Abstract
A high prevalence of systemic amyloidosis was documented in the black-footed cat (Felis nigripes) based on a retrospective review of necropsy tissues (n = 38) submitted as part of ongoing disease surveillance. Some degree of amyloid deposition was present in 33 of 38 (87%) of the examined cats, and amyloidosis was the most common cause of death (26/38, 68%). Amyloid deposition was most severe in the renal medullary interstitium (30/33, 91%) and glomeruli (21/33, 63%). Other common sites included the splenic follicular germinal centers (26/31, 84%), gastric lamina propria (9/23, 39%), and intestinal lamina propria (3/23, 13%). Amyloid in all sites stained with Congo red, and in 13 of 15 (87%) cats, deposits had strong immunoreactivity for canine AA protein by immunohistochemistry. There was no association with concurrent chronic inflammatory conditions (P = .51), suggesting that amyloidosis was not secondary to inflammation. Adrenal cortical hyperplasia, a morphologic indicator of stress that can predispose to amyloid deposition, was similarly not associated (P = .09) with amyloidosis. However, adrenals were not available from the majority of cats without amyloidosis; therefore, further analysis of this risk factor is warranted. Heritability estimation suggested that amyloidosis might be familial in this species. Additionally, tissues from a single free-ranging black-footed cat had small amounts of amyloid deposition, suggesting that there could be a predilection for amyloidosis in this species. Research to identify the protein sequence of serum amyloid A (SAA) in the black-footed cat is needed to further investigate the possibility of an amyloidogenic SAA in this species.
Black-footed cats (Felis nigripes), native to arid steppe and savannah habitats in southern Africa, are among the smallest of the felid species and are currently scheduled as vulnerable in the IUCN Red List (2006) and are also listed under Appendix I by the Convention on the International Trade of Endangered Species (CITES). 18 In contrast to many species that live longer in captive settings where food is abundant and predators scarce, black-footed cats tend to have shorter life spans in captivity than in the wild. The majority of captive adult black-footed cat deaths occur between 2 and 5 years, 18 whereas free-ranging cats have been estimated to live approximately 5–6 years (A. Sliwa, personal communication). A retrospective review of necropsy reports suggested that amyloidosis was a significant cause of mortality and an important factor in the decreased survival of captive black-footed cats (Lamberski, unpublished data).
The amyloidoses are a group of diseases that are characterized by intracellular or extracellular deposition of insoluble fibrillar protein. Over 20 different precursor proteins have been identified in the various forms of amyloidosis. Despite differences in the nature of the precursor protein, all amyloidoses have similar homogeneous eosinophilic histologic appearance when stained with hematoxylin and eosin (HE) and share affinity for certain histologic stains such as Congo red. Biochemically, proteins that assemble into amyloid fibrils all show a propensity to form β-pleated sheet secondary structures in at least 1 domain of the protein sequence. Processes that may be involved in the development of amyloidosis include overproduction of an amyloidogenic protein, production of a mutant protein that has increased propensity to form amyloid fibrils, failure to excrete or properly traffic an intact protein that has a concentration-dependent tendency to misfold, abnormal proteolysis generating an amyloidogenic fragment from a normal precursor, and inadequate proteolytic cleavage due to either overproduction of the substrate or insufficient degradation. 3
The purpose of this study was to characterize the lesions associated with amyloidosis, identify the type of amyloid, and investigate possible risk factors to better understand the pathogenesis of amyloidosis in the black-footed cat.
Materials and Methods
Necropsy tissues or histopathology slides from black-footed cats (n = 38) that died between 1989 and 2005 representing the North American, European, and South African captive populations were reviewed. In addition, necropsy samples from 1 free-ranging juvenile were also examined. Cause of death or reason for euthanasia was determined for each individual by 2 of the authors (KAT and LM) on the basis of histologic lesions in combination with gross necropsy findings, when available. All tissues were processed routinely for histology, and sections were stained with HE. Amyloid deposition and tissue distribution were confirmed by the presence of compatible lesions and identification of characteristic “apple green” birefringent fibrils in Congo red (CR)–stained sections of affected tissues. Amyloid was graded on a semiquantitative scale in the kidney as follows: renal medulla, − = no amyloid detected, + = minimal to mild deposition within the interstitium affecting <25% of the medulla, ++ = moderate multifocal deposition within the interstitium affecting between 25 and 50% of the medulla, and +++ severe multifocal to diffuse deposition affecting >50% of the interstitium; renal glomeruli, − = no amyloid detected, + = mild segmental deposition within <25% of glomeruli, ++ = moderate segmental to global deposition affecting 25–50% of the glomeruli, and +++ = severe segmental to global deposition affecting >50% of glomeruli. For nonrenal tissues, amyloid was graded as follows: − = no amyloid detected, + = minimal to mild deposition with no evident tissue disruption, ++ = moderate multifocal deposition with mild disruption of normal tissue architecture, and +++ = severe multifocal to diffuse deposition with disruption of normal tissue architecture, evidence of secondary tissue damage, or both. Prevalence of amyloid deposition in specific organs was determined on the basis of the numbers of cats for which that tissue was available for evaluation (not all cases included a complete set of tissues). Concurrent lesions were also documented to determine whether there were any associations between amyloidosis and other disease conditions.
