Abstract
Helicobacter spp. have been implicated in a variety of gastrointestinal tract diseases, including peptic ulcer disease, gastric cancer, and inflammatory bowel disease (IBD), in humans and animals. Although most models of IBD are experimentally induced, spontaneous or natural models of IBD are rare. Herein, we describe a long-term study of chronic, progressive lesions that develop in the distal portion of the large bowel of unmanipulated Syrian hamsters naturally infected with Helicobacter spp. Twenty-four Syrian hamsters of three age groups (group A, 1 month [n = 4], group B, 7–12 months [n = 12], group C, 18–24 months [n = 12]), underwent complete postmortem examination. Results of microbial isolation and polymerase chain reaction and restriction fragment length polymorphism analyses confirmed the presence of Helicobacter spp. infection in the distal portion of the large bowel of all animals. Additionally, confounding pathogens, such as Clostridium difficile, Lawsonia intracellularis, and Giardia spp. that can cause proliferative enteritis, were absent in the hamsters of this study. Histopathologic scores for inflammation (P < 0.01), hyperplasia (P < 0.01), and dysplasia (P < 0.05) were significantly higher in the ileocecocolic (ICC) junction of animals in group C, relative to group A. Dysplastic lesions of various grades were detected in 5 of 11 hamsters in group C. Interestingly, the segment of the bowel that is usually colonized by Helicobacter spp. in hamsters had the most severe lesions. One hamster of group C developed a malignant fibrous histiocytoma, whereas another hamster developed a round cell sarcoma originating from the ICC junction. Thus, lesions in the distal portion of the large bowel of aging hamsters naturally colonized with Helicobacter spp. warrants developing the hamster as an animal model of IBD and potentially IBD-related cancer.
Inflammatory bowel disease (IBD), including ulcerative colitis and Crohn's disease, is a chronic inflammatory disease of the human gastrointestinal tract of nonspecific etiology that is associated with high risk of developing cancer. 3 The etiology of IBD is considered to be multi-factorial, and includes factors such as host genetics, diet, enteric microflora, and oral tolerance. 13 Most of the animal models used to study IBD are genetically modified or immunodeficient. 2, 54 However, there is a paucity of spontaneous models of IBD, which currently include cotton top tamarins (Saguinus oedipus) and C3H-HeJBir and SAMP1/Yit mice. 2, 6, 52 Unfortunately, use of these spontaneous models is limited, as cotton-top tamarins are an endangered species and the murine models do not recapitulate the human disease completely. The multi-factorial nature of IBD and the paucity of spontaneous models of IBD, therefore, give an impetus to developing new animal models.
Helicobacter spp. have been implicated in a variety of gastrointestinal tract diseases, including peptic ulcer disease, gastric cancer, IBD, and colon cancer, in humans and animals. 1, 10, 29, 36, 50, 51 Although several Helicobacter species have been linked to typhlocolitis and enterohepatic cancers in murine models, 4, 11, 30, 48, 56 most of the available rodent models of Helicobacter-induced IBD are genetically manipulated and/or immunodeficient. 2, 54 Hamsters, however, are naturally infected with several enterohepatic Helicobacter spp. 14, 18, 26, 37, 49 We reported earlier that hamsters naturally infected with Helicobacter spp. develop chronic gastritis and precursor lesions of gastric cancer. 37 Despite colonization of the ileocecocolic (ICC) junction by Helicobacter spp., the long-term pathologic changes in the large bowel of hamsters are largely unknown. Here, we report the findings from a long-term study and describe chronic, progressive lesions that develop in the distal portion of the large bowel of unmanipulated Syrian hamsters naturally infected with multiple Helicobacter spp.
Materials and Methods
Animals
Twenty-four Syrian hamsters (Mesocricetus auratus) of three age groups (group A, 1 month [n = 4], group B, 7–12 months [n = 12], and group C, 18–24 months [n = 12] were studied. Group A was composed of only female animals; group B had seven females and five males; and group C was composed of three females and nine males. Animals from a commercial vendor were housed in an Association for the Assessment and Accreditation of Laboratory Animal Care-approved animal facility at the Massachusetts Institute of Technology. All animals were housed in solid-bottom polycarbonate cages with hard wood bedding, and were fed LabDiet 501 (RPMI Nutritional International Inc., Brentwood, MO) and water ad libitum. Animals were euthanatized by means of CO2 inhalation and underwent complete postmortem examination.
Histologic examination
Representative tissue sections from all of the organ systems, including the entire gastrointestinal tract, were collected at necropsy. Tissues were immersion-fixed in neutral-buffered 10% formalin and embedded in paraffin, and 5-µm-thick sections were stained with hematoxylin and eosin (HE). Changes in the ICC junction and colon were scored by a veterinary pathologist (PRN) for inflammation and hyperplasia on the basis of: degree of severity—0 (absent), 1 (mild), 2 (moderate), 3 (marked), and 4 (severe); and distribution—1 (focal), 2 (multifocal), 3 (locally extensive/segmental), and 4 (diffuse). Histologic criteria from earlier publications 24, 45 were modified and used to subclassify dysplasia as mild (1) and moderate (2). Briefly, hypercellular, slightly irregular crypts with nuclei confined to the basal half of the cell and moderate decrease in goblet cells; mild-to-moderate nuclear pseudostratification, minimal anisokaryosis, and mild-to-moderate increase in the nucleus-to-cytoplasm ratio were classified as mildly dysplastic crypts. Similarly, moderate dysplastic lesions were classified on the basis of increased severity of the aforementioned features, including tortuous-to-cribriform crypts, marked decrease or complete loss of goblet cells, and haphazard nuclear pseudostratification with nuclei between the basal and apical halves. Atypical lesions associated with active colitis or erosions/ulcers were precluded from the dysplasia category. 24, 45 Distribution scores for dysplasia were similar to those of inflammation and hyperplasia. Products of lesion (inflammation, hyperplasia, dysplasia) severity and distribution scores were used as the final score for each animal. To test significance among the groups, a nonparametric Kruskal-Wallis test followed by Dunn's test for multiple comparisons were performed using Prism 4 statistics software package (GraphPad, San Diego, CA). A value of P < 0.05 was considered significant.
