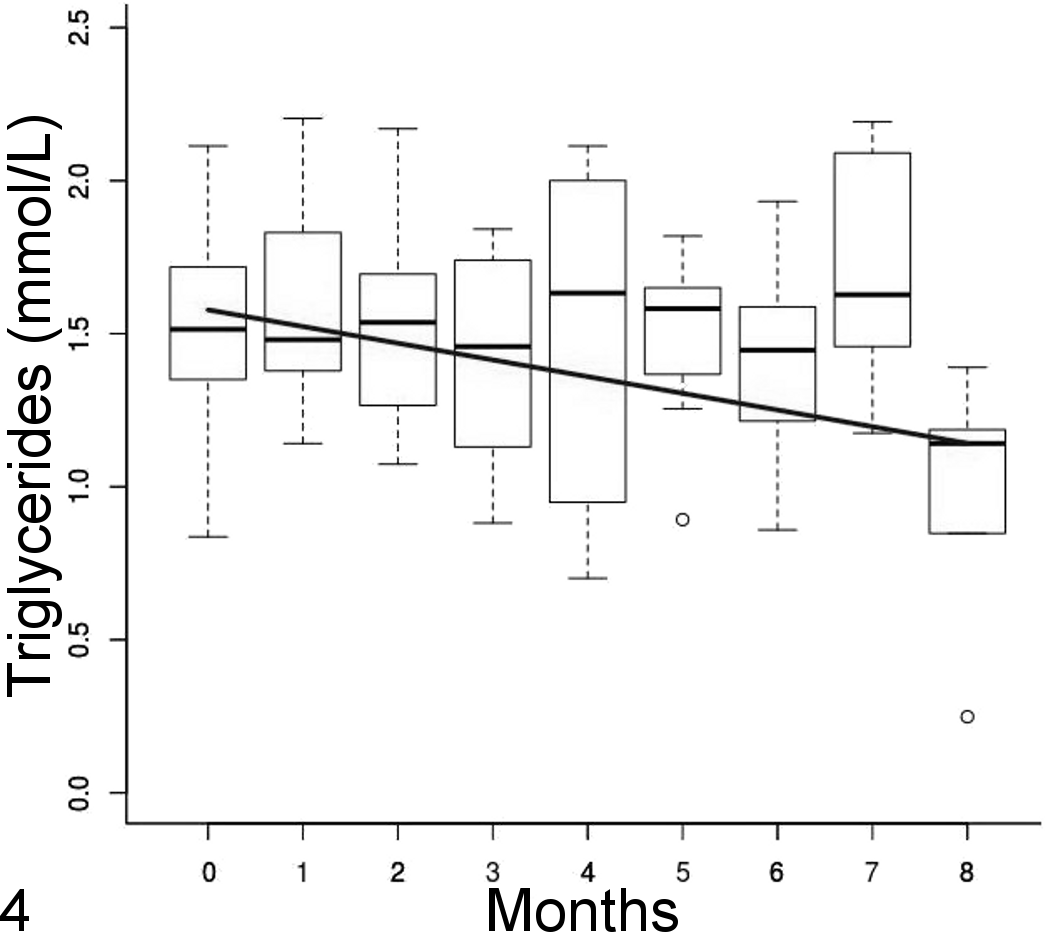
Triglycerides concentration over time with a linear regression line represented by side-by-side boxplots. Mean concentration decreased linearly over time.
No other biochemical and hematologic parameters showed any significant difference over time between control and treatment groups (all P > .05).
CETP activity was detected in all Quaker parrots’ plasma samples. Although CETP activity was generally greater in parrots on the atherogenic diet than on the control diet, the difference was not significant (Kruskal-Wallis test, P = .298) (Fig. 5). The median (interquartile range) was 45.80 (38.9–65.0) nmol/mL/h and 99.40 (46.10–148) nmol/mL/h in the control and treatment groups, respectively.
