Abstract
Two sibling Boxer puppies presented with severe suppurative myocarditis in the absence of additional disseminated suppurative foci. The identification of gram-negative bacteria within areas of myocarditis in both puppies and the pure growth of large numbers of Citrobacter koseri from the myocardial lesions in one of the dogs were consistent with a bacterial etiology. The fact that C. koseri is an opportunist pathogen suggested intercurrent immunosuppression. The finding of a concomitant bacterial myocarditis in two canine siblings is novel. The case is also unusual in that syncope could be related to the myocardial injury.
Keywords
Myocarditis is common in a variety of systemic animal diseases but is rarely found in the absence of disseminated disease.12,16 Bacterial, viral, and protozoal etiologies have been described in the dog.17 Citrobacter koseri is an occasional inhabitant of human and animal intestines and is an opportunistic pathogen of immunosuppressed and neonatal human patients, causing bacteremia and suppurative meningoencephalitis.4 In this report, we describe two 12-week-old sibling Boxer puppies that presented with a severe bacterial myocarditis and from one of which a pure, heavy growth of C. koseri was cultured from the myocardial lesions.
A litter of two 12-week-old female Boxer puppies had a 5-week history of ill thrift and recurrent, intermittently hemorrhagic diarrhea. During this time, the animals maintained normal appetite and were fed a highly digestible prescription diet. Both puppies had been treated orally on separate occasions with anthelmintics (piperazine citrate and praziquantel–pyrantel embonate–febantel) and antimicrobials (streptomycin–phthalylsulphathiazole and sulphadiazine–trimethoprim, 5 day course of each). Both puppies were clinically normal when vaccinated against canine distemper virus, canine adenovirus 2, canine parvovirus, canine parainfluenza virus 2, Leptospira canicola, and L. icterohaemorrhagiae (Eurican DHPPi & L, Merial Animal Health, Dublin, Ireland) at 12 weeks of age.
Puppy No. 1 had a syncopal episode 2 days after vaccination. The animal collapsed suddenly and exhibited bilateral vertical nystagmus before gradually recovering. When examined by a veterinarian 15 minutes later, the puppy had a subnormal rectal temperature, extremely pale mucous membranes, marked tachycardia, and moderate dyspnea and tachypnea. Two further syncopal episodes occurred during the examination approximately 10 minutes apart. The animal died during a further syncopal episode 25 minutes later.
Puppy No. 2 exhibited multiple, similar syncopal episodes in the following 7 days, during which time the animal was intermittantly dyspneic and tachypneic. On presentation at the Veterinary Teaching Hospital, University College Dublin, puppy No. 2 was in a state of collapse with a very weak pulse, cyanotic mucous membranes, dyspnea, tachypnea, and tachycardia. The animal was euthanatized because of its deteriorating clinical condition and the necropsy findings for puppy No. 1.
At necropsy, both puppies were in poor nutritional condition with diffusely congested, edematous lungs. Puppy No. 2 had approximately 500 ml, 250 ml, and 75 ml of translucent fluid in the peritoneal, pleural, and pericardial cavities, respectively. The hearts of both animals were enlarged. The left and right apical ventricular surfaces in puppy No. 1 each exhibited one raised, firm, ovoid lesion approximately 1 cm in diameter, which were white and red, respectively (Fig. 1). A solitary lesion of similar shape, consistency, and dimensions but pale yellow in color was visible on the left apical ventricular surface in puppy No. 2. On sectioning the cardiac chambers, these lesions extended transmurally within the ventricular free walls. Similar lesions were observed at higher locations deep within both septal and left and right ventricular free walls in both animals and involving papillary muscles and ventral aspects of the atrial walls. No other significant gross abnormalities were observed.
