Abstract
We describe the main pathologic changes in small ruminants affected by AA amyloidosis, together with the partial sequence of the protein involved. Twenty-one sheep and one goat were selected for presenting macroscopic kidney lesions compatible with systemic amyloidosis. Available tissue samples were studied by histologic, immunopathologic, and ultrastructural means. Renal lesions were characterized grossly by pale cortical surfaces with scattered, miliary, whitish-yellow foci and on cut cortical surfaces by straight, whitish-yellow striations. Gangrenous pneumonia was observed in 16 out of 21 affected sheep (76.2%), although other chronic inflammations were also observed. Amyloid was detected in all grossly affected kidneys using Congo red staining, lesions being most remarkable in glomeruli, affecting 95.5% of animals studied. Congophilic deposits were also observed in intertubular interstitium (68.2%) and medulla (57.1%). All amyloid-affected animals presented proximal convoluted tubule lesions, mostly characterized by an increase in diameter and by hyaline granular degeneration that were responsible for the macroscopic appearance of the kidney. Histologically, amyloid was also seen in blood vessels, spleen, liver, lymph nodes, gastrointestinal tract, and adrenal glands. All amyloid deposits demonstrated greenish-yellow birefringence with polarized light, and the antisera prepared against goat amyloid extracts specifically reacted with birefringent congophilic deposits of both sheep and goats. Ultrastructurally, these deposits were formed by masses of straight, nonbranching fibrils located predominantly in the basement membranes of glomerular capillaries and in the mesangium. Partial sequence of the protein in sheep and goats indicated a high degree of homology with the previously reported sequence of sheep Serum Amyloid A.
Amyloidosis is a general term describing a group of diseases characterized by the pathologic deposition of fibrillar protein in various tissues that is seen as hyaline aggregates by hematoxylin and eosin (HE) staining. These deposits, called amyloid, regardless of their chemical nature, show affinity for Congo red (CR), revealing orange-colored staining, and they show green birefringence when CR-positive areas are viewed in cross-polarized light. 5 Ultrastructurally, this material is composed of characteristic nonbranching, straight, approximately 10-nm-thick fibrils of indeterminate length. Amyloid fibrils have a high content of beta-pleated sheet secondary structure, as shown by X-ray diffraction and infrared spectroscopy. 41
In human pathology, at least 20 different proteins are known to have the ability to aggregate, insolubilize, and deposit in tissue as amyloid. 25 The AL amyloid type, which is derived from immunoglobulin light chains, is the most common form in humans. 29 In animals, at least eight different amyloid precursors have been described, and the AA form of amyloid is the one that is most frequently found. 15,22,48 AA amyloid is derived from an acute-phase protein called serum amyloid A (SAA) that normally plays a role in cholesterol transport 19 and as a chemoattractant in the inflammatory process. 2 When the concentration of this molecule is increased, typically as a result of chronic inflammation, certain isoforms of SAA are partially cleaved into fragments that have an increased propensity to form fibrillar aggregates (amyloid) that are deposited systemically, mainly in the kidney, liver, and spleen. 15
AA amyloidosis occurs in a wide variety of domestic animal species including canine, equine, bovine, avian, and less frequently porcine and feline. 4,7,12,13,20,36,48 This type of amyloidosis has also been documented in various wild mammalian and avian species such as cheetah (Acinonyx jubatus), 16,24 Siberian tigers (Panthera tigris altaica), 35 mink (Mustela vison), 27 Dorcas gazelle (Gazella dorcas), 32 mountain gazelle (Gazella gazella), 21 and anatidae. 34 Among small ruminants, AA amyloidosis has been reported in wild bighorn sheep (Ovis canadensis) 11 and Dall sheep (Ovis dalli); 18 however, descriptions in domestic sheep and goats are rare and normally refer to single cases. 6,9,15,33,39 Remarkably, systemic amyloidosis in small ruminants has been diagnosed at the Veterinary Faculty of Zaragoza for decades, and it is relatively common. The objectives of the present study are to: 1) describe the salient pathologic features of spontaneous, systemic AA amyloidosis in domestic sheep and goats, and 2) establish immunohistochemically and chemically the AA nature of the amyloid deposits. The study included cases observed at routine necropsy and cases collected prospectively from an abattoir during a 5-year period.