Paraffin-embedded sections of amyloid-positive (on the basis of CR staining) kidney, spleen, or stomach from 15 black-footed cats from whom appropriate tissue was available were sectioned at 5 μm and placed onto Probe-On Plus microscope slides (Fisher Scientific, Pittsburgh PA). Sections were deparaffinized, rehydrated, and immunohistochemically labeled using the avidin–biotin–peroxidase complex method. Endogenous peroxidase was quenched with 3% hydrogen peroxide for 15 minutes, and sections were rinsed. Nonspecific binding was blocked with normal goat serum (1 : 10 in Tris-buffered saline) for 15 minutes followed by incubation overnight at 4°C with primary antibody (rabbit anti-dog AA at 1/400 dilution). 12 After rinsing, sections were incubated with biotinylated goat anti-rabbit (cat. No. LSAB2 Kit, DAKO) for 15 minutes. Sections were again rinsed and incubated with streptavidin–horseradish peroxidase label (LSAB2 kit, Dako) for 15 minutes, rinsed, and incubated with 3-amino-9-ethylcarbazole (AEC ready-to-use kit, Dako) with hydrogen peroxide for 10 minutes. Sections were then rinsed for 5 minutes in distilled water followed by running tap water rinse for 5 minutes and counterstained with Mayer's hematoxylin (Sigma) for 2–5 minutes. Negative staining control consisted of the above procedure with the primary antibody omitted. As a nonspecific staining control, the samples were also stained as above, substituting rabbit anti-human islet amyloid polypeptide (Peninsula Laboratories, San Carlos, CA) as the primary antibody. Positive control staining for the primary antiserum was performed on sections of dog kidney with confirmed AA amyloid deposits.
Comparisons of age at death and sex between affected and unaffected individuals were performed using a t-test assuming unequal variances. Association between concurrent conditions and amyloidosis was assessed by the Fisher Exact test excluding cases with incomplete tissue sets.
To assess whether amyloidosis was familial in this species, pedigrees of affected individuals were determined to the level of founders (wild-caught individuals). A total of 86 animals were included in this analysis, including all study animals as well as related animals on the basis of captive breeding records. Although it was not possible to evaluate all animals included in the pedigree for amyloidosis, inclusion of related individuals was essential to determine relationships between affected and unaffected animals for estimation of heritability. For the purposes of this analysis, animals were classified as known positive, known negative, or unknown amyloid status. Known positive animals had histologically confirmed amyloidosis, whereas known negative animals were confirmed histologically not to have any evidence of amyloid deposition. Animals for whom no tissues were available to evaluate (e.g., founders and some ancestors of the study population) or animals from whom tissues available did not include all of the commonly affected sites were classified as unknown. The heritability of amyloidosis, scored as a binary phenotype, was estimated in a threshold model in a manner similar to that used for the inheritance of deafness in the Dalmatian described in Cargill et al. 4 This model took advantage of a Bayesian approach to the estimation of the unknown genetic parameters through Gibbs sampling, in which the computations were facilitated through the public domain software MTGSAM. 28
Results
Cases included 19 males, 15 females, and 4 animals whose gender could not be determined from the submitted tissues or history. Animals ranged from 299 days to 13 years with average and median ages at death of 3.77 and 3.75 years, respectively. Some degree of amyloid deposition was present in 33 of 38 (87%) of the examined cats (Table 1). Amyloidosis was the most common cause of death in the study animals (26/38, 68%), usually because of renal deposition. In 4 cats (Nos. 9–12), minimal renal amyloid deposition was not considered of sufficient severity to have caused the renal failure. In a single cat, gastrointestinal amyloidosis without renal involvement was diagnosed as the cause of death. There was no difference in the ages at death of affected and unaffected cats (P = .28). There was no apparent sex predilection.
Signalment, cause of death, predisposing conditions, and grade and distribution of amyloid in black-footed cats.
