Abstract
Neprilysin is an amyloid-β-degrading enzyme localized in the brain parenchyma. The involvement of neprilysin in the pathogenesis of Alzheimer's disease has recently received much attention. We examined the localization of neprilysin and amyloid-β, as well as the activity of neprilysin, in the brains of dogs and cats of various ages to clarify the relationship between neprilysin activity and amyloid-β deposition. The distribution of neprilysin was almost identical in dogs and cats, being high in the striatum, globus pallidus, and substantia nigra, but very low in the cerebral cortex. The white matter and hippocampus were negative. Neprilysin activity in the brain regions in dogs and cats was ranked from high to low as follows: thalamus/striatum > cerebral cortex > hippocampus > white matter. Amyloid-β deposition was first detected at 7 and 10 years of age in dogs and cats, respectively, and both the quantity and frequency of deposition increased with age. In both species, amyloid-β deposition appeared in the cerebral cortex and the hippocampus. In summary, the localization of neprilysin and neprilysin activity, and that of amyloid-β, were complementary in the brains of dogs and cats.
Keywords
Alzheimer's disease (AD) is a neurodegenerative disorder that currently affects nearly 2% of the population in industrialized countries. 30 The risk of AD dramatically increases in individuals beyond age 70 years. It is predicted that the incidence of AD will increase threefold within the next 50 years, because of the aging of the population and that it will become a serious social problem. Patients with AD lose their memory and cognitive abilities, and, later, their personality. 30
AD is characterized by pathologic features such as extracellular senile plaques, intracellular neurofibrillary tangles, synaptic loss, and brain atrophy. The senile plaques are mainly composed of the amyloid-β peptide (Aβ) of 40–43 amino acids. 7, 14, 22 Amyloid deposition and senile plaque formation were observed in aged dogs, 12, 45, 46 cats, 20, 35 monkeys, 34 bears, 10, 40, 47 coyotes, 41 and camels. 36 Dogs may be a particularly appropriate model for monitoring the development of, and analyzing the pathogenesis of, AD, because they have a longer life span than experimental rodents and share a common environment with humans. 11, 21 The major constituent of senile plaques in dogs and human patients is Aβ. 21 In aged dogs, dementia-like conditions are frequently observed, 39 and, except for a lack of neurofibrillary tangles, the brain lesions in such dogs are very similar to those in patients with AD. 13, 19 Therefore, dogs are widely used as an animal model for studies on AD pathology. 18, 39, 43 In addition to dogs, cats recently became another candidate for an animal model of AD. 8, 20
Neprilysin (NEP) (EC 3.4.24.11) is a type-II membrane metalloendopeptidase that is composed of approximately 750 residues, with an active site that contains a zinc-binding motif (HEXXH) at the extracellular carboxyl terminal domain. It exists on the plasma membrane as a noncovalently associated homodimer. 44 NEP is also commonly referred to as neutral endopeptidase–24.11 or enkephalinase, and is identical to leukocyte cell surface antigen 10 (CD10) or common acute lymphoblastic leukemia antigen. It is expressed in a wide range of tissues, including the kidney, prostate, testis, and lung, and cleaves a range of peptide hormones, such as substance P, 15 endothelin, 1 bradykinin, 38 and atrial natriuretic peptide. 27 Within the brain, where NEP is expressed pre- and postsynaptically, 5, 16 it performs an important physiologic role in the hydrolysis of neuropeptides. 42 Recent studies revealed that NEP is distributed in the hippocampus and cortical regions, where it may serve to degrade Aβ. 2, 16, 24 Aβ catabolism was reduced when selective NEP inhibitors were administered to rats, 25 and the accumulation of Aβ in NEP-deficient mice could be reversed by the administration of exogenous NEP. 24 Other studies of mouse models 16 and human postmortem tissues 2, 31, 50, 51, 53 suggest that NEP expression is inversely related to the extent of AD pathology.
Because of the considerable differences in the metabolism, life span, and brain pathology between humans and rodents, the use of rodent species for studies on aging is controversial. In the present study, we examined the immunohistochemical localization of NEP expression and Aβ, and also measured NEP activity by using a fluorescent substrate in the brains of dogs and cats of various age groups to clarify the relationship between NEP and Aβ deposition. In addition, we compared the present results with those that appeared in recent reports that dealt with NEP expression and activity in humans and other animal species.
Materials and Methods
Animals
The brains of 25 dogs (ranged from 1 year and 5 months old to 14 years and 3 months old, cases C1 to C25) and 14 cats (ranged from 7 months old to 20 years old, cases F1 to F14) were examined. The breed, sex, age, and pathologic diagnoses of the dogs and cats are shown in Tables 1 and 2, respectively. The brains were cut into 2 parts at the center. The left half was routinely fixed in 10% phosphate-buffered formalin for immunohistochemistry. The right half was frozen and stored at −80°C to assay NEP activity.
Cases examined in the study (canine).
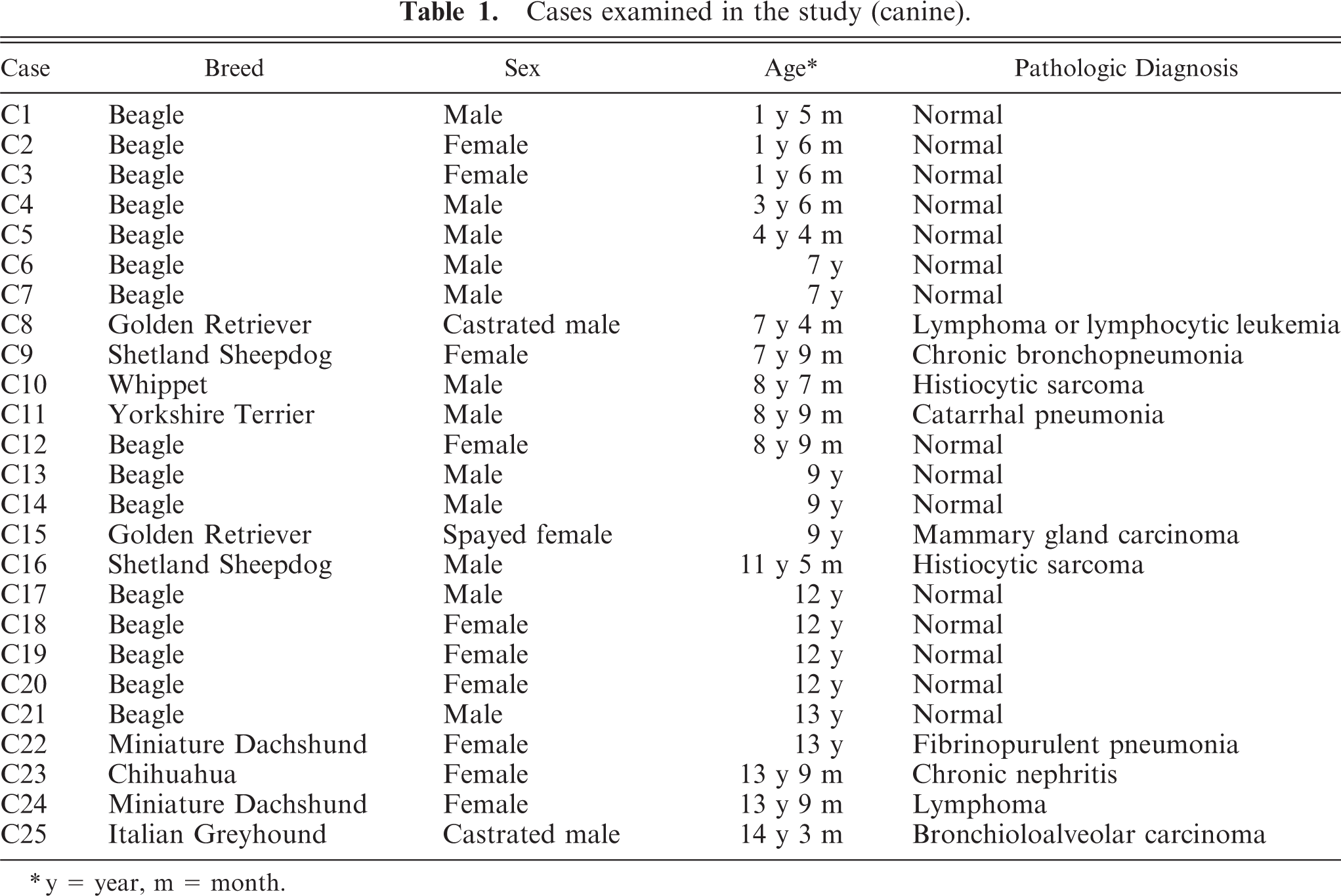
y = year, m = month.