Materials and Methods
Animals and samples
Tissue specimens from 28 sheep and two goats were studied. Samples were classified in six groups, and details of the animals studied are shown in Table 1. Sheep in groups I (Nos. 1–15) and II (Nos. 16–21) were selected for the presence of macroscopic renal lesions compatible with AA amyloidosis (see results). For group I, the available samples included only kidney, lungs, heart, and sometimes liver and spleen. Sheep in group III (Nos. 22–26) had lesions of chronic interstitial nephritis or tubulonephrosis, or both, which were macroscopically similar to AA amyloid–induced lesions. Group IV consisted of two control, healthy, adult, female Rasa Aragonesa sheep (Nos. 27–28). Group V included one goat (No. 29) that presented renal lesions that were macroscopically compatible with amyloidosis, and group VI consisted of one control goat (No. 30).
Clinical and pathological data on the 30 animals included in this study.
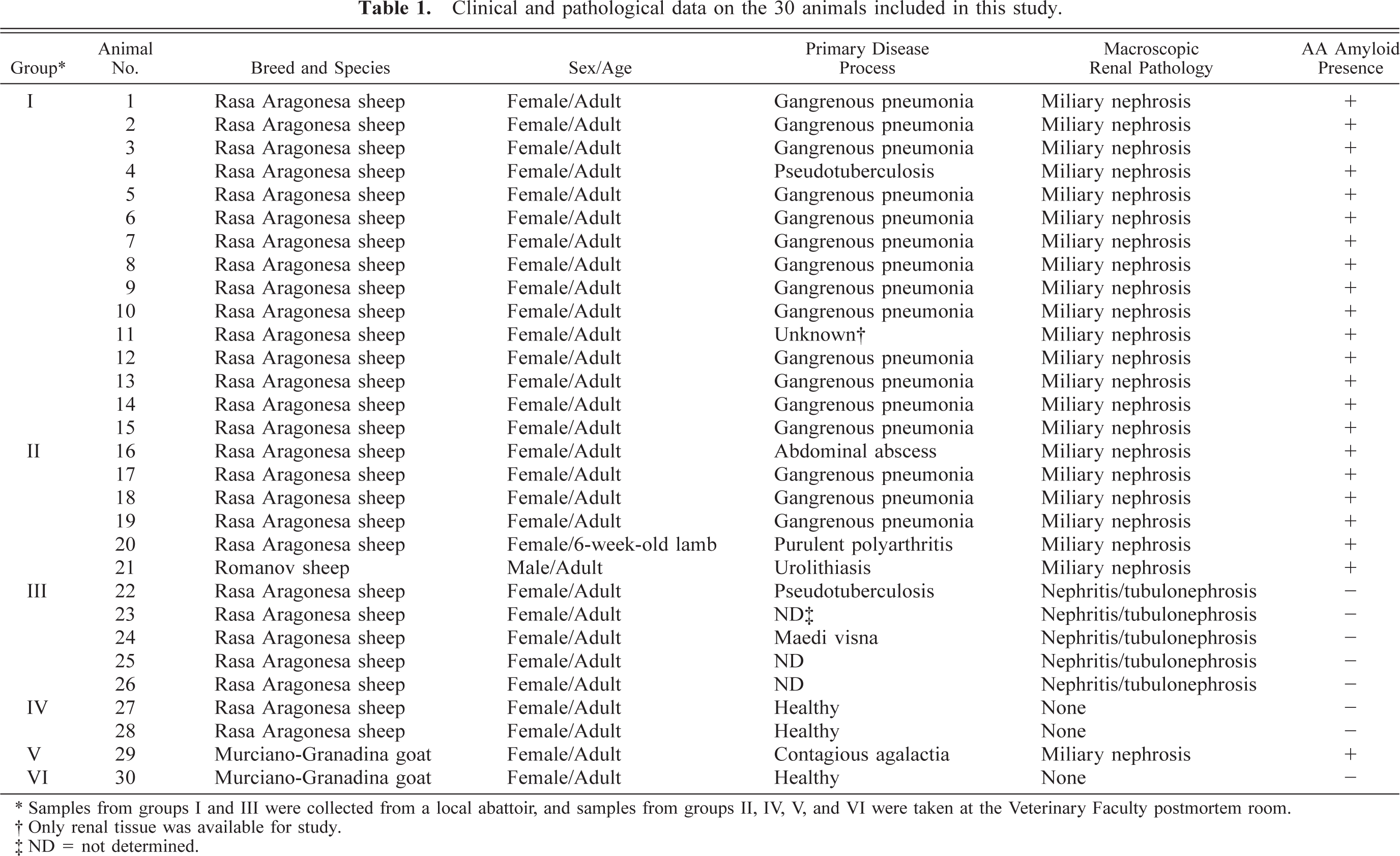
∗ Samples from groups I and III were collected from a local abattoir, and samples from groups II, IV, V, and VI were taken at the Veterinary Faculty postmortem room.