Helicobacter spp. culture and isolation
Culture for microaerophilic bacteria was performed on harvested cecal tissue from 4, 10, and 5 animals of groups A, B, and C, respectively. Tissues were rinsed in sterile phosphate-buffered saline, and were homogenized in sterile Brucella broth (Difco Laboratories, Detroit, MI) containing 5% fetal bovine serum (Summit Biotechnology, Ft. Collins, CO). Samples were inoculated onto culture media as described. 38 Plates were then incubated at 37°C under microaerobic conditions of 10% H2, 10% CO2, and 80% N2 for up to 21 days. Helicobacter spp. were identified on the basis of gram staining, phase contrast microscopic, and biochemical test results. 37, 38
Polymerase chain Reaction (PCR) and restriction fragment length polymorphism (RFLP) analyses
To definitively confirm genus and species of the isolated Helicobacter spp., PCR and RFLP analyses were performed. Briefly, DNA was extracted from cecal tissue and/or cecal contents using a High Pure PCR Template Preparation Kit (Roche Diagnostics, Penzberg, Germany) and a QIAamp® DNA Mini Kit (Qiagen Inc., Valencia, CA), respectively. Helicobacter spp. primers C97 and C05 were used to generate 16s rRNA amplicons of 1.2-kb size using conditions described earlier. 29 The 1.2-kb PCR products were electrophoresed on a 1% agarose gel, and were visualized under ultra-violet (UV) illumination after ethidium bromide staining.
The Helicobacter species-specific, 1.2-kb PCR products were also subjected to RFLP analysis. Briefly, 27 µl of the PCR products was digested with 10 µl each of restriction endonucleases AluI and HhaI and 3 µl of restriction buffer (New England Biolabs, Beverly, MA) at 37°C overnight. The samples were then electrophoresed through a 6% Visigel separation matrix (Stratagene, Cedar Creek, TX), stained with ethidium bromide, and visualized under UV illumination.
To rule out Lawsonia intracellularis as a cause of proliferative enteritis, PCR-based analysis using L. intracellularis-specific primers was performed on cecal and/or ileal DNA samples as described. 7, 8 The DNA extracted directly from a culture of L. intracellularis was used as a positive control. The 182-bp PCR products were electrophoresed on a 6% Visigel matrix, stained with ethidium bromide, and visualized under UV illumination.
Immunohistochemical analysis
Immunohistochemical (IHC) analysis of formalin-fixed, paraffin-embedded sections from the ICC junction, including the intestinal neoplasms, was performed using an automated processor (BioGenex, San Ramon, CA). All slides were subjected to heat-based antigen retrieval using 10 mM sodium citrate buffer (pH 6) prior to incubation with the following primary antibodies: pancytokeratin rabbit polyclonal (DakoCytomation, Carpinteria, CA; diluted 1 : 800), vimentin mouse monoclonal (DakoCytomation, 1 : 50), CD3 rabbit polyclonal (DakoCytomation, 1 : 300), CD79a mouse monoclonal (DakoCytomation, 1 : 50), and F4/80 rat monoclonal antibodies (Caltag laboratories, Burlingame, CA, 1 : 150). Appropriate murine and hamster tissue sections (lymphocyte, macrophage and epithelial cells) served as external and internal positive controls, respectively. Biotinylated goat anti-rabbit secondary antibody (DakoCytomation) was used at a 1 : 1,000 dilution for pancytokeratin and CD3 antibody. For F4/80 and CD45-B220 antibodies, biotinylated goat anti-rat (Sigma-Aldrich, Inc., St. Louis, MO; 1 : 150) and biotinylated multiple absorbed goat anti-rat secondary (BD Biosciences Pharmingen, San Diego, CA; 1 : 100), respectively, were used. The ARK kit (DakoCytomation) was used for vimentin and CD79a, as per the manufacturer's instructions. For all antibodies, streptavidin-horseradish peroxidase (1 : 100) was used as the enzyme, and antigen-antibody binding was visualized using diaminobenzidine as the substrate. All slides were counterstained with hematoxylin.
Cytotoxicity assay
To rule out Clostridium difficile as a cause of proliferative typhlocolitis, we used a cytotoxicity assay kit (Kit B-1029-70, Bartels, Carlsbad, CA) to detect the presence of Cl. difficile toxin B, as per the manufacturer's instructions. 46 Briefly, cecal contents or cecal tissue were diluted 1 : 1 in sterile phosphate-buffered saline and filtered through a 0.2-µm filter (Pall Corporation, Ann Arbor, MI). Samples were incubated with HeLa cells and toxin in a microtitration dish at 37°C overnight. The maintenance or lack of the typical actinomorphic morphology of the HeLa cells indicated the absence or presence of toxin in the sample, respectively. Additionally, toxin-specific antibody was used as a control to verify the efficacy of the assay.
Results
Gross and histopathologic findings
Hamsters of group A were clinically normal, and gross lesions were not observed. Similarly, hamsters of group B did not manifest clinical signs of disease; however, 2 of 12 had mildly to moderately thickened ICC junction. In group-C hamsters, 12 of 12 were in fair or poor body condition, with history of chronic weight loss. Infiltrative gastric adenocarcinoma, most likely associated with chronic helicobacter-associated gastritis, was diagnosed in 1 of the 12 hamsters from group C. 34 Six of 12 hamsters had grossly thick ICC junctions with a few serosal cysts (Fig. 1A). The cecal mucosa of these hamsters had a cobblestone appearance, and the lamina propria of the ICC junctions was expanded by variable numbers of lymphocytes, plasma cells, macrophages, heterophils, eosinophils, and cystic crypts (Fig. 1B). These cellular infiltrates frequently caused separation, haphazard arrangement, and submucosal crypt herniation. A few of the inflammatory infiltrates extended into the submucosa and muscular layer (Fig. 1B, D). Animals of group C with high-grade inflammation often had multifocal crypt abscesses and heterophilic infiltrates (Fig. 1C). Presence of an effacing round cell tumor in the ICC junction of one hamster precluded evaluation of inflammation or crypt dysplasia, and was, therefore, dropped from the data set for inflammation and dysplasia. Dysplastic lesions were noted in the ICC junction of 5 of 11 hamsters in group C (Fig. 1E, G). These dysplastic crypts ranged from mild-to-moderate severity, and had histologic appearance similar to dysplasia described in humans with chronic IBD. Crypts considered dysplastic were not surrounded by active inflammatory incidents such as crypt abscesses and/or overlying, abutting epithelial ulcers (Fig. 1E, G). However, in Fig. 1F, although atypia noted in colonocytes warrants a diagnosis of dysplasia, their association with heterophils and focal erosion precluded such diagnosis.