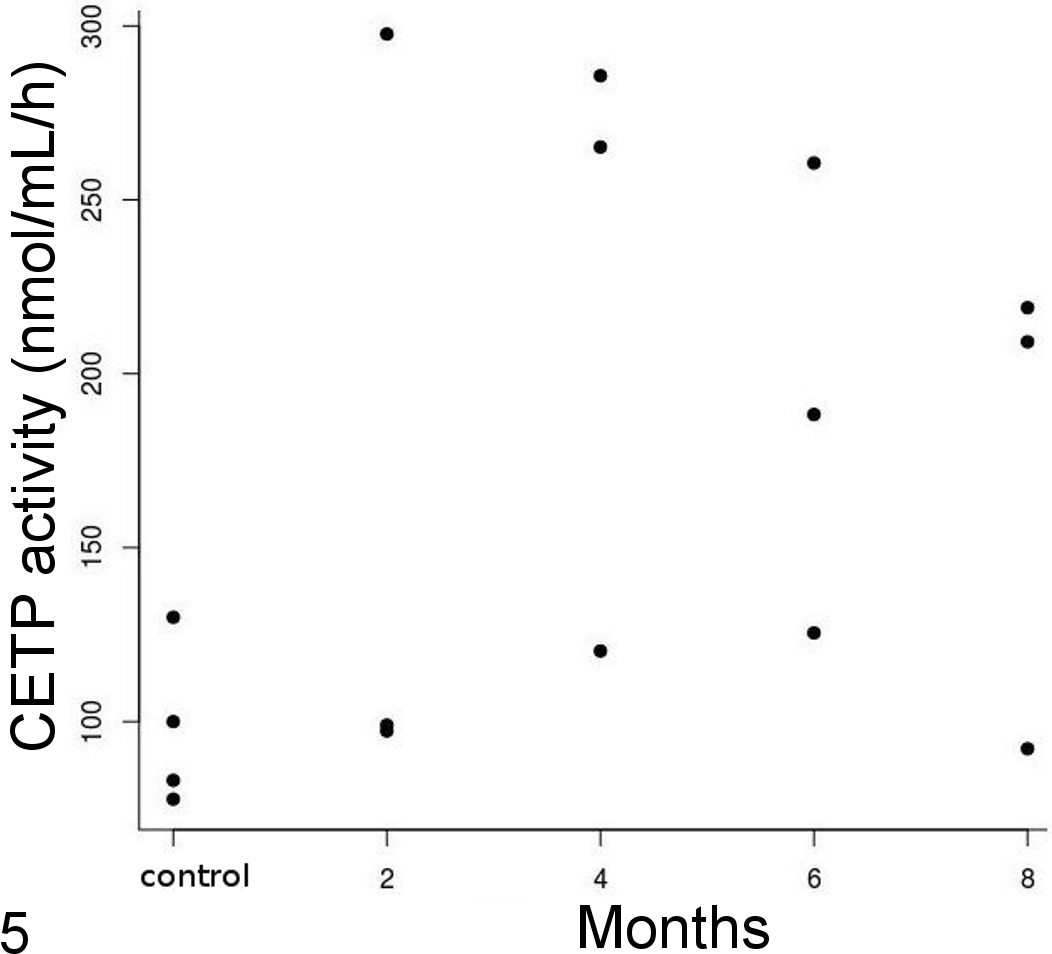
Plasma cholesterylester transfer protein (CETP) activity over time. Although plasma CETP activity was greater in treated birds than in control birds, differences were not significant.
For hepatic cholesterol concentrations, there were only significant differences between the control group and 4- to 8-month groups (Kruskal-Wallis test, P < .001) (Fig. 6). Arterial cholesterol concentrations linearly increased from control birds to the 8-month group (Fig. 7) with a significant treatment effect (F = 31.74, P < .001) but no difference between arteries (F = 0.72, P = .41). One outlier was removed to have an adequate fit and validate model assumptions. The outlier was an aortic sample in the 6-month group that differed markedly from the brachiocephalic sample within the same bird (Fig. 7). The variability in the pulmonary arterial samples was high due to the small quantity of artery that could be harvested and used in the assay, and the values were not considered reliable and thus not included in the analysis. However, pulmonary arterial samples appeared to have less cholesterol than other elastic arteries with a median (interquartile range) of 23.17 (18.16) μmol/g. There was a significant moderate correlation between plasma cholesterol at euthanasia and atherosclerotic lesion type (τ = 0.50, P < .001) and arterial cholesterol content (ρ = 0.64, P < .001), as well as a high correlation with liver cholesterol content (ρ = 0.81, P < .001). Arterial cholesterol content was highly correlated to the type of lesions (τ = 0.80, P < .001).
