Abstract
This paper is a retrospective morphologic study of 7 young Boxer dogs, showing end-stage kidney lesions compatible with chronic pyelonephritis with severe segmental cortical atrophy and fibrosis, associated with chronic tubulointerstitial inflammation of varying degree. Azotemia was observed in 6 of the 7 cases. The gross kidney lesions were as follows: bilateral small kidneys with numerous segmental cortical scars causing depression of the renal cortical surface. Histologic examination revealed salient atrophy of nephrons, including paucity of glomeruli, glomerulocystic lesions, colloid-filled tubular microcysts, and a conspicuously increased occurrence of arteries with narrowed lumina caused by intimal thickening. These segmental abnormalities were accompanied by pronounced interstitial fibrosis. All but 1 dog showed salient tubulointerstitial lympho-plasmacytic infiltration, which in 3 cases also included diffuse infiltration of polymorphonuclear neutrophilic leukocyte (PMN)-cells and occurrence of tubular PMN-casts. Morphologic signs of abnormal metanephric differentiation (renal dysplasia) were observed in all cases in the form of atypical tubules or asynchronous nephronic development (immature glomeruli) or both. However, other morphologic primary dysplastic features were absent. Based on the morphologic features, it is concluded that the end-stage kidney disease in these young Boxer dogs was the result of chronic atrophic nonobstructive pyelonephritis, most probably caused by vesico-ureteral reflux, compatible with reflux nephropathy causing segmental hypoplasia (Ask-Upmark kidney) in man. It is proposed that atypical tubular epithelium in the form of adenomatoid proliferation of collecting duct epithelial cells should be considered an acquired compensatory lesion, rather than the result of disorganized metanephric development.
Keywords
Introduction
Chronic renal failure with end-stage kidney lesions in the juvenile, adolescent, or young adult dog is often congenital and often inherited. 9, 11, 13 Such so-called canine juvenile nephropathies may have many causes.
Both the well-defined hereditary nephropathies and the less well-understood juvenile nephropathies manifest themselves as small kidneys in the juvenile, adolescent, or young adult dogs suffering from chronic renal failure. While the congenitally small kidneys in cases of canine familial nephropathies should be considered to represent acquired small, atrophic kidneys, the small kidneys of the canine juvenile nephropathies frequently are considered to represent congenitally small, anomalously underdeveloped kidneys. Renal hypoplasia, a quantitative renal defect, is considered to occur infrequently. 11 Thus, the prevailing comprehension in veterinary nephropathology is that most small kidneys probably are secondarily scarred or the result of disorganized development of the renal parenchyma because of anomalous differentiation, i.e., renal dysplasia. 11 In the veterinary medical literature there has been a steadily increasing number of reports on canine renal dysplasia affecting a large number of different breeds. There have been relatively few reports from the Boxer breed, but most of these have concluded that renal dysplasia is also a cause of the juvenile nephropathies observed in this breed. 5, 7, 10 We report here our results from a retrospective study of renal lesions observed in young Boxer dogs with end-stage renal disease in Norway.
Materials and Methods
Four carcasses of uremic and/or azotemic Boxer dogs were submitted for autopsy to the National Veterinary Institute in Norway between January 1998 and October 2005 (dog Nos. 1, 5, 6, and 7). Formalin-fixed whole kidneys were submitted for pathologic evaluation from 2 dogs (dog Nos. 2 and 4), whereas only formalin-fixed coronal cut kidney slices were obtained from 1 dog (dog No. 3). The dogs were 4 intact females and 3 intact males, aged from 2 months to 5 years (median age 24 months). Six of the cases had a complete clinical record, including blood analyses (serum biochemistry and hematology), and a partial or complete urinalysis had been performed in 5 of the cases.
At autopsy, coronal slices from the kidneys were fixed by immersion in phosphate-buffered 4% formaldehyde, dehydrated in graded alcohols, and embedded in paraffin. Four-micrometer-thick sections were stained with hematoxylin and eosin (HE), elastin van Gieson (evG), periodic acid–Schiff (PAS), Perls' stain, and the von Kossa stain. Sections were examined under the light microscope, and the severity of lesions was assessed semiquantitatively using a scale from 0 to 3 (0 = normal, 1 = mild, 2 = moderate, 3 = severe). Microphotographs were captured using a Nikon DS-5M-U1 digital camera mounted on a Nikon Eclipse 50i light microscope (Nikon Instech Co., Kawasaki, Kanagawa, Japan).
Results
Disease history and physical examination findings
The records of the dogs, derived from 5 different clinics, were reviewed retrospectively. The range of ages at first clinical signs was 2 months to 2.5 years with a median of 13 months.
The most common clinical signs were poor body condition (6 dogs), anorexia, vomiting (5 dogs), and fatigue (4 dogs). Polydipsia and dermatological lesions were reported in 3 dogs. Other signs included polyuria, nocturia, ascites, dyspnea, altered behavior, seizures, ataxia, and death.
Complete blood analyses with serum biochemistry and hematology were available in 6 of the 7 Boxers, all of which were azotemic. Common findings were hyperphosphatemia and anemia (Table 1).
Results of blood analyses, blood chemistry, and hematology.