† Only renal tissue was available for study.
‡ ND = not determined.
Histopathologic and electron microscopy studies
Samples were fixed in 10% buffered formalin, dehydrated, and embedded in paraffin wax. Four-micrometer-thick serial sections were stained with HE, periodic acid–schiff (PAS), and CR. All CR-stained sections were viewed in cross-polarized light, and the resistance of all CR-treated kidneys to destaining by potassium permanganate was determined as described previously. 46 Additionally, kidney samples of sheep Nos. 2–5 were fixed by immersion in 2.5% glutaraldehyde, postfixed in 1% OsO4, dehydrated through a graded series of alcohols to propylenoxid, and embedded in Epon 812® (Fluka Chemie AG, Buchs, Switzerland). Fifty-nanometer-thick sections were stained with uranyl acetate and lead citrate and examined under a Philips CM-10 transmission electron microscope.
Amyloid extraction and characterization
Amyloid fibrils were extracted according to standard methods 31 and isolated by reverse-phase high-performance liquid chromatography (HPLC) 8 from approximately 1 gm portions of sheep and goat kidneys (Nos. 6 and 29, respectively) obtained at necropsy and maintained at −80 C. The HPLC-purified proteins were reduced, pyridylethylated, and cleaved enzymatically with trypsin (Worthington Biochemical, Freehold, NJ) at an enzyme to substrate ratio of 1:100 (wt/wt), as described previously. 8 The resultant tryptic peptides were separated by HPLC and their amino acid sequences determined using an ABI Model 494 Procise gas-phase sequencer (Applied Biosystems, Foster City, CA).
Immunohistopathologic studies
Antisera to fibrils extracted from the amyloid-laden goat kidney (No. 29) were prepared by immunizing a rabbit following standard methods 30 and obtaining preimmune and postimmune sera. For immunohistochemistry (IHC), the polyclonal antibody was used at a dilution 1:500, and the reaction was developed using a commercially available streptavidin–biotin–peroxidase complex kit with 3-amino-9-ethylcarbazole as substrate (LSAB®, DAKO, Glostrup, Denmark). Positive control sections were obtained from kidneys of sheep No. 16 that showed a severe and disseminated AA amyloidosis and that was strongly positive for immunolabeling. Two positive control sections from sheep No. 16 were tested in all experiments, one with preimmune sera and the other with postimmune sera. Negative control sections were obtained from kidneys of animals showing other, non-AA–related renal lesions (group 3) or from control ewes (group 4). All these sections were tested with the postimmune rabbit sera only. Additional negative controls were provided by substitution of primary polyclonal antibody for normal rabbit serum or phosphate-buffered saline (PBS) on amyloid-laden sections. Presence of amyloid in samples either by CR staining or by IHC was estimated using a semiquantitative approach where deposits were classified as 0 (absent), + (trace), ++ (moderate), and +++ (severe).
Results
Macroscopic findings
The kidneys of the 22 animals comprising groups I, II, and V, although typical in size (7–9 cm as longitudinal diameter, except for sheep No. 13, where it measured 13 cm), appeared grossly abnormal and were characterized by a pale, smooth renal surface that notably contained scattered, miliary whitish-yellow foci (Fig. 1). The extent of these foci varied in amount from only a few scattered throughout the parenchyma to numerous and sometimes coalescing, giving the kidney a whitish homogeneous appearance. When the kidneys were sectioned, similar miliary foci, as well as straight whitish-yellow striations, were found within the cortex (Fig. 2). No obvious lesions were observed in the renal medulla, except in two cases (sheep Nos. 5 and 16), in which it was very pale (almost white) due to massive amyloid deposition, as evidenced afterward microscopically. No macroscopic lesions suggestive of amyloid deposition were observed in other organs, e.g., adrenal glands, liver, spleen, or gastrointestinal tract.
