Abstract
The histomorphologic characteristics and chemical composition of the crystals associated with suspected pet food-induced nephrotoxicosis in 3 dogs are described. Kidney specimens from 2 dogs, a 3-year-old Parson Russell Terrier and a 3-year-old Bernese Mountain Dog, were examined. Both developed acute renal failure after eating canned pet food on the 2007 Menu Foods recall list. The third case was a kidney specimen from a 1-year-old mixed-breed dog from a similar 2004 outbreak of canine renal failure in Taiwan, which occurred after eating a commercial dog food. Hematoxylin and eosin (HE), 72-hour Oil Red O (ORO72h), Alizarin Red S (pH 4.1–4.3), and Von Kossa stains; infrared (IR) spectroscopy; and scanning electron microscopy with energy dispersive X-ray analysis (SEM/EDXA) were performed to determine the histomorphologic characteristics and chemical composition of the crystals observed in each case. Histomorphologic findings in each case included acute, marked tubular degeneration and necrosis with many intratubular birefringent crystals, and lymphoplasmacytic interstitial nephritis. In each case, most of the crystals were rough, pale brown, and stained with ORO72h but did not stain with Alizarin Red S (pH 4.1–-4.3) or Von Kossa stains; these features were consistent with a plastic or lipid. IR spectroscopy and SEM/EDXA results were consistent with melamine-containing crystals. A second crystal type identified in each case was smooth and platelike with staining characteristics and IR spectroscopy and SEM/EDXA results consistent with calcium oxalate crystals. Melamine-containing crystals have distinct light microscopic, histochemical, and SEM/ EDXA characteristics that facilitate their identification in tissue.
Keywords
Introduction
On March 16, 2007, Menu Foods Inc., a Canadian manufacturer of wet pet food, issued a recall of more than 60 million containers of cuts and gravy style pet food because of concerns about the effects of the product on the renal health of pets. 2, 4 The recalled food was manufactured between December 3, 2006, and March 6, 2007. 4 The recall was initiated because of consumer complaints that began on February 20, 2007, and the deaths of animals in routine palatability studies. 4 Food-related acute renal failure was diagnosed in dogs and cats in veterinary clinics across the country. Aminopterin, an antagonist of folic acid, was initially suspected to be the toxin contaminating the recalled pet food; however, this finding was not confirmed by the US Food and Drug Administration (FDA), and testing for other contaminants continued. 3, 4 Melamine was later found in the recalled products. The FDA confirmed the finding and detected melamine in wheat gluten originating from China that was subsequently added to the food. 4 This led to the expansion of the recall to include dry diets, treats, and more wet products that could contain the contaminated wheat gluten. 3 Melamine was subsequently found in rice protein concentrate as well. 15 The FDA later determined that the contaminated wheat gluten and rice protein were mislabeled. Both products were actually wheat flour contaminated with melamine and melamine-related products. 2, 17 It is believed that melamine, which has a high nitrogen content, was added to the wheat flour to increase the apparent protein content. 1– 3 The FDA later announced that cyanuric acid as well as melamine had adulterated ingredients from China that went into the pet food. 2, 16
The Cornell University Animal Health Diagnostic Center subsequently identified melamine in pet food, in urine from cats that ate the food, and in the kidney of 1 cat from the palatability studies. 4 The University of Guelph Laboratory Services Division formed melamine–cyanuric acid complex crystals in cat urine in vitro by adding melamine and cyanuric acid (http://www.labservices.uoguelph.ca/urgent.cfm crystals). Further analysis of the crystals revealed that they were composed of approximately 70% cyanuric acid and 30% melamine and were extremely insoluble. 16 Ammelide and ammeline—2 other melamine related substances—may also play a role in pet food–associated nephrotoxicity and are currently under investigation. 16
The March 2007 pet food–associated renal failure outbreak in North America bears striking similarities to a March 2004 outbreak of pet food–associated renal failure that occurred in Asia. 1, 8, 9 In the 2004 Asian outbreak, over 6,000 dogs and fewer cats reportedly developed nephrotoxic renal failure after ingesting a certain brand of commercial pet food. 1 Green–yellow nephroliths were detected in the renal pelvis of some of the dogs involved in the outbreak. 9 Additionally, necrosis, calcification, and calculi were detected in the renal tubules. 9 The crystals present in the kidneys of animals involved in the 2004 outbreak in Asia were green–yellow and round with many lines radiating from their centers and histomorphologically identical to the crystals observed in the kidneys of animals involved in the 2007 pet food–associated outbreaks of renal failure. 1, 9 The 2004 outbreak of pet food–associated renal failure was attributed to contamination of raw materials with mycotoxins (especially ochratoxin and/or citrinin) in a pet food manufacturing plant in Thailand. 1, 8, 9
Melamine is used in the production of plastics worldwide and as a fertilizer in Asia. 2, 3 In mice and rats, melamine causes urolithiasis. Chronic irritation of the urothelium by the calculi induces urothelial hyperplasia and subsequent urinary bladder neoplasia. 7, 13, 18, 19, 21 Melamine has been reported to cause crystalluria in mice, rats, and dogs; diuresis in rats and dogs; and nephrosis, fatal uremia caused by crystalluria, and hemorrhagic cystitis in sheep. 5, 11– 13, 18 There is very little literature available on the effects of melamine in companion animals other than causing crystalluria and having a diuretic effect in dogs. 12, 13, 18
Materials and Methods
Description of cases
Renal tissue from 3 dogs was submitted to the Armed Forces Institute of Pathology, Washington, DC. Two of the cases were submitted by Idexx Veterinary Services, West Sacramento, California, for light microscopic examination and characterization of the chemical composition of crystals observed previously in hematoxylin and eosin (HE)–stained sections. The third case had been submitted from the Animal Technology Institute Taiwan, Division of Animal Medicine, Chunan, Miaoli, Taiwan, as a Wednesday Slide Conference case submission for the 2004–2005 academic year and was retrieved retrospectively. Case No. 1 was a 3-year-old, female, Parson Russell Terrier that developed acute renal failure after eating canned food on the Menu Foods recall list. There was no known prior history of renal disease or ethylene glycol exposure. Significant clinical pathologic findings included azotemia and hyperphosphatemia consistent with acute renal failure. Additionally, leukocytosis and neutrophilia were present. Liver enzyme values were within normal limits. The patient was nonresponsive to treatment and died. Significant necropsy findings included a perforated gastric ulcer, severe bruising and bleeding of the gastric wall, and green/yellow fluid with green crystals within the renal pelvis. Case No. 2 was a 3-year-old, spayed female, Bernese Mountain Dog that also developed acute renal failure after eating canned food on the Menu Foods recall list. There was no known prior history of renal disease or ethylene glycol exposure. Significant clinical pathologic findings included azotemia, hyperphosphatemia, hyperkalemia, decreased TCO2 (bicarbonate), and increased anion gap consistent with acute renal failure and concurrent metabolic acidosis. Liver enzyme values were within normal limits. The patient was nonresponsive to treatment and was euthanized. Only the kidneys were collected at necropsy. Case No. 3 was a 1-year-old, male, mixed-breed dog that developed renal failure after eating a commercial dog food for several months. Significant gross findings included prominent white powderlike deposits on the kidney. Significant clinical pathologic findings included leukocytosis, neutrophilia, and marked azotemia consistent with renal failure.
Histology
Sections of kidney were fixed in 10% neutral-buffered formalin, routinely processed, embedded in paraffin, and sectioned at approximately 5 μm. HE; 72 hour Oil Red O (ORO72h), a stain for lipids and plastics; Alizarin Red S (pH 4.1–4.3), a stain for divalent cations; and Von Kossa, a stain for phosphates, carbonates and oxalates of calcium, iron, and other ions were performed by routine methods. 22
Crystal analysis
Tissue sections were cut from paraffin-embedded formalin-fixed kidney from both dogs involved in the 2007 outbreak of pet food–associated renal failure (dog Nos. 1 and 2) and mounted on aluminum-coated glass slides for infrared (IR) spectral analysis or on carbon disks for scanning electron microscopy with energy dispersive X-ray analysis (SEM/EDXA). An unstained section of kidney on a glass slide from the dog involved in the 2004 outbreak of pet food–associated renal failure (dog No. 3) was transferred to an aluminum-coated glass slide for IR spectroscopy and another unstained section of kidney on a glass slide from dog No. 3 was examined by SEM/EDXA.