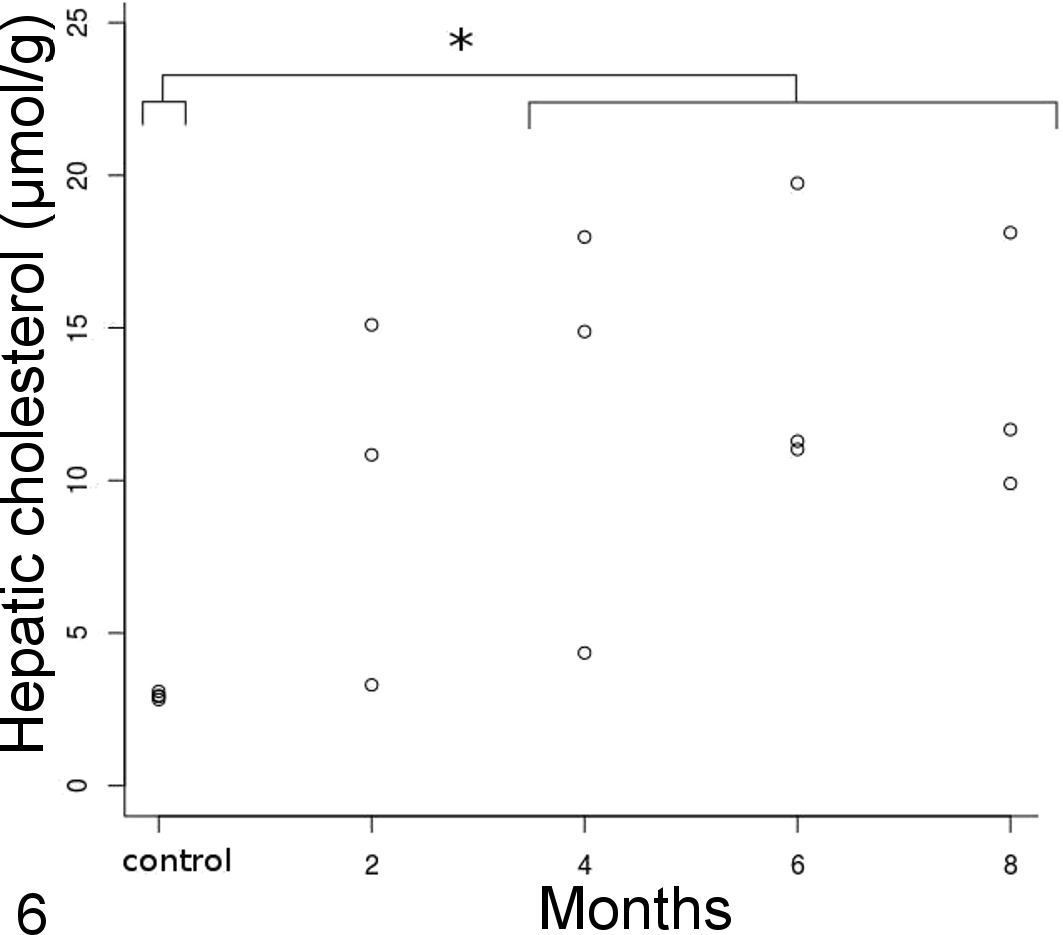
Hepatic cholesterol concentration over time. The 4- to 8-month group significantly differed from controls (*).
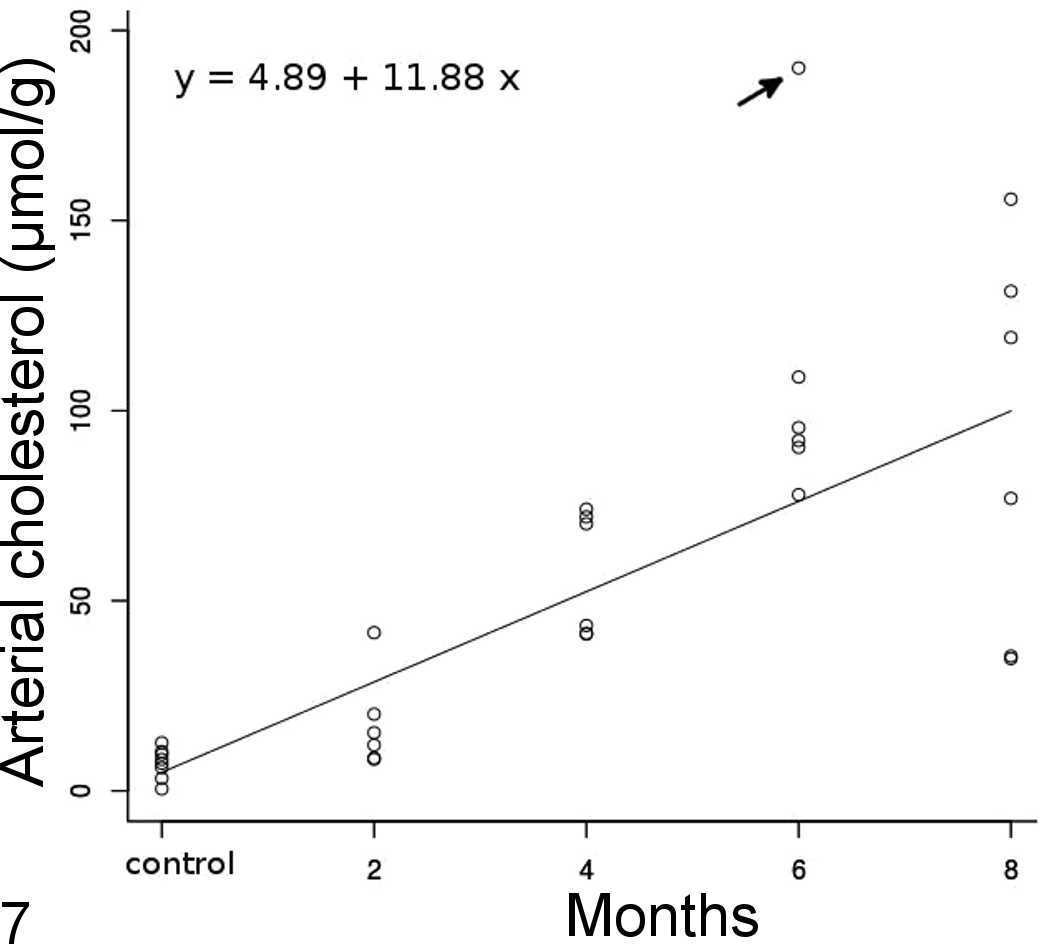
Arterial cholesterol concentration over time with a linear regression line (obtained from the fixed component of the mixed model; arrow: outlier). Each time point has 6 dots (some values may overlap) that correspond to 2 arterial samples, the aorta and brachiocephalic arteries, for 3 birds. Arterial cholesterol concentration linearly increased over time.
On histopathologic examination of harvested arterial samples, the 4 control birds did not have any evidence of atherosclerotic changes in any artery (Fig. 8). At 2 months on a 1% cholesterol diet, 1 bird had atherosclerotic lesions of type III in the aorta and brachiocephalic arteries and a type I lesion in the pulmonary arteries. The type III lesions were extensive, proliferative, and diffuse; encompassed the full circumference of the arteries; and displayed significant fibrosis as evidenced by the trichrome stain (Fig. 9). The 2 remaining birds had type I lesions in the aorta and brachiocephalic arteries, mainly characterized by mild ORO-positive staining and scattered lipid vacuoles on toluidine blue stain but no evidence of lesion or lipid in the pulmonary arteries. At 4 months, all treated birds exhibited advanced lesions of type IV, extending to the whole circumference of the aorta and brachiocephalic arteries and characterized by fatty proliferation, intense ORO staining, stenosis of the lumen, mild calcification (on von Kossa stain), and high but diffuse collagen expression (Figs. 10, 11). Pulmonary arteries displayed none to mild lesions of types 0 to II. At 6 months, all birds had type IV to V lesions in the aorta and brachiocephalic arteries with a well-delimited lipid core, mild calcification, and the presence of a thin fibrous cap overlying the atheroma in 1 bird. Pulmonary arteries exhibited type II lesions in 2 birds and type I in 1 bird. At 8 months, 2 birds developed advanced lesions of type V with a fibrous cap and moderate calcification in some areas of the atheroma (Figs. 11, 12, 13). The pulmonary arteries had only mild lesions of type II. However, 1 bird developed only mild lesions despite being 8 months on the atherogenic diet with type I to II lesions in the aorta and brachiocephalic arteries and no lesion in the pulmonary arteries.