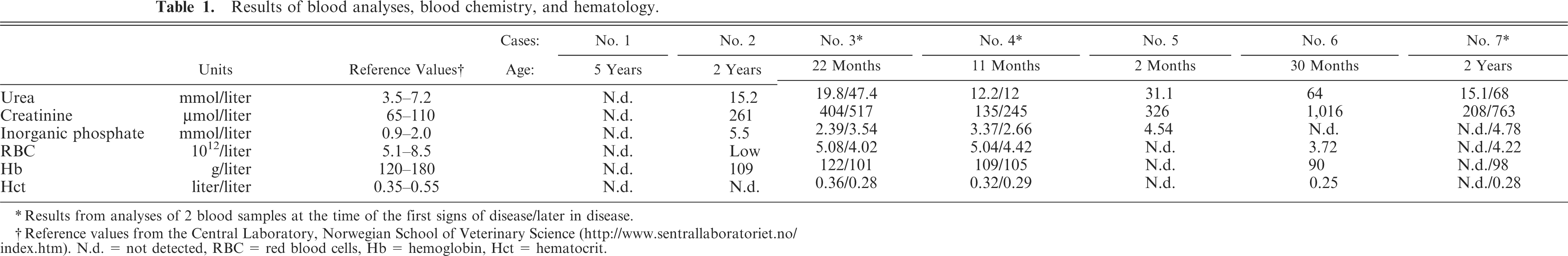
Results from analyses of 2 blood samples at the time of the first signs of disease/later in disease.
Reference values from the Central Laboratory, Norwegian School of Veterinary Science (http://www.sentrallaboratoriet.no/index.htm). N.d. = not detected, RBC = red blood cells, Hb = hemoglobin, Hct = hematocrit.
Urinalysis was performed in 5 of the 7 cases. Hyposthenuric urine (urine specific gravity, USG = 1,007) was found in 2 dogs (Nos. 3 and 4), isosthenuric urine (USG = 1,010) in 2 dogs (Nos. 5 and 6), and minimally concentrated urine (USG = 1,015) in 1 dog (No. 2). Four of the 5 dogs had proteinuria (Nos. 3, 4, 5, and 6) and 2 had hematuria (Nos. 3 and 4). Moderate numbers of white blood cells, red blood cells, and epithelial cell casts were found on urine sediment analysis from dog Nos. 3 and 4. Bacteriologic evaluation of urine was not performed in any of the dogs.
Six of the 7 Boxers had a history indicating kidney disease, based on blood analysis and classic signs of renal disease. However, Boxer dog No. 1 showed no obvious signs of renal disease. At an age of 13 months, this dog developed posterior ataxia and received a long-term glucocorticoid treatment, without a specific etiologic diagnosis. At 5 years of age the dog developed dyspnea and ascites, and eventually died.
Four dogs received different kinds of supportive therapy (diets, intravenous fluids, antibiotics, vitamin B, and/or nandrolone) for periods ranging from 3 days to 8 months. Two dogs were not treated, and 1 dog died before a diagnosis was made. The others were euthanatized.
Genetic relationship
The pedigrees of the dogs were studied to identify possible genetic relationships. Six dogs were registered in the Norwegian Kennel Club and 1 dog in the Swedish Kennel Club. Six of the 7 dogs were related (Fig. 1) through one mating (“P-generation”), representing generations F2–F4. Both males and females were potential carriers. The unrelated dog (No. 1), which had a different set of clinical signs, was imported from the United States at 7 months of age and was the only dog with a non-European origin.
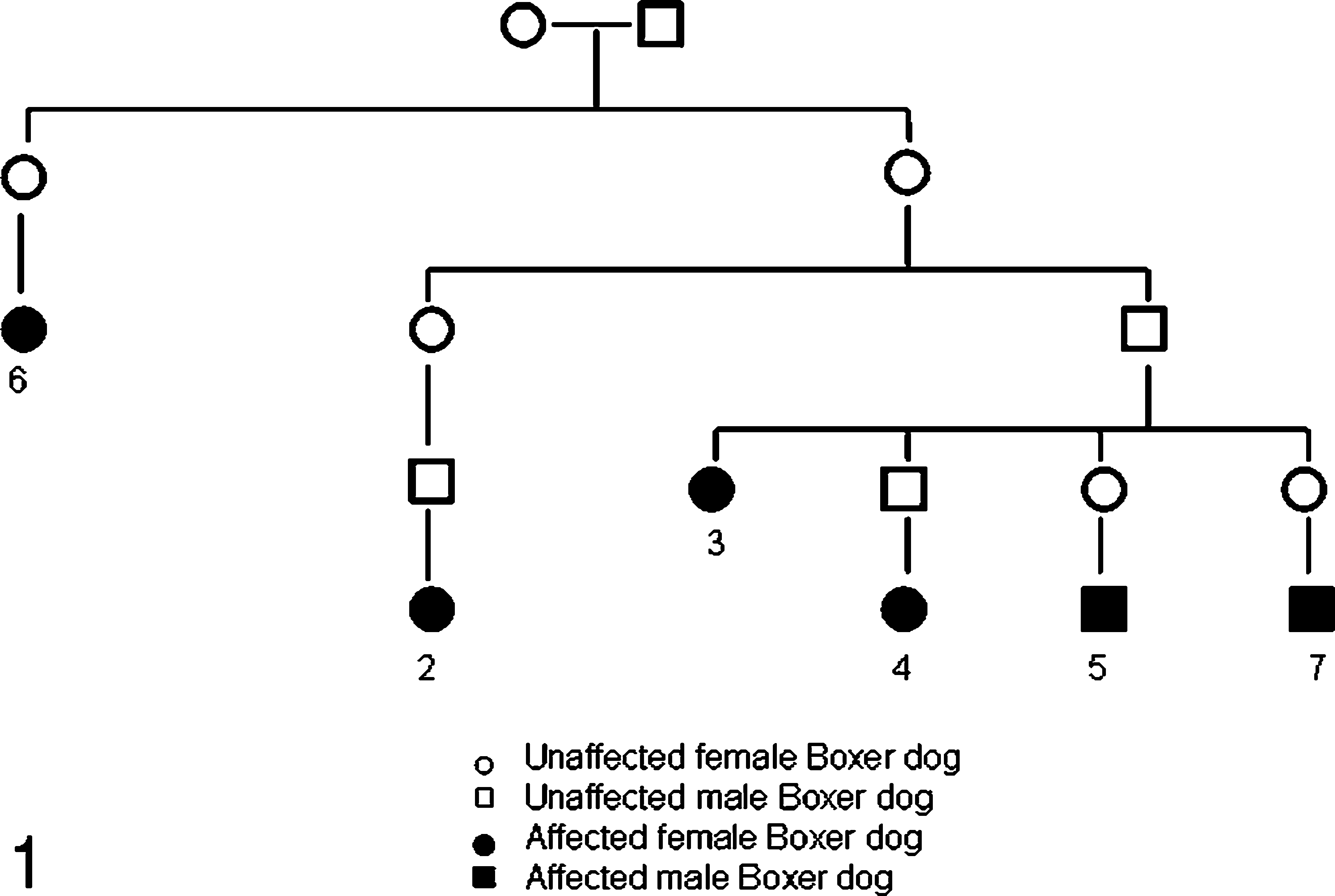
The genetic relationship between 6 of the 7 Boxer dogs in the study. The 6 related dogs were all ancestors from 1 mating. Numbers refer to the dog's number in the study. Siblings are not included.