Infrared spectra were obtained by using a Nicolet IRuS microspectrometer or a Nicolet Continuum infrared microscope attached to a Nicolet model 860 Fourier transform infrared (FT-IR) spectrometer (ThermoNicolet, Madison, WI). A reflective Cassegrainian objective (32×) was used to focus the infrared light onto tissue sections, which were mounted on aluminum-coated glass slides. The infrared spectrometers were operated in the “reflection” mode wherein the light passed through the sample, reflected off the aluminum surface, and passed through the sample a second time before being directed to a liquid-nitrogen-cooled mercury-cadmium-telluride (MCT) detector. Spectra of the specimens were ratioed to background spectra obtained from blank regions on the aluminum-coated slide, and apertures (between 20 and 50 μm) were used to direct the infrared beam to the areas of interest. Spectra were recorded at 4-cm−1 spectral resolution, and 128 scans were co-added to reduce noise levels and provide adequate spectral quality. The spectrometers were purged with dry air to minimize the amounts of atmospheric water vapor and carbon dioxide in the instruments.
To replicate the crystals formed by the University of Guelph, which were created from a combination of cyanuric acid and melamine in cat urine, melamine, cyanuric acid, and urea (all Sigma Chemical Company, St. Louis, MO) were mixed at room temperature in vitro in varying proportions. In addition, 10% neutral buffered formalin was added to combinations of cyanuric acid and melamine in vitro to observe its effect on crystal formation.
A Hitachi model S-3500N scanning electron microscope (Hitachi Instruments, Inc., San Jose, CA) equipped with a ThermoNoran (ThermoNoran, Madison, WI) SEM-EDXA accessory was used to determine the elemental composition of the materials in the tissue. The electron beam was produced with a 15-kV accelerating voltage and was directed at the specimens mounted on carbon disks for dog Nos. 1 and 2 and a section of kidney mounted on a glass slide for dog No. 3. Characteristic X-rays produced by the elements in the specimen were directed to the liquid-nitrogen-cooled silicon detector.
Results
Histomorphology and histochemistry
In dog No. 1, approximately 90% of the renal tubules were either pale, swollen and vacuolated (degenerate), or were occasionally hypereosinophilic and shrunken with nuclear pyknosis or karyolysis (necrosis) on the HE-stained slide (Fig. 1). The basement membranes of tubules in the inner cortex were thickened and basophilic. There were numerous crystals evenly distributed throughout the cortex and medulla within renal tubules and collecting ducts (Fig. 2). The crystals demonstrated bright birefringence when viewed under polarized light (Fig. 3). Approximately 75% of the crystals were round, pale brown, and appeared to have a rough surface as a result of smaller crystalline structures being arranged radially and more randomly within the entire birefringent crystal (Fig. 2). Some of these crystal structures were arranged in concentric circles. Occasionally, the centers of these concentric circles were empty (Fig. 4). The crystals were present within renal tubular epithelial cells and in the lumens of tubules, where they filled and in some areas distended the tubules. The crystals measured up to approximately 80 μm in diameter. Birefringent crystals were also present in the renal calyces. A second type of crystal comprising approximately 25% of the birefringent crystals was also present within tubule lumens. These crystals often had a blue tinge because of a prismatic effect (Fig. 3). They had various sizes and shapes, and a smoother appearing surface (Fig. 2). Less commonly, basophilic nonbirefringent particles were also noted. Some of these appeared to be in the walls of blood vessels, in addition to the lumens of renal tubules. Low numbers of lymphocytes and plasma cells multifocally infiltrated the renal interstitium. Similar lesions and crystals were observed on HE-stained sections in dog Nos. 2 and 3; however, only approximately 60% of the renal tubules were affected in dog No. 2. Tubular basement membrane mineralization and basophilic nonbirefringent particles were also present in dog No. 3 but were absent in dog No. 2. Similar to dog No. 1, the crystals in dog Nos. 2 and 3 were almost exclusively within renal tubules. In dog No. 2, occasional crystals within the renal interstitium elicited a granulomatous inflammatory response with multinucleate giant cells (Fig. 5). In dog Nos. 2 and 3, the rough pale brown crystals measured up to approximately 100 μm in diameter. Multifocally, there were aggregates of many lymphocytes, plasma cells, and fewer neutrophils within the interstitium of dog No. 2. Prominent interstitial fibrosis was present in dog No. 3.
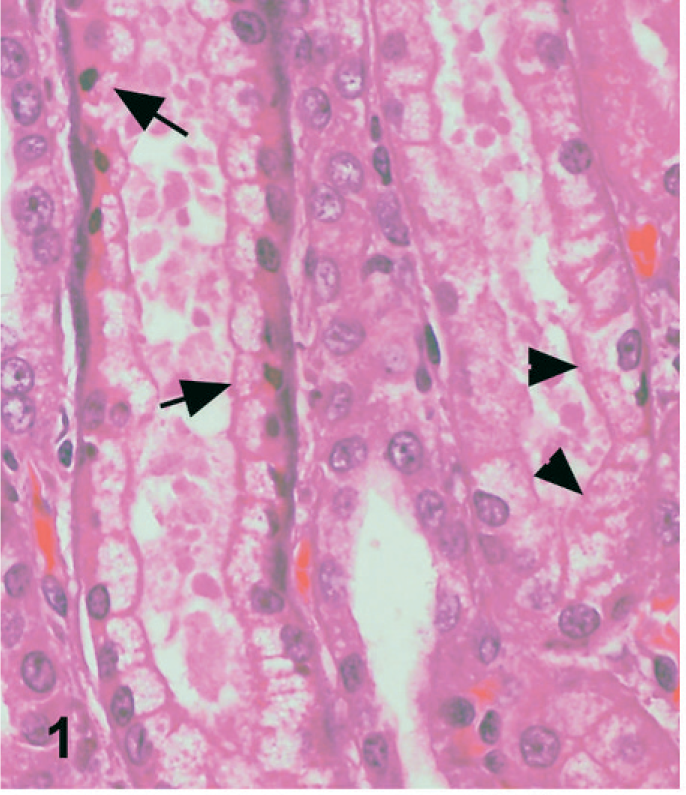
Kidney; dog No. 1. Tubular degeneration (arrowheads) and necrosis (arrow). HE.
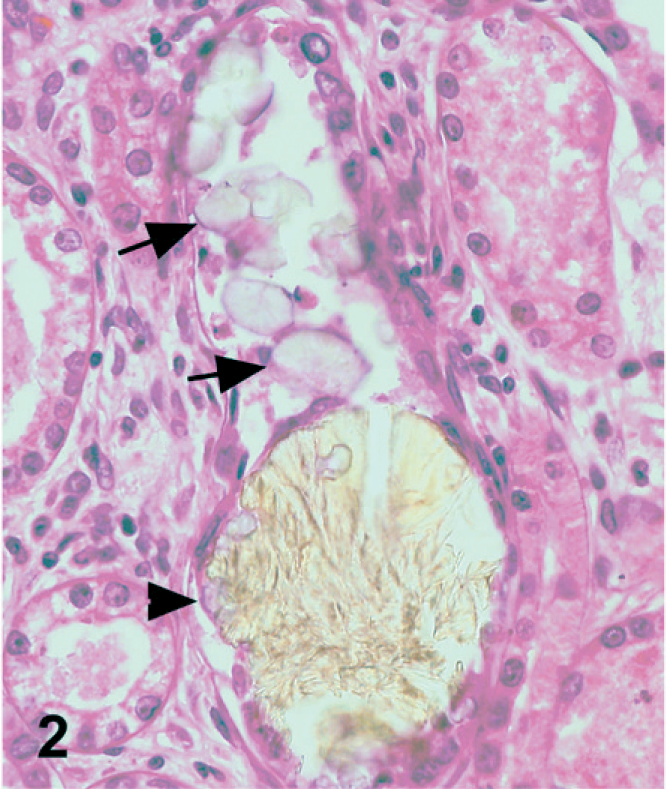
Kidney; dog No. 1. Pale brown, rough, melamine-containing crystals (arrowhead) and smoother oxalate crystals (arrow) are present within a renal tubule. HE.