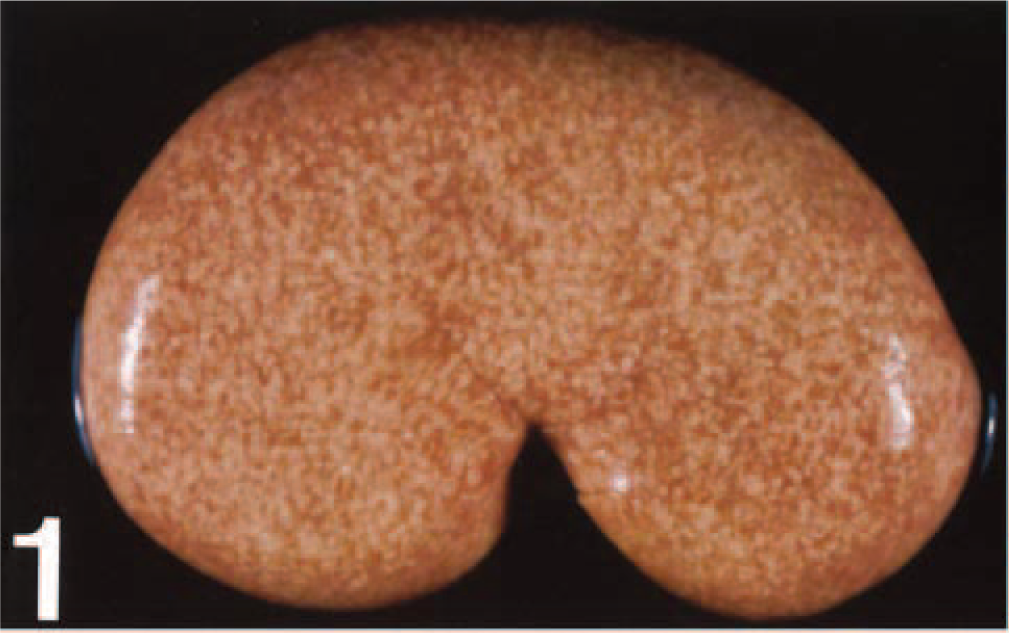
Kidney; sheep No. 2. Severely affected animal. Pale renal surface with presence of numerous, homogeneously distributed miliary yellowish foci.

Kidney; sheep No. 5. Sectioned renal cortex showing numerous whitish-yellow foci and striations. This gross appearance corresponds to a severe nephrosis affecting the convoluted proximal tubules.
Sixteen of the 21 affected sheep (76.2%) had gangrenous pneumonia that consisted of intraparenchymal areas of gangrenous necrosis with accumulation of soft, pale, necrotic, foul-smelling exudate. Remarkably, foreign bodies were not detected in the necrotic content or in the airways. Other lesions noted in the rest of the cases (n = 5) included pulmonary pseudotuberculosis (sheep No. 4), a large intra-abdominal abscess of undetermined origin (sheep No. 16), purulent polyarthritis (sheep No. 20), and urolithiasis (sheep No. 21). In the case of sheep No. 11, only renal tissue was available for study. The affected goat (No. 29) showed lesions compatible with contagious caprine agalactia, (e.g., chronic arthritis, mastitis, and keratitis), and Mycoplasma spp. was isolated from this animal.
Macroscopic lesions found in kidneys of animals of group III (sheep Nos. 22–26) were classified as chronic interstitial nephritis, or tubulonephrosis, or both. Additionally, sheep No. 22 had pseudotuberculosis lesions in multiple lymph nodes, and sheep No. 24 showed interstitial pneumonia compatible with maedi-visna infection. Macroscopic lesions were not observed in animals from groups IV and VI (sheep Nos. 27 and 28, and goat No. 30).
Histopathology
Microscopically, the presence of amyloid deposits was detected in kidney and other organs and structures. Irrespective of its location, amyloid was observed, by HE, as extracellular eosinophilic deposits that compressed surrounding tissues and that were constantly CR-positive and PAS-negative.
Regardless of species, breed, or sex, the anatomical pattern of amyloid deposition within the kidney was similar and differed only in severity. Lesions were most notable in the glomeruli (Table 2), where amyloid was present in capillary walls and mesangium in 21 of 22 sheep (95.5%).