Cases examined in the study (feline).∗
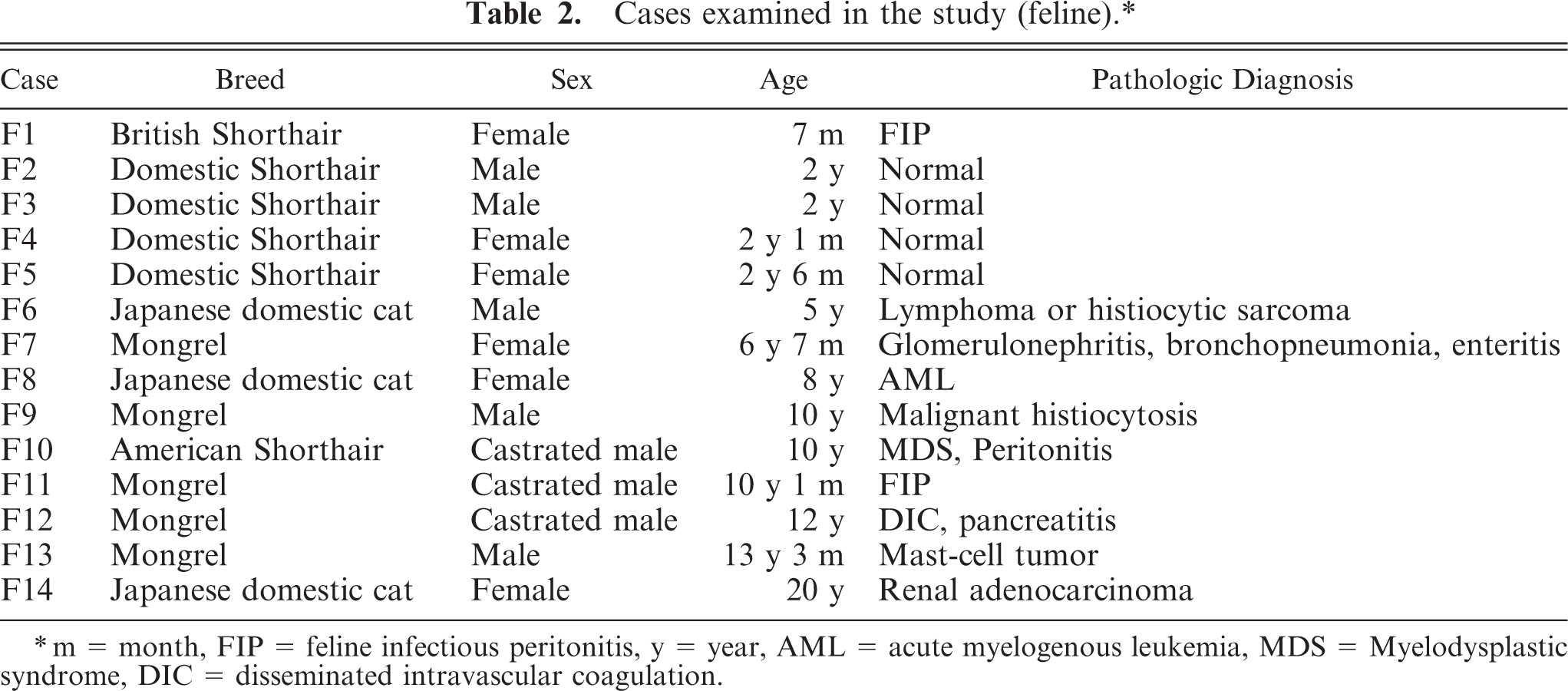
m = month, FIP = feline infectious peritonitis, y = year, AML = acute myelogenous leukemia, MDS = Myelodysplastic syndrome, DIC = disseminated intravascular coagulation.
Immunohistochemistry
The left half of the brain was cut transversely from the frontal lobe to the cerebellum at intervals of approximately 5 mm, and 7–10 slices were obtained. The tissue slices were routinely embedded in paraffin wax and cut into 4-µm-thick sections. Deparaffinized sections were stained with hematoxylin and eosin. Primary antibodies used for immunohistochemistry were mouse anti-NEP monoclonal antibody (Novocastra, Newcastle, UK) and rabbit anti-Aβ 1–42 polyclonal antibody (BioSource International, Camarillo, CA).
After deparaffinization and rehydration, sections were heated by using an autoclave at 121°C for 20 minutes in 10 mM sodium citrate buffer (pH 6.0) for antigen epitope retrieval. After they were rinsed in Tris-buffered saline solution (TBS), the sections were treated with 0.3% hydrogen peroxide in methanol for 30 minutes to inactivate endogenous peroxidase. To block nonspecific immunoreactivity, the sections were then treated with TNT buffer containing blocking reagent (TNB) Blocking Buffer (PerkinElmer, Shelton, CT) for 1 hour. They were subsequently incubated with anti-NEP monoclonal antibody diluted with TNB (1 : 50) or with TNB only (negative control) and were left to react overnight at 4°C. After rinsing in TBS buffer, the sections were sequentially incubated with biotinylated goat anti-mouse immunoglobulin (Ig) G (1 : 400 dilution : KPL, Guildford, UK) for 1 hour at room temperature, horseradish peroxidase (HRP) conjugated streptavidin (1 : 300; DAKO, Carpinteria, CA) for 1 hour at room temperature, and biotinylated tyramide for 10 minutes at room temperature. Tyramide-enhanced immunoreactivity was visualized with HRP-conjugated streptavidin (1 : 100; PerkinElmer) for 30 minutes at room temperature and then with diaminobenzidine (DAB) for 10 minutes at room temperature. Counter-staining was performed with hematoxylin. Murine brains and canine kidneys were used as positive controls for the immunostain, because the thalamus and striatum of murine brain 24 and the tubular epithelium of canine kidneys are strongly positive for NEP.
Immunohistochemical staining for Aβ 1–42 was performed by using the labeled streptavidin biotinylated antibody method. After deparaffinization and rehydration, sections were pretreated, first with 0.3% hydrogen peroxide in methanol and next with 99% formic acid at room temperature for 5 minutes. After they were rinsed in TBS, the sections were then treated with 8% skim milk for 30 minutes to block nonspecific immunoreactivity and then were incubated with anti-Aβ 1–42 polyclonal antibody diluted with TBS (1 : 500) or with TBS only (negative control) overnight at 4°C. Then, the sections were sequentially incubated with biotinylated goat anti-rabbit IgG (1 : 400; KPL) for 40 minutes at room temperature and with HRP-conjugated streptavidin (1 : 300; DAKO) for 40 minutes at room temperature, and, finally, the reaction products were visualized with DAB. Counter-staining was performed with hematoxylin. Aβ-laden aged canine brains were used as positive controls for the immunostain.
Assay for NEP activity
The frozen right half of the brain was divided into 4 parts (cerebral cortex, white matter, thalamus/striatum, and hippocampus). Each part was homogenized in ice-cold Solution A (10 mM Tris–HCl buffer [pH 8.0], which contained 0.15 M NaCl, 1 µg/ml leupeptin and 1 µg/ml Pepstain A) by using an ULTRA TURRAX T25-Basic homogenizer (IKA, Werke, Staufen, Germany). The homogenates were centrifuged at 500 × g for 5 minutes, and the supernatants were further centrifuged at 100,000 × g for 30 minutes. Pellets were solubilized in the Solution A that contained 1% Triton X-100 (v/v), and the solution was recentrifuged at 100,000 × g for 15 minutes. The resultant clear supernatants were then used as the membrane fractions. NEP activity was estimated spectrophotometrically as follows. The assay mixture consisted of 20 µg of membrane fractions, 0.1 mM Succinyl-Ala-Ala-Phe-AMC (Bachem, Bubendorf, Switzerland) as a substrate, and 50 mM N-2-hydroxyetylpiperazine-N′-ethanesulphonic acid (HEPES; pH 7.2) in a total volume of 100 µl. The reaction was initiated by adding the substrate to the assay mixture and kept at 37°C for 1 hour. Ten microliters of 50 mM HEPES that contained 0.4 mU of leucine aminopeptidase (Sigma, St. Louis, MO) and 0.1 mM phosphoramidon were then added to the reaction mixture, followed by further incubation at 37°C for 20 minutes to remove a phenylalanine residue from Phe-AMC. The liberated 7-amino-4-methylcoumarin was photometrically measured with excitation 355 nm and emission 460 nm by using a WALLAC ARVO SX 1420 MULTILABEL COUNTER (PerkinElmer). The NEP-related neutral endopeptidase activity was determined based on the decrease in the rate of digestion caused by 0.2 mM phosphoramidon, a specific inhibitor of NEP. 44 Protein concentrations were determined by using a DC protein assay kit (BIO-RAD, Hercules, CA).
Statistics
For comparison of NEP activity and age, Pearson's correlation coefficients were calculated. Differences between NEP activity in the different brain regions were assessed by one-way analysis of variance (ANOVA) and the Bonferroni test. Statistical analyses were performed by using the statistical software package SPSS, version 15.0 (SPSS Japan, Tokyo, Japan).
Results
Distribution of NEP and Aβ in the brain
The results of immunohistochemistry for NEP and Aβ are summarized in Tables 3 and 4. The distribution of NEP in the brain was almost identical in dogs and cats, being particularly rich in the caudate nucleus, putamen, globus pallidus, and substantia nigra. In contrast, the cerebral cortex stained very weakly, and the white matter, hippocampus, and cerebellum were negative for NEP (see Figs. 1–20). In all the positive regions, both neuronal cell bodies and dendrites were stained in a fine-granular pattern (see Figs. 5–10 and 15–20). No differences were observed in NEP immunoreactivity among the different age groups.
Immunohistochemistry for NEP and Aβ in brain (canine).∗
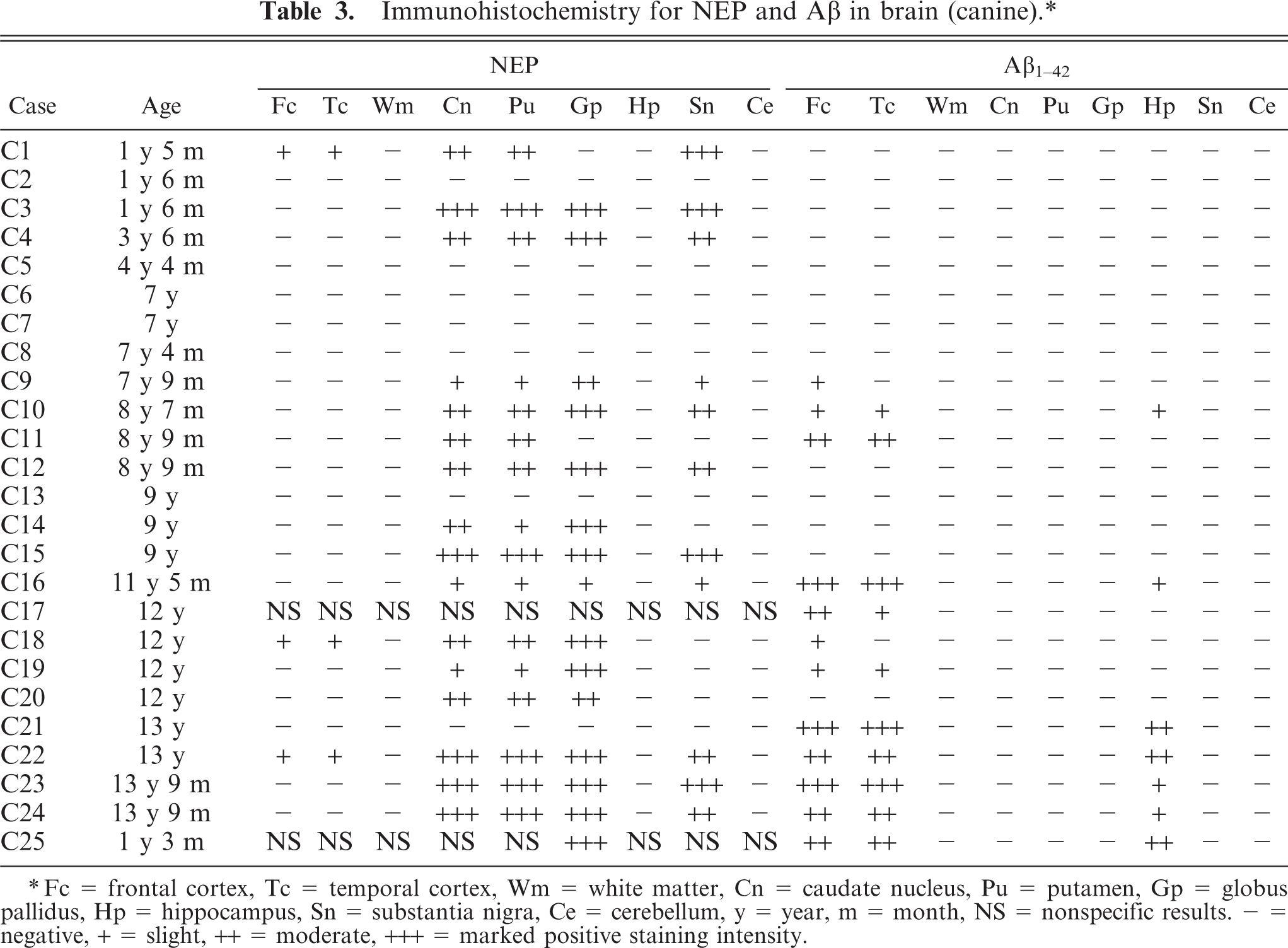
Fc = frontal cortex, Tc = temporal cortex, Wm = white matter, Cn = caudate nucleus, Pu = putamen, Gp = globus pallidus, Hp = hippocampus, Sn = substantia nigra, Ce = cerebellum, y = year, m = month, NS = nonspecific results. - = negative, + = slight, ++ = moderate, +++ = marked positive staining intensity.