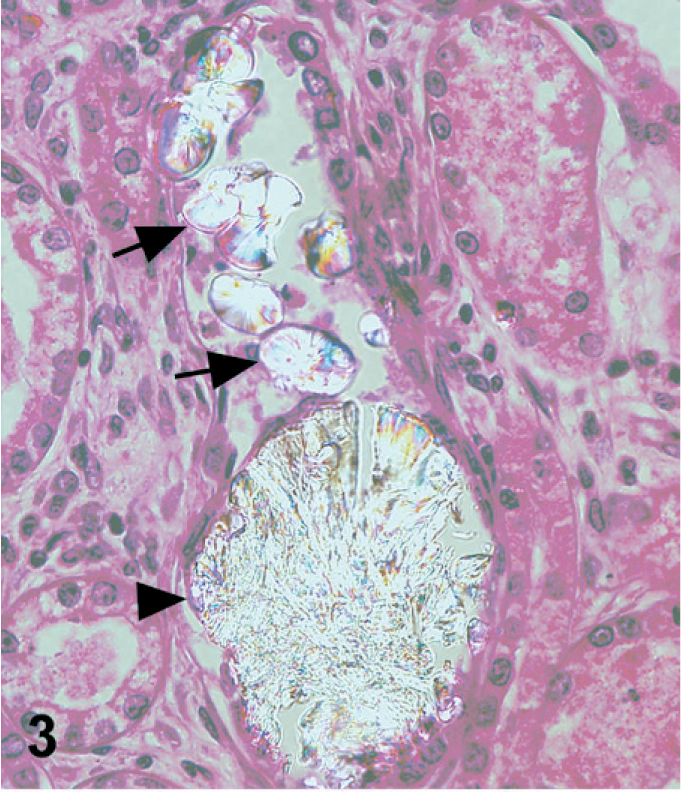
Kidney; dog No. 1. Birefringent melamine-containing crystals (arrowhead) and oxalate crystals (arrows) are present within a renal tubule. The oxalate crystals have a blue tinge as a result of a prismatic effect. Same field of view as Fig. 2 under polarized light.
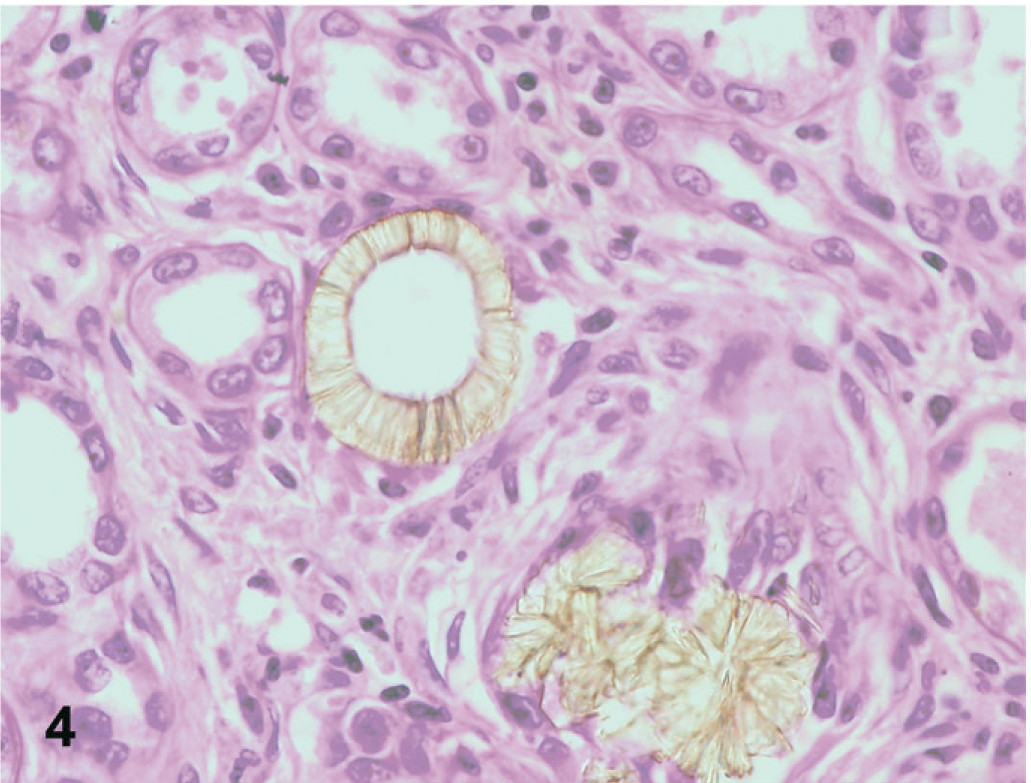
Kidney; dog No. 2. Central drop out is present in some examples of the melamine-containing crystals. HE.
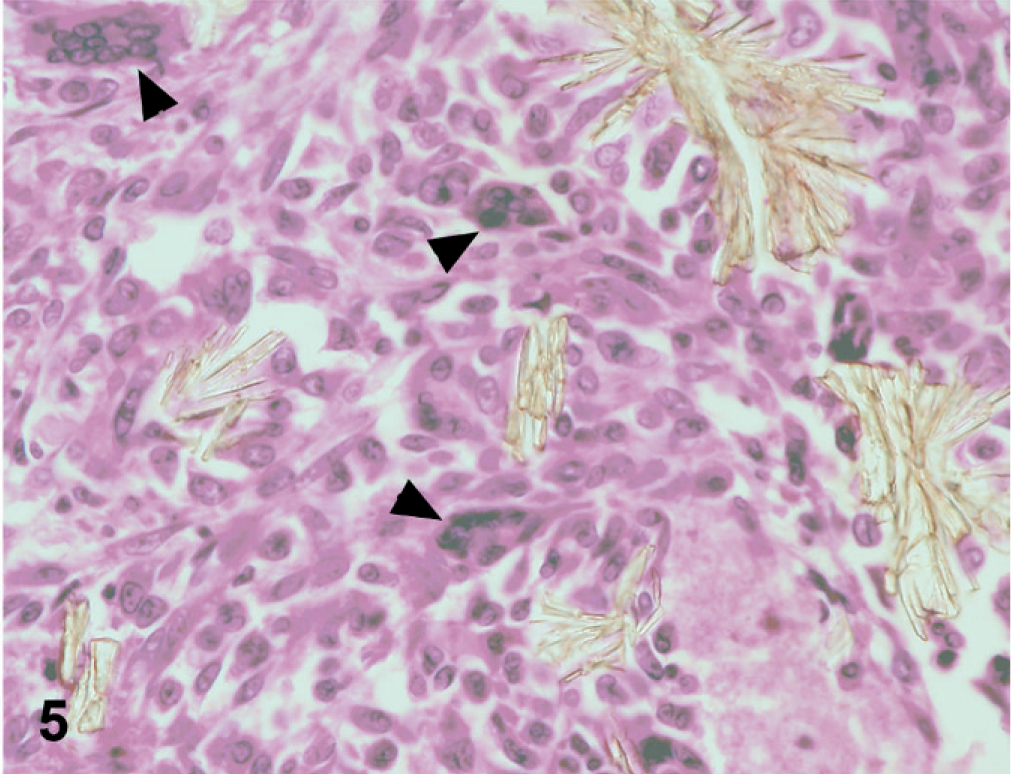
Kidney; dog No. 2. In some areas, crystals within the interstitium elicit a foreign body–type giant cell (arrowheads) reaction. HE.
In all 3 cases, an ORO72h stain demonstrated variable degrees of positive staining of the pale brown, rough-textured crystals, indicating a plastic or lipid origin, but did not stain the smoother platelike crystals (Fig. 6). 23 Oil Red O staining performed at 24 and 48 hours yielded similar results.
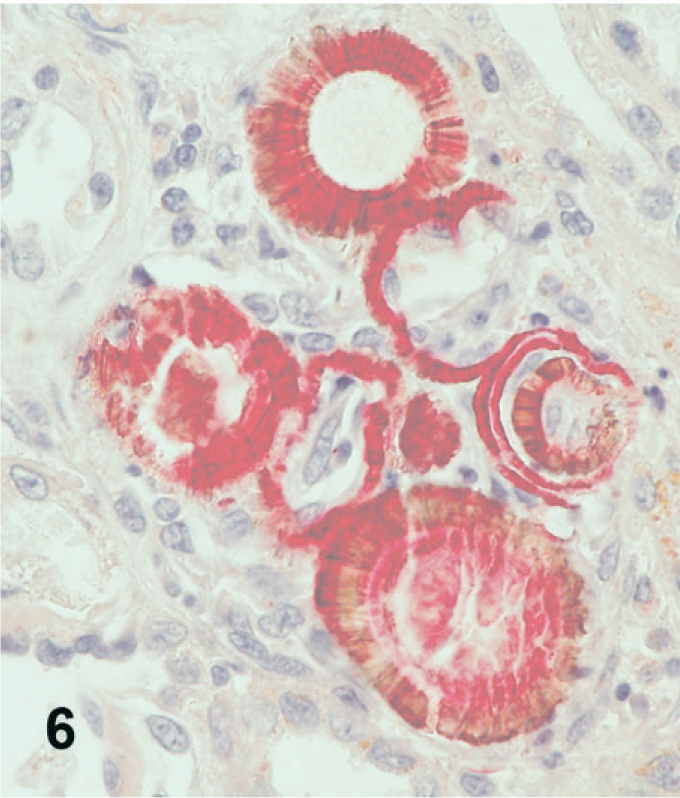
Kidney; dog No. 2. Melamine-containing crystals stain positively with ORO72h.