Location and severity of renal AA amyloid deposition and presence of proximal convoluted tubulonephrosis in 22 small ruminants.

∗ Total number of animals with amyloid located in the specified structure/total amyloid-affected animals.
† Total number of animals affected by cortical tubulonephrosis/ total amyloid-affected animals.
Amyloid was initially observed as segmental, eosinophilic, CR-positive deposits that usually became global and occupied the entire glomerular tuft, compressing and distorting the normal architecture (Fig. 3). Although amyloid deposits were hypocellular, a periglomerular mononuclear cell interstitial infiltrate was occasionally noted around the most severely damaged glomeruli. Deposits were not observed in the cortical interstitium by HE, but when CR and IHC techniques were used, 15 animals (68.2%) demonstrated amyloid deposition in the intertubular interstitium. In 12 animals (55.1%), the renal medulla was affected multifocally by amyloid deposits that were located around the vasa recta and the venous plexuses, with atrophy and disappearance of adjacent collecting tubules and loops of Henle (Fig. 4). The CR staining within these areas was most intense in the vicinity of blood vessels. In 20 animals (91.0%), congophilic material also was found in the tunica intima and the tunica media of the arcuate arteries. These deposits, not detected by HE, were severe in only two cases (sheep Nos. 11 and 16). Proximal convoluted tubule lesions were variable in severity and were characterized by a significant increase in tubular diameter, epithelial hypertrophy, intracytoplasmic hyaline granular degeneration of tubular epithelial cells, and the presence of a homogeneous eosinophilic proteinaceous fluid within the lumen (hyaline casts). These tubular lesions were found in all animals that had renal amyloid deposition (Fig. 5). However, proximal tubules, intracytoplasmic hyaline, and casts were consistently CR-negative.

Kidney; sheep No. 9. Diffuse congophilic, amyloid deposit in a glomerulus that shows hypocellularity, increase in size, and pressure atrophy of normal structures. Note the congophilic arteriole. Congo red. Bar = 56 μm.

Kidney; sheep No. 10. Amyloid deposit in the renal medulla causing pressure atrophy of normal structures. Rabbit polyclonal antibody against goat amyloid A protein. Streptavidin–biotin–peroxidase complex method, AEC chromogen, Mayer's hematoxylin counterstain. Bar = 26 μm.

Kidney; sheep No. 3. Proximal convoluted tubule showing increase in diameter, epithelial hypertrophy, intracytoplasmic hyaline granular degeneration, and homogeneous eosinophilic proteinaceous fluid within the lumen. HE. Bar = 33 μm.
Although no amyloid-related macroscopic lesions were observed in organs other than the kidney, the presence of amyloid was demonstrated microscopically in many organs and tissues by CR staining (Table 3). In 16 of 18 animals (88.8%), the amyloid was present in the tunica media of extrarenal vessels (Fig. 6). Further, amyloid was present within the splenic germinal centers in seven of nine cases (77.7%), forming an “amyloid ring” around the lymphoid follicles. In two instances (sheep No. 16 and goat No. 29), severe amyloid deposits also involved the wall of the venous sinus (red pulp). Nine of 16 animals (56.3%) had hepatic amyloid deposits located in the space of Disse; in one animal (sheep No. 7), this was severe enough to cause partial pressure atrophy of the surrounding hepatocytes. Four of 11 cases (36.7%) were found to have small amounts of amyloid deposited in lymph node subcapsular sinuses and follicles. The gastrointestinal tract was examined in only four animals; in two animals (50.0%), amyloid deposits were observed in the entire digestive tract including the esophagus, the forestomachs, the small and large intestines, and the rectum. In one case (sheep No. 16), this process was most extensive in the duodenal lamina propria, submucosa, and Peyer's patches. Amyloid deposition was also present in the adrenal cortex in four of seven animals (57.1%). With the exception of the vascular congophilic material, the lung and heart were not affected, andamyloid could not be detected in the central nervous system.
Location and severity of extrarenal amyloid deposits among 21 small ruminants naturally affected by AA amyloidosis.∗

∗ Tissue available for 21 of the 22 animals with AA amyloidosis in the kidney.
† Total number of animals with amyloid deposits/total number of samples.

Duodenum; sheep No. 16. Greenish-yellow birefringence located within the tunica media of submucosal vessels due to the presence of amyloid. Viewed in cross-polarized light. Congo red. Bar = 12 μm.