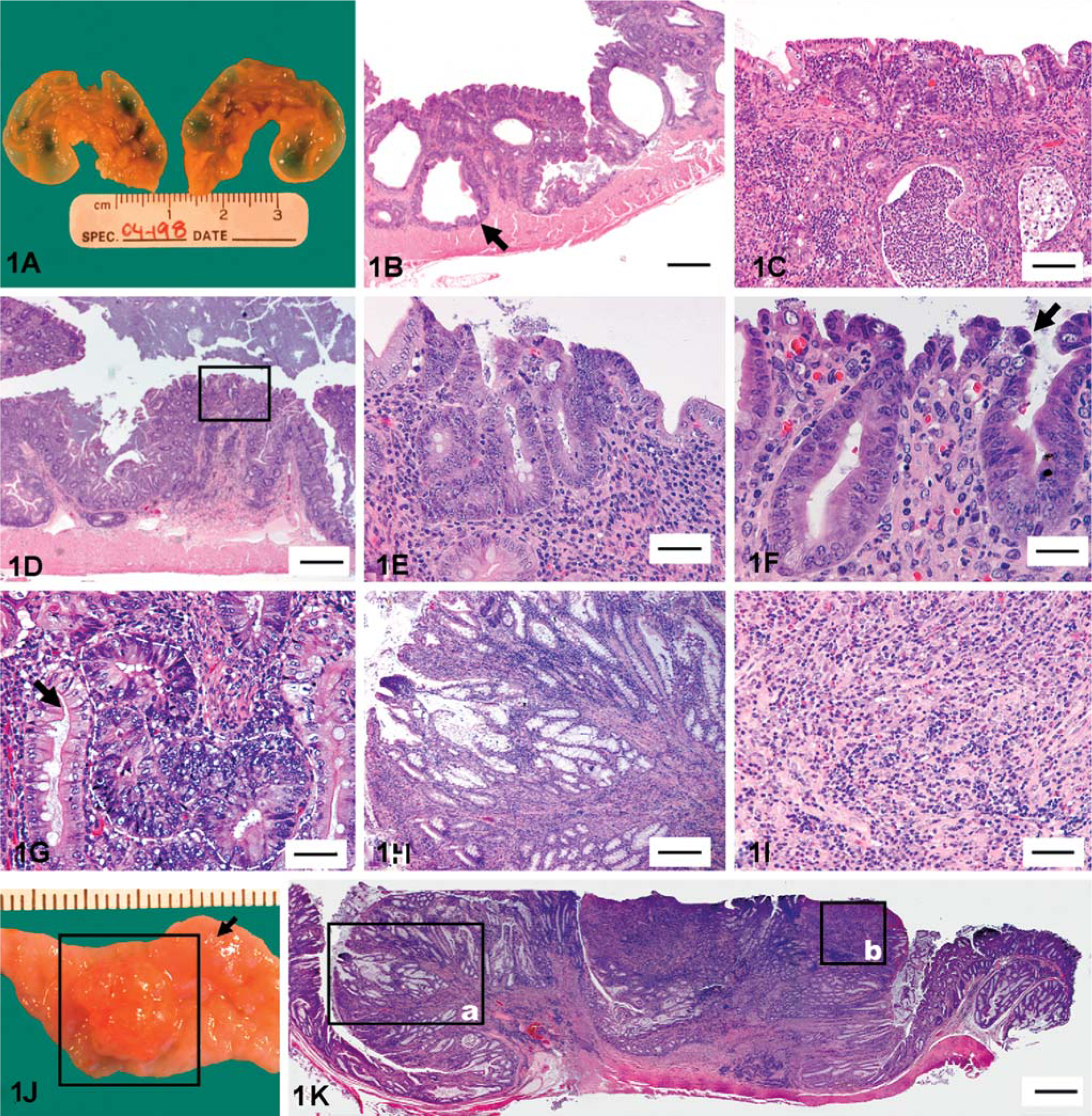
Ileocecocolic (ICC) junction; gross and histomorphologic appearance of the ICC junction of 2-year-old Syrian hamsters naturally infected with Helicobacter spp.
One hamster had a focal, 9-mm-diameter, red, polypoid mass within the cecum (Fig. 1J). This polypoid nodule was composed of well differentiated, tall, tortuous crypts with marked goblet cell hyperplasia that was especially prominent along the peripheral zones of the mass (Fig. 1H, K). At the central and superficial zones, these hyperplastic crypts were frequently intersected by moderate-to-marked fibrovascular stroma admixed with large numbers of macrophages, lymphocytes, plasma cells, heterophils, and eosinophils (Fig. 1H). Furthermore, along the superficial and central zones, there was complete replacement of the crypts by connective tissue stroma that was infiltrated with dense inflammatory cell infiltrates (Fig. 1I). The inflammatory cells infiltrated into the underlying prolapsed submucosa and multifocally into the serosa. In the abdominal cavity of two hamsters from group C, there were multiple nodular masses associated with the mesentery and intestinal serosa. The first neoplasm, diagnosed as metastatic round cell sarcoma, originated at the ICC junction, and the second neoplasm was a malignant fibrous histiocytoma associated with the intestinal serosa and mesentery (Fig. 2).