In dog Nos. 1 and 3, Alizarin Red S (pH 4.1–4.3) did not stain either birefringent crystal type. However, it did stain the basement membranes that were basophilic on HE section (Fig. 7). This finding was consistent with the deposition of a calcium salt other than calcium oxalate, likely calcium phosphate. There were also scattered Alizarin Red S positive crystals, which likely also represented calcium phosphate. In dog No. 2, there were scattered foci of Alizarin Red S staining only in the outer cortex, some of which may have represented other calcium salts such as calcium phosphate, and some of which may have represented stain artifact.
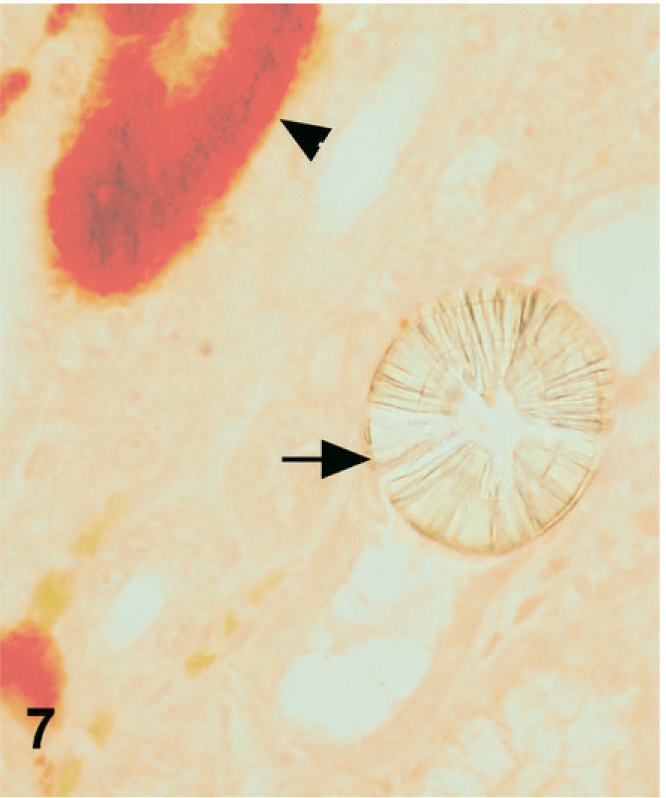
Kidney; dog No. 1. Mineralized tubular basement membranes stain positively with Alizarin Red S (arrowhead) while melamine-containing crystals (arrow) do not stain with Alizarin Red S.
In dog Nos. 1 and 3, Von Kossa stained the smooth crystals and the basophilic crystals consistent with calcium oxalate and calcium phosphate, respectively, but did not stain the pale brown, rough crystals (Fig. 8). Both Von Kossa–stained crystals and nonstained crystals were present in the same tubule in some areas, and occasionally the 2 crystals appeared intermingled. Similar results were obtained in dog No. 2. In dog No. 1, in a band running around the inner cortex, the tubular basement membranes demonstrated foci of circumferential staining. The histochemical characteristics are summarized in Table 1.
Special histochemical staining characteristics of melamine-containing crystals, calcium oxalate, and calcium phosphate crystals.

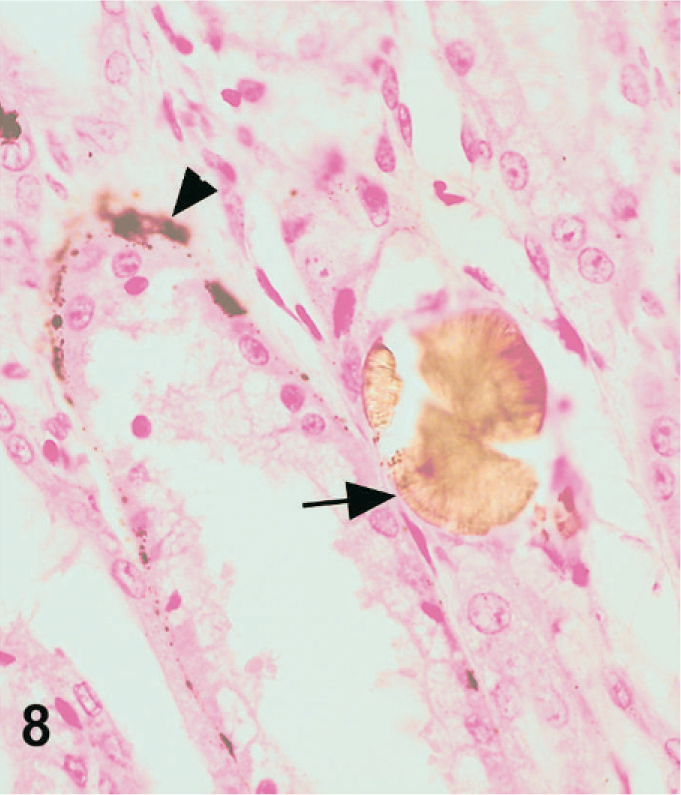
Kidney; dog No. 1. Mineralized tubular basement membranes stain with Von Kossa (arrowhead), while the melamine-containing crystals do not stain with Von Kossa (arrow).
Infrared microspectroscopy
In all 3 cases, the smooth birefringent crystals demonstrated spectra characteristic of whewellite (calcium oxalate monohydrate) by IR spectroscopy (Fig. 9). The basophilic crystals and the basophilic tubular basement membrane material both demonstrated the IR spectral characteristics of a calcium phosphate, possibly carbonate apatite. The rough, pale brown birefringent crystals demonstrated the IR spectral characteristics of melamine-containing material (Fig. 10). The infrared spectra of the pale brown crystals did not match those of pure melamine or pure cyanuric acid but were very similar to spectra obtained at the University of Guelph of a melamine–cyanuric acid complex (http://www.labservices.uoguelph.ca/urgent.cfm crystals). To produce the same complex, we mixed aqueous solutions of melamine and cyanuric acid in varying relative amounts. When the solutions were mixed, spontaneous precipitation of crystals caused the solutions to appear cloudy, but the addition of urea (or formalin) to the solution significantly enhanced the formation of precipitates. The recovered crystals were found to exhibit infrared spectra similar to those obtained from the kidney tissue of the 3 dogs. The spectra were also the same as those reported at the University of Guelph for urinary crystals from cats and melamine–cyanuric acid crystals formed in vitro in cat urine (http://www.labservices.uoguelph.ca/urgent.cfm crystals).