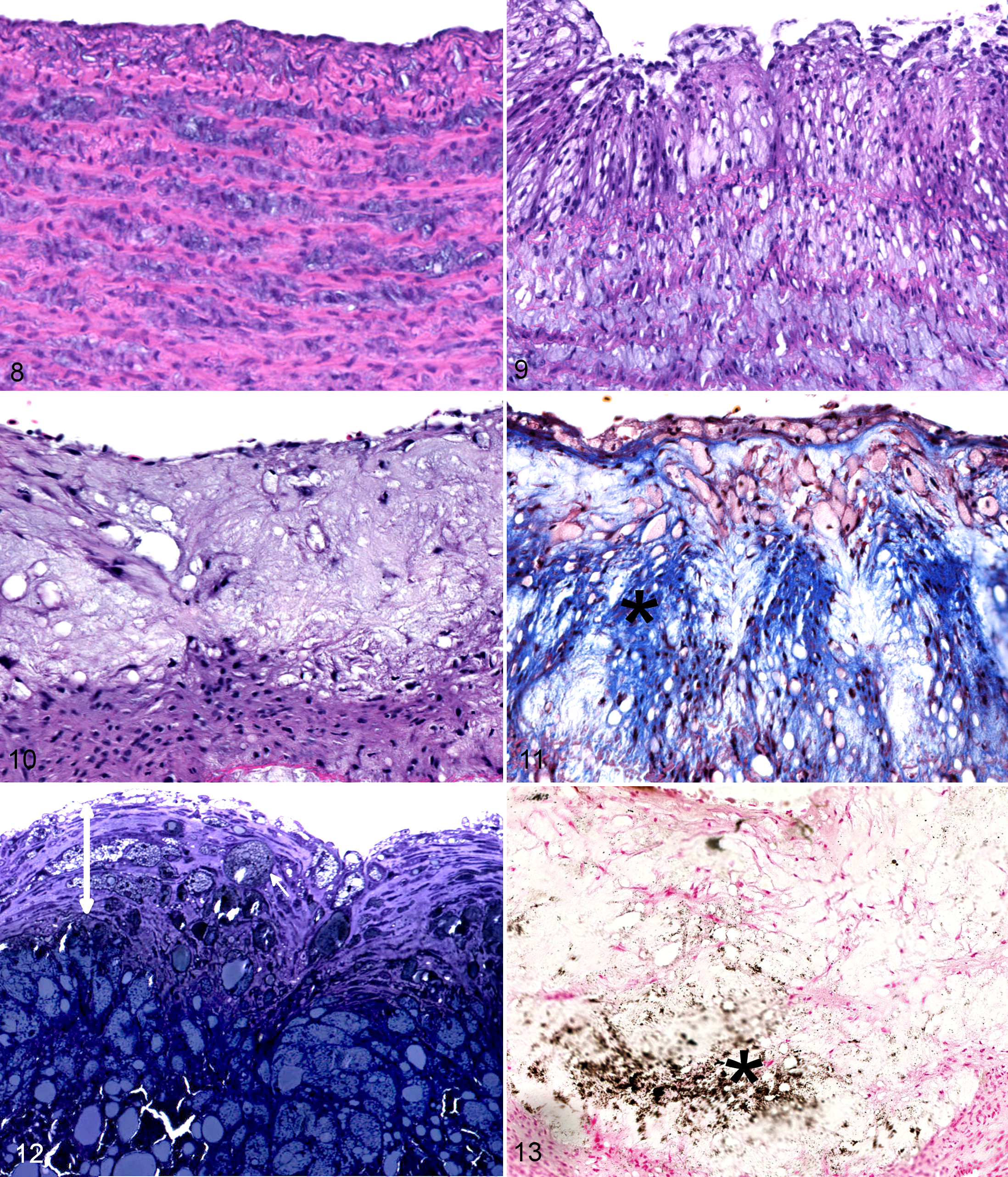
Spontaneously occurring lesions in 16 other Quaker parrots appeared more organized, and the different areas of the fibroatheromas were better defined when compared with experimental lesions. Also, experimental lesions of types III to IV were more diffuse, vacuolated, and typically involved the entire circumference of the arteries, whereas spontaneous lesions of these types were usually more circumscribed and less vacuolated. Finally, calcification was often more advanced in spontaneous lesions than in experimental lesions, and chrondroid metaplasia and ossification were not observed in the latter. Advanced lesions characterized as type V appeared histologically similar between spontaneous and experimental atherosclerosis in Quaker parrots.
No histologic abnormalities, except coronary atherosclerotic lesions, were evident in the hearts of any of the 16 birds. The birds in the control and 2-month groups had no observed atherosclerosis in the coronary arteries. At 4 months, all birds had atherosclerotic changes, mainly type III lesions, but with complete obstruction of the arterial lumen in some coronary arteries by some type III to IV lesions. At 6 months, 2 birds had advanced type IV lesions, characterized by segmental arterial walls almost transmurally replaced by foam cells and extracellular lipid, but no atherosclerotic lesions were observed in 1 bird. At 8 months, only 1 bird demonstrated coronary atherosclerotic lesions of type III. In the hepatic sections, prominent hepatocellular vacuolization was observed in 2 birds, 1 at 6 months and 1 at 8 months.