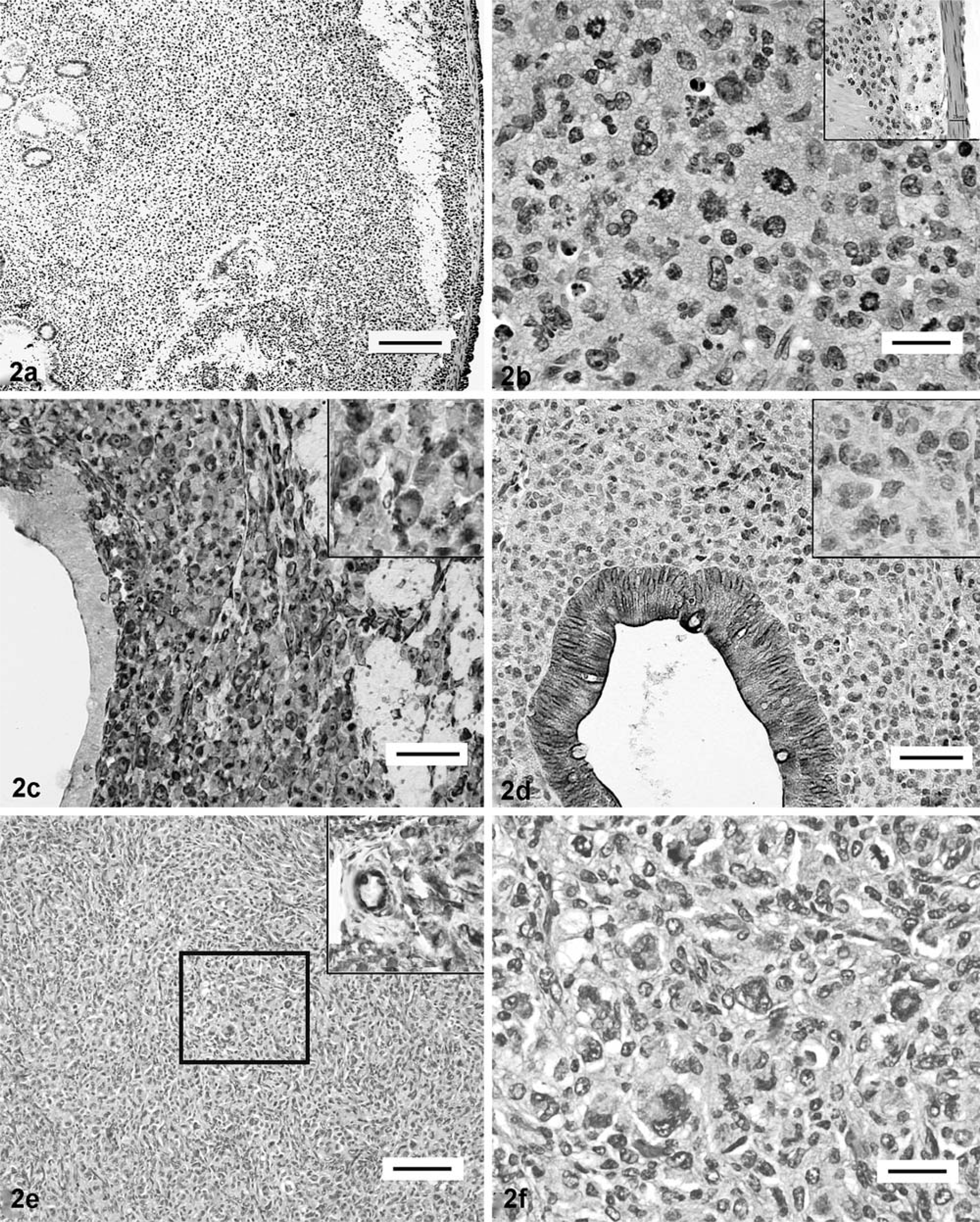
Intestines; histomorphologic and immunohistochemical (IHC) analyses of the nonepithelial neoplasms in aging Syrian hamsters naturally infected with Helicobacter spp.
In the hamster with the intra-abdominal metastatic round cell tumor, there was diffuse, severe expansion of the lamina propria in the ICC junction and separation of the crypts by large numbers of mononuclear cells (15–20 µm) arranged in sheets with minimal fibrovascular stroma (Fig. 2A, B). This infiltrative cell population was transmural, with multifocal involvement of all the layers of the intestinal wall including the serosa (Fig. 2A). The neoplastic cells had marked anisocytosis and faintly discernible cell borders; pale, amphophilic, finely vacuolated cytoplasm; and central-to-eccentric, moderately anisokaryotic, and oval-to-reniform, vesiculated, hyperchromatic nuclei. Nucleoli were singular and basophilic. Karyomegaly, with 4–6 mitotic figures/high-power field that were occasionally bizarre, was evident (Fig. 2B). A few monomorphic populations of neoplastic cells were found within a few lymphatics in the tunica muscularis (Fig. 2B) and local lymph nodes, infiltrating the mesenteric adipose tissue and attached to the splenic capsule. The neoplastic cells were intensely positive for vimentin (Fig. 2C), but were negative for pancytokeratin (Fig. 2D), B-cell (CD 79a), T-cell (CD3), and macrophage (F4/80) markers on the basis of results of IHC analysis (data not shown). Appropriate subsets of cells (lymphocytes, macrophages and epithelial cells) in the ICC junction stained positive to their respective antibodies (internal positive control).
The mesenchymal tumor in the second hamster was histologically consistent with a malignant fibrous histiocytoma. This neoplasm was composed of a pleomorphic population of spindle-shaped, and to a lesser extent, histiocyte-like cells. Neoplastic cells were arranged in whorls, storiform pattern, and a few sheets, and were principally restricted to the serosa of multiple intestinal loops and the splenic capsule, with metastasis to the mesenteric lymph nodes (Fig. 2E, F). There were a few multinucleate giant cells with karyomegaly and bizarre nuclei throughout the mass (Fig. 2F). The tumor cells stained intensely positive for vimentin (Fig. 2E [inset]), but negative for pancytokeratin, B-cell (CD 79a), T-cell (CD3), and macrophage (F4/80) markers on the basis of results of IHC analysis (data not shown).