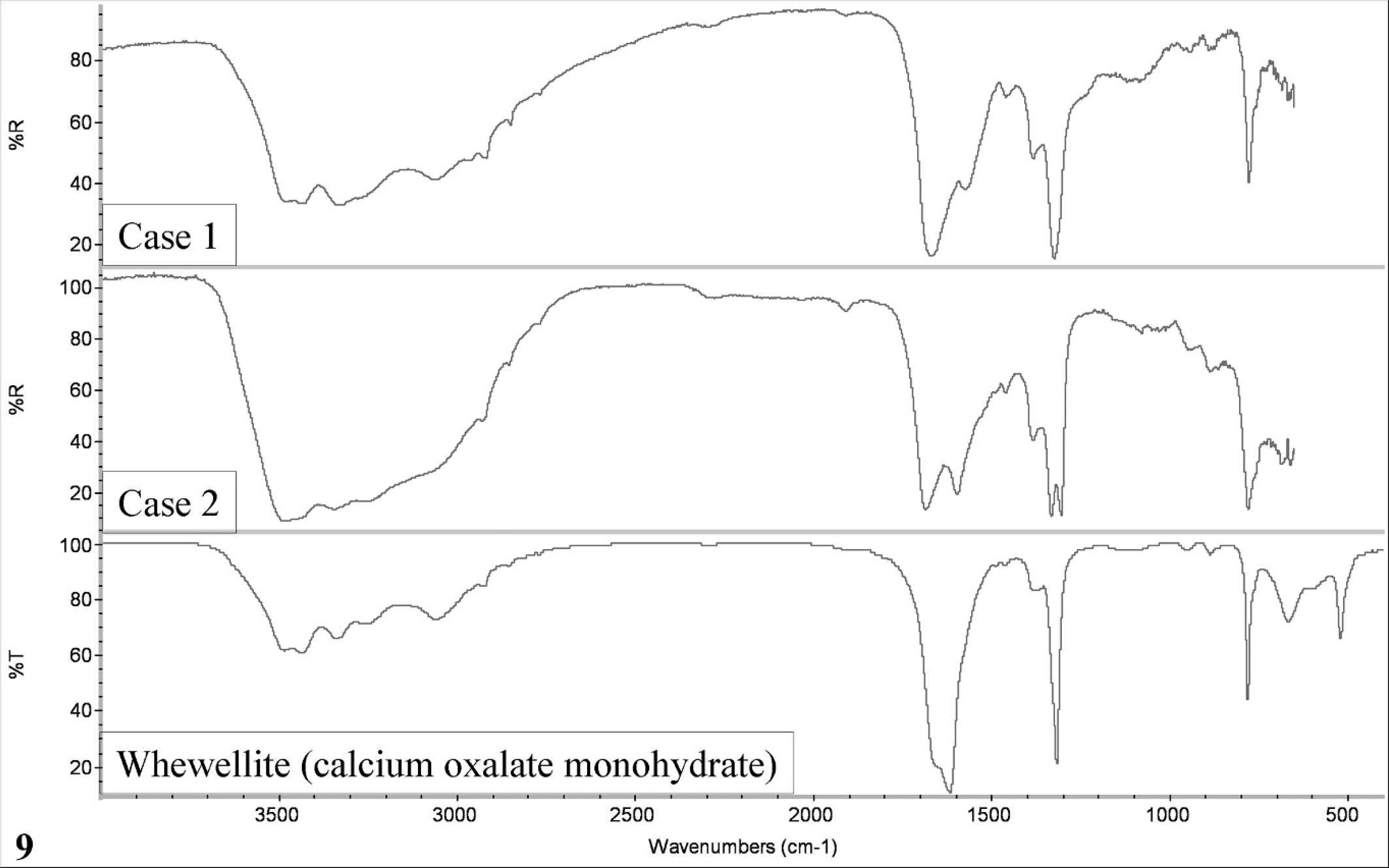
Kidney; dog Nos. 1 and 2. IR spectra obtained from crystals in dog Nos. 1 and 2 compared with a library spectrum of whewellite (calcium oxalate monohydrate).
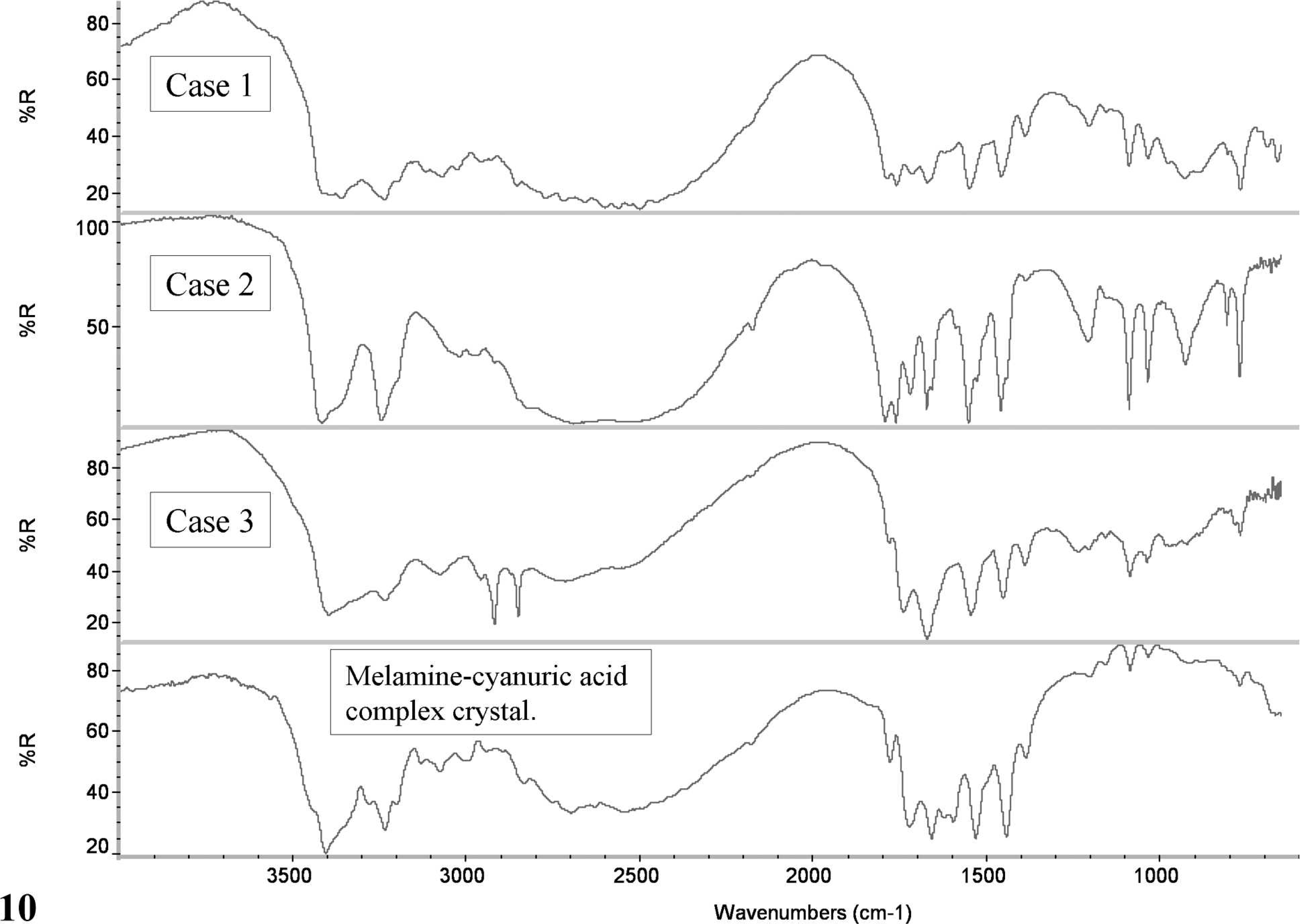
Kidney; dog Nos. 1, 2, and 3. IR spectra obtained from melamine-containing crystals from dog Nos. 1, 2, and 3 compared with the spectra of melamine–cyanuric acid complex crystals prepared in vitro.
Scanning electron microscopy with energy dispersive X-ray analysis
In all 3 cases, SEM/EDXA demonstrated the colocation of calcium, oxygen, and carbon in some crystals consistent with calcium oxalate (Figs. 11, 12). Areas of background tissue demonstrated peaks for carbon and oxygen only (Fig. 13). Other crystals demonstrated peaks for carbon, nitrogen and oxygen consistent with melamine-containing crystals (Figs. 14, 15). The presence of a nitrogen peak in the melamine-containing crystals reflected concentrations of nitrogen in excess of those seen in normal tissue protein (Fig. 13). In dog Nos. 1 and 3, the basophilic thickening of the basement membranes of tubules demonstrated peaks for calcium, phosphorus, oxygen, magnesium, and carbon. A small peak for magnesium was also identified in this area in dog No. 1. There were also particles demonstrating the presence of calcium, phosphorus, oxygen, and carbon consistent with calcium phosphate. In dog No. 2, small peaks for phosphorus and chlorine were present in addition to calcium, oxygen, and carbon.