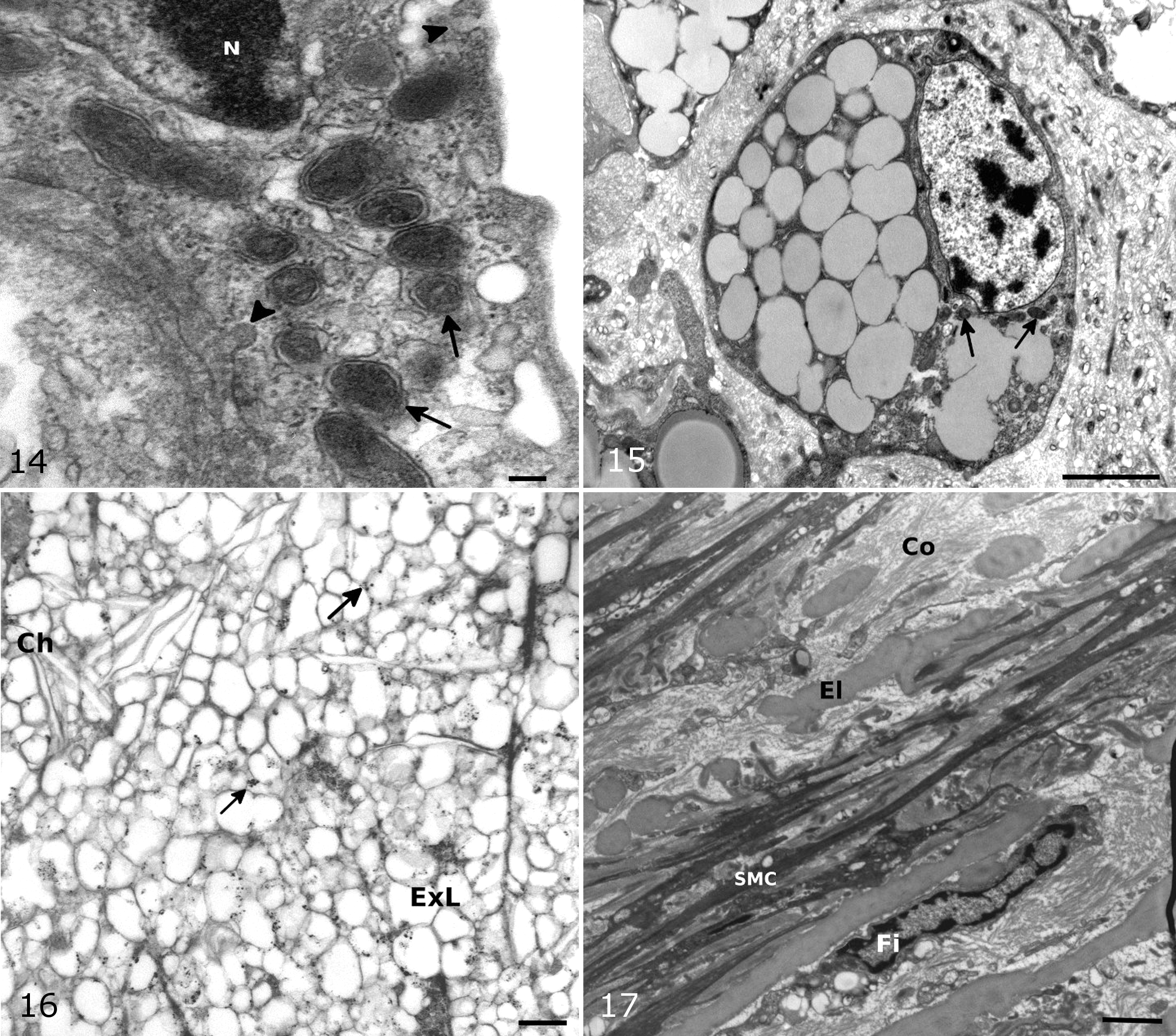
On TEM, the normal intima showed a single layer of endothelial cells with various organelles, plasmalemmal vesicles, and short microvilli. The media were mainly composed of 2 types of cells: smooth muscle cells and interlaminar fibroblast-like cells. Overall, extracellular fat accumulation seemed to precede the presence of foam cells. In type I lesions, foam cells were rare, but extracellular lipid vacuoles were present throughout the intima and media. Some endothelial cells in treated birds seemed to have more cytoplasm, filopodia, vacuoles, and Weibel-Palade bodies (which are spherical and granular in birds) than in control birds (Fig. 14). In addition, a few monocytes were seen to adhere to endothelial cells in some sections. Some smooth muscle cells (SMCs) showed phenotypic changes to a more synthetic state with an increased presence of smooth and rough endoplasmic reticulum and plasmalemmal vesicles. There also seemed to be an increased number of fibroblasts. In type II to III lesions, there were moderate to large pools of extracellular lipid containing small and large lipid droplets, cholesterol crystals, cellular debris, myelin figures with disorganization of elastin and collagen fibers, and increased extracellular matrix. Although lipid remained primarily extracellular, characteristic foam cells were present, mainly in type III lesions (Fig. 15). The cellular origin of foam cells was difficult to determine based on morphology alone, but both SMC and macrophage-derived foam cells were present. In the type IV to V atherosclerotic lesions, there was major disorganization of the arterial ultrastructure with large areas devoid of normal cells filled with lipid and extracellular matrix components, cellular debris, and foam cells (Fig. 16). The fibrous cap was composed of collagen, elastin fibers, and fibroblast-like cells (Fig. 17). Large numbers of foam cells were present immediately subjacent to the fibrous cap.