Macroscopic renal lesions
Both kidneys from 6 of the dogs were generally small, firm, and pale with irregular capsular surfaces. The capsular irregularities had a coarse nodular appearance caused by numerous segmental cortical indentations or depressions interposed between hypertrophic remnants of renal parenchyma. In 4 dogs (Nos. 2, 4, 6, and 7), the kidneys were asymmetric. The right kidneys in dogs 2, 4, and 6 were almost half the size of the left kidneys, whereas the left kidney was the smallest in dog 7. Only fixed kidney slices were available from dog No. 3, and no data were provided regarding the size of these kidneys.
The renal capsules were firmly adherent to the kidney surfaces in all of the 4 autopsied cases. On the cut surfaces, the cortices were unevenly thinned and contained irregular, pale, scarred radial bands that caused segmental depressions of the cortices (Fig. 2).
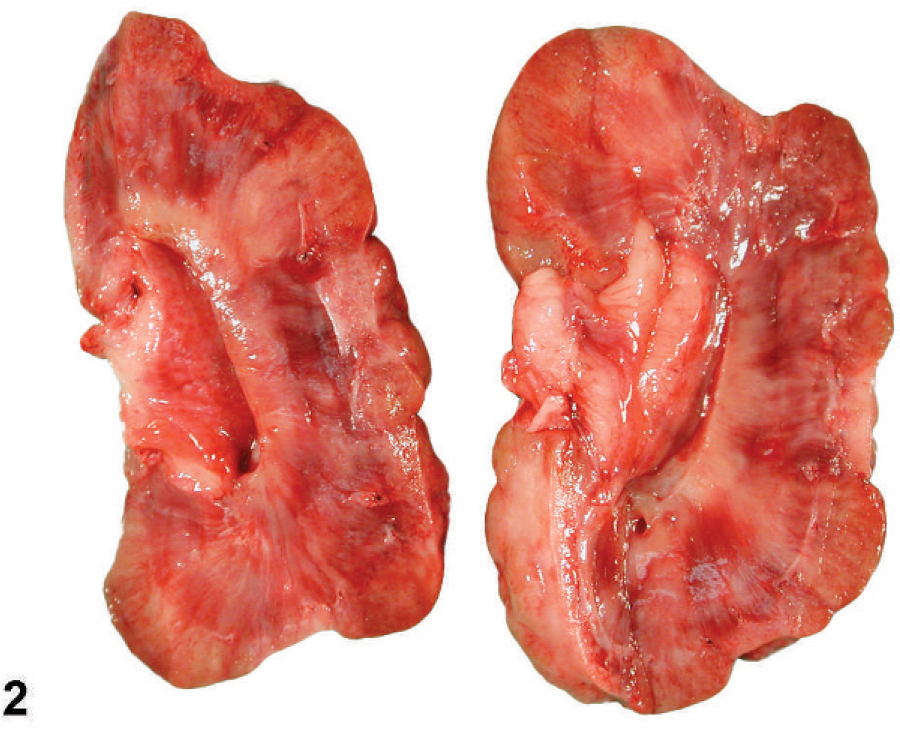
Kidney; Boxer dog No. 6. Cut surfaces of the left kidney. Note the irregular depressions of the cortices.
Larger cortical cysts (5–15 mm) were seen both on the surface and on the cut surfaces in 2 dogs (Nos. 4 and 7), and numerous smaller cysts (<1 mm) were seen on the cut cortical surfaces in 4 of the dogs (Nos. 2, 3, 4, and 7).
In the dogs submitted for autopsy, no obvious macroscopic lesions were found in the renal pelvis or lower urinary tracts, except in dog No. 6, in which both ureters were dilated and filled with a gray, pus-like mucous. In this case the vesica contained urine mixed with mucoid material. In all autopsied cases, the vesico-ureteral junctions were judged macroscopically to be normal.