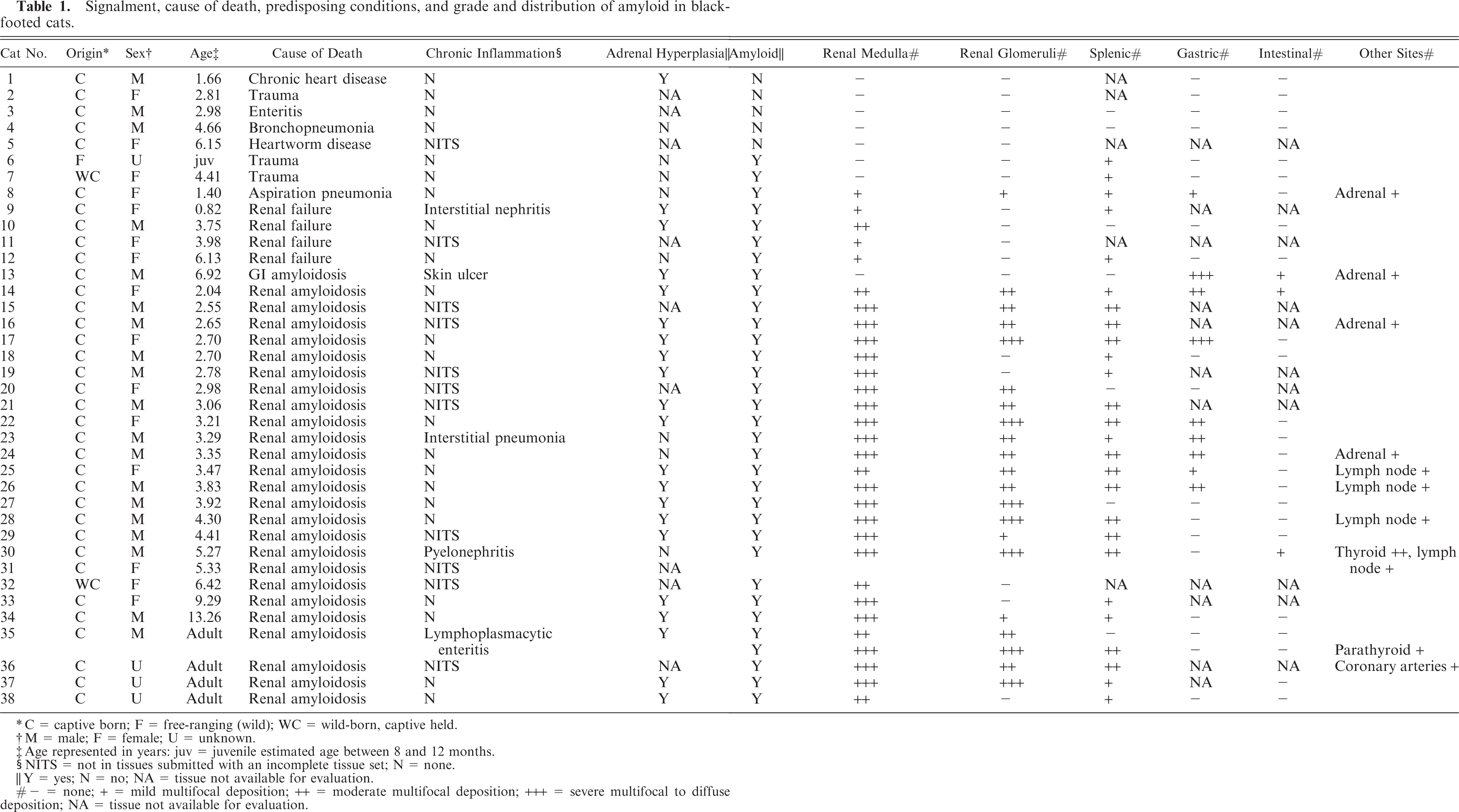
C = captive born; F = free-ranging (wild); WC = wild-born, captive held.
M = male; F = female; U = unknown.
Age represented in years: juv = juvenile estimated age between 8 and 12 months.
NITS = not in tissues submitted with an incomplete tissue set; N = none.
Y = yes; N = no; NA = tissue not available for evaluation.
- = none; + = mild multifocal deposition; ++ = moderate multifocal deposition; +++ = severe multifocal to diffuse deposition; NA = tissue not available for evaluation.
Amyloid deposition was most commonly present within the renal medulla (30/33, 91%). In the renal medulla, amyloid was present between tubules of both the outer and inner medulla (Fig. 1). Tubules within affected areas were typically atrophied, and interstitial fibrosis was prominent in some cats. Papillary necrosis was present in 7 of 33 (21%) cats with amyloidosis and was only present in animals with severe renal medullary amyloid deposition. Amyloid was also present within the renal glomeruli (21/33, 63%) with segmental to global expansion of the mesangium (Fig. 2). Tubular hyaline casts were evident in 19 of 33 (58%) cats with amyloidosis, all of which had some degree of glomerular amyloid deposition. Histologic evidence of uremia (mineralization within the stomach, tongue, lungs) was present in 7 of 33 (21%) cats with amyloidosis. Other common sites of amyloid deposition included splenic follicular germinal centers and tunica media of splenic arterioles (26/31, 84%) (Fig. 3); gastric lamina propria (9/23, 39%) (Fig. 4); small intestinal, colonic lamina propria, or both (3/23, 13%); adrenal cortex (4/27, 15%); and lymph nodes (4/33, 12%). Uncommon sites in which deposition was only noted in a single animal included the tunica media of the coronary arteries, thyroid, and parathyroid glands.