Immunohistochemistry for NEP and Aβ in brain (feline).∗

Fc = frontal cortex, Tc = temporal cortex, Wm = white matter, Cn = caudate nucleus, Pu = putamen, Gp = globus pallidus, Hp = hippocampus, Sn = substantia nigra, Ce = cerebellum, y = year, m = month, NS = nonspecific results. - = negative, + = slight, ++ = moderate, +++ = marked positive staining intensity.
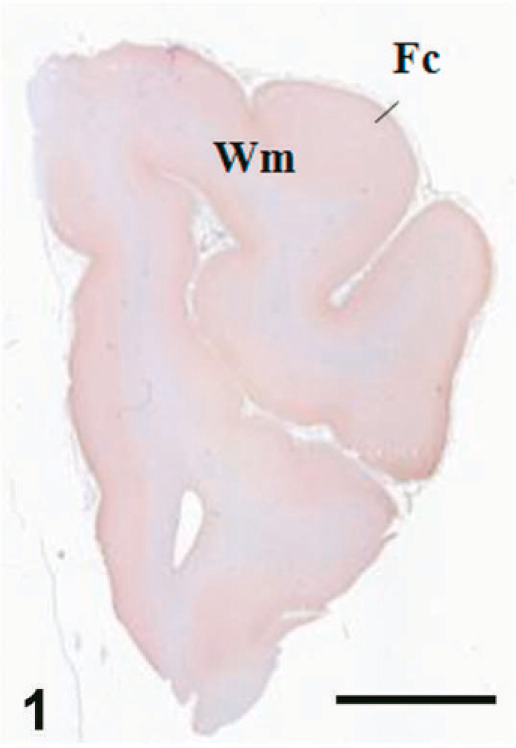
Case C22. Immunohistochemistry for NEP of a canine brain. Transverse section, including the cerebral cortex and white matter. Scale bar = 5 mm. Fc = frontal cortex, Wm = white matter.
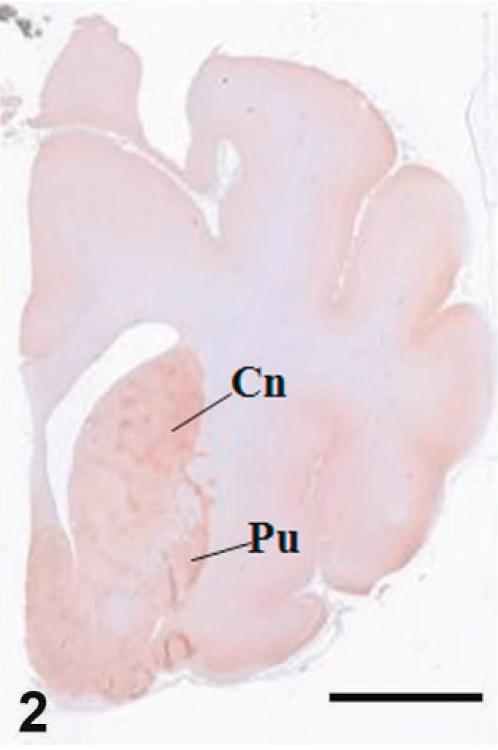
Case C22. Immunohistochemistry for NEP of a canine brain. Transverse section, including putamen and caudate nucleus. Scale bar = 5 mm. Cn = caudate nucleus, Pu = putamen.
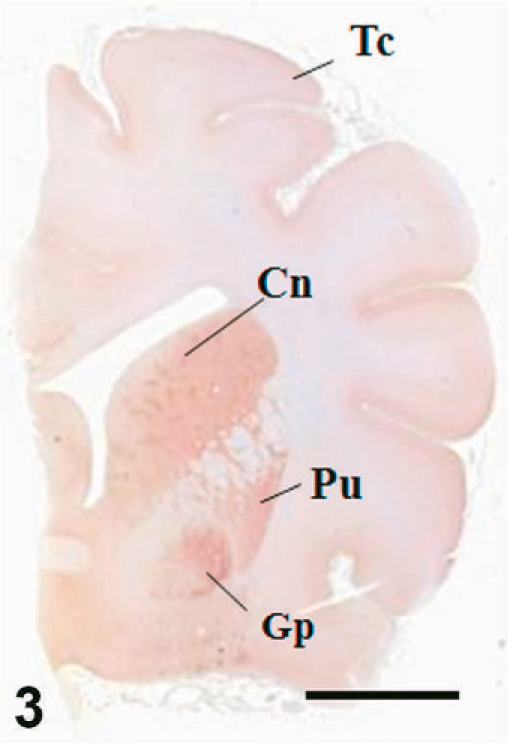
Case C22. Immunohistochemistry for NEP of a canine brain. Transverse section, including putamen, caudate nucleus, and globus pallidus. Scale bar = 5 mm. Tc = temporal cortex, Cn = caudate nucleus, Pu = putamen, Gp = globus pallidus.
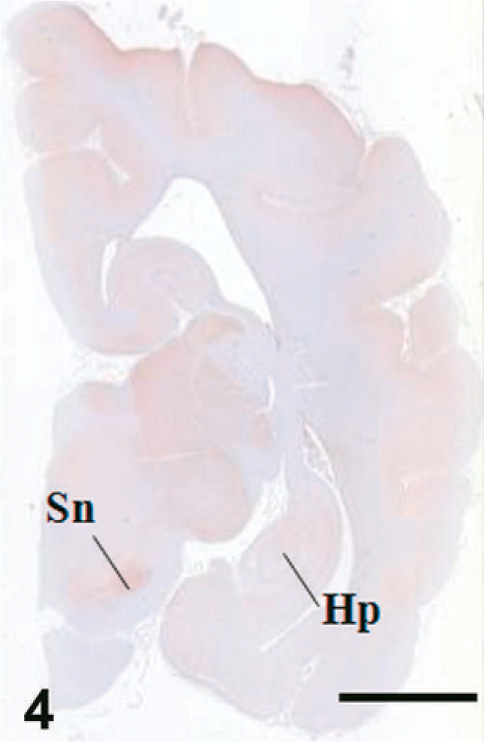
Case C22. Immunohistochemistry for NEP of a canine brain. Transverse section, including hippocampus and substantia nigra. Scale bar = 5 mm. Hp = hippocampus, Sn = substantia nigra.
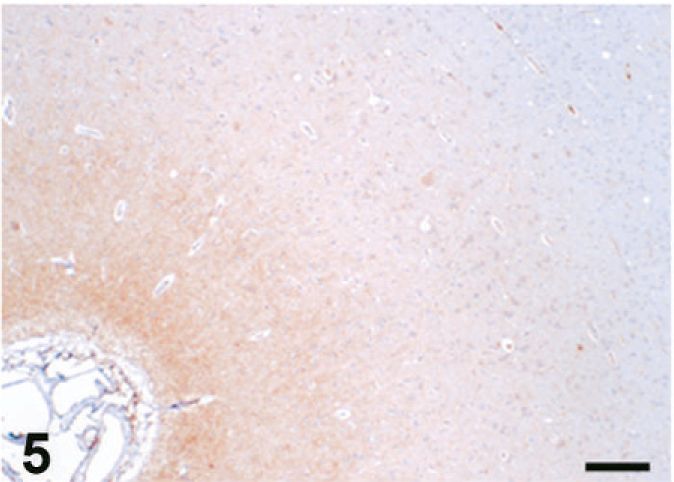
Case C22. Immunohistochemistry for NEP of a canine brain. Photograph represents a high-power view of the cerebral cortex. Scale bar = 200 µm.
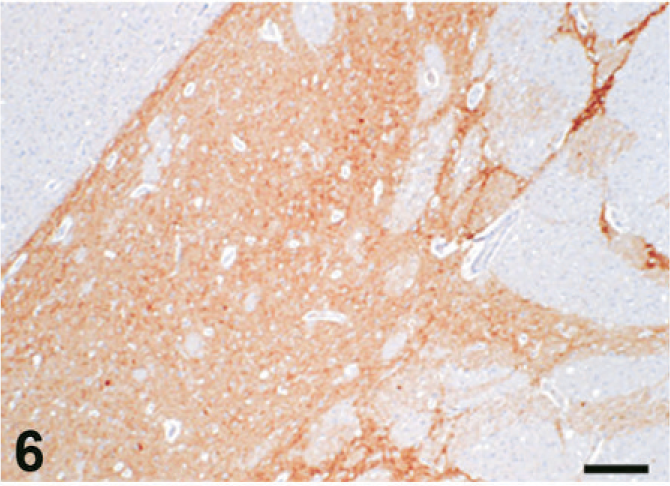
Case C22. Immunohistochemistry for NEP of a canine brain. Photograph represents a high-power view of the putamen. Scale bar = 200 µm.