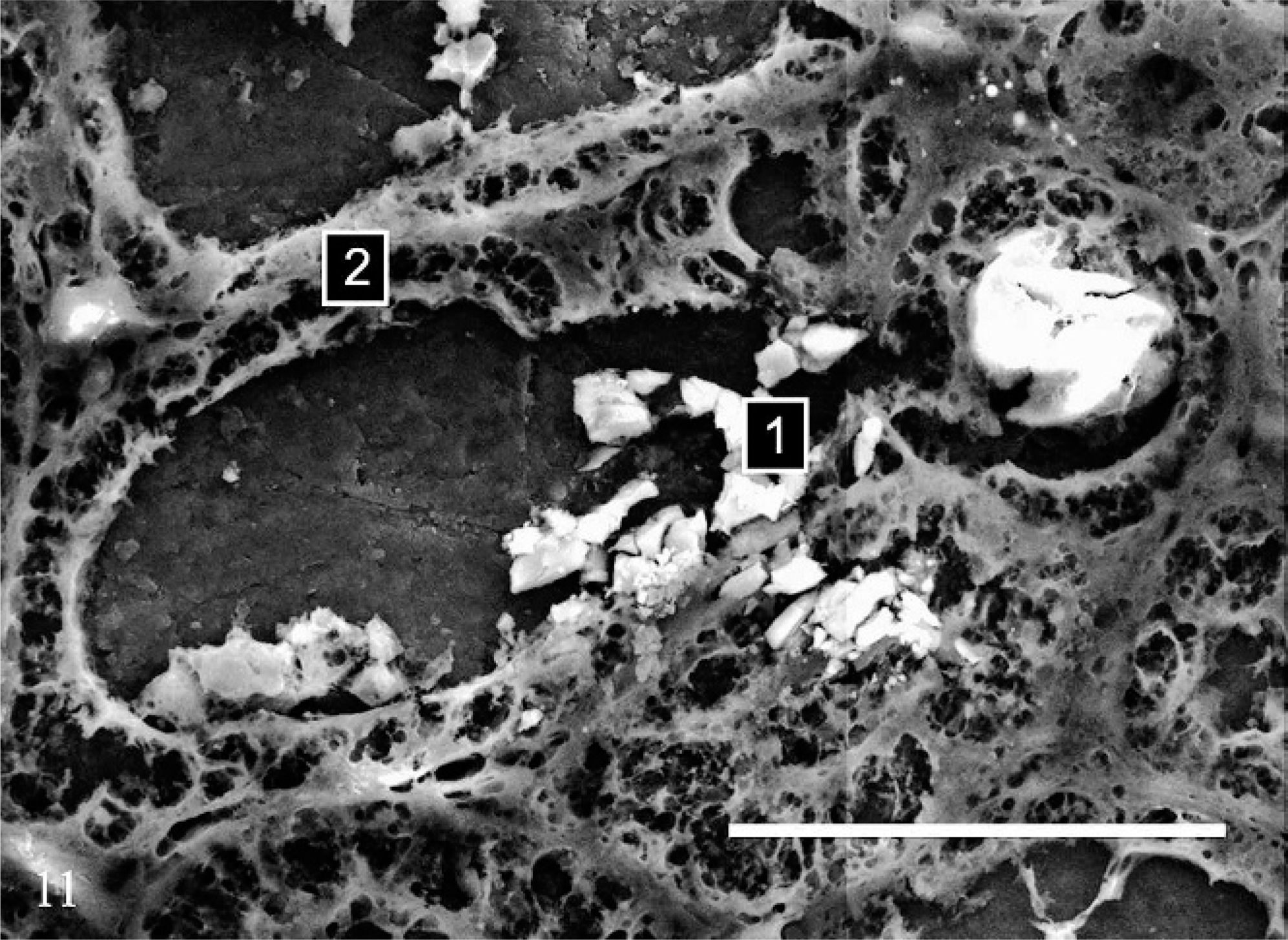
Kidney; dog No. 1. SEM showing calcium oxalate crystal in renal tubule [1] and tissue protein [2]. Bar = 50 μm.
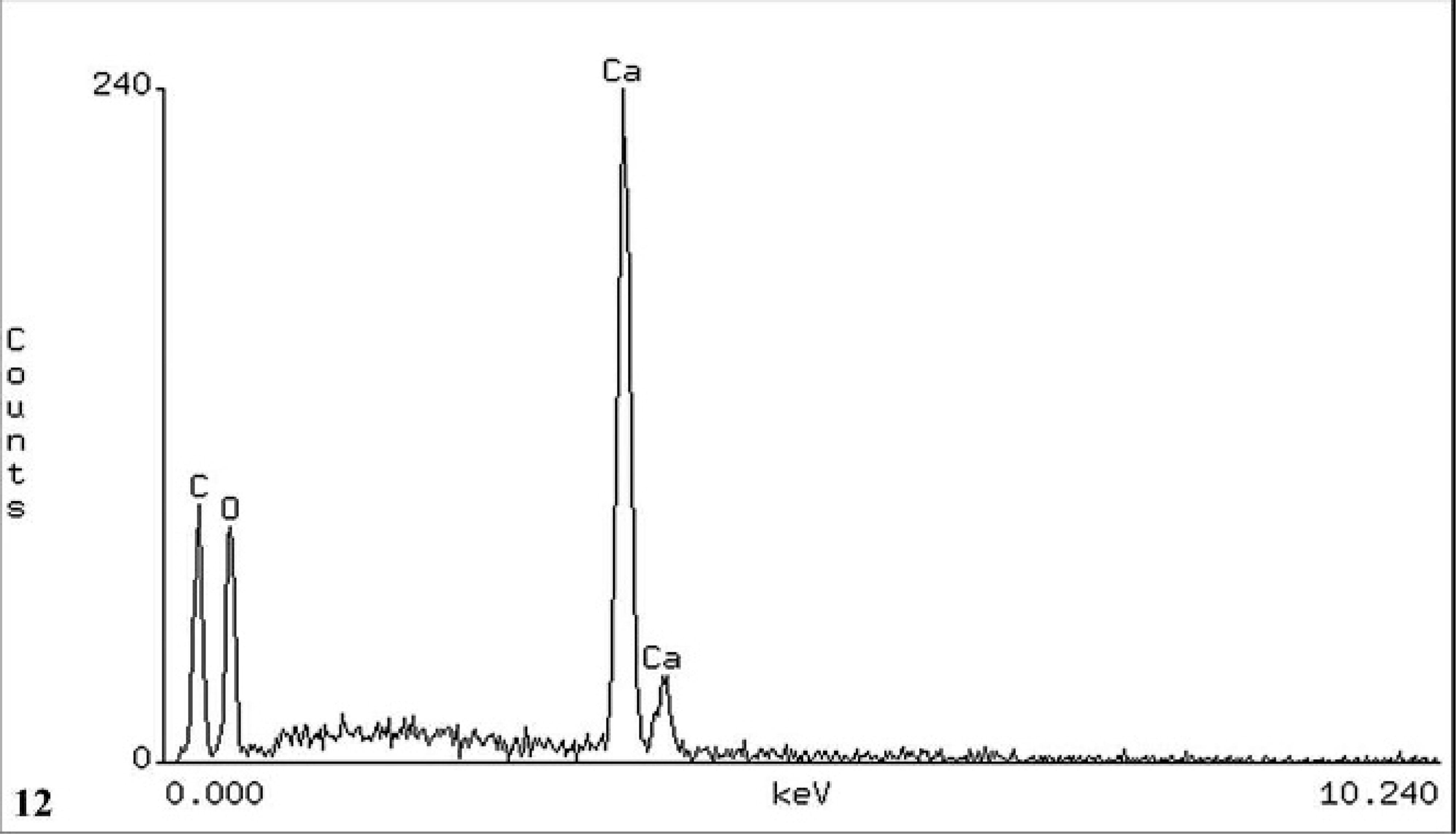
Kidney; dog No. 1. EDXA result from position [1] showing peaks for the elements carbon (C), oxygen (O), and calcium (Ca) consistent with calcium oxalate.