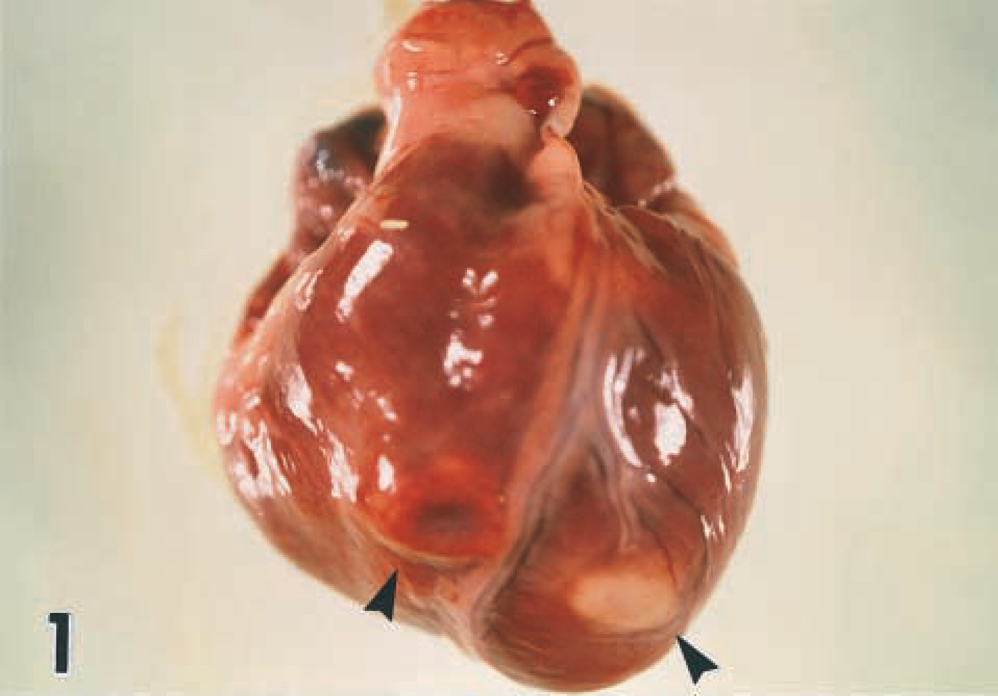
Heart; puppy No. 1. Well-demarcated, raised, ovoid lesions, red and white in color within the apical aspects of right and left ventricular free walls, respectively (arrowheads).
Histopathologic examination of affected cardiac tissue from both dogs revealed severe distruption of cardiac myofiber alignment by extensive inflammatory cell infiltrates with accompanying mild to moderate interstitial fibroplasia and mineralization of hyalinized myofiber fragments (Fig. 2). Staining by the Van Gieson and Von Kossa methods indicated a similar degree of fibroplasia in both animals and more extensive segmental mineralization in puppy No. 2, respectively. The inflammation was transmural in some areas and consisted predominantly of admixed intact and degenerate neutrophils and macrophages with smaller numbers of lymphocytes and plasma cells. Inflammatory cells were intermingled with haphazardly orientated fibroblasts and short strands of collagen. Anichkov cells were noted in these areas in puppy No. 2. Individual gram-negative bacilli within macrophages in addition to extracellular scatterings of single short chains and small clusters of similar bacilli were detected in areas of myocarditis in both animals (Fig. 3). Organisms were more numerous in puppy No. 2.