The scores for all criteria including inflammation (P < 0.01), hyperplasia (P < 0.01), and dysplasia (P < 0.05) were significantly higher in group C, relative to group A (Fig. 3). Although there was significantly greater inflammation in group-B than group-A hamsters, median scores for hyperplasia and dysplasia were low and not significantly different (P > 0.05). The overall dysplasia scores in group-C hamsters were significantly different (P < 0.05) from those for hamsters of groups A and B (Fig. 3C). Thus, although the degree of inflammation increased with age and plateaued around 1 year of age, the degree of dysplasia increased concurrently with age.
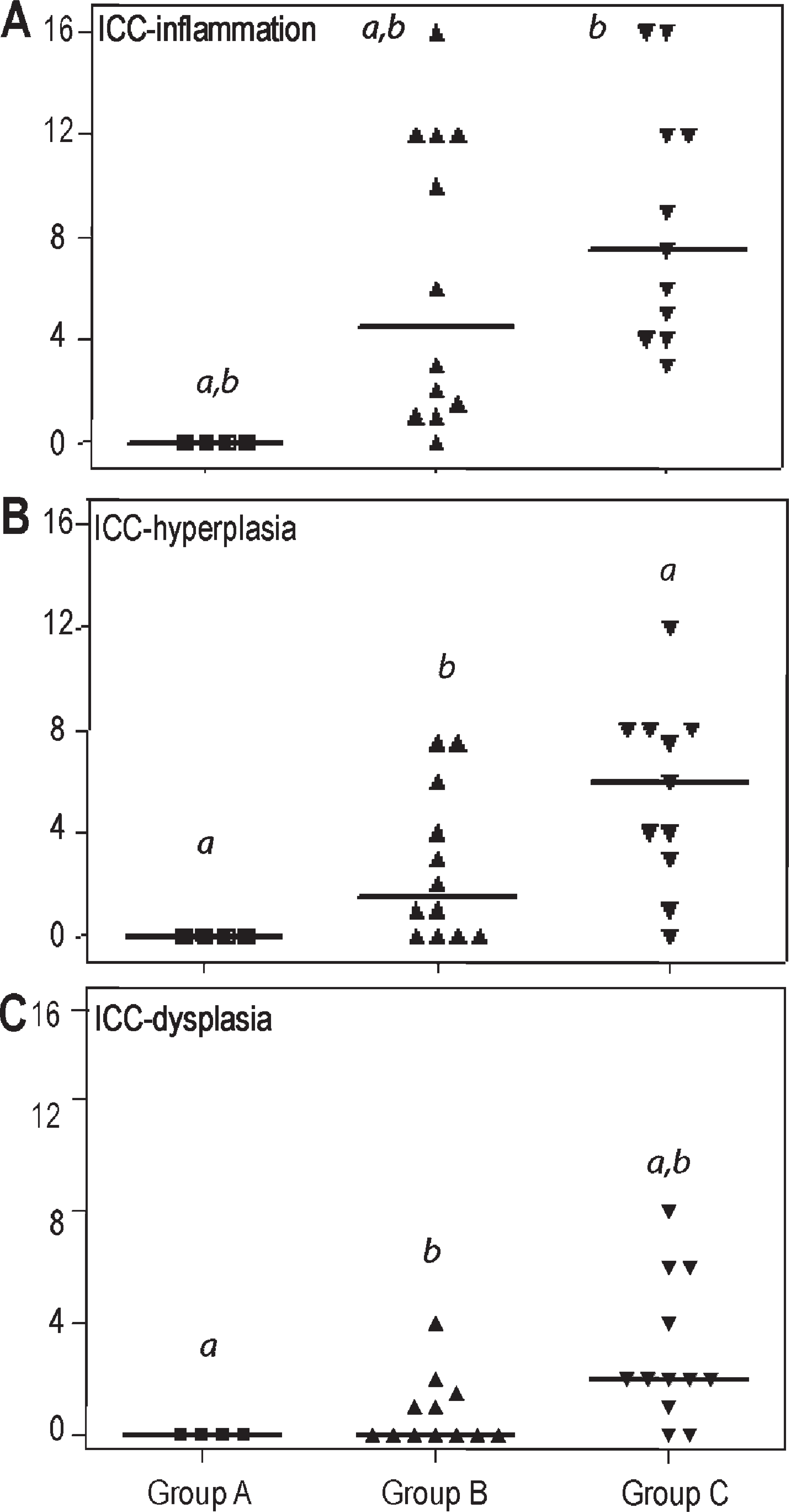
Comparison of inflammation (
Helicobacter spp. isolation and characterization
Similar to our previously published results, Helicobacter aurati and two unnamed Helicobacter species were isolated in culture from the cecum of helicobacter-positive animals. 38 Furthermore, animals of all three age groups were positive by means of PCR analysis using Helicobacter genus-specific primers (Fig. 4). The presence of H. aurati (data not shown) and two unnamed Helicobacter spp. (Fig. 5) was confirmed in all positive samples by results of RFLP analysis of the PCR products. The banding patterns noted