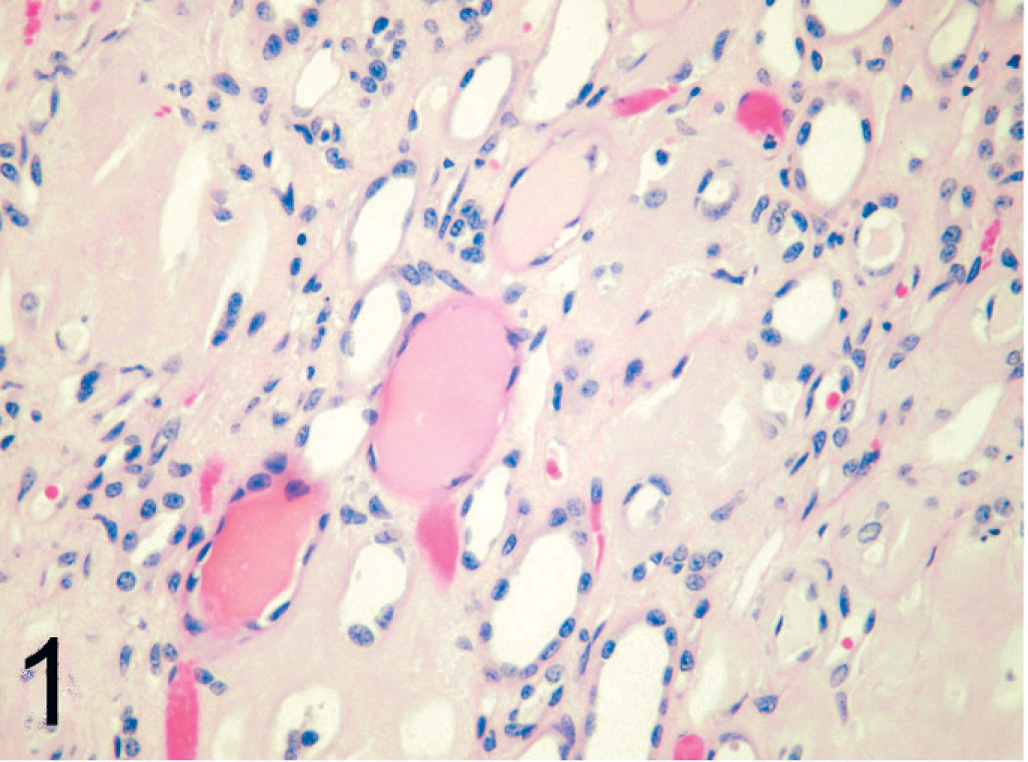
Kidney; black-footed cat No. 17. Amyloid deposition within the renal medulla. Multiple tubular hyaline casts are also evident. HE.
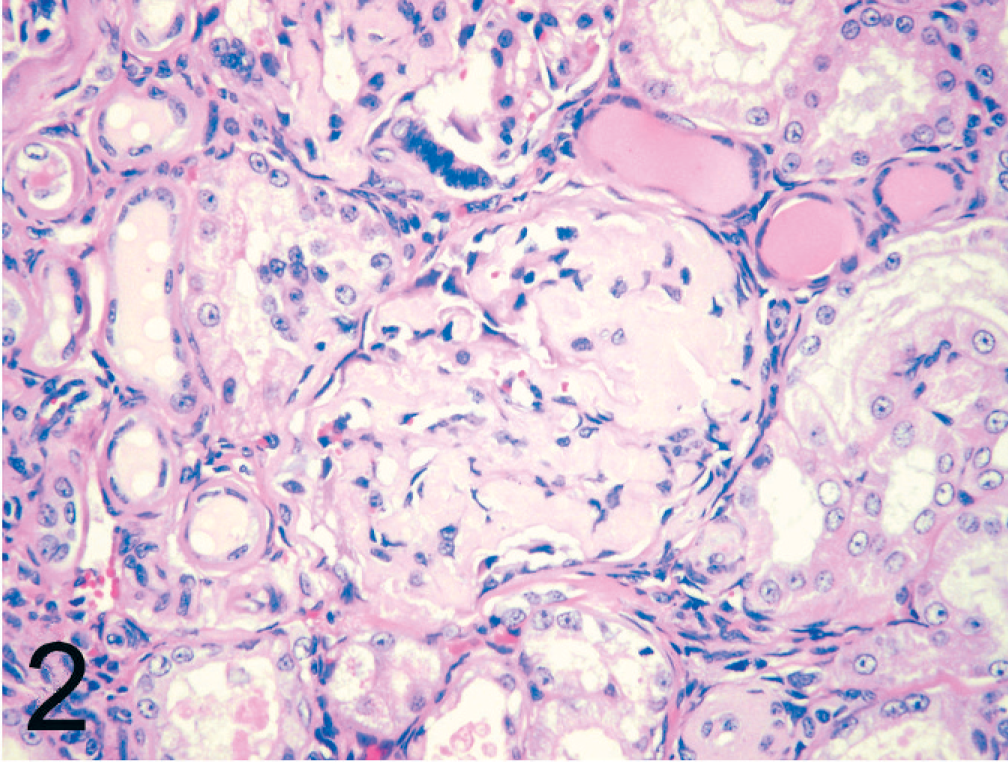
Kidney; black-footed cat No. 17. Global deposition of amyloid within the glomerular mesangium. Tubular hyaline casts are also evident. HE.