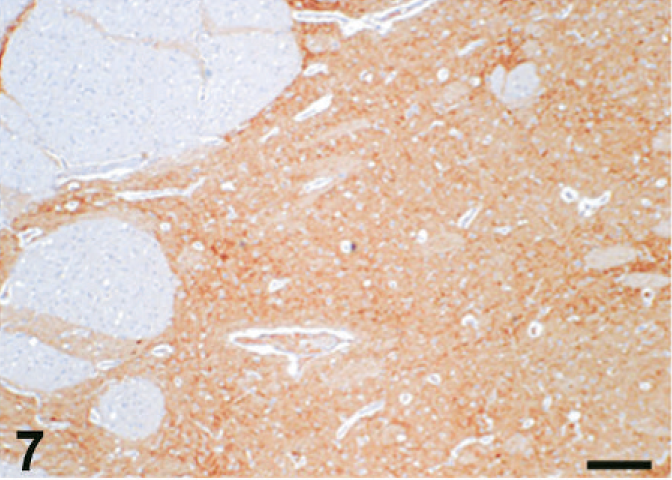
Case C22. Immunohistochemistry for NEP of a canine brain. Photograph represents a high-power view of the caudate nucleus. Scale bar = 200 µm.
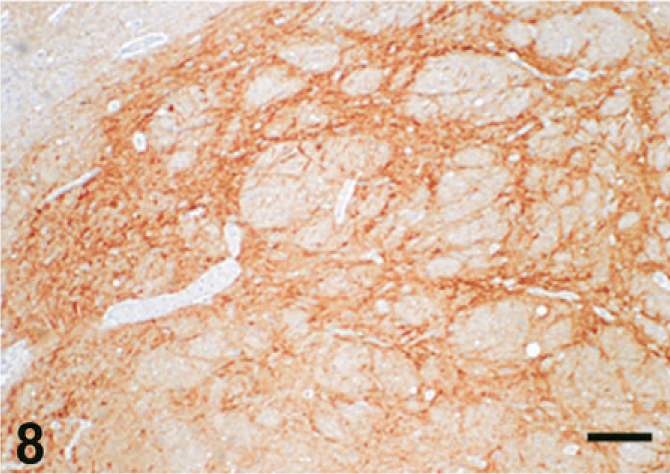
Case C22. Immunohistochemistry for NEP of a canine brain. Photograph represents a high-power view of the globus pallidus. Scale bar = 200 µm.
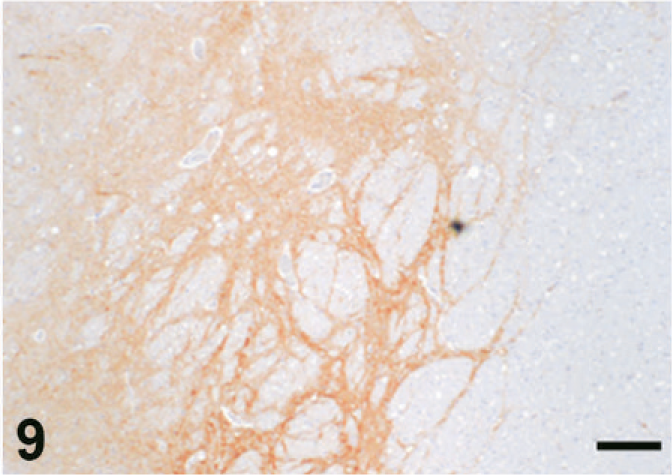
Case C22. Immunohistochemistry for NEP of a canine brain. Photograph represents a high-power view of the substantia nigra. Scale bar = 200 µm.
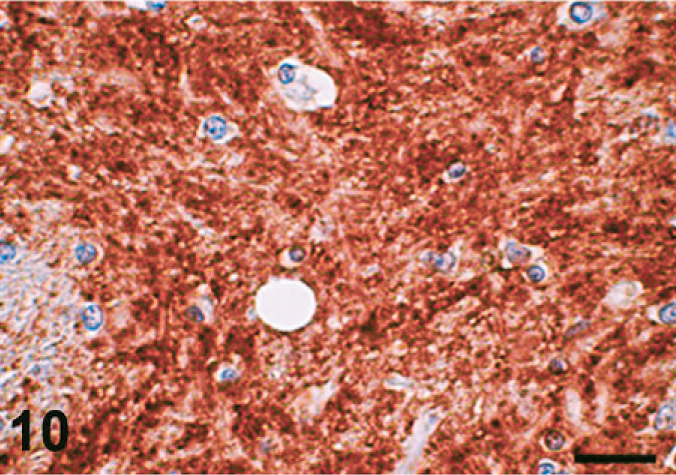
Case C22. Immunohistochemistry for NEP of a canine brain. Photograph represents a high-power view of the globus pallidus. Scale bar = 20 µm.
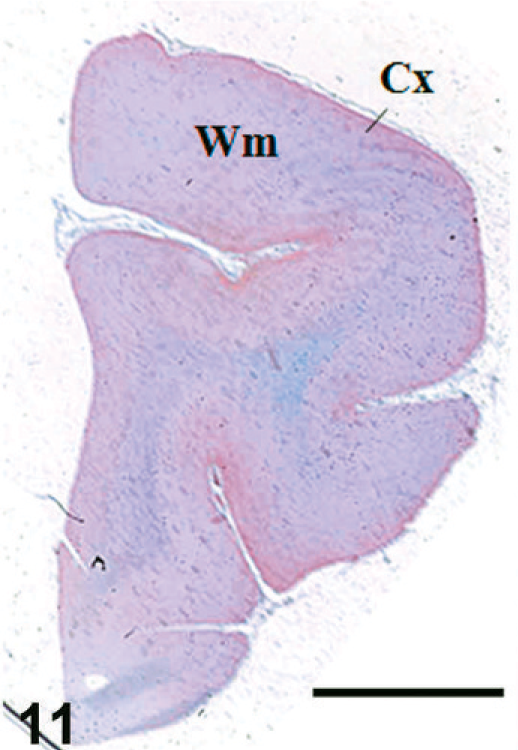
Case F11. Immunohistochemistry for NEP of a feline brain. Transverse section, including the cerebral cortex and white matter. Scale bar = 5 mm. Fc = frontal cortex, Wm = white matter.
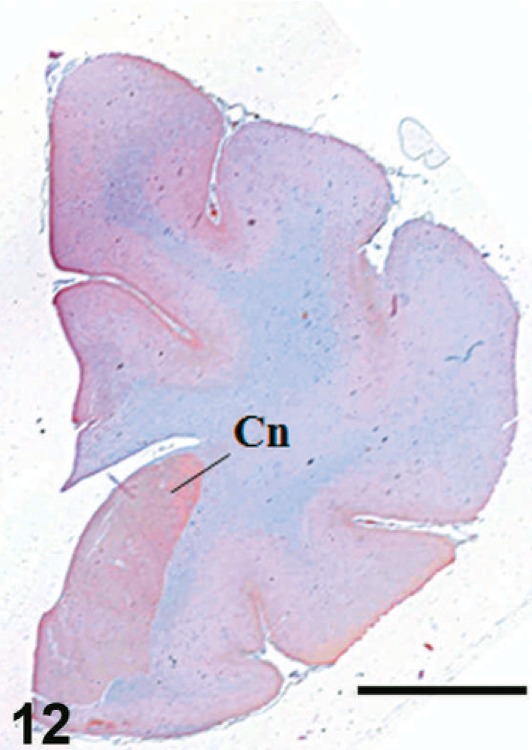
Case F11. Immunohistochemistry for NEP of a feline brain. Transverse section, including caudate nucleus. Scale bar = 5 mm. Cn = caudate nucleus.
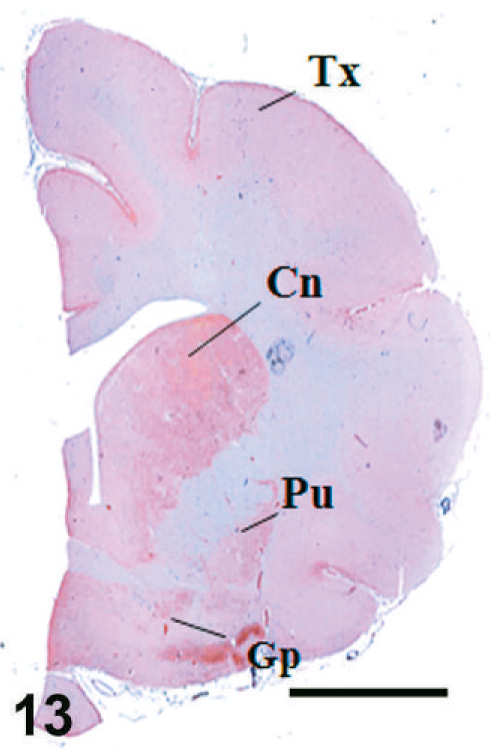
Case F11. Immunohistochemistry for NEP of a feline brain. Transverse section, including putamen, caudate nucleus, and globus pallidus. Scale bar = 5 mm. Tc = temporal cortex, Cn = caudate nucleus, Pu = putamen, Gp = globus pallidus.
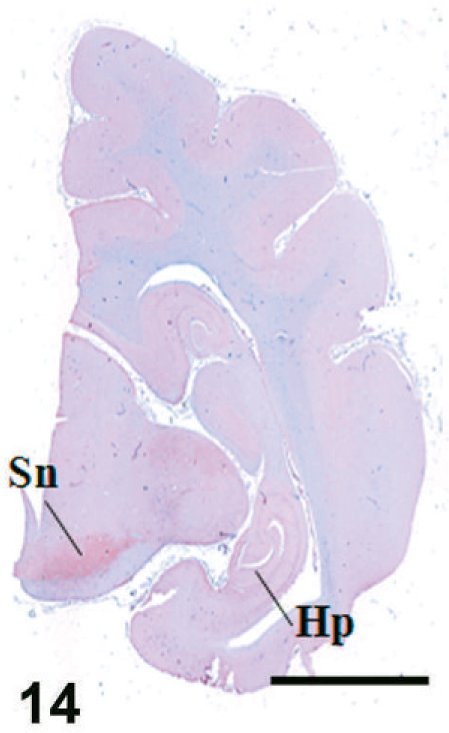
Case F11. Immunohistochemistry for NEP of a feline brain. Transverse section, including hippocampus and substantia nigra. Scale bar = 5 mm. Hp = hippocampus, Sn = substantia nigra.