Light microscopic lesions
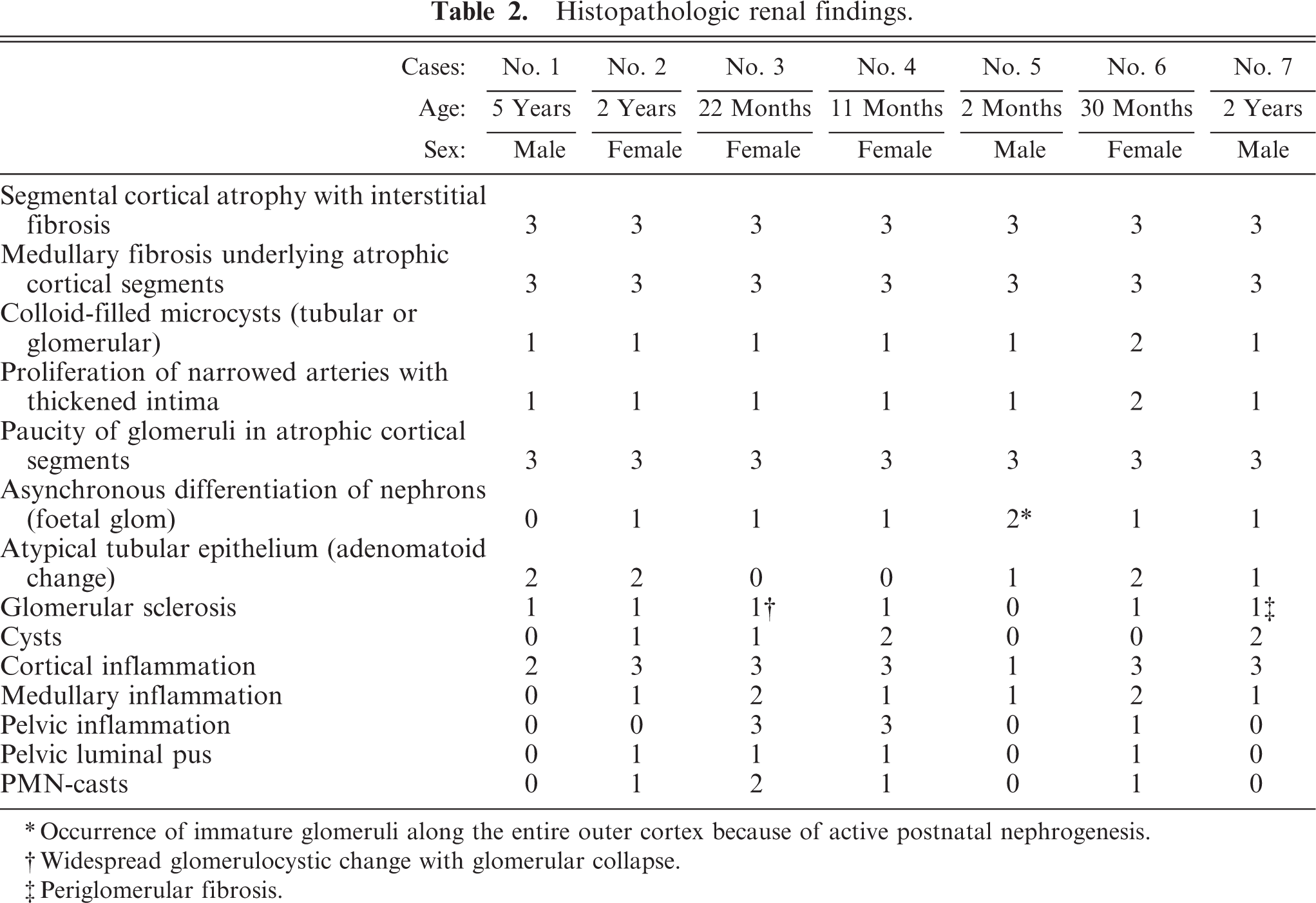
The summarized results of the light microscopic examination of the kidneys are presented in Table 2.
Histopathologic renal findings.
Occurrence of immature glomeruli along the entire outer cortex because of active postnatal nephrogenesis.
Widespread glomerulocystic change with glomerular collapse.
Periglomerular fibrosis.
The fibrotic segments of the cortices in all kidneys were characterized by tubular atrophy with a paucity of glomeruli; abundant narrow, hyperplastic small arteries and arterioles, often with intimal thickening (Fig. 3); and pronounced interstitial fibrosis. The scarred cortical segments often contained microcysts of tubular or glomerular origin, containing colloid fluid (Fig. 4). A conspicuous diffuse fibrosis in the underlying medulla was seen consistently.
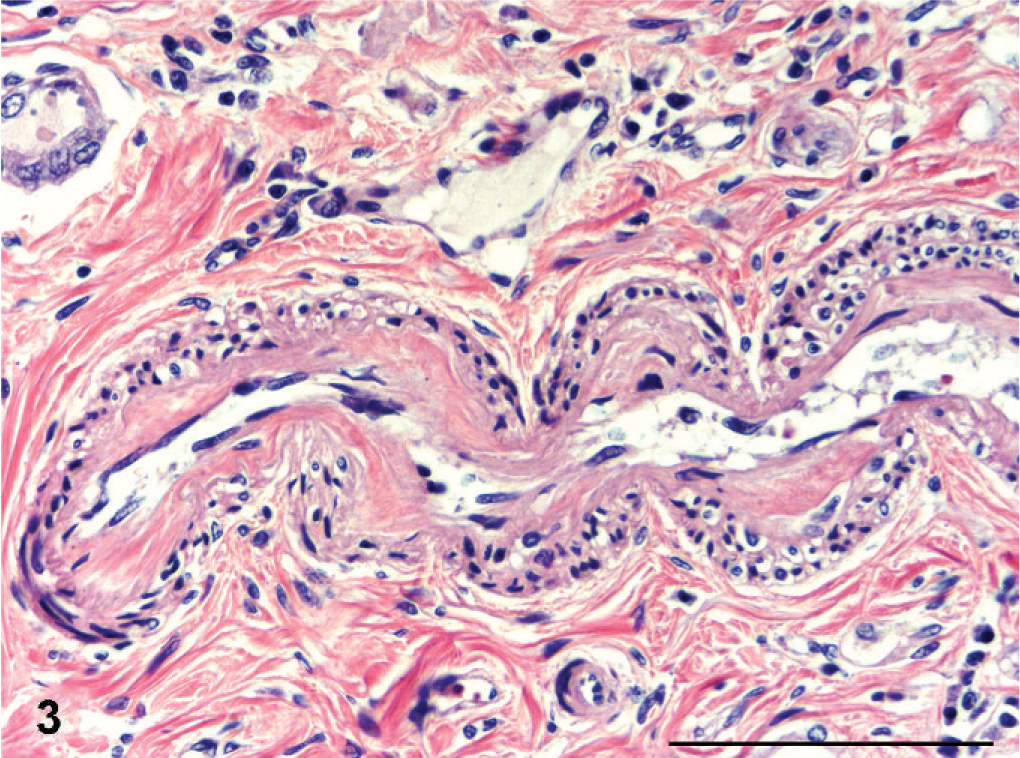
Kidney; Boxer dog No. 6. Longitudinal section of a narrow and hyperplastic cortical artery with intimal thickening. HE. Bar = 100 µm.