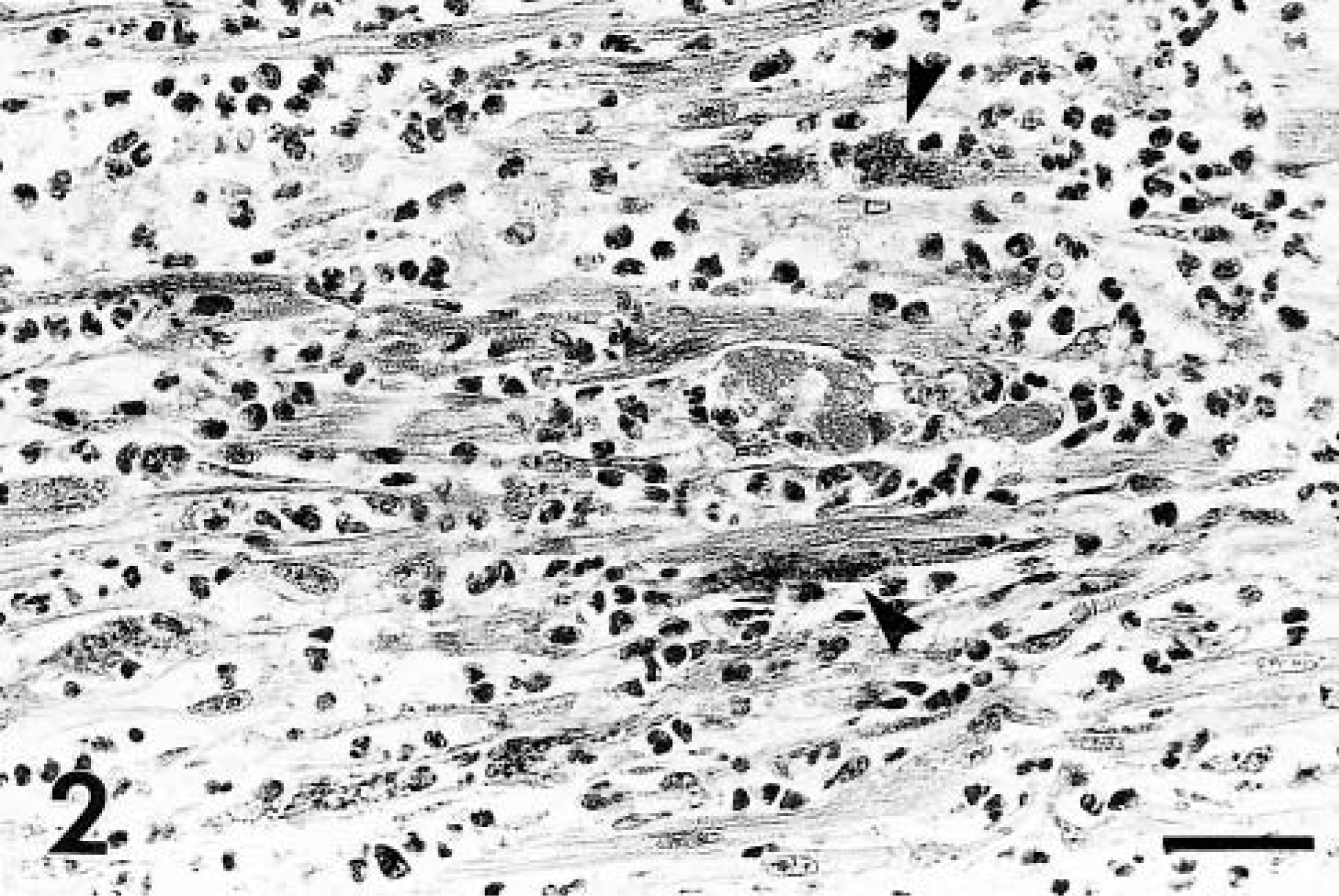
Myocardium; puppy No. 2. Severe disruption of cardiac myofiber alignment by admixed intact and degenerate neutrophils and macrophages with segmental fiber fragmentation and mineralization (arrowheads). HE. Bar = 25 μm.