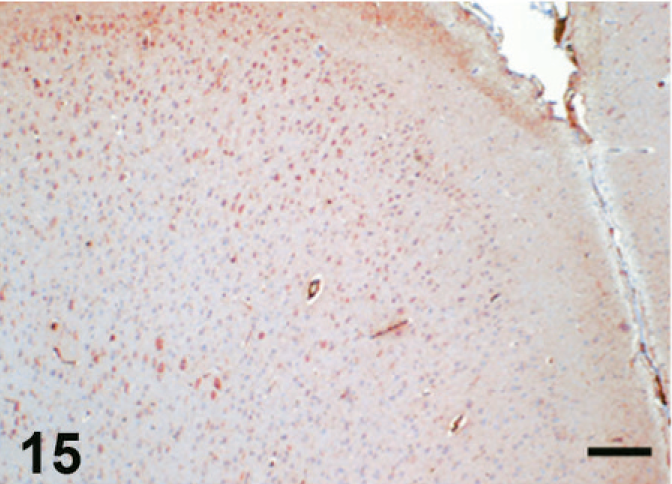
Case F3. Immunohistochemistry for NEP of a feline brain. Photograph represents a high-power view of the cerebral cortex. Scale bar = 200 µm.

Case F3. Immunohistochemistry for NEP of a feline brain. Photograph represents a high-power view of the putamen. Scale bar = 200 µm.

Case F3. Immunohistochemistry for NEP of a feline brain. Photograph represents a high-power view of the caudate nucleus. Scale bar = 200 µm.
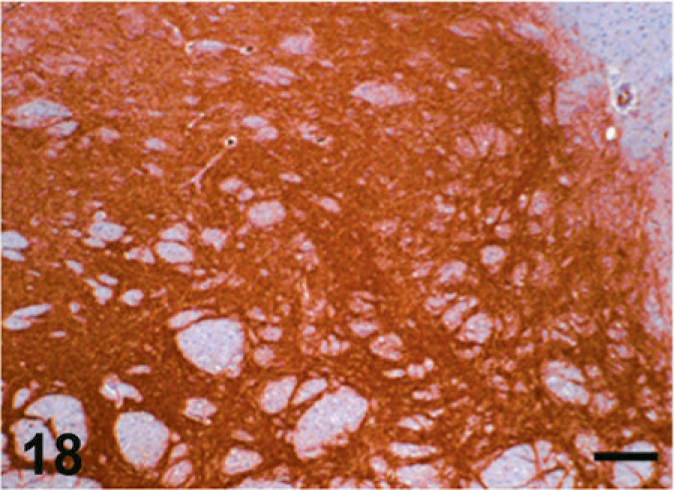
Case F3. Immunohistochemistry for NEP of a feline brain. Photograph represents a high-power view of the globus pallidus. Scale bar = 200 µm.
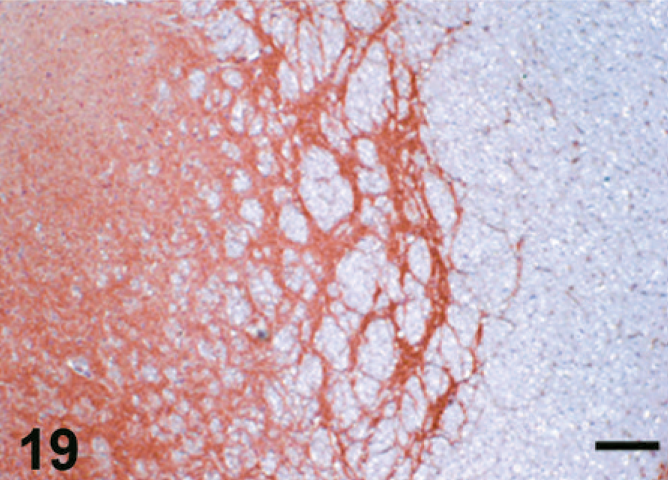
Case F3. Immunohistochemistry for NEP of a feline brain. Photograph represents a high-power view of the substantia nigra. Scale bar = 200 µm.
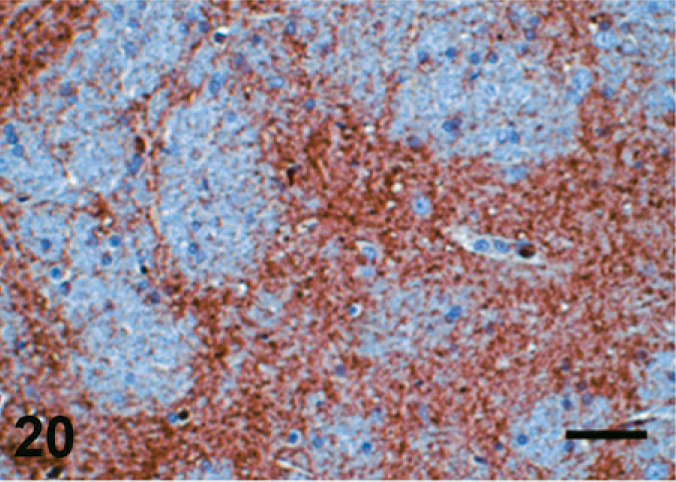
Case F3. Immunohistochemistry for NEP of a feline brain. Photograph represents a high-power view of the globus pallidus. Scale bar = 20 µm.
Cerebral Aβ deposition was first detected at 7 years and 9 months in the dog and at 10 years old in cat, and both the quantity and frequency of deposition increased with age. In dogs, Aβ deposits were observed in the cerebral cortex, hippocampus, and meningeal vessel wall. In cats, the deposits appeared in the cerebral cortex and hippocampus but not in the meningeal vessel wall. Aβ deposition occurred in 2 patterns: diffuse accumulation (see Figs. 21–24) and diffuse-type senile plaques (see Figs. 25 and 26) in the cerebral parenchyma of both dogs and cats. No Aβ deposition was observed in the white matter, caudate nucleus, putamen, globus pallidus, substantia nigra, or cerebellum of either species.
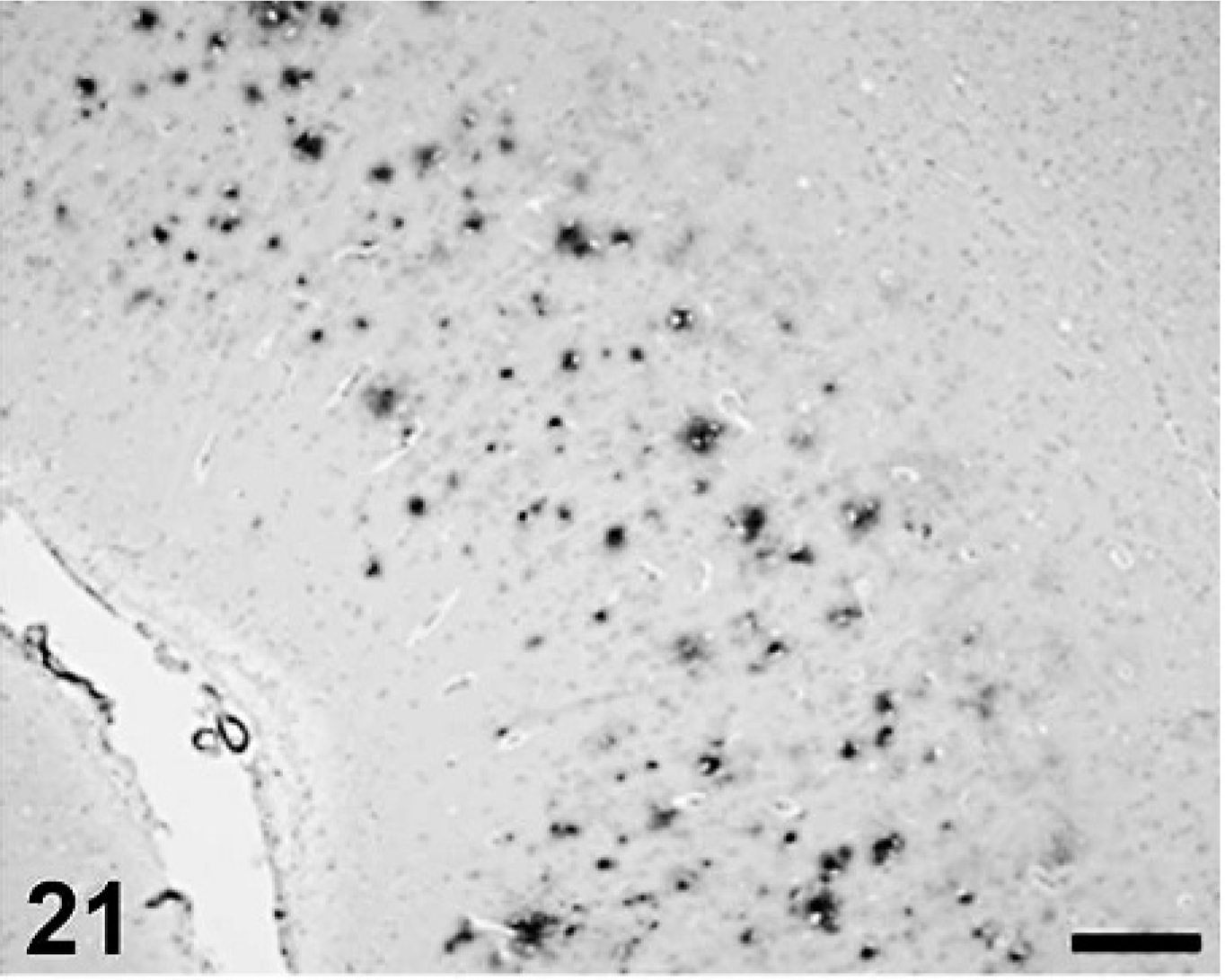
Aβ deposition in canine frontal cortex. Scale bar = 200 µm.