Discussion
This study showed that atherosclerosis could be induced in Quaker parrots in as little as 2 months with a 1% cholesterol diet. Moreover, advanced atherosclerotic lesions developed by 4 months and were characterized by a large circumferential lipid core and a fibrous cap. At 6 months, lesions appeared histologically analogous to spontaneous advanced lesions in the same parrot species and showed significant luminal stenosis. The chronology of lesion progression in the aorta and brachiocephalic arteries seemed to follow the previously established psittacine classification system, with the type V lesions being present only at 6 and 8 months. 8 Coronary lesions also developed at 4 months, but owing to the small sizes of these arteries (about 40–250 μm) in Quaker parrots, categorization of the lesions was less precise. Lesion severity and arterial cholesterol content increased linearly with time, and both were significantly correlated to plasma cholesterol concentration. Pulmonary arteries displayed only mild to moderate lesions that never progressed to advanced lesions. Although severe pulmonary lesions have been described in association with clinical signs in psittacine birds, 37 only mild to moderate lesions were noted in this group of birds. Spontaneous lesions appeared more organized, more calcified, and less diffuse than experimental lesions, possibly reflecting the slow development of spontaneous lesions compared with the accelerated rate of experimental lesion formation. The different appearance of atherosclerotic lesions has also been noted in pigeons fed with different concentrations of cholesterol. 20 Atherothombosis, plaque rupture, and cardiac ischemia were not encountered, but it is possible that complicated lesions eventually would have developed. On the other hand, these complications are common in White Carneau pigeons. 33,41 A recent experiment in budgerigars (M. undulatus) revealed that in vivo carotid artery injury did not lead to the formation of occlusive thrombi contrary to mice, in which carotid diameter and blood flow are similar to budgerigars. 36 Avian thrombocytes seem less capable of forming shear-resistant arterial thrombi and do not usually form 3-dimensional aggregates. The physiologic differences between avian thrombocytes and mammalian platelets may partly explain the clinical differences observed in the nature and prevalence of atherothrombotic diseases. In addition, coronary arteries were significantly stenotic in a few instances, which could theoretically lead to chronic ischemic cardiac lesions with longer periods of cholesterol feeding. However, arteries were not fixed under physiologic pressure, which renders difficult the quantification of the degree of coronary stenosis.
During this research investigation, the treated Quaker parrots also exhibited dyslipidemia with a shift from HDL to LDL as their main cholesterol carrier lipoproteins. The dyslipidemia developed quickly and stabilized at about 3 to 4 months, at which total and LDL cholesterol reached a plateau. This shift from lipoprotein profiles and similar levels of cholesterol were also observed in quails and pigeons after cholesterol feeding. 2,5,15,18,28,30 As with these parrots, change in plasma HDL is usually not observed in birds during cholesterol feeding. 2,5,15,17,19,35 Furthermore, the Quaker parrots displayed a high plasma CETP activity, which, although not statistically significant, increased on the atherogenic diet. This enzyme transfers cholesteryl esters to LDL from HDL, and an increase in its activity has been described with atherogenic dyslipidemia in both humans and animal experimental models. 42 The response to dietary cholesterol varies among species and individuals, and different concentrations of this compound have been used (0.05%–5%) in formulated bird diets to induce atherosclerotic lesions with variable severity, prevalence, and rate of development. 5,18,34,38 Within a same species, the induction of atherosclerosis depends on the line of birds, the level of dietary cholesterol, and the duration of the feeding period. 5,18,34,38 It appears from our study that advanced atherosclerotic lesions can be induced relatively quickly in Quaker parrots with 1% dietary cholesterol as all birds in the 2-month group had significant arterial lesions. In budgerigar, significant atherosclerotic lesions were induced by feeding a diet that contained 2% cholesterol and 15% lard for 6 months, but there was concurrent development of hepatic lipidosis. 12 A concentration of 0.5% to 1% is usually required to generate significant atherosclerotic lesions in a short time span (months), but more physiologic levels (0.15% cholesterol, Western-type diet) are also being used to better mimic the slow progression of the disease when time is not a constraint. 5 Japanese quails have been particularly bred and selected to produce atherosclerosis rapidly with less dietary cholesterol. 5,35,38 Similar results may be achievable in Quaker parrots through selection to obtain a more homogeneous population (variability was high here), which would allow the investigator to reduce the number of animals required to achieve statistically relevant results, cholesterol level, and time to induce atherosclerotic lesions. Other dietary modifications have been used in conjunction to cholesterol in avian models, including increased saturated fat level and cholic acid supplementation, which induces an inflammatory state and aids in cholesterol absorption. 5,35 In our study, only the cholesterol content of the diet was altered and the formula was controlled for caloric and micronutrient intake to limit potential confounding factors of inflammation induced by cholic acid, increased weight and fat reserve on metabolism, and dilution of other nutrients, as well as to provide more reproducible dietary modifications.