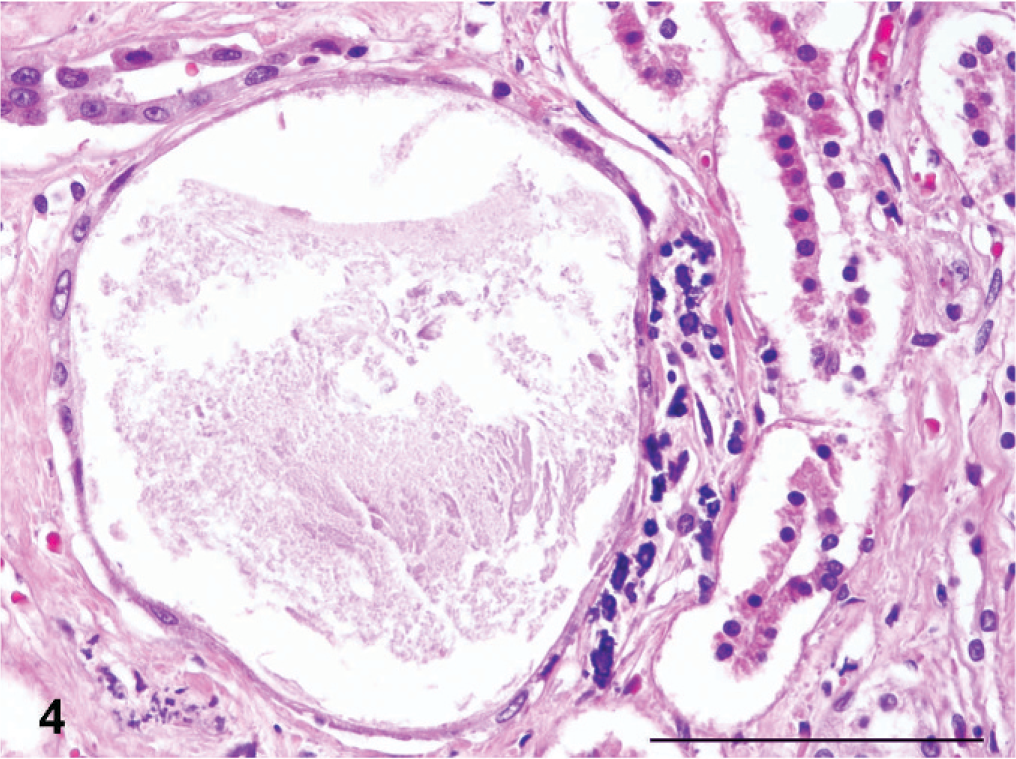
Kidney; Boxer dog No. 1. Colloid-filled glomerular microcyst in a scarred cortical segment. HE. Bar = 100 µm.
Lesions compatible with primary microscopic features of canine renal dysplasia 14 were present in all kidneys, either in the form of asynchronous differentiation of nephrons evident as immature or fetal glomeruli within the segmental cortical scars (Fig. 5), or as atypical tubular epithelium represented by medullary or less frequently cortical “adenomatoid” proliferation of collecting duct epithelium (Fig. 6). Other primary dysplastic morphologic features of canine renal dysplasia were never encountered.
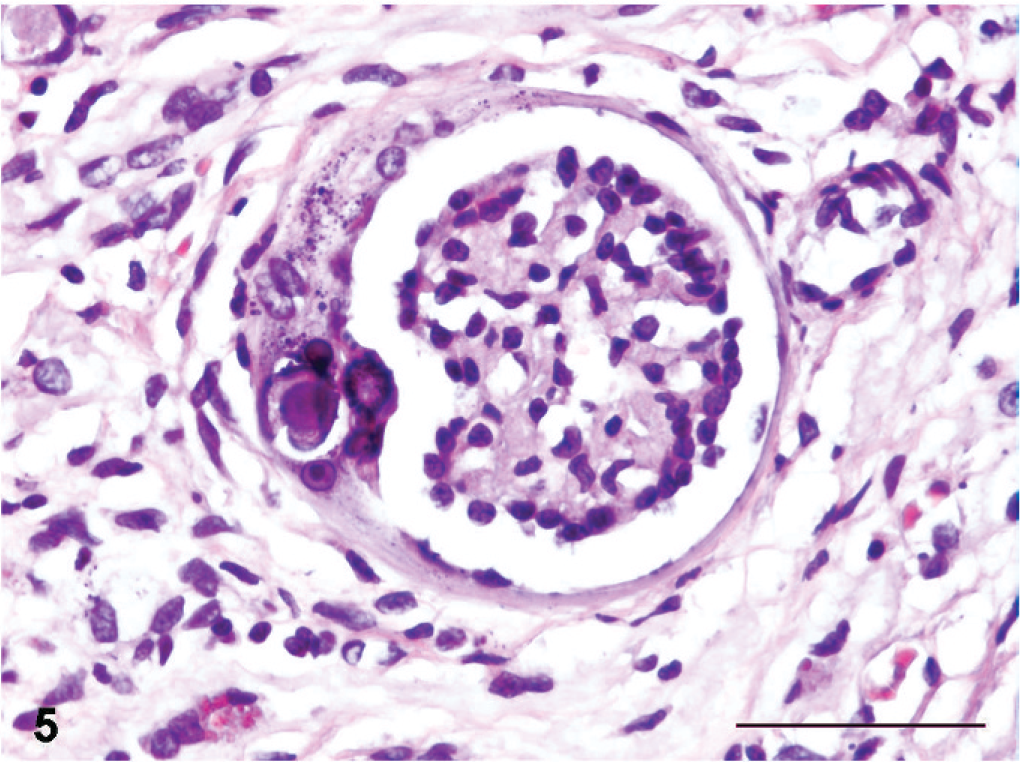
Kidney; Boxer dog No. 7. Immature glomerulus with focal mineralization of the Bowman's capsule. HE. Bar = 50 µm.