in lanes 1, 3, and 5 (by use of restriction enzyme AluI), and lanes 2, 4, and 6 (by use of restriction enzyme HhaI), were consistent with those for the previously described unnamed Helicobacter spp. The unnamed Helicobacter sp. was reported to be phylogenetically in the “Flexispira rappini” taxon (unpublished results) similar to the helicobacter isolates from cotton-top tamarins. 47
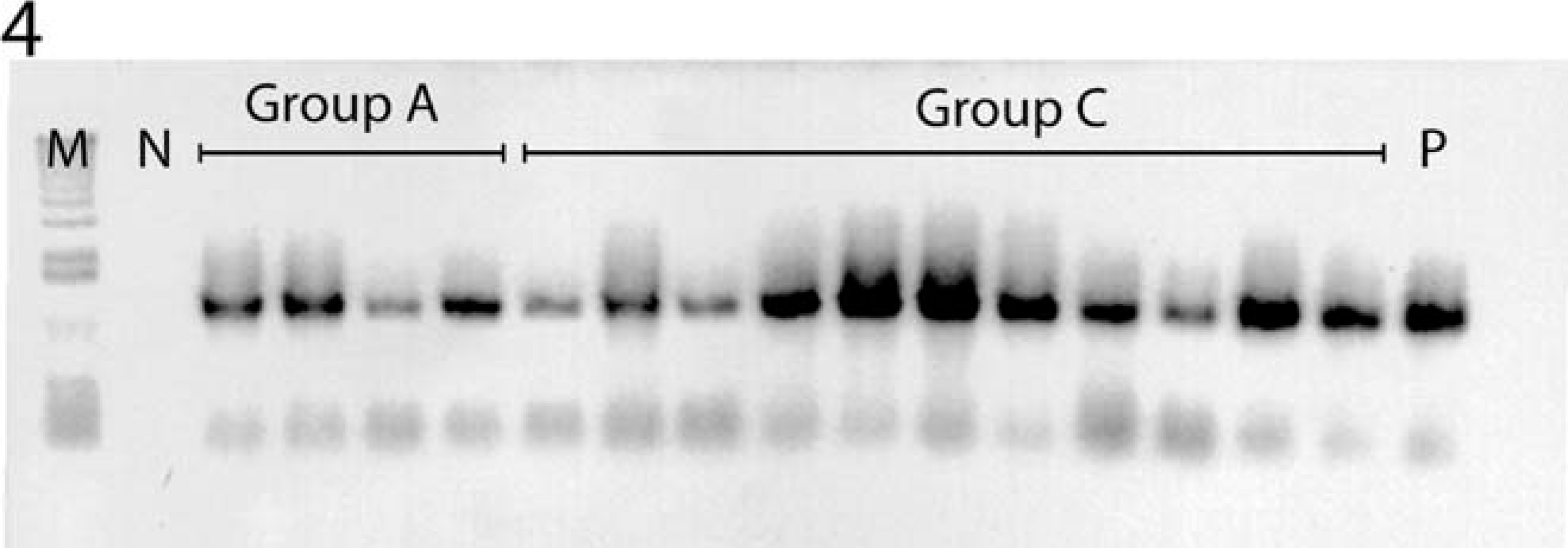
Polymerase chain reaction (PCR)-based detection of Helicobacter spp. in ileocecocolic (ICC) junctions of 1-month-old (group A) and 18- to 24-month-old (groups C) Syrian hamsters. M = 1-kb+ molecular ladder; N = PCR negative control; P = positive control (DNA extracted from Helicobacter spp. culture). The 1.2-kb PCR products were detected in all test samples.
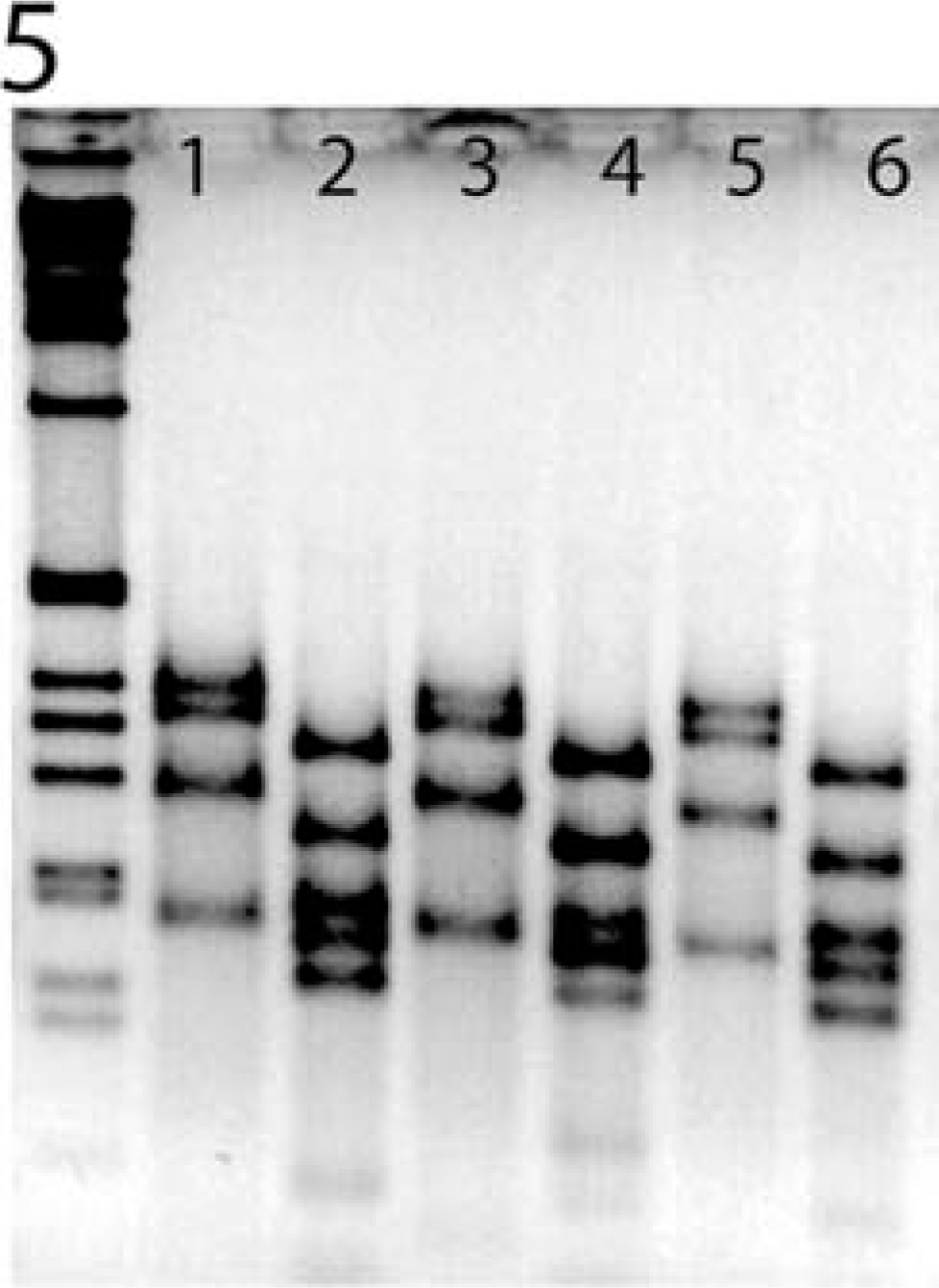
Representative restriction fragment length polymorphism (RFLP) analysis of the 1.2-kb PCR products generated by using genus-specific helicobacter primers. The observed banding patterns are consistent with the novel unnamed Helicobacter sp. reported previously.37,38 Lanes 1-6 represent samples from three hamsters of group C (18-24 months old). Lanes 1, 3, 5, and 2, 4, 6 represent PCR products that were cut with AluI and HhaI restriction enzymes, respectively.