Among avian models of atherosclerosis, histopathology and ultrastructural pathology have mainly been reported in White Carneau pigeons and Japanese quails. 3,16,18 –20,23,38 Although parrots and quails principally develop lesions in the ascending aorta and brachiocephalic arteries, pigeons preferentially acquire lesions in the lower thoracic aorta, especially at the celiac bifurcation. 4,18,20
Spontaneous and experimental lesions in parrots, pigeons, and quails are similar to human lesions with the presence of foam cells, extra- and intracellular lipid, a lipid core, a fibrous cap, and calcification. 8,41 Early lesions in pigeons are characterized by endothelial activation and monocyte adherence to the endothelium. 20,22,24,43 Endothelial changes noted in this study suggested endothelial activation and degeneration with adherent monocytes being observed in a few instances. In parrots, as in pigeons, SMC- and macrophage-derived foam cells are present in spontaneous and experimental lesions. 8,16,19,23 The large amount of extracellular lipid present in early lesions (types I–II) in the Quaker parrots contrasts with spontaneous early lesions in the same species of parrots that are primarily characterized by foam cells and intracellular lipids. 8 More characteristic foam cells were present in further advanced experimental lesions. Similar to pigeons, quails, and other birds but in contrast to mammalian species, interlaminar fibroblast-like cells were present in the media and may actively participate in fibrosis and synthesis of constituents of the extracellular matrix during atherogenesis. 16,21,29,44,48 As evidenced in lesions from parrots, remodeling with increased deposition of collagen is a hallmark of atherosclerotic lesion maturation and fibrous cap formation and is similar to other avian models. 27,45
Even if the advanced lesions seem identical between diet-induced and spontaneous atherosclerosis, the mechanisms of lesion formation and progression may differ owing to the compressed experimental time frame. Birds also have major differences in lipoprotein metabolism (absence of apolipoprotein B48, apolipoprotein E, and chylomicrons; HDL as the main carrier lipoprotein; and drastic changes during egg laying) and arterial microanatomy (no SMC in the intima and fibroblasts in the media) compared with humans. 1 Nevertheless, all experimental models have limitations, and even with animals more closely related to humans, some findings may not translate well to the human disease. 25 For instance, transgenic mice primarily develop lesions in the proximal great vessels and rarely get lesion complications, as observed in parrots. 25 Similarities in plasma biochemical changes, lesion composition, pathology, ultrastructure, and progression suggest that the pathogenesis of atherosclerosis in Quaker parrots is comparable to other avian models investigated and humans. Therefore, Quaker parrots could be used to study some aspects of the pathogenesis and therapeutics of atherosclerosis. The Quaker parrot is certainly a good model to study spontaneous atherosclerosis in psittaciformes, which commonly develop atherosclerosis-related diseases under standard captive conditions and may share common risk factors with humans.
In conclusion, Quaker parrots may be a useful model of atherosclerosis for parrots and humans as this species is small, has low food consumption (10 g/d), and rapidly develops advanced atherosclerotic lesions with cholesterol feeding that resemble lesions found in both humans and other avian models. In addition, advanced atherosclerotic lesions (type V) induced experimentally are similar to spontaneous-developing lesions within the same species.
Footnotes
Acknowledgements
We thank Mr Rich Bonkowski and Dr Randal Brue (Kaytee) for formulating and providing the atherogenic and control diets, the LSU-Division of Laboratory Animal Medicine for their excellent care of this Quaker parrot colony, Dr João Brandão for his technical help, Dr Ying Xiao (Miscroscopy Center) for processing tissues for transmission electron microscopy, Dr Brian Marx (Experimental Statistics, LSU) for his advice on statistical analysis, and Dr Drury Reavill (Zoo/Exotic Pathology, Sacramento) for providing the spontaneous atherosclerosis cases in Quaker parrots.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Kaytee Avian Foundation and a VCS corp grant (LSU-SVM).