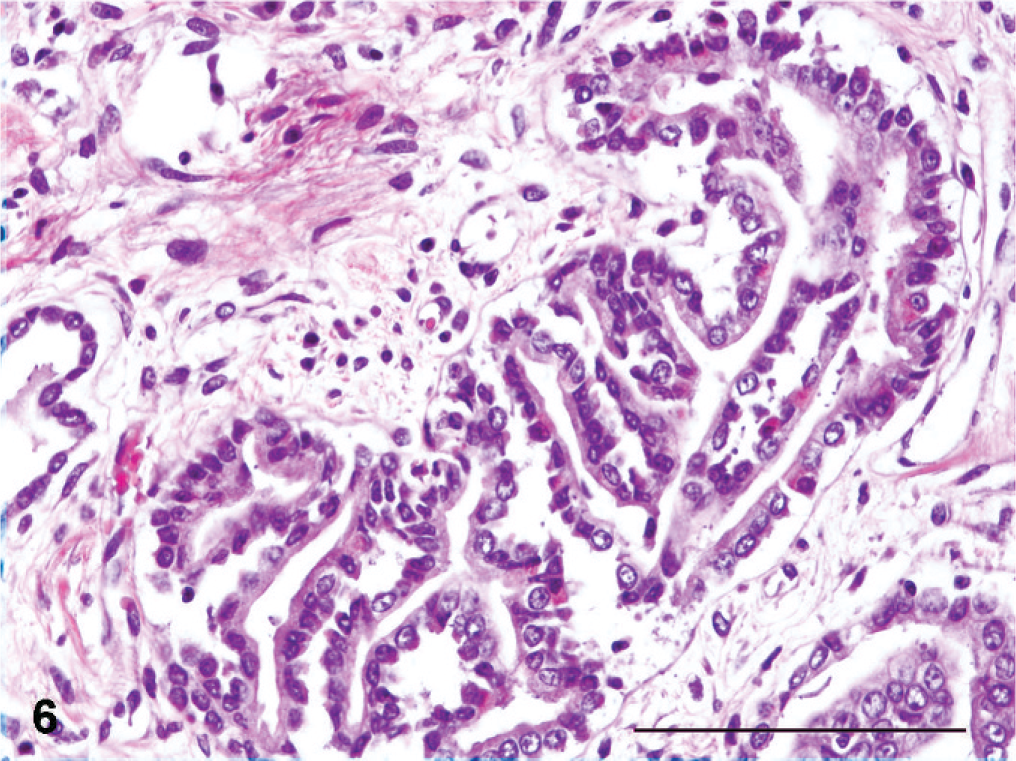
Kidney; Boxer dog No. 7. Atypical tubulus with the so-called adenomatoid proliferation of collecting ducts. HE. Bar = 100 µm.
There was generally a relatively sharp demarcation between the segmental cortical scars and the adjacent, better-preserved cortical tissues. However, glomeruli in the less-affected areas revealed signs of segmental or global sclerosis. Dilation of the Bowman's spaces was frequently encountered, often associated with fibrotic thickening of the Bowman's capsules. One dog (No. 3) showed widespread occurrence of glomerulocystic change combined with frequent glomerular collapse. Evidence of glomerular hyperfiltration injury with global insudative lesions was prominent in some glomeruli (Fig. 7).
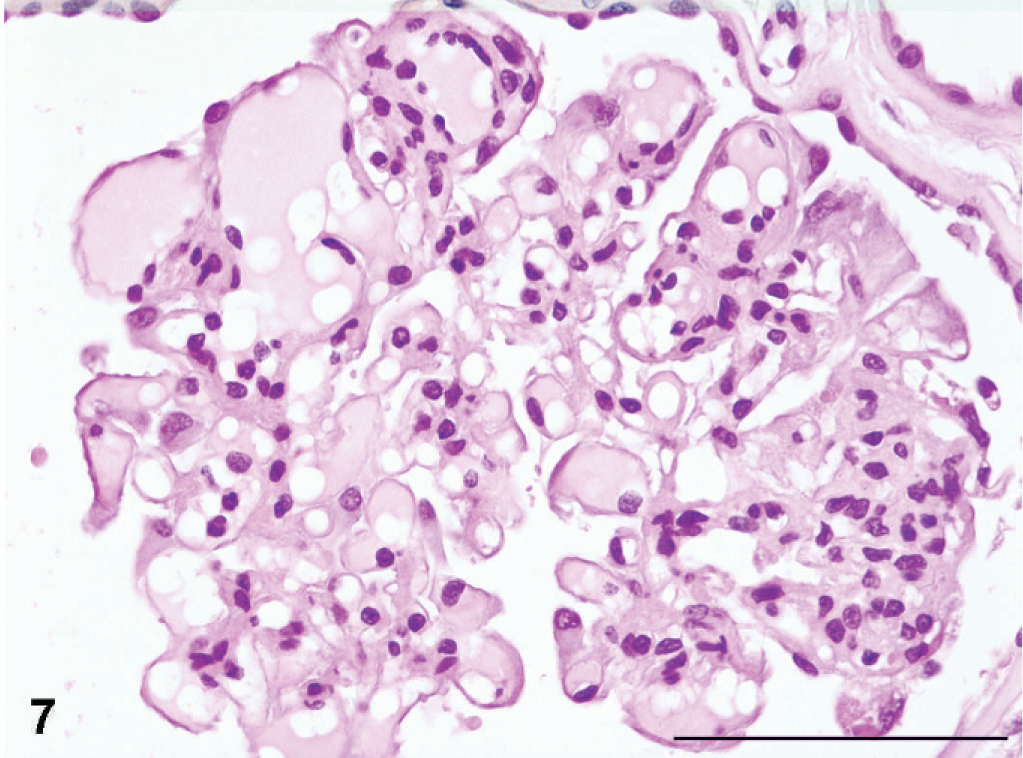
Kidney; Boxer dog No. 3. Focally sclerotic glomerulus with extensive hyperperfusion injuries (global insudative lesions). HE. Bar = 100 µm.