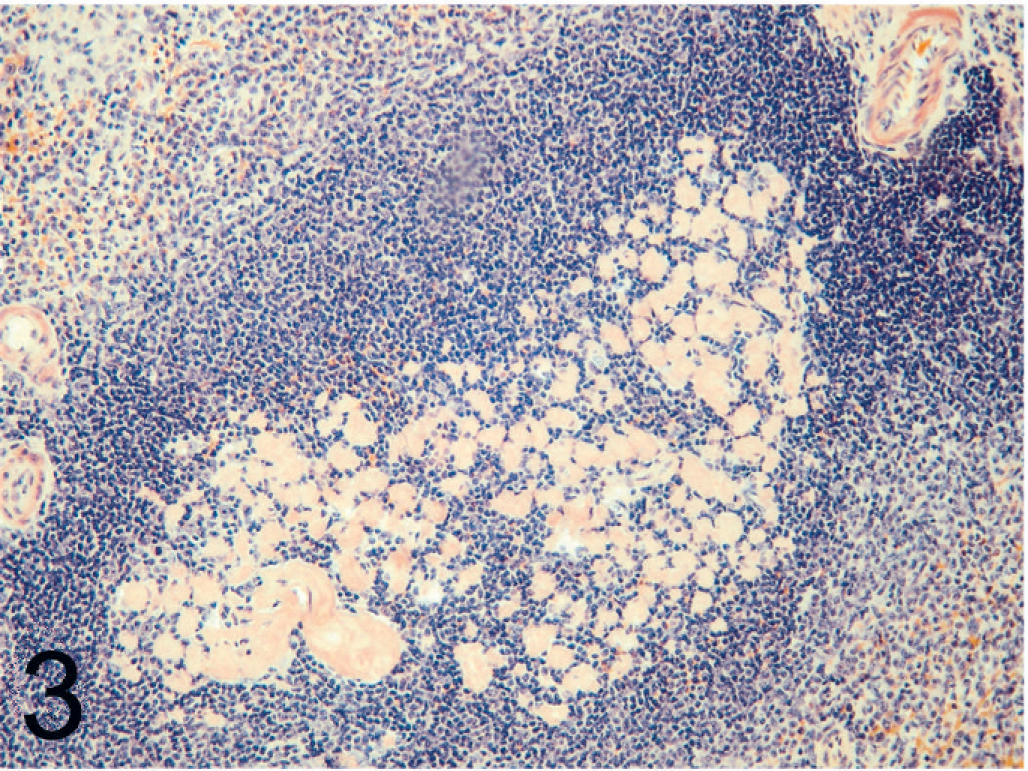
Spleen; black-footed cat No. 12. Amyloid deposition within a splenic lymphoid follicle. CR.