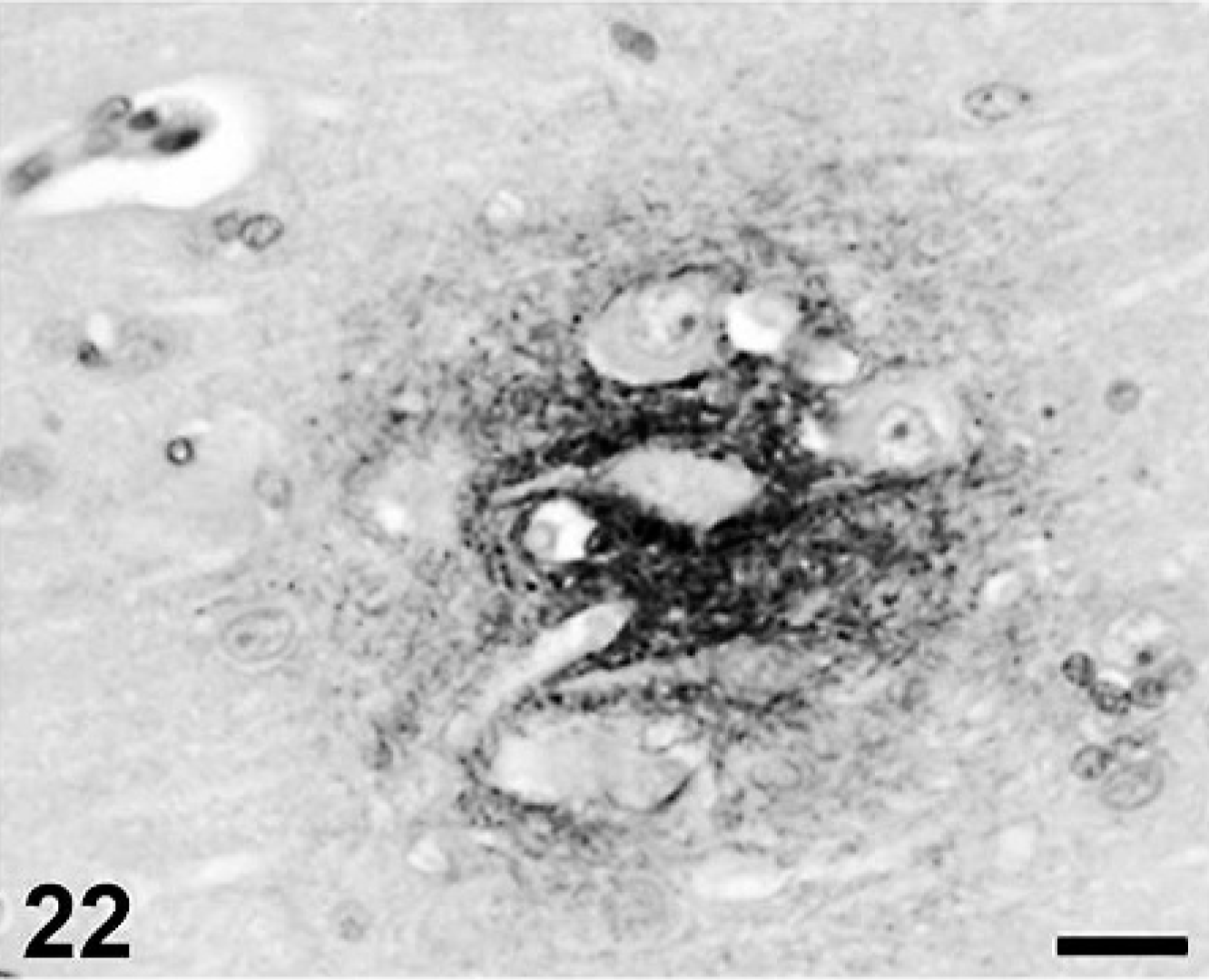
Aβ deposition in canine frontal cortex. Photograph is a high-power view of Fig. 23. Scale bar = 20μm.
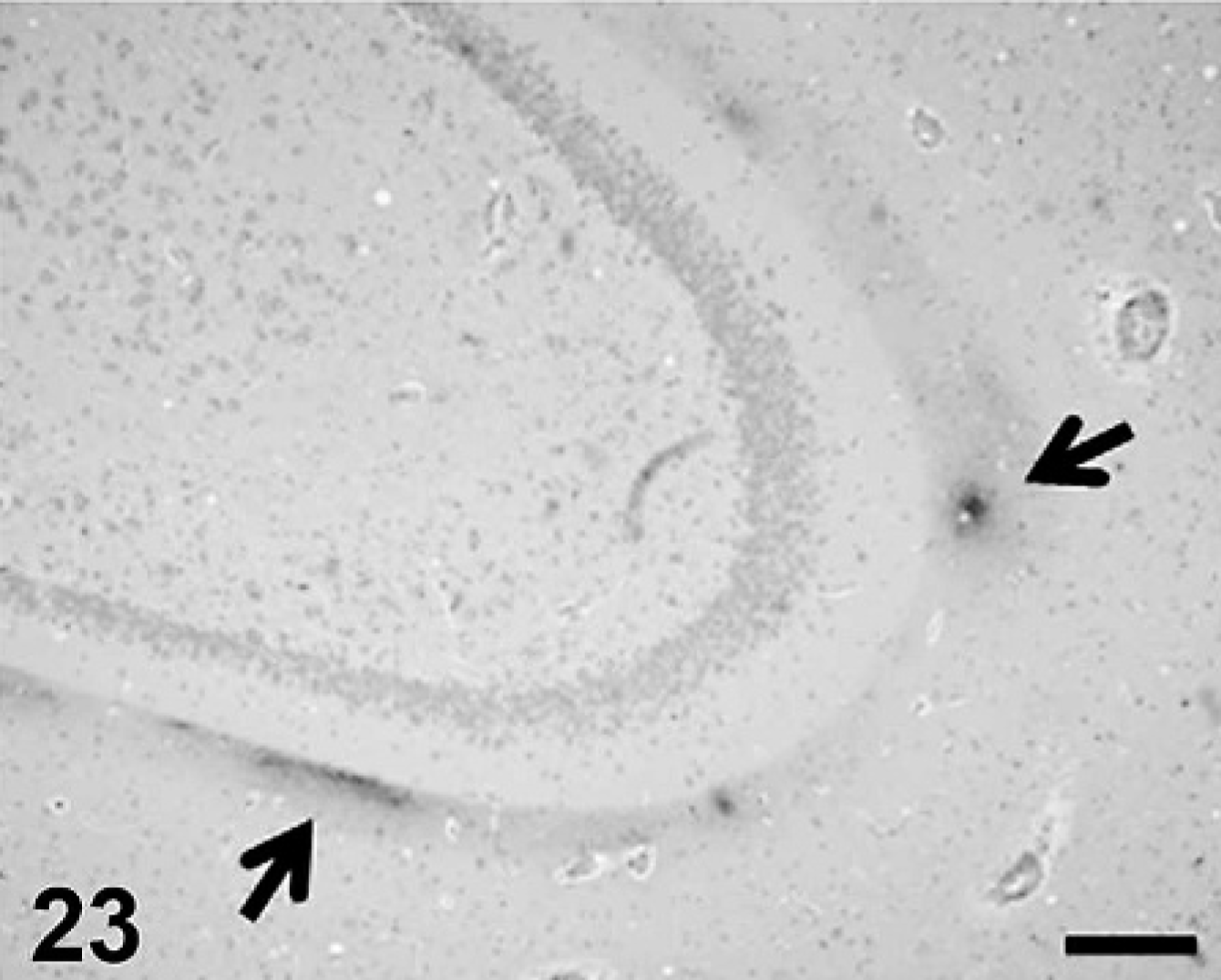
Aβ deposition in canine hippocampus (arrows). Scale bar = 200μm.

Aβ deposition in feline frontal cortex. Scale bar = 200 µm.
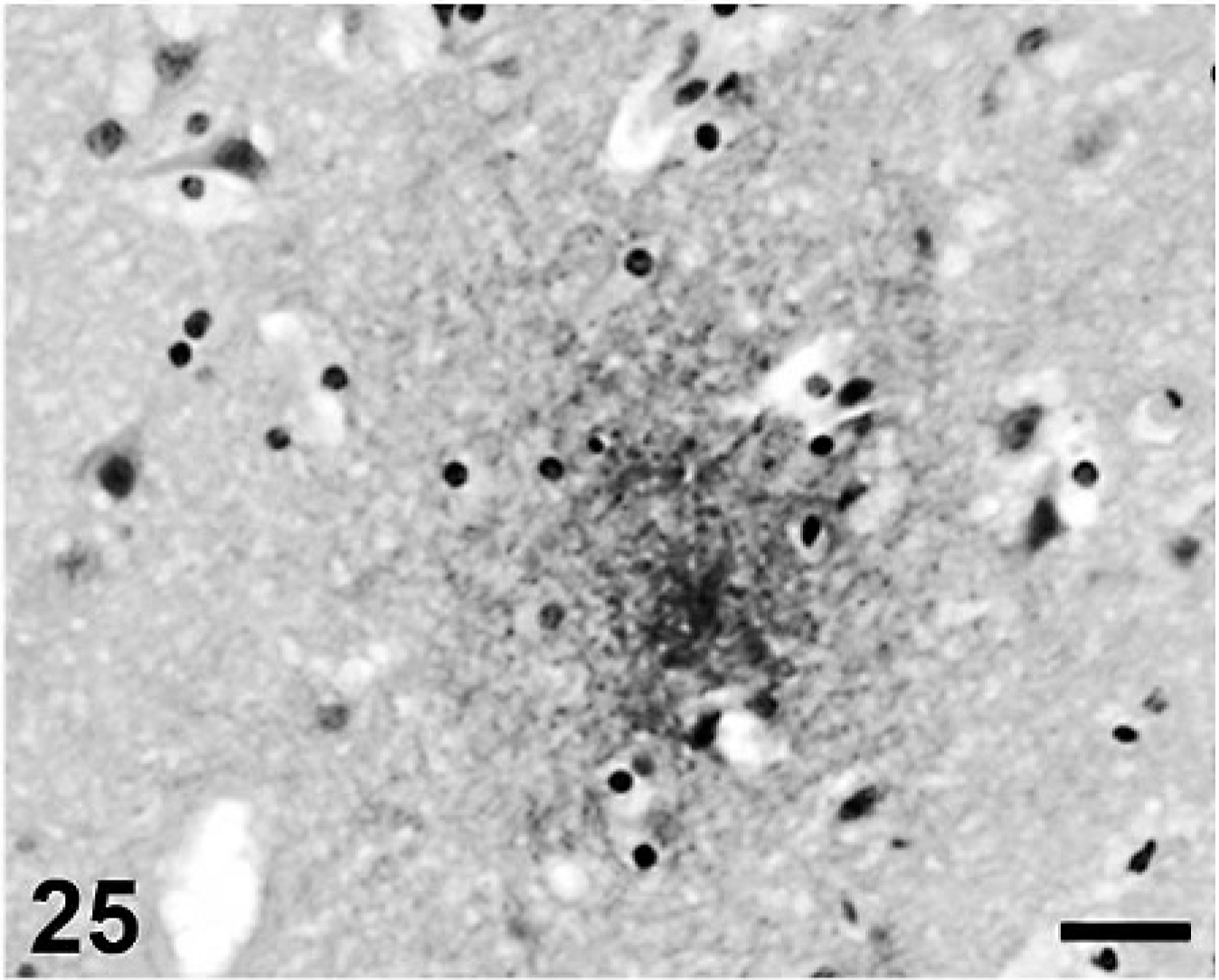
Aβ deposition in feline frontal cortex. Photograph is a high-power view of Fig. 26. Scale bar = 20 µm.