All CR-positive areas, irrespective of their location, showed greenish-yellow birefringence of variable intensity when observed under polarized light (Fig. 6). Remarkably, a constant staining in both affected and control animals was observed using CR in the intima of arteries and arterioles. This staining was negative for birefringence; therefore, it was not considered to be amyloid. Treatment with potassium permanganate abolished the congophilia in all cases. Tissue samples from groups III, IV, and VI were free of amyloid deposits.
Immunohistochemistry
The antisera prepared against the goat amyloid extracts specifically immunolabeled the green birefringent congophilic deposits found in all animals. Notably, in many instances, the anti-AA antisera reacted with the noncongophilic filtrate within the Bowman's capsule, as well as with the plasma within blood vessels and the tunica media of blood vessels of all organs. Controls in which primary antiserum was replaced with preimmune rabbit serum or PBS were consistently negative. In addition, staining was not observed when the postimmune rabbit polyclonal serum to goat AA was used on control sections. The amyloid deposits in the spleen, liver, adrenal glands, lymph nodes, and digestive tract, recognized on the basis of CR staining, demonstrated positive immunohistochemical staining using antiserum to goat AA. No positive reactions were observed in tissue samples from animals of groups III, IV, and VI.
Electron microscopy
Renal amyloid deposits were shown by electron microscopy to be composed of masses of fibrils located predominantly in the basement membranes of glomerular capillaries and mesangium (Fig. 7). Less frequently, this material also was present in similar areas in interstitial capillaries and, occasionally, around tubules. In the case of glomeruli, the amyloid deposits always were located subendothelially (Fig. 8) and partially occluded the capillary lumen. Additionally, thickening of glomerular capillary basement membranes, slight edema of podocytes, and foot process fusion were noted (Figs. 7, 8).

Kidney; sheep No. 5. Fibrillar amyloid deposit (asterisk) in a glomerular capillary. Note the slight edema of podocytes and foot process fusion (arrow). Bar = 0.4 μm.
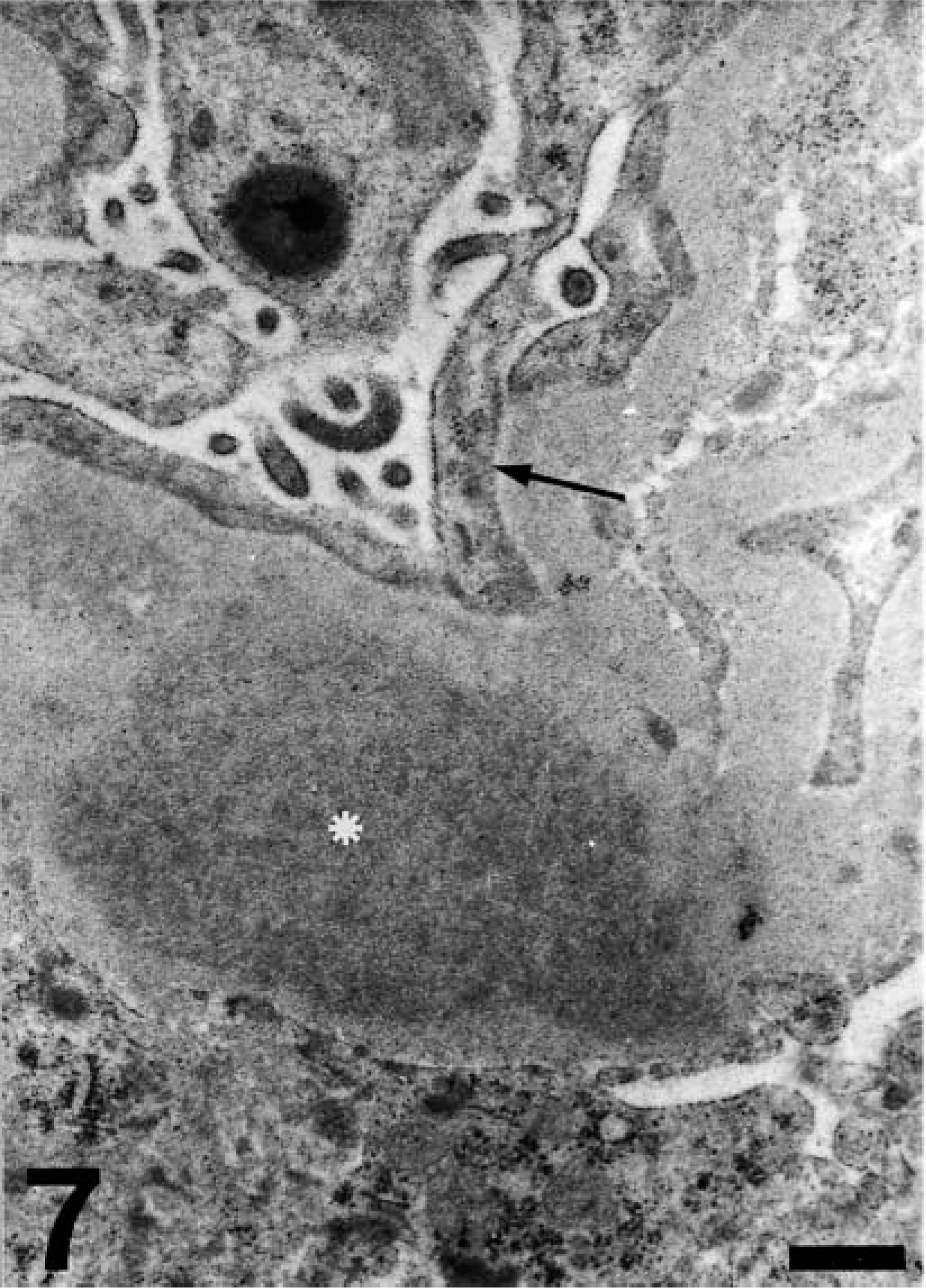
Kidney; sheep No. 4. Subendothelial amyloid deposits (asterisk) in a thickened basal membrane of a glomerular capillary. Note the partial occlusion of the capillary lumen and the foot process fusion of podocytes (arrow). Bar = 0.9 μm.
Characterization of sheep and goat amyloid