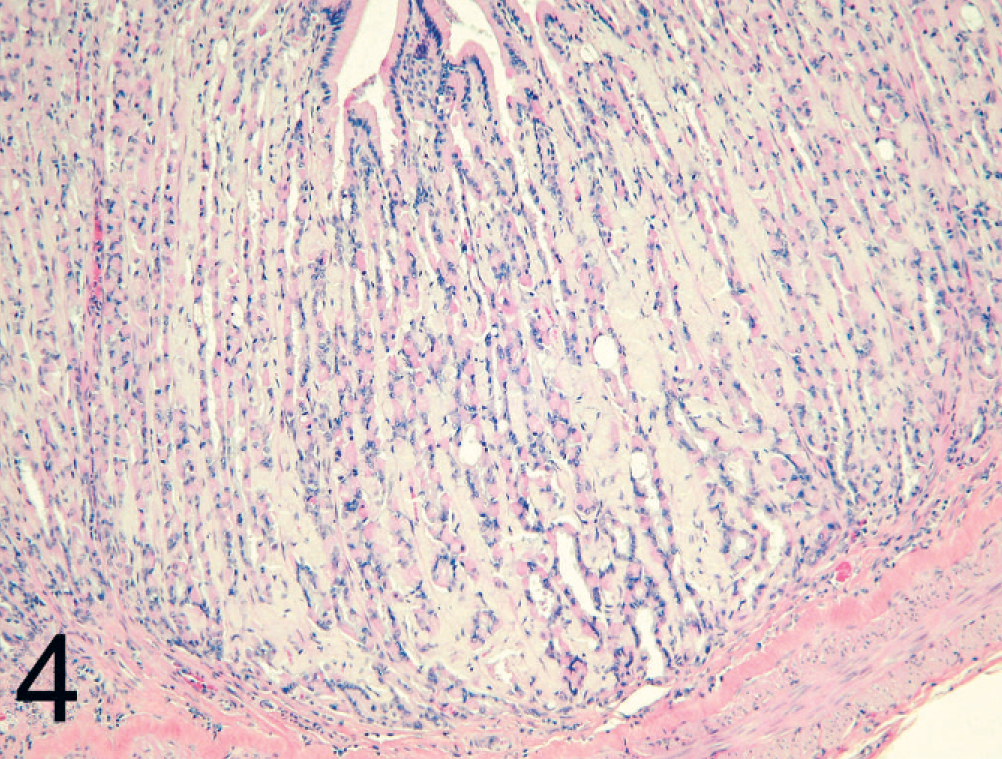
Stomach; black-footed cat No. 13. Amyloid deposition within the gastric lamina propria. HE.
All but 3 animals with amyloidosis (cat Nos. 6, 7, 13) had some degree of amyloid deposition within the kidneys. In cat No. 13, amyloid deposition was confined to the gastrointestinal tract, whereas in cat Nos. 6 and 7 amyloid was only noted within splenic follicles.
There was no association (P = .51) between concurrent chronic inflammation and amyloidosis. Only 4 of 23 (17%) black-footed cats from whom complete tissue sets were available had a chronic inflammatory condition. Adrenal cortical hyperplasia was also not associated with amyloidosis (P = .09); however, adrenal glands were only available from 2 unaffected cats. Many cats also had iron accumulation within Kupffer cells and splenic macrophages, although this also was not correlated with amyloidosis (P = .48).
Strong immunoreactivity with a polyclonal antibody against canine AA protein was noted in amyloid deposits within sections of kidney, spleen, lymph node, and stomach from 13 of 15 (86%) cats (Figs. 5, 6). Staining was equivocal in cat No. 14 and negative in cat No. 7, although both cats had positive CR staining. No immunoreactivity was noted with a polyclonal antibody to human IAPP.
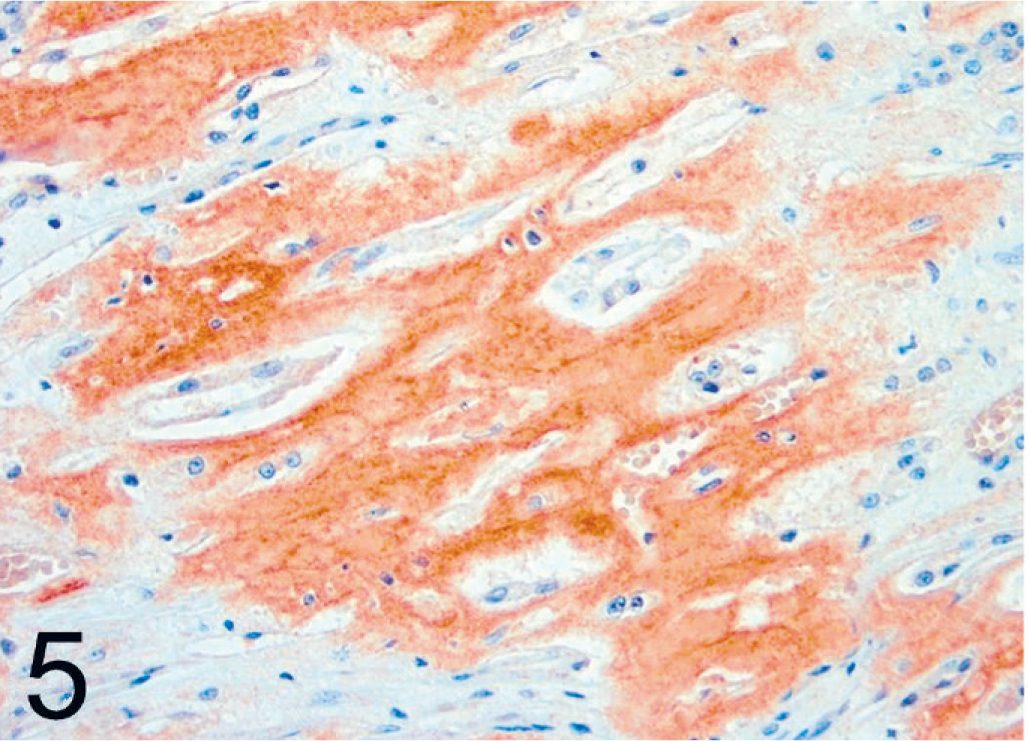
Kidney; black-footed cat No. 17. Immunoreactivity of renal medullary amyloid using a polyclonal antibody against canine AA. LSAB/AEC, Mayer's hematoxylin counterstain.