Differential diagnosis
Clostridium difficile, L. intracellularis and Giardia spp. are common etiologic agents that have been reported to induce proliferative enteritis in hamsters. Therefore, all animals included in this study were screened for these agents. All animals were negative for Cl. difficile toxin B by results of a cytotoxicity assay and for L. intracellularis by results of PCR analysis. Furthermore, none of the animals had histologic evidence of proliferative ileitis consistent with Lawsonia sp. infection. Fecal specimens were negative for Giardia spp. by results of examination of direct smears of the material from all animals. In addition, there was no evidence of any protozoal organisms in the examined ICC junction sections.
Discussion
The use of animal models has greatly enhanced our knowledge of IBD pathogenesis, especially in dissecting the various components that contribute to the disease. Although the exact cause of IBD is unknown, multiple factors, including genetic background, microbial flora, mucosal integrity, oral tolerance, and diet, have been implicated in the pathogenesis of IBD. 13 The most commonly used models of IBD are genetically manipulated and/or immunodeficient rodents. 51 Similarly, most of the murine models describing helicobacter-associated typhlocolitis and distal large bowel cancers were conducted using genetically manipulated and/or immunodeficient mouse strains. 4, 30, 48, 53, 56 Although experimentally induced models allow evaluation of one variable at a time, spontaneous models permit the study of complex interactions between the host and its microenvironment. Because of the paucity of spontaneous animal models of human IBD, research highlighting IBD-like disease in unmanipulated laboratory animals is highly desirable. 2, 4 In the study reported here, we describe progressive typhlocolitis and associated dysplasia in aging hamsters naturally infected with Helicobacter spp.
In this study, hamsters from all three age groups (groups A–C) were positive for Helicobacter spp. This was consistent with our previous survey of hamsters from multiple vendors and research facilities, wherein Helicobacter spp. were detected in 100% of the animals by use of either PCR-based techniques or microbial isolation. 38 As evidenced by the positive culture results from the 1-month-old hamsters, Helicobacter infection occurred early in life. Hamsters are coprophagic and, therefore, young hamsters are infected by ingesting feces originating from the adults (i.e., fecal-oral route of transmission). This is a common route of helicobacter transmission in mice. 15, 16, 31, 44, 55 The 1-month-old hamsters did not have large bowel inflammation, and the inflammation scores were significantly higher in the older animals (groups B and C). Although the inflammation scores in group-C hamsters were higher than those in group-B hamsters, this difference was not statistically significant. Thus, although the degree of inflammation increased with age, it appears to plateau after approximately one year of age.
In humans with chronic IBD, the risk of developing precancerous and cancerous lesions increases markedly with duration of the illness. Typically, patients with chronic IBD (diagnosed in their second or third decade of life) develop dysplasia and cancer in their fourth or fifth decade of life. 21 Similarly, in the geriatric hamsters with chronic typhlocolitis, dysplasia scores were significantly higher. In fact, 45% of the hamsters in group C developed dysplastic lesions, and 100% of the animals had various degrees of typhlocolitis. However, unlike that in humans, it must be stressed that the prognostic value and significance of dysplasia in hamsters is uncertain, as they did not develop cancer. Additionally, in humans, distinguishing true dysplasia from severe atypia associated with active colitis or regenerative hyperplasia often is difficult. Because significance of dysplastic lesions in hamsters is uncertain, necessity of discriminating dysplasia from mucosal changes associated with acute inflammation or regenerative hyperplasia appears limited at this time. However, to be more conservative, we only categorized lesions as dysplasia when atypical mucosa was independent of active colitis or erosions/ulcers.
In an earlier report, although the occurrence of sporadic intestinal adenocarcinoma in aging hamsters (2–2.5 years) was reported to be rare (<1%), 12, 22 approximately 39–83% of the hamsters had concurrent chronic typhlocolitis or “atypical proliferative enteritis” of unknown etiology. 12, 57 This high incidence of inflammatory changes was reported to frequently complicate diagnosis and interpretation of intestinal neoplasia. 12 Chronic proliferative colitis, cystadenomatous hyperplasia, and spontaneous intestinal adenocarcinoma in the cecum and colon of hamsters also were described. 42, 43, 57 Importantly, the photomicrographs of the “well-differentiated intestinal adenocarcinoma” were suggestive of inflammation-associated crypt herniation and dysplasia, rather than true neoplasia. 43 It is important to note that the infection status (L. intracellularis or Helicobacter spp.) of the hamsters in those studies was not reported. 42, 43, 57 Therefore, it is possible that spontaneous inflammation and neoplasia that have been historically reported in geriatric hamsters similar to those of our study, may have been associated with helicobacter-induced chronic inflammation.