Amyloid was extracted from the kidneys of sheep No. 6 and goat No. 29 that contained large amounts of green birefringent, potassium permanganate–sensitive, congophilic deposits that immunostained with the anti-AA antisera. The protein extracts were purified by reverse-phase HPLC where, in both instances, several well-defined peaks were evident. These materials were subjected to direct amino acid sequence (automated) analyses, but no sequence was obtained that was consistent with a “blocked” N-terminal residue, i.e., pyroglutamate—a cyclic form of glutamine found among AA proteins of other species. 14 However, when the major component was digested with trypsin, several peptides were obtained that could be sequenced directly. In the case of the sheep-derived material, two such peptides contained residues that were identical to those found at positions 47–58 and 62–71 of sheep SAA. 38 In the case of the goat, the sequences of two tryptic peptides were found to match positions 15–24 and 30–54 of sheep SAA (at position 55, the goat AA contained lysine rather than glutamic acid) (Fig. 9).

Comparison of the amino acid sequence of the sheep SAA with those of tryptic peptide fragments of amyloid extracted from the kidneys of a sheep and goat with AA amyloidosis.
Discussion
In this article, a detailed pathologic study on AA amyloidosis in small ruminants is presented. We have demonstrated that the amyloid deposits found in the animals studied were composed of SAA-derived protein and were therefore classified as AA amyloid. Microscopically, the amyloid material showed positive CR staining and birefringence under cross-polarized light, but when these deposits were treated with potassium permanganate, staining was removed. In addition, immunohistochemical analyses using polyclonal antisera developed against fibrils extracted from the kidneys of one such animal reacted specifically with amyloid deposits in sheep and goats that were initially identified on the basis of congophilia. Further, chemical studies of protein extracted from the kidneys of an affected sheep and goat substantiated the AA nature of the amyloid. Although the complete primary structures of these proteins were not determined, it was evident that the amino acid sequences of peptides obtained by trypsin digestion of the sheep amyloid extract were identical to those of the corresponding regions of sheep SAA. Thirty-five of the 36 residues of the goat amyloid also were the same as those of the corresponding regions of the amyloidogenic precursor protein.