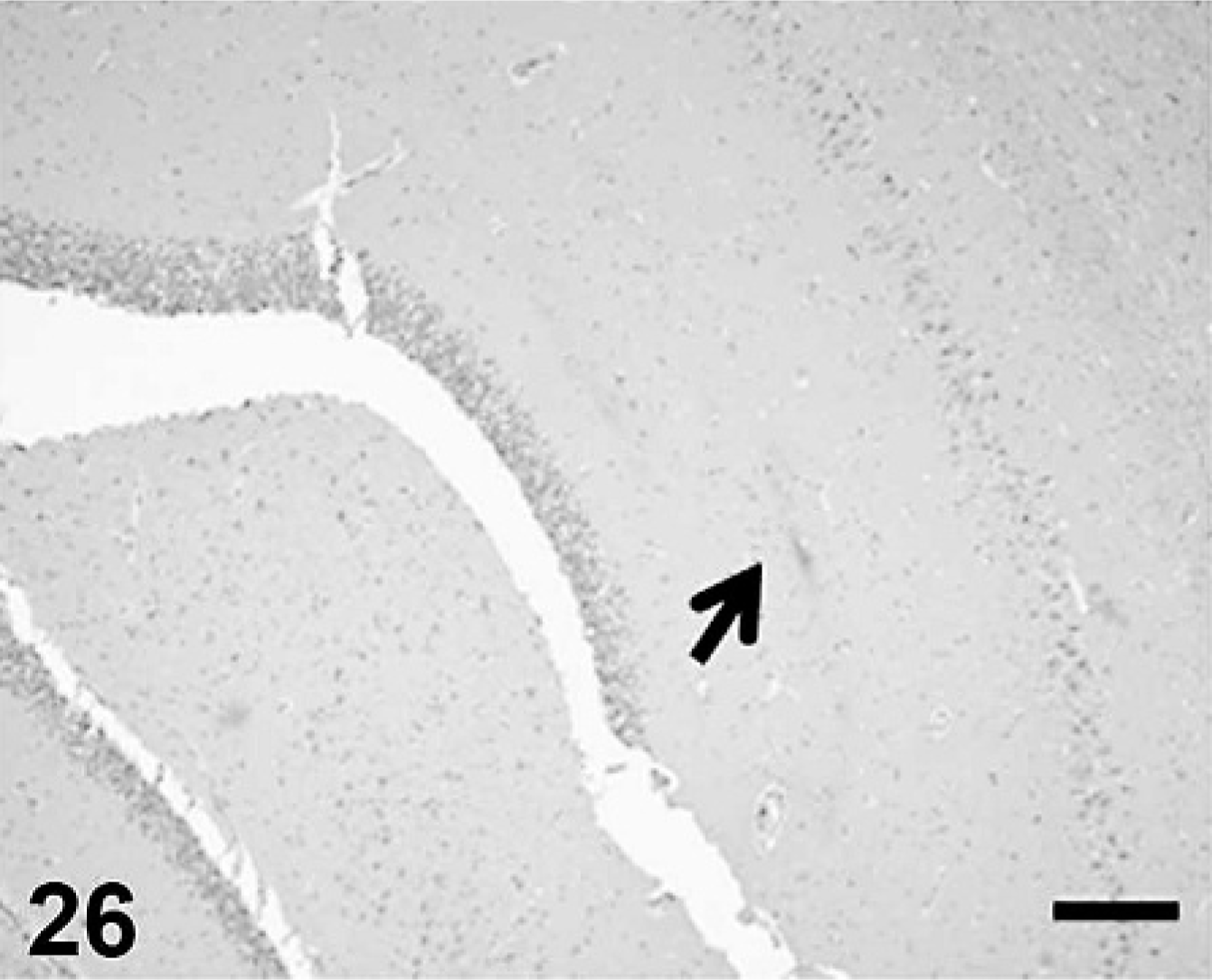
Aβ deposition in feline hippocampus (arrow). Scale bar = 200 µm.
NEP activity in the brain
The results of the NEP activity assay in the dog brain are shown in Fig. 27. The animals were divided into 4 age groups: 1) 4 years or less, 2) 4 to 8 years, 3) 8 to 12 years, and 4) more than 12 years (see Fig. 27). A negative correlation was found between NEP activity in the thalamus/striatum and age (r = −0.69; P < .05) (see Fig. 27A). However, there were no correlations between NEP activity in the cerebral cortex (r = −0.08; P = .7), white matter (r = −0.1; P = .65), or hippocampus (r = 0.08; P = .72), and age. In all groups, the NEP activity in the various brain region was ranked from strong to weak; in the following order, thalamus/striatum >> cerebral cortex > hippocampus ≥ white matter.
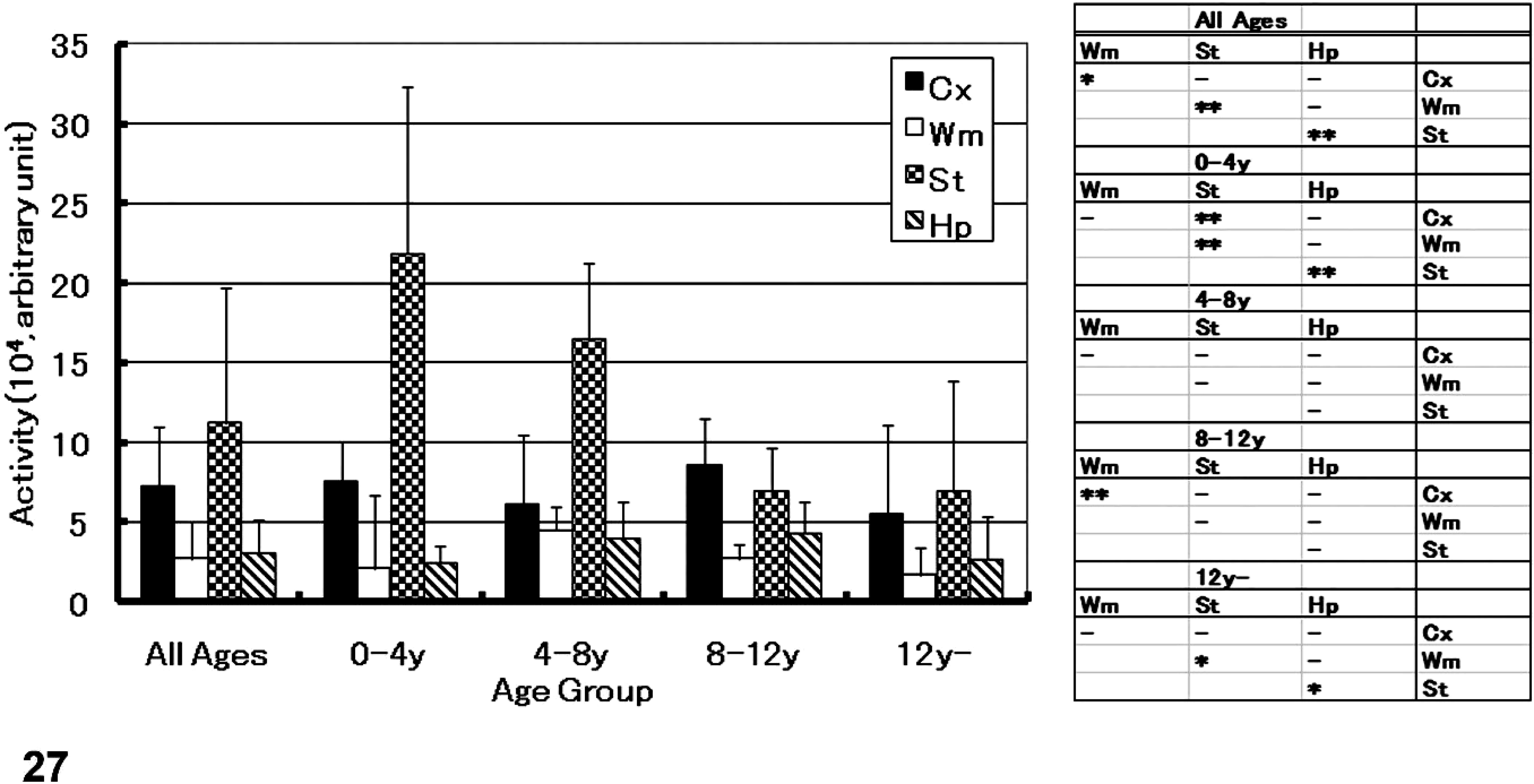
NEP activity in the canine brain; NEP activity in brain regions of each age group and results of statistical analyses, in which the significance of each difference between brain regions are indicated. Values in each region are represented as mean (SD). ∗ = P < .05, ∗∗ = P < .01, (–) = not significant (one-way ANOVA and Bonferroni test), Cx = cerebral cortex, Wm = white matter, St = striatum, and Hp = hippocampus.
The results of the NEP activity assay in the cat brain are shown in Fig. 28. There were no apparent correlations between NEP activity in the various brain regions and age (cerebral cortex [r = −0.47; P = .13], white matter [r = 0.17; P = .59], thalamus/striatum [r = 0.04; P = .89], and hippocampus [r = −0.07; P = .83]). The NEP activity of the 4 feline age groups were the following: thalamus/striatum >> cerebral cortex > hippocampus > white matter. The activity was not changed according to aging in cats (see Fig. 28A).