To rule out other pathogens in hamsters that could mimic lesions described in our study, we screened animals in all three groups for the presence of Cl. difficile, L. intracellularis, and Giardia spp. 8, 25, 46 Clostridium difficile overgrowth subsequent to antibiotic therapy (antibiotic-associated colitis), typically causes a necrotizing, fatal typhlocolitis. Additionally, in adult hamsters without exposure to antibiotics, Cl. difficile infection has been linked to proliferative typhlocolitis. 5, 46 Animals of our study were not treated with antibiotics, were negative for Cl. difficile toxin, and did not develop lesions consistent with Cl. difficile infection. Cecal mucosal hyperplasia in suckling and weanling hamsters has been suggested to represent the recovery phase of clostridial enteropathy. 39 However, the helicobacter status of the hamsters in these studies was not reported. Lawsonia intracellularis-induced lesions (proliferative enteritis with histiocytic lamina propria and submucosal infiltrates) are common in younger animals (<10 weeks old) and are frequently restricted to the distal portion of the ileum. 40 The age of the affected hamsters and distribution of the lesions described in our study are not consistent with L. intracellularis-induced diseases. Giardia spp. also have been implicated in causing enterotyphlocolitis in aging hamsters. 41 Examination of wet mount preparations and histologic evaluation of the small and large intestines of the 2-year-old hamsters in our study failed to detect any protozoal organisms. Thus, our results from PCR, toxin, and fecal analyses indicate that the hamsters of this study developed IBD-like diseases independent of these confounding pathogens.
Hamsters, known to be reservoirs of Campylobacter jejuni, typically do not develop disease. 17 It is unlikely that C. jejuni plays a role in IBD-like disease in hamsters because only two of seven hamsters in group C were positive for C. jejuni by use of culture (data not shown). There was no correlation between infection and inflammation, and inflammation-associated dysplasia (data not shown). In our previous report, there was no correlation between colonization of the stomach by C. jejuni and inflammation. 37 Additionally, results of studies of different strains of immunocompetent mice infected with various C. jejuni strains have indicated intestinal colonization, and in some instances, bacteremia; however, there has been a lack of consistent development of gastroenteritis. 58
Conclusive evidence implicating Helicobacter spp. in proliferative typhlocolitis and associated dysplasia can only be documented by means of experimentally induced infection in helicobacter-free hamsters. Because of the enzootic distribution of Helicobacter spp. in hamster populations and the difficulty in establishing helicobacter-free hamster colonies, fulfillment of Koch's postulates remains problematic. The ability to generate helicobacter-free hamster colonies is limited due to unsuccessful embryo rederivation, and because antibiotic-based helicobacter eradication programs are associated with fatal Cl. difficile colitis. Nevertheless, the role of helicobacter in causing typhlocolitis and colitis-associated cancer has been unequivocally documented by results of infection studies in helicobacter-free, genetically engineered or immunodeficient rodent species. 11, 56 Recently, the role of Helicobacter spp. in IBD, which was mostly restricted to animal models, has been extended to some cases of ulcerative colitis in humans and cotton-top tamarins. 47, 50 Additionally, several characteristics of human IBD pathophysiology strongly suggest a microbial component. 20 From those studies, we can make an association, albeit not definitive, between helicobacter colonization and IBD-like disease in hamsters as well. 4, 28
The findings of round cell sarcoma and malignant fibrous histiocytoma in a setting of chronic inflammation in one hamster each are intriguing. The tumor cells in both cases were positive for vimentin and were negative for B-cell, T-cell, and histiocyte markers. Although, the round cell sarcoma has a histiocytic morphology, we could not definitively establish the cell of origin. The failure of immunophenotypic analyses to establish definitively the cell of origin led to a generic diagnosis of a round cell sarcoma. Histiocytic sarcoma and lymphoma topped our differential diagnosis. The malignant fibrous histiocytoma had histologic and immunophenotypic features similar to a previously described lesion in the subcutis of a 2-year-old Djungarian hamster. 9 It is possible that these two tumors were incidental findings associated with aging, and were not associated with chronic inflammation. However, the intra-abdominal site and predominant localization of the round cell sarcoma to the ICC junction suggests that the tumors may have emerged within an area of chronic inflammation. Other round cell tumors such as gastric lymphomas (MALToma) have been associated with H. pylori in humans, H. mustelae in ferrets, and H. felis in mice. 28 A similar link between immunoproliferative disease of the small intestine and enteric microflora (i.e., C. jejuni infection) in humans has been recently established. 27
Thus, we have described a spontaneous model of IBD with several features of human IBD. Unfortunately, none of the hamsters of our study developed cancer with metastasis. This lack of tumor development can be compensated for, as hamsters are susceptible to carcinogen-induced gastrointestinal track tumors. The model described here allows a system to test the interaction effects of chronic inflammation with carcinogen(s) treatment in the context of the large bowel in hamsters. 23, 32 Indeed, synergistic effect on gastrointestinal track tumorigenesis between the carcinogen 3,2′-dimethyl-4-aminobiphenyl and atypical proliferative enteritis has been reported. 57 Alkylating carcinogens, such as azoxymethane, have been reported to selectively induce colonic tumors in susceptible mouse strains. 19, 33, 35 Use of such mutagenic colonotropic carcinogens may decrease the latency period of IBD-like disease in this model and even induce colitis-associated cancer and metastasis—features that were not identified in this study, even after two years. In conclusion, the model of IBD described herein will allow the study of multiple aspects including micro-environmental factors in IBD and potentially IBD-associated cancer.
Footnotes
Acknowledgements
This work was supported in part by RR07036 (JGF), R01A137750 (JGF), and P30 ES02109 (JGF). We thank Dr. Connie Gebhart at the University of Minnesota, for providing Lawsonia intracellularis DNA. We also want to acknowledge the technical support received from Jeff Bajko, Erinn Stefanich, Elaine Robbins, and Ellen Buckley. We want to thank Drs. Arlin Rogers, Courtnye Jackson, and Laura Lemke for their assistance with this manuscript.
Authors PRN and SMK contributed equally to this manuscript.