Interstitial inflammatory reaction with lympho-plasmacytic infiltration, either diffuse or multifocal, was seen in the kidneys of all dogs. Infiltration of inflammatory cells was less pronounced in the medullary tissue. Three dogs had a salient chronic pelvic inflammatory reaction with widespread pelvic submucosal lymphoid follicle formation (Nos. 3, 4, and 6). In these dogs and 1 additional dog (No. 2), pelvic luminal pus was observed at the histologic examination and revealed occurrence of tubular PMN-cell casts.
Focal granulomas containing numerous cholesterol clefts were present in the renal medulla of 1 dog (No. 1). Polarization microscopy revealed a moderate occurrence of birefringent crystals characteristic of calcium-oxalate in the tubular luminae of 2 dogs (Nos. 1 and 7). Hemosiderin pigments were noted in 5 dogs (Nos. 1, 3, 4, 6, and 7). A modest multifocal mineralization of renal tissue, which stained positively for calcium salts with the von Kossa stain, was observed along the tubular and glomerular basal laminae in 5 dogs (Nos. 1, 2, 3, 4, and 7).
The most significant extrarenal lesions associated with chronic renal failure in the autopsied dogs consisted of soft and pliable ribs (Nos. 6 and 7), gastric mucosal hyperemia (No. 7), mineralization of the intercostal pleura (No. 7) and of the alveolar wall (No. 1), pulmonary edema (Nos. 1, 5, 6, and 7), peritoneal effusion (No. 1), and enlarged parathyroid glands (No. 6).
In 2 dogs (Nos. 1 and 7) an enlarged heart was found with multifocal myocarditis, with dog 1 also showing an aortic valvular endocarditis caused by Escherichia coli.
In the 3 cases where only the kidneys (Nos. 2 and 4) or slices of the kidneys were available for examination (dog No. 3), no extrarenal lesions were described by the referring clinician.
Discussion
The patho-anatomic examination of the kidneys from our cases of young Boxer dogs suffering from chronic renal failure revealed in all cases a macroscopic feature of bilaterally small kidneys with coarse segmental scarring, always involving both cortex and medulla. Between the scarred segments, the remnants of less-affected renal parenchyma were hypertrophied, giving these kidneys a coarse lobular or nodular appearance. The microscopic findings were compatible with chronic pyelonephritis, with a segmental distribution of lesions consisting of pronounced interstitial fibrosis with salient atrophy of the renal parenchyma, and with a varying degree of tubulointerstitial inflammation.
When we compare our own findings with the few other papers on cases of chronic renal failure in young Boxers, we observe striking similarities in the described renal lesions in these cases.
The first published paper on chronic renal failure in an 18-month-old Boxer dog, 10 described the kidneys as reduced in size and distinctively lobulated with unevenly pale and narrowed cortices caused by wide radial bands of fibrous tissue extending from the capsular surface to the renal pelvis. A modest lympho-plasmacytic inflammation and microscopic features of renal dysplasia were also reported. The authors of this report 10 also mentioned similar findings in a biopsy from another 18-month-old Boxer. Of the examined 13 young dogs belonging to 10 different breeds, comprising only 1 Boxer, these authors found the renal pathology in the Boxer to be so characteristic that it was described to define a separate subgroup of renal lesions within their material, termed “atypical connective tissue changes without cyst formation.” 10 Similar renal lesions have been described in 2 more recent reports on young Boxers with chronic renal failure. 7, 12 Both these reports included descriptions of small kidneys with segmental lesions with radiating bands of connective tissue, revealing some of the microscopic features of canine dysplasia evident as immature glomeruli and atypical tubular structures. One of these papers also described evidence of focal mononuclear inflammation. Comparing these 3 previous reports with our present data, there seems to be a striking conformity in the renal lesions in all described young Boxer dogs with chronic renal failure. In a recent paper by Chandler et al., 5 ultrasound examination of the kidneys from 36 young Boxers revealed irregular shape of the kidneys in 18 dogs, small renal size in 11 dogs, and the presence of cysts in 7 dogs. Histologic examination of needle biopsies from 8 dogs and autopsy material from 1 other revealed pericapsular and interstitial fibrosis (8 dogs), presence of inflammatory cells (3 dogs), dilated tubuli (3 dogs), and sclerotic glomeruli (2 dogs).
The previous reports of renal lesions in young Boxers with chronic renal failure by Lucke et al. 10 and Hoppe et al., 7 both concluded that these were cases of canine renal dysplasia, 7, 10 whereas Chandler et al. 5 concluded that the histologic findings in only 1 from their 9 examined dogs was consistent with renal dysplasia. None of the previous referred reports, however, reported a complete range of dysplastic lesions within their material, including primitive metanephric ducts and dysontogenic chondroid metaplasia, 3, 14 to prove anomalous metanephric differentiation as the cause of the observed renal lesions. Peeters et al. 12 also abstain from defining the juvenile nephropathy in young dogs as cases of renal dysplasia, since the findings are not consistent with the very restrictive definition of renal dysplasia in man.