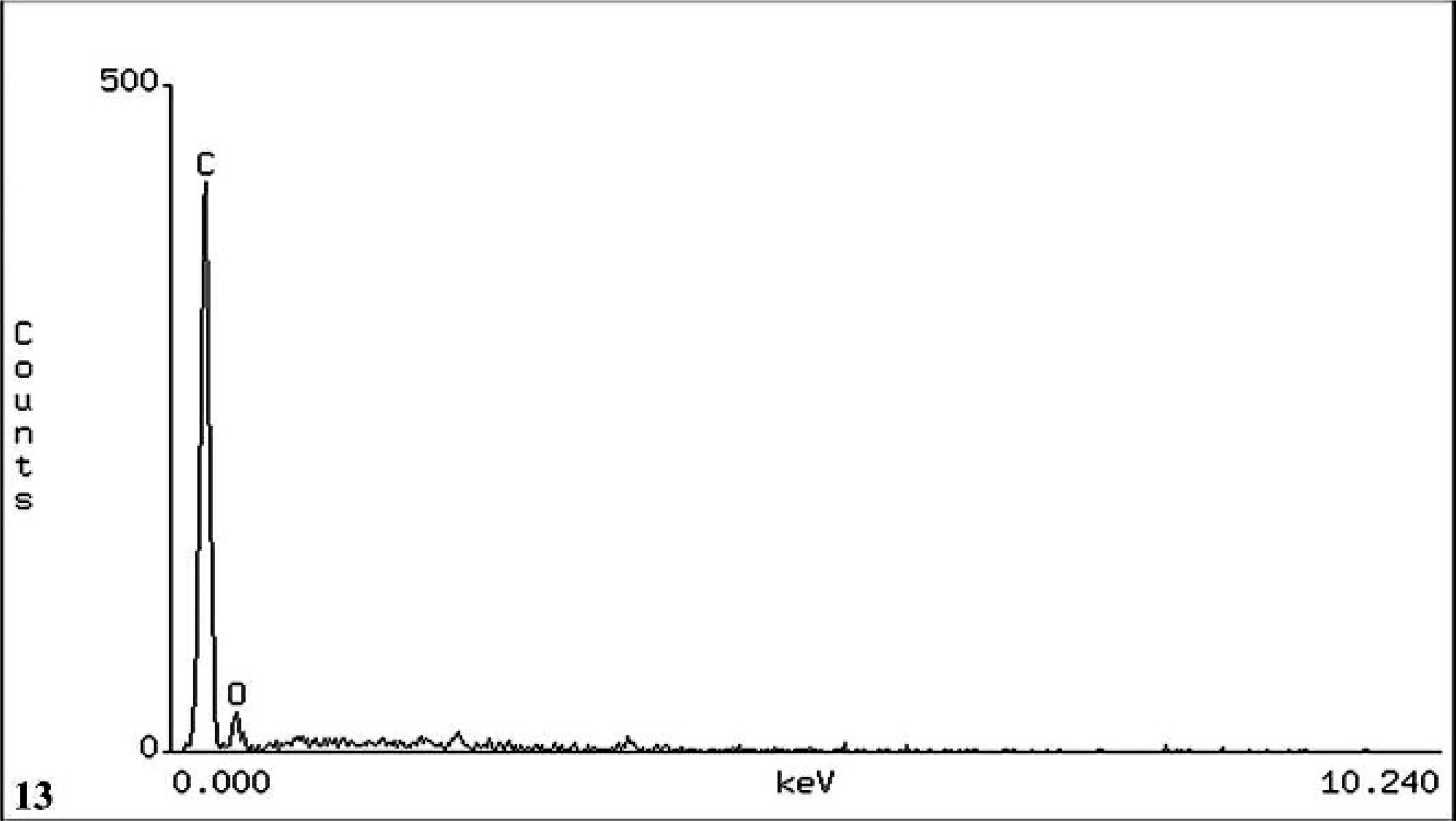
Kidney; dog No. 1. EDXA result from position [2], renal tubular tissue showing peaks for the elements carbon (C) and oxygen (O). Note the absence of a nitrogen peak in normal tissue protein compared with melamine–cyanuric acid complex crystals.
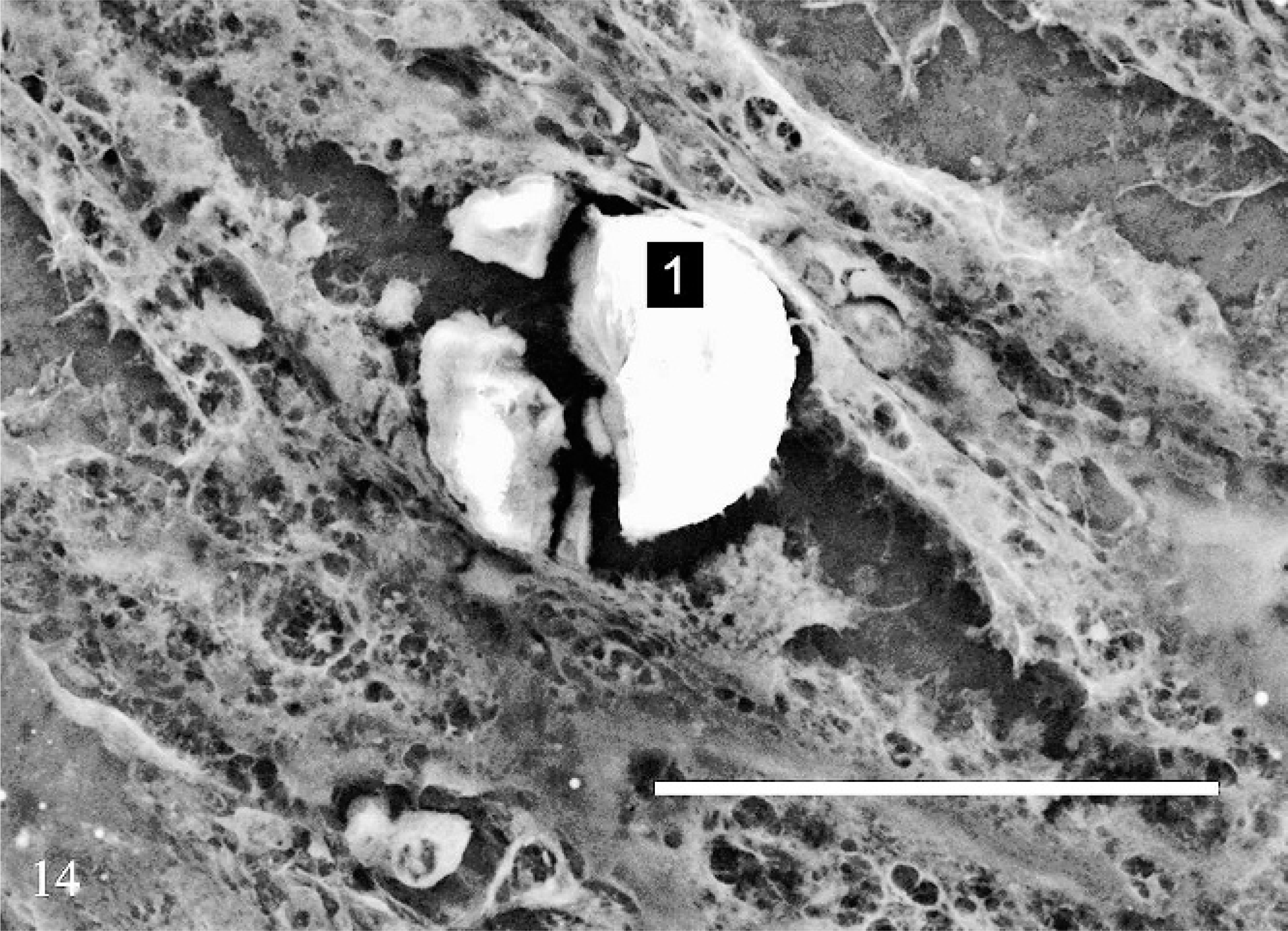
Kidney; dog No. 1. SEM showing melamine-containing crystal within a renal tubule. Bar = 50 μm.