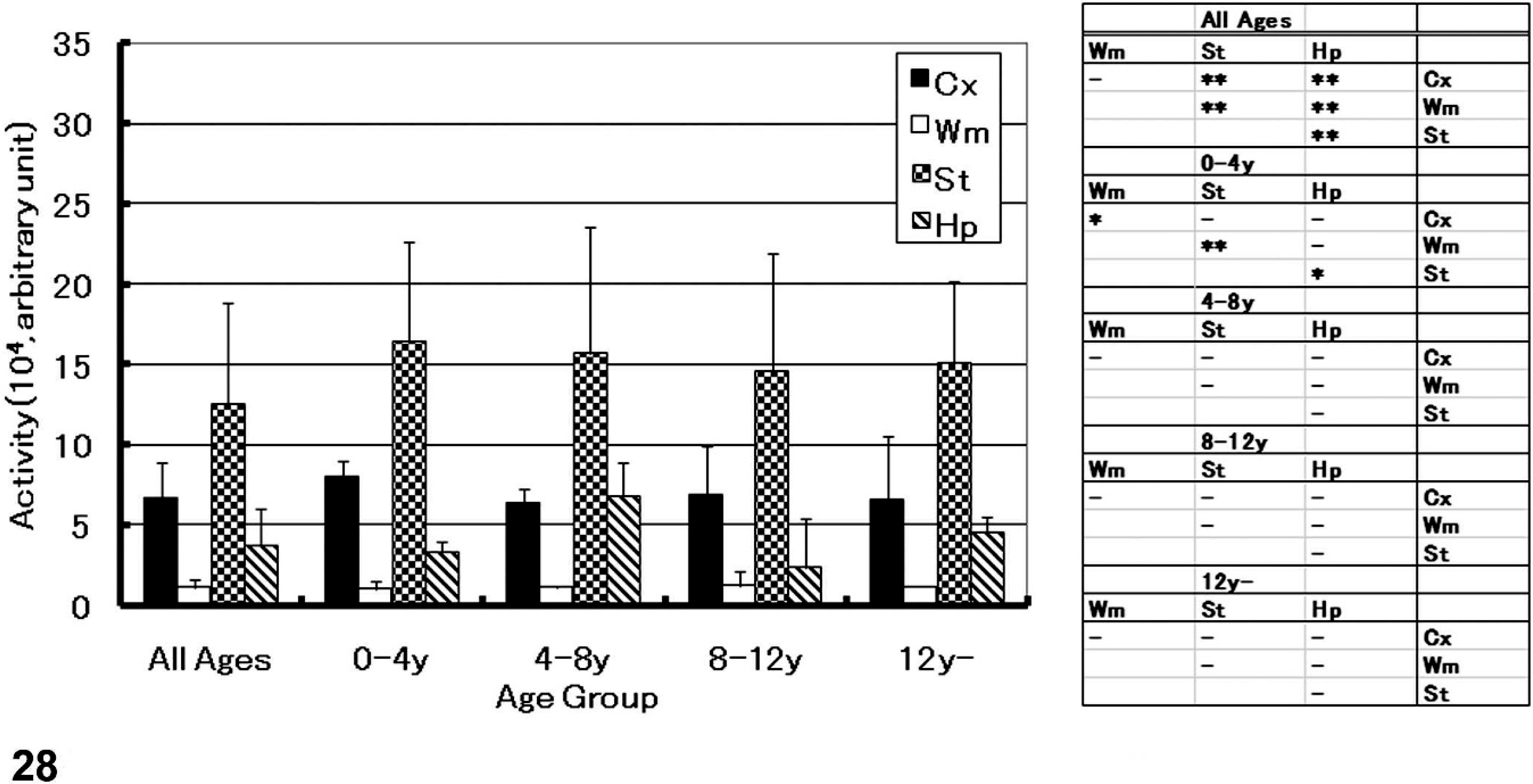
NEP activity in the feline brain; NEP activity in brain regions of each age group and results of statistical analyses, in which the significance of each difference between brain regions are indicated. Values in each region are represented as mean (SD). ∗ = P < .05, ∗∗ = P < .01, (–) = not significant (one-way ANOVA and Bonferroni test), Cx = cerebral cortex, Wm = white matter, St = striatum, and Hp = hippocampus.
Discussion
In the present study, the age of the youngest canine case of Aβ accumulation in the brain was approximately 8 years old, and the accumulation appeared in the cerebral cortex, hippocampus, and meningeal vessel wall. Diffuse-type senile plaques were found mainly in the cerebral cortex. These findings were consistent with those in several previous reports. 12, 19, 45
Senile plaques were reported to appear in cats aged more than 18 years old, and the plaques were reported to be morphologically different from those in dogs. 35 However, in the present study, feline Aβ accumulation appeared in cats as young as 10 years old. The localization of the accumulation was the same as in dogs (excluding the meningeal vessels), and the morphologic characteristics of feline senile plaques were more similar to those of dogs than had previously been thought. Brellou et al. 8 reported recently that Aβ accumulation in the brain appeared in a cat as young as 7.5 years old.
It, therefore, is concluded that Aβ accumulation in both dogs and cats might first appear at middle age (8–10 years old). The accumulation is detected in the cerebral cortex and hippocampus as diffuse plaques. It is currently considered that the life span of dogs and cats is approximately 20 years. Thus, the aging process and the rate of Aβ accumulation may be similar in these 2 animal species.
In the present study, NEP was abundantly detected in the striatum (caudate nucleus and putamen), globus pallidus, and substantia nigra, but was less abundant in the cerebral cortex and was never detected in the white matter of either dogs or cats. Such NEP distribution in these species is consistent with the reported distribution in of mice, 16 pigs, 29 and humans. 2 However, whereas NEP immunoreactivity was not detected in the hippocampus of dogs and cats, it was found in the hippocampus of mice 16 and pigs. 4
On microscopic examination, NEP was distributed as fine granules in neuronal cell bodies and dendrites, as is reported in other species. 2, 16, 29 This synaptic and axonal localization suggests that, after it is synthesized in the neuron body, NEP, a membrane-integrated protein, is axonally transported to the presynaptic terminals, where Aβ degradation is likely to take place. 22, 44
NEP activity in various brain regions in both dogs and cats was ranked from high to low as follows: thalamus/striatum (including striatum and globus pallidus) > cerebral cortex > hippocampus > white matter. These findings were identical to those in mice. 16 The distribution of NEP protein, as determined by immunohistochemistry, correlated well with the distribution of NEP activity in the present study. In the cerebral cortex and the hippocampus, NEP immunoreactivity was low or negative, although NEP activity was observed. Therefore, we think that immunohistochemistry is less sensitive for identification of NEP compared with the activity assay.
In some animals (C2, C5–8, C13, C21, F1, F2, F5, F6, F8, and f12), NEP was not expressed in any brain region. In the present study, neither living environment nor breed was considered as a factor for inclusion or exclusion of cases for evaluation. Therefore, it is possible that NEP expression in the brain might be different among breeds.
NEP expression and activity in the striatum, globus pallidus, and substantia nigra of both species were higher than those in other brain regions. The striatum, globus pallidus, and substantia nigra are parts of the striatal system. In addition to the strong expression of NEP in this system, substance P, a substrate of NEP, is abundantly present there. 2, 6, 17 NEP activity in this system decreases with age in dogs but not in cats. In the striatal system of dogs and cats, we detected no changes of NEP protein expression with aging. In the striatal system of human patients with AD, NEP shows substantial expression, 2 and AD-related lesions, such as senile plaques and neurofibrillary tangles, and synaptic loss are milder than in the cerebral cortex and the hippocampus. 32 In addition, Iwata et al. 24 reported that Aβ density in the thalamus/striatum is lower than that in the cerebral cortex and the hippocampus of NEP-deficient mice. Therefore, it is possible that Aβ production itself is lower in the striatal system of dogs and cats than in other brain regions.
There was a very low NEP expression in the cerebral cortex, and no expression in the hippocampus in dogs and cats. NEP activity in these regions was low as well and was not changed by aging. In contrast, NEP expression and activity decrease with age in the mouse hippocampus. 9, 23 NEP expression is also decreased in the cerebral cortex of patients with AD but not in the normal aged brain. 2, 51 Taken together, these facts suggest that, because NEP expression and activity in the cerebral cortex are lower than those in the striatum, Aβ is not sufficiently degraded and consequently accumulates in the cortex.
Although NEP was not detected and NEP activity was very low in the white matter of dogs and cats, Aβ deposition was not observed there. Little Aβ deposition is observed in the corresponding region of patients with AD. 48 Thus, Aβ deposition is not necessarily observed in the regions where NEP activity is low. In fact, Aβ is degraded by not only NEP but also endothelin-converting enzyme, insulin-degrading enzyme (IDE), and angiotensin-converting enzyme. 49 NEP cannot efficiently degrade some kinds of Aβ variants, for example, those with the familial AD-linked Flemish-, Arctic-, Italian-, and Dutch-type mutations in their peptide sequence. However, IDE is able to degrade these Aβ variants, as well as Iowa-type mutant Aβ, although its activity toward the Arctic- and Dutch-type mutants is low. Therefore, IDE rather than NEP would be expected to play a key role in the removal of such hereditary variants of Aβ from the cerebral vessel wall or brain parenchyma. 22, 33 NEP is thought to be capable of degrading monomers and oligomers of Aβ but not fibrilized Aβ. 22, 26 In this respect, phagocytosis of Aβ by microglia 3, 37 and/or intake of Aβ by astrocytes 28, 52 appear to be critically involved in the clearance of fibrillar Aβ from the brain parenchyma rather than the degradation by NEP.
In conclusion, the distribution of NEP and NEP activity and that of Aβ were observed to be complementary in the brains of dogs and cats. Interestingly, the NEP distributions in these species were almost identical to one another and to those of the human, pig, and mouse. Thus, the Aβ clearance pathway in the brain might be preserved among mammalian species. Because dogs and cats share their living environments with humans, they might be the best-suited animal model for the pathogenesis of AD.
Footnotes
Acknowledgements
We thank Dr. Sunao Manabe, director of Medicinal Safety Research Laboratories, Sankyo Co., Ltd., for the generous donation of 10 dog brains.