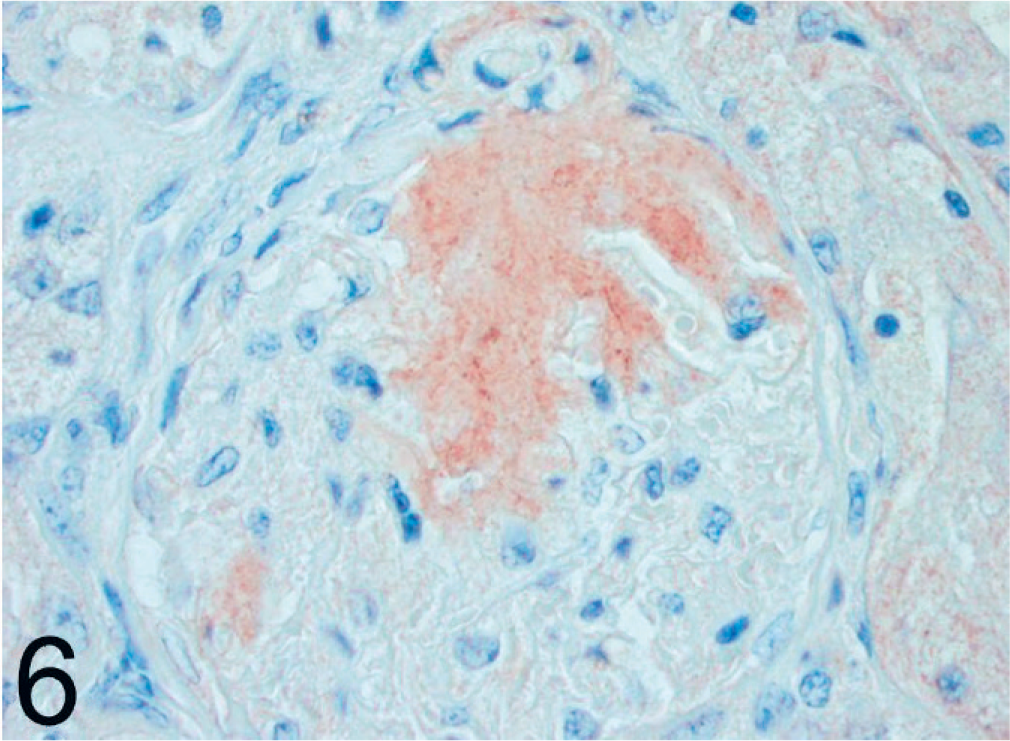
Kidney; black-footed cat No. 17. Immunoreactivity of glomerular amyloid using a polyclonal antibody against canine AA. LSAB/AEC, Mayer's hematoxylin counterstain.
Making use of the Gibbs sample, generated by the MTGSAM software, the mean estimate for the heritability of amyloidosis was 0.81 ± 0.08. Accordingly, although the sample size is small compared with data sets typically used for the estimation of genetic variances, there appears to be sufficient evidence to consider this disorder to be heritable. In addition, the analysis revealed that there was not a significant difference between the sexes in the incidence of disease.
Discussion
This study identified amyloidosis and resultant renal failure as an important cause of mortality in black-footed cats. Amyloid deposition was predominately renal, but extra-renal deposition was common in the spleen. One cat had extensive gastrointestinal tract deposition that was considered of sufficient severity to have resulted in clinical disease and death. Interestingly, hepatic amyloid deposition was not noted in any of the study animals. By immunohistochemistry, amyloid fibrils were determined to be AA type, although staining was weak in the case of gastrointestinal amyloidosis.