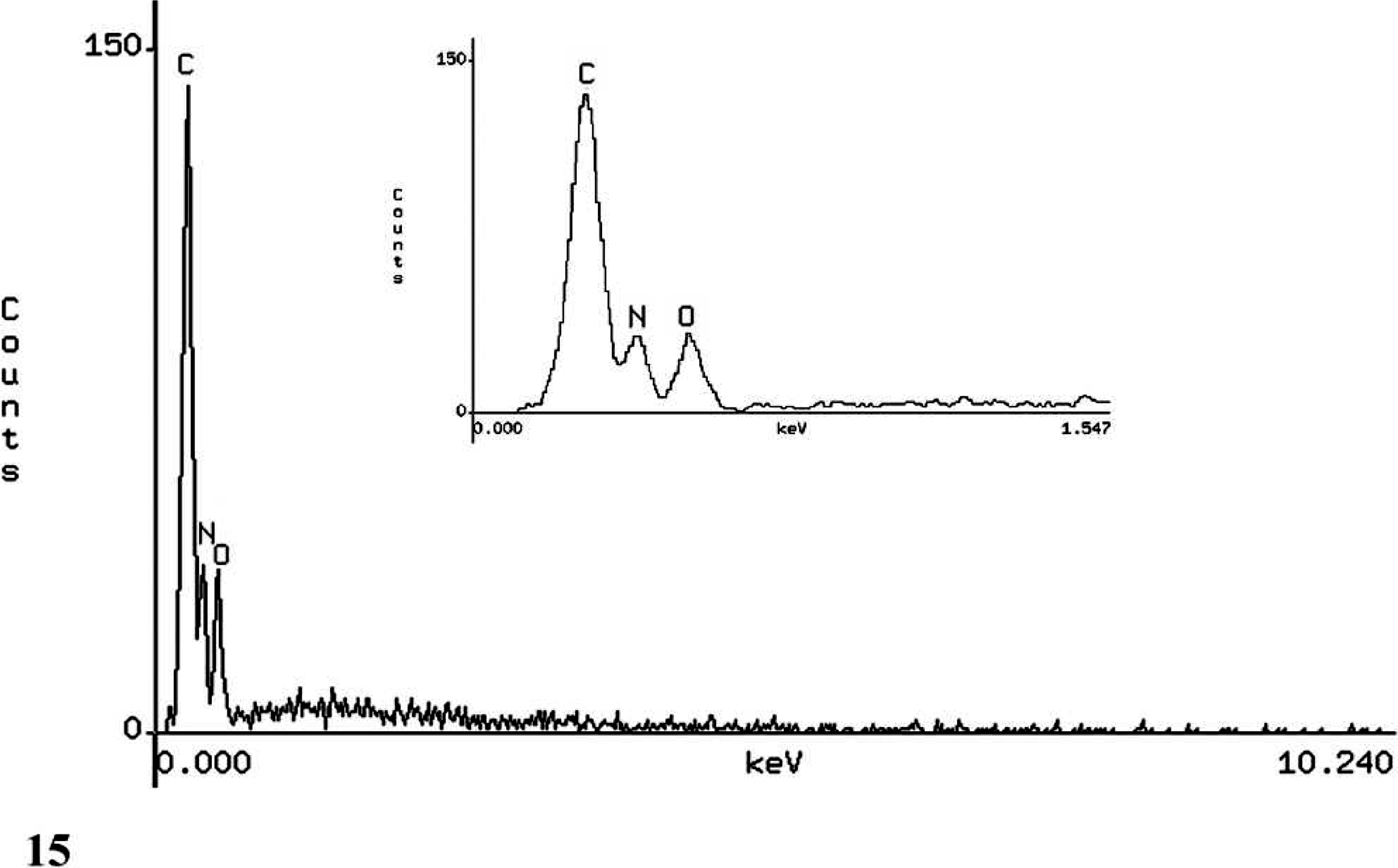
Kidney; dog No. 1. EDXA result from position [1] on melamine-containing crystal showing peaks for the elements carbon (C), nitrogen (N), and oxygen (O). Inset: Closeup of carbon (C), nitrogen (N), and oxygen (O) peaks.
Discussion
Both calcium oxalate monohydrate and melamine-containing crystals were identified in all 3 cases. Melamine and cyanuric acid were reported to have been identified in the recalled pet food and were present in urinary crystals and kidney obtained from cats. 1, 2, 4 Researchers at the University of Guelph, Ontario, produced crystals by adding melamine and cyanuric acid to cat urine in vitro (http://www.labservices.uoguelph.ca/urgent.cfm crystals). Both of these crystals had the same infrared (IR) spectral characteristics, which resemble the IR spectra of the pale brown crystals that we observed in the kidney tissue in all 3 of our cases.
We agree with the previous report by Brown et al. that the pet food–associated renal failure outbreaks in 2004 and 2007 share clinical, histologic, and toxicologic findings; this provides compelling evidence that they share the same causation. The report by Brown et al. acknowledges that it can be difficult to distinguish between calcium oxalate and melamine–cyanuric acid complex crystals by light microscopy alone. Thus, we have provided further support for this argument by obtaining IR spectra and SEM/EDXA of crystals in the kidneys of dogs from both outbreaks yielding results that are consistent with melamine–cyanuric acid complex crystals.
In this report we have illustrated the histologic and histochemical features of the melamine-containing crystals, which help to distinguish them from calcium oxalate and calcium phosphate. As previously stated, Alizarin Red S is a histochemical stain that is used to demonstrate the presence of divalent cations in tissue. In physiologic situations, the cations are usually calcium and therefore the stain is used for the detection of calcium. At pH 4.1–4.3, calcium oxalate does not react to form the red precipitates seen with other calcium salts such as calcium phosphate. 22 At pH 7.0, Alizarin red S will reportedly stain calcium oxalate, and repeating the stain at both pH values has been suggested to be a simple method for the histochemical detection of calcium oxalate. 20 Phosphates, carbonates, and oxalates of calcium, iron, and other ions can be demonstrated in tissue with the Von Kossa stain, which develops black deposits when positive. 22 Oil Red O stain for paraffin sections is primarily used as a stain for lipids. However, it has been noted to be a useful stain for many plastics that may be colorless or colored in routine HE-stained sections. 23 As the chemical composition of melamine includes many C-H groups, it was thought likely that it would stain with Oil red O. Calcium phosphate and calcium oxalate do not stain with Oil-red O even after a 72-hour exposure. The melamine-containing crystals do not stain with Alizarin Red S or Von Kossa but do stain with Oil Red O. The combination of the HE-appearance, birefringence, and histochemical features of the melamine-containing crystals are quite characteristic and distinguish them particularly from calcium oxalate and calcium phosphate. However, when confirmation is deemed necessary, crystal characterization by infrared (IR) spectroscopy is advisable.
Calcium oxalate dihydrate crystals can be found in the urine of healthy animals and occasionally in cases of ethylene glycol toxicosis. Calcium oxalate monohydrate crystals are more commonly associated with ethylene glycol toxicosis. 6 The presence of calcium oxalate monohydrate crystals in the kidney of all 3 dogs raises the possibility of ethylene glycol/oxalate poisoning in addition to the melamine-related pathology. However, occasional calcium oxalate crystals are commonly seen in urine sediment of normal dogs and in renal tubules with chronic tubular obstruction. 14 Importantly, calcium oxalate monohydrate crystals were a minority of the total number of birefringent crystals in all 3 of our cases. The melamine-containing crystals were in all cases the predominant crystal type seen. The presence of relatively small numbers of calcium oxalate crystals in these cases is most likely a result of secondary oxalosis associated with tubular injury as reported by Brown et al. 1
The calcium phosphate deposition in dog Nos. 1 and 3 and the mild lymphoplasmacytic interstitial nephritis noted in all 3 cases is interesting, and we wonder whether a subclinical chronic renal condition preceded the acute renal failure in these dogs. The selective deposition of calcium phosphate in the basement membrane of tubules in a band running around the inner cortex in dog No. 1 may have been a result of the pH gradient in this area and most likely represents metastatic calcification secondary to renal failure as in dog No. 3. It is well known that metastatic calcification principally occurs in the interstitial tissues of the gastric mucosa, kidneys, lungs, systemic arteries, and pulmonary veins since all of these tissues lose acid and therefore have an internal alkaline compartment that predisposes them to metastatic calcification. Mineral salts typically do not cause clinical dysfunction in the tissues in which they are deposited; however, massive deposits in the kidney (nephrocalcinosis) may cause renal damage over time. 10
Melamine has a relatively high safety margin, and it is considered unlikely that melamine itself directly causes renal failure. 3 It is currently believed that a combination of chemicals (melamine, cyanuric acid, possibly others) form insoluble crystals in animal kidneys with subsequent physical damage to renal tubules. 16 As previously stated, 2 other melamine by-products—ammeline and ammelide—may play a role and are currently under investigation. 16
The SEM/EDXA characteristics of melamine-containing crystals have not been reported to our knowledge. The interesting finding was the peak for nitrogen, which is not seen when examining tissue protein under normal operating conditions. This finding highlights the high nitrogen content of both melamine and to a lesser extent cyanuric acid. SEM/EDXA of calcium oxalate and calcium phosphate showed the expected colocation of elements, which also differs from the elemental composition of the melamine-containing crystals. The origin of the magnesium in dog No. 1 is undetermined but is probably an exogenous source.
We have demonstrated that the crystals seen in kidneys of dogs that ingested pet food on the 2007 Menu Foods recall list and in the 2004 Taiwan pet food–related outbreak of renal failure have characteristic light microscopic and histochemical properties that facilitate their recognition in tissue. Furthermore, we have positively identified distinct crystal types that can be reliably differentiated by routine methods. We have also demonstrated that they have supportive and somewhat distinctive SEM/EDXA findings. To our knowledge, this is the first report characterizing the special histochemical, IR spectral, and SEM/EDXA characteristics of melamine-containing crystals observed in cases of suspected pet food–induced nephrotoxicosis in dogs.
Footnotes
Acknowledgements
We are grateful to Mr. Anthony Shirley and Mr. Doug Landry for their photographic support. We would also like to thank Dr. Thelma Lee Gross for reviewing the manuscript, Dr. Anandita A. Datta, Department of Nephropathology, for her consultative support, and Dr. Florabel G. Mullick, The Director, Armed Forces Institute of Pathology. The work was supported in part by the American Registry of Pathology.