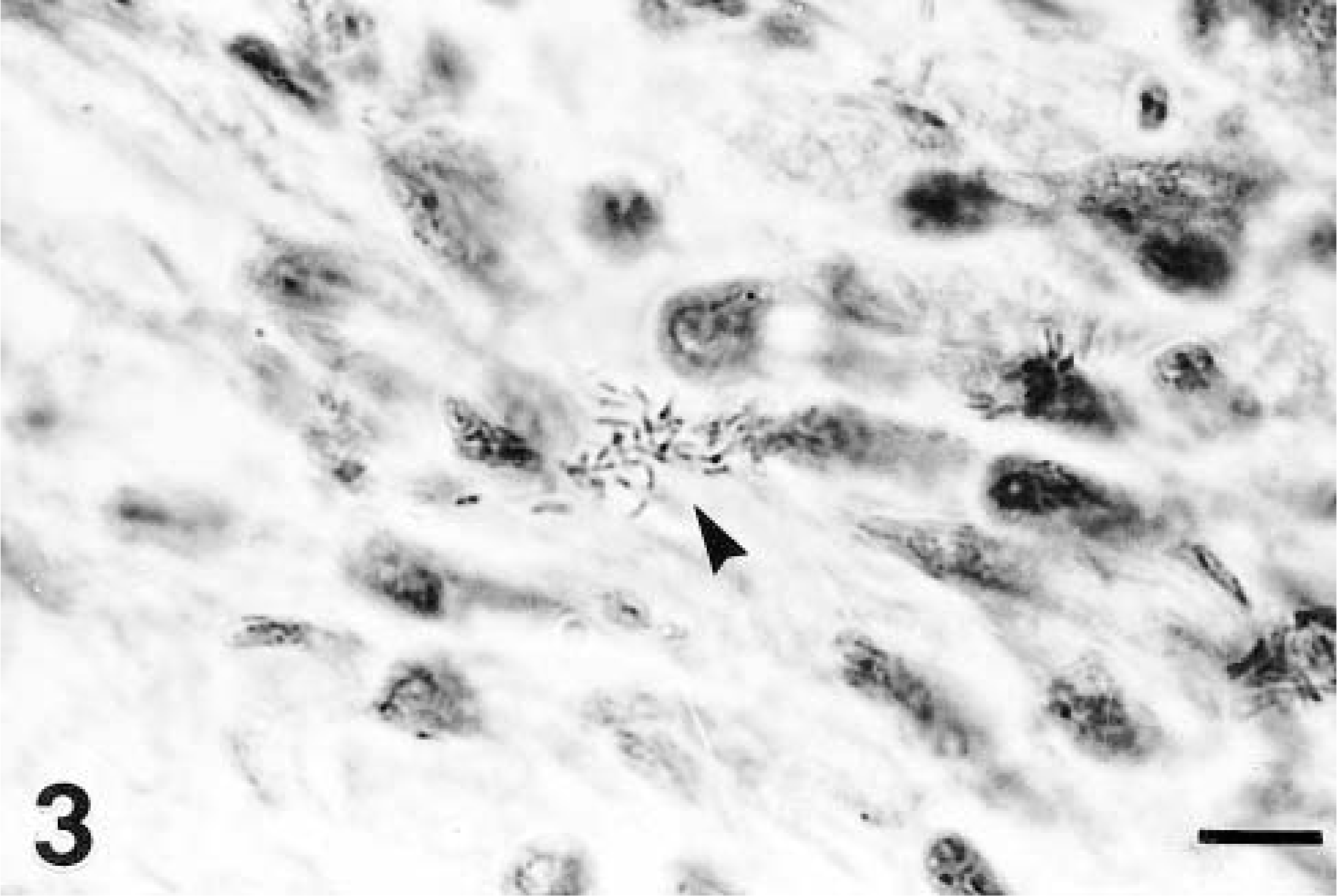
Myocardium; puppy No. 2. A cluster of gram-negative bacilli (arrowhead) within an area of inflammation. Bar = 2 μm.
No significant abnormalities were observed in the brain of either animal or in thymic and renal tissue from puppy No. 1 and puppy No. 2, respectively. Thymic lymphoid tissue in puppy No. 2 was severely depleted with moderate distension of interlobular and subcapsular areas by fibrinohistiocytic exudate. The kidney of puppy No. 1 was not examined microscopically. Puppy No. 2 exhibited periacinar hepatocyte necrosis and distension of portal lymphatics. There was villous atrophy of the small intestine with loss of surface enterocytes. Predominantly intact macrophages, neutrophils, lymphocytes, and plasma cells infiltrated the intestinal lamina propria and crypt abscesses were present. A similar inflammatory cell infiltrate was noted in this puppy's colonic lamina propria.
The cardiac lesions, small intestine, and a pericardial swab from puppy No. 2 were cultured for bacteria. A heavy, pure growth of C. koseri (formerly Citrobacter diversus) was recovered from the cardiac lesions on McConkey and blood agar. Identification was based on colony morphology and biochemical profile using an api strip (api 20E, Biomérieux SA, Lyon, France). Escherichia coli and Proteus spp. were isolated from the small intestine, and no organisms were cultured from the pericardial swab. No samples were cultured from puppy No. 1.