AA amyloidosis is the most common type of amyloid in mammals and birds and often results in hepatic or renal failure due to physical disruption of normal cellular and organ processes. AA amyloid is derived from the normal serum protein AA (SAA), produced mainly by the liver. SAA production is increased in response to inflammation and therefore AA amyloidosis is often secondary to chronic inflammation. Among nondomestic species, cheetahs (Acinonyx jubatus), 19 Dorcas gazelles (Gazella dorcas), 21 and anseriformes (e.g., ducks and geese) 5, 9, 15 appear to be uniquely susceptible to develop AA amyloidosis secondary to chronic infections. Concurrent disease conditions were also considered the most important predisposing factors for AA amyloidosis in the black-footed ferret (Mustela nigripes); however, the authors did not rule out a genetic contribution to disease development given the inbred nature of this population. 7 In the current study of black-footed cats, there was no association between chronic inflammation and amyloidosis. Concurrent inflammatory processes were generally acute and of mild severity.
AA amyloidosis has also been associated with chronic stress in a variety of species including laboratory mice, 17 hamsters, 8 and herring gulls. 11 Significant increases in serum amyloid A have been noted in domestic pigs after transport, 20 and in domestic cattle housed on certain types of flooring. 1 Psychological and physical stressors in rats can elevate plasma interleukin-6, an important mediator of the acute phase response. 16, 29 Stressful stimuli can also induce the secretion and production of glucocorticoids. Although glucocorticoids are generally thought to have anti-inflammatory properties, they can enhance the transcriptional activation of serum amyloid A by certain cytokines, most notably interleukins 1 and 6, in humans. 25 In mice, restraint stress has been demonstrated to increase expression of serum amyloid A, but that expression is attenuated in IL-6 knockout mice. 10 Captive black-footed cats are presumed to experience some degree of chronic stress under captive conditions. Adrenal cortical hyperplasia, a morphologic indicator of stress, was present in the majority of study animals but was not statistically associated with amyloidosis, most likely because adrenal glands were available for evaluation from only 2 of the black-footed cats without amyloidosis. Therefore, further research will be needed to determine whether chronic stress is a predisposing or contributing factor for the development of amyloidosis in the black-footed cat.
AA amyloidosis can also occur as a familial or breed-specific disease in the absence of chronic inflammation. In animals, familial amyloidosis has been best described in Shar-Pei dogs 6, 12 and Abyssinian 2 and Siamese 26 cats. In both the Abyssinian and Siamese cats, there are specific amino acid sequence variations in the SAA protein that may render the protein more amyloidogenic, but additional factors affecting production or processing of the precursor protein may also be involved in these familial AA amyloidoses. 14, 27 The cheetah AA protein sequence has amino acid variations similar to those found in the Abyssinian cat (compared with domestic short-haired cats), which, in combination with increased production secondary to chronic inflammation and chronic stress, might predispose the cheetah to systemic amyloidosis. 13, 24 The SAA protein sequence has not yet been determined for black-footed cats.
In this study, heritability estimation suggested that amyloidosis in black-footed cats might be an inherited trait. However, this calculation is influenced by inbreeding and small sample sizes, both of which are present in the study population. For the animals considered in this sample, 26 were inbred. Among the inbred animals, the average inbreeding coefficient was .226 (with a range among the nonzero values from .059 to .3125). Cats available for this study were those submitted to the Association of Zoos and Aquarium Black-footed Cat Species Survival Plan (SSP) pathologist (KAT) for evaluation by collaborating zoos and from colleagues working with the European and South African captive populations. Because of concern about amyloidosis in the captive population, SSP submissions may be biased toward cases of amyloidosis; thus, the cats in this study represent a nonrandom sampling of all black-footed cat postmortem examinations.
The free-ranging black-footed cat with amyloidosis was part of a field study on the ecology and behavior of these cats conducted between 1992 and 1998 close to Kimberley, central South Africa. 22, 23 Although only a juvenile, there was a small amount of amyloid present in the splenic follicles. No renal amyloid was noted. Amyloid in this case stained with CR but was negative by immunohistochemistry. Failure of immunolabeling might have been an artifact of sectioning in that only a small amount of amyloid was deposited in this case. The presence of amyloid in a free-ranging black-footed cat provides additional evidence for a species predilection and supports the existence of a possible familial type of amyloidosis in the black-footed cat.
Footnotes
Acknowledgements
The authors thank Gea Olbricht, Drs. Alex Sliwa and Lorna Bolton, and the following institutions for submitting material: Central Florida Zoo, Sanford, FL; Cincinnati Zoo & Botanical Garden, Cincinnati, OH; Happy Hollow Zoo, San Jose, CA; Johannesburg Zoo, Johannesburg, South Africa; The Living Desert Palm Desert, CA; Riverbanks Zoo and Garden, Columbia, SC; Sacramento Zoo, Sacramento, CA; San Antonio Zoo, San Antonio, TX; Zoo of Wuppertal, Wuppertal, Germany; and Zoological Society of San Diego, San Diego, CA. We also thank Drs. Sliwa and Jennifer Landolfi for critical review of this manuscript. This study was funded by a grant from the American Association of Zoo Veterinarians Mazuri Fund.