The kidney lesions of the Boxers in our data contained immature glomeruli, atypical tubules and “proliferative arterioles” as primary features of renal dysplasia. 14 The prevailing view is, however, that the occurrence of immature or fetal glomeruli within scarred renal cortices should be interpreted with caution, since such lesions may merely indicate arrested cortical maturation caused by renal ischemia or scarring in intrauterine or early postnatal life. 11 The other primary feature of dysplasia that we could observe in the kidneys of our dogs was atypical tubular epithelium in the form of proliferated medullary or cortical collecting duct epithelia, the so-called adenomatoid change. 14 We are, however, reluctant to interpret this lesion as a primary dysplastic microscopic feature for 2 important reasons. Firstly, this identical lesion is occasionally seen in old dogs with end-stage renal lesions resulting from a variety of causes unrelated to renal dysplasia, 11 and in juvenile dogs with end-stage renal lesions resulting from causes other than renal dysplasia, e.g., primary hyperoxaluria 6, 8 and in hypercalcemic nephropathy (J. H. Jansen, unpublished observation). Secondly, a similar lesion with salient proliferation of medullary collective duct epithelium has been observed as part of the renal lesion in sheep accidentally or experimentally poisoned with anhydrous calcium chloride used for dust control on gravelled roads (Professor Emeritus K. Nordstoga, personal communication). This observation is intriguing since it may indicate that this is a secondary compensatory mechanism caused by the development of a hyperchloremic acidosis in sheep poisoned with calcium chloride. Thus, it may well be that the renal tubular “adenomatoid change” actually represents a compensatory mechanism with proliferation of intercalated cells of the cortical collective ducts and/or proliferation of the inner medullary collective duct cells, as an attempt to increase hydrogen ion secretion in the metabolic acidotic, uremic dog.
When comparing the renal pathological findings observed in our data from young Boxers with the literature of human renal pathology, our findings closely resemble the cardinal macroscopic and histopathologic features of nonobstructive chronic pyelonephritis or so-called reflux nephropathy, 15 originally described as juvenile malignant nephrosclerosis and subsequently known as the Ask-Upmark kidney. 2 The Ask-Upmark kidney or so-called segmental hypoplasia of human kidneys is characterized by unilateral or bilateral occurrence in the juvenile or adolescent human being of abnormally small kidneys with so-called segmental hypoplasia, and the presence of 1 or more grooves on the capsular surfaces. Microscopically this lesion is characterized by scarred cortical segments with tubular atrophy and paucity of glomeruli, colloid-filled microcysts and prominent arteries with varying degrees of intimal thickening. Microscopic features of abnormal metanephric differentiation may also be seen in such kidneys, indicating an intrauterine origin of the abnormality. 1, 3 These lesions are progressive, even if the vesicoureteric reflux is corrected and urinary tract infection is prevented. 16 The macroscopic and histologic descriptions of canine juvenile progressive nephropathies hold many similarities with the features of human reflux nephropathy. The third microscopic primary feature of canine renal dysplasia that we encountered in the kidneys of our Boxers was the so-called “proliferative arterioles.” 14 This lesion may also prove to be identical to the prominent arterial blood vessels that are associated with human reflux nephropathy. The progressive nature of the canine juvenile nephropathies may also indicate that vesicoureteric reflux may be involved in the development of such disorders. Reflux nephropathy in man and the pig is known to primarily affect the renal poles, 15 whereas the observed lesions in our Boxers had a diffuse distribution throughout the affected kidneys. This difference in the observed distribution of the cortical atrophic lesions may be a result of the species difference in renal anatomy. Both man and the pig have multipapillary kidneys with compound papillae vulnerable to urinary reflux at the kidney poles, whereas the canine species has a unipapillary kidney with a single crest-like papilla caused by fusion of the renal lobes. 11 This difference in renal anatomy between the canine and the human and porcine species may explain the more diffuse distribution of the lesions in the dog as opposed to the tendency toward polar affection in man and pig.
Vesico-ureteral reflux was not proven clinically in any of our cases, but we find that comparing the renal lesions in these dogs with descriptions of the renal lesions in reflux nephropathy in man makes a pathogenesis of reflux nephropathy most probable.
In the study by Chandler et al. 5 more than half of the Boxer dogs in the study had urinary incontinence, likely caused by urethral sphincter incompetence in neutered female dogs and by the increased volume of urine produced by the defect kidneys. The female Boxer dogs in our study were not neutered; however, Boxer dogs are mentioned to be overrepresented as a breed for sphincter mechanism incompetence. 4
A possible cause of the kidney lesions in our Boxers may be concomitant incompetence of the muscles of the ureters, thereby increased reflux, possibly complicated by bacterial invasion, and positive urine cultures were obtained in 9 of the 30 Boxer dogs examined by Chandler et al. 5
The possibility of an inherited origin for renal lesions in our study may be suspected in 6 of the dogs (Nos. 2–7), since the pedigrees of these dogs confirmed genetic relationships. Breeding trials will be necessary to characterize the mode of inheritance.
Footnotes
Acknowledgements
We thank Drs. Reidun Heiene, Astrid Indreb⊘, Ellen Eide Kjellman, and Vigdis Korbi, and Ms. Trine Aar⊘e for providing information on clinical parameters and pedigrees. We are grateful for the excellent histotechnical assistance provided by Ms. Randi Terland, for assistance with the layout of illustrations by Mr. Kim Egenes, and for the language corrections by Dr. David Griffiths.