No antibodies to Borrelia burgdorferi were detected on serologic examination of three additional dogs from the household (Snap® Lyme Antibody Test Kit, Idexx Laboratories, Buckinghamshire, UK).
Suppurative myocarditis is rare in dogs particularly in the absence of disseminated disease.12,15,16 The identification of gram-negative bacilli within areas of predominantly suppurative inflammation is consistent with a bacterial etiology. The random, multifocal distribution and intramyocardial location of the lesions suggest that the organisms were of hematogenous origin. The heavy, pure growth of C. koseri cultured from the myocardial lesions in puppy No. 2 is significant because this gram-negative bacillus is an opportunistic pathogen of immunosuppressed and neonatal human patients.4 C. koseri is an occasional inhabitant of human and animal intestines and of soil, water, sewage, and food.4 Although disease caused by this organism has not previously been reported in domestic animals, the bacteremia, meningitis, and multifocal brain abscessation described in humans has been experimentally reproduced in a murine model.14 Although it is not clear why C. koseri infection became established in these two puppies, immune naivety or intercurrent immunosuppression may have been contributory factors. Immune naivety could have been age related or, given that the animals were siblings, could have had a genetic component. Intercurrent diarrhea or recent vaccination or both may have contributed to immunosuppression. Chronic enteritis depresses both cellular and humoral immune responses in animals because of malabsorption and endogenous nutrient loss and transient immunosuppression can follow the administration of polyvalent vaccines to dogs.10,13 The recurrent diarrhea in both puppies from 7 weeks of age and the microscopic evidence of atrophic enteritis and thymic depletion in puppy No. 2 suggests these animals may have had parvoviral infection, which would account for the immunosuppression and intestinal mucosal injury necessary to facilitate systemic invasion by intestinal C. koseri.15 The fibroplasia within the myocardial lesions may also indicate previous parvoviral injury on which bacteria-induced suppurative inflammation was superimposed. Canine parvovirus specifically targets myocardium in animals up to 8 weeks of age, possibly increasing the susceptibility of this tissue to subsequent bacterial colonization, which may explain why the lesions induced by the invading C. koseri were restricted to this site.2 The absence of intranuclear inclusions in cardiac myofibers in these puppies is insufficient to rule out parvovirus infection because these lesions are not consistently observed.2,11
Other causes of myocarditis in dogs include B. burgdorferi infection, toxoplasmosis and neosporosis. Lyme disease (caused by the spirochetal agent B. burgdorferi) has been associated with myocarditis and syncope in dogs.7,17 Although the myocarditis of Lyme disease is similar microscopically to that seen in these puppies, the gram-negative bacteria within inflammatory infiltrates and the absence of antibodies to B. burgdorferi in three other dogs in the household suggest spirochetes were not involved. Immunohistochemical or silver staining for B. burgdorferi were not performed because of the diagnostic insensitivity and interpretative difficulties, respectively, associated with these techniques.3,6 Toxoplasmosis and neosporosis were ruled out based on the absence of intralesional bradyzoites and the restriction of the inflammation to the myocardium.9
The syncope was probably related to the extensive myocardial lesions. Frequently, the pathogenesis of syncope in animals remains undetermined because the transient nature of the attacks provides insufficient opportunity for diagnostic investigation.1 The syncope probably resulted from interruptions to cerebral blood flow secondary to myocardial inflammation and fibroplasia impeding ventricular output or secondary to cardiac dysrhythmias triggered by repeated depolarization of injured cardiac myofibers.1,5,12 The size and the fibroplastic component of the myocardial lesions were consistent with long-standing inflammation,8 which suggests that although a slightly less extensive and severe myocarditis was probably present when puppy No. 1 was vaccinated such lesions did not manifest clinically. The infection in these puppies provides further evidence of the potential pathogenicity of Citrobacter spp. and indicates a possible trophism of these organisms for the canine myocardium.
Footnotes
Acknowledgements
We acknowledge the excellent technical assistance of Mr. M. Nugent and Mr. C. King and the assistance and advice of Mrs. C. McQuillen, Mr. G. O'Dowd, M.R.C.V.S., and of Profs. B. J. Sheahan (Dublin), P. J. Quinn (Dublin), and D. F. Kelly (Liverpool).