To the best of our knowledge this is the first comprehensive pathologic study on AA amyloidosis in domestic small ruminants. AA amyloidosis has been previously reported incidentally in domestic sheep and goats. 33,39 Amyloidosis was also seen in a goat infected by caprine arthritis encephalitis virus, although no detailed description was provided. 6 Some sheep that were experimentally inoculated to obtain hyperimmune sera and a goat that suffered numerous protein inoculations also developed amyloidosis. 9,13 Remarkably, the only detailed study on reactive amyloidosis in small ruminants was done in wild bighorn sheep and in captive Dall sheep, where a pathologic condition similar to the one described in this study was presented. 11,18,45
We found in our sheep and goat that, as previously observed in these species as well as in cows, 16,18,26,33,39 bighorn sheep, 11 and Dorcas gazelles, 32 the organ most involved was the kidney. The kidney was the only organ harboring macroscopic lesions that consisted of miliary foci on the cortical surface of the kidney and, on cut surfaces, radial striations located only in the renal cortex. We attribute these features to a resorptive, proteinaceous tubulonephrosis that affected the convoluted proximal tubules and occurred as a consequence of protein loss through impaired glomeruli by the deposition of amyloid. Therefore, the macroscopic picture did not directly reflect the amount of renal amyloid but merely represented the degree of proximal convoluted tubulonephrosis (notably, the tubular epithelial cells were not congophilic). A similar conclusion had been made in the case of bovine renal AA amyloidosis. 10,26
Remarkably, our animals exhibited no gross amyloid-related abnormalities in organs besides the kidney. This finding is in contrast to the pattern of AA deposition in other species of small ruminants, as well as horses and experimental rodents, where amyloid was found to be most pronounced in spleen, liver, adrenal glands, and gastrointestinal tract. 9,11,22,40,45,47 This difference in tissue distribution of AA amyloid among animal species suggests that particular host factors, e.g., accessory molecules such as proteoglycans that codeposit with the amyloidogenic protein, 37 may be involved in this phenomenon. Alternatively, the presence of multiple, SAA isoforms in sheep and goats, such as found in mice, cows, and humans, may account for organ selectivity. 1,43
Microscopically, we found that the amyloid deposits in sheep and goat were predominantly located within the glomeruli and, to a lesser degree, the medulla, as found in dogs and occasionally in cattle. 7,16,26 However, AA amyloid deposits were observed almost exclusively in the medulla of Dorcas gazelles, 32 and others authors have found it most frequently in the renal medulla of cows. 10 Initially, the amyloid in our animals was localized to the glomerular capillary subendothelium, as also seen in the cow, 28 but as the disease progressed, involved additional areas of the kidney. Ultrastructurally, the lesions were similar to those described in other species and included thickening of the basement membrane and fusion of podocyte foot processes. 23,32 Only in the most advanced cases, with the glomerulus being in a terminal stage, was a mononuclear cell interstitial infiltrate noted.
AA amyloidosis characteristically develops in humans and animals as a consequence of a long-standing, chronic infectious or inflammatory process. Most of our affected sheep had gangrenous pneumonia, an acute process that normally became chronic in sheep (L. Luján, personal observation). However, other animals in the study were free of this type of pneumonia and were affected by other primary inflammatory foci such as pseudotuberculosis or abscesses. These results are concordant with causes previously studied for domestic and wild small ruminants, where chronic suppurative pneumonias, Corynebacterium pseudotuberculosis, and Actinomyces pyogenes, were the primary inflammatory diseases. 11,18,32,39,45 Gangrenous pneumonia typically develops when contaminated foreign bodies are introduced into the airways, 17 but we were unable to find any recognizable foreign material within the lesions. However, over the years, we have found several cases with vegetal fragments in the bronchi causing pulmonary gangrene and amyloidosis. This process seems to be relatively high in adult sheep of our geographic area. In countries where such pneumonias are not important, the appearance of AA amyloidosis is likely to be minimal.
Another amyloid form described in ruminants is APrPsc, the amyloid detected in animals suffering from transmissible spongiform encephalopathies. This form of amyloidosis is observed in scrapie of sheep and goats and also in a few cases of bovine spongiform encephalopathy. 3,42,44 In our study, no CR-positive areas were seen in the nervous system parenchyma thus indicating that these two amyloid forms seem to be unrelated.
Further investigations into the different SAA isoforms, their involvement in amyloidogenesis, and the experimental reproduction of the gangrenous pneumonia and systemic AA amyloidosis in sheep are currently being performed.
Footnotes
Acknowledgements
We thank the veterinarians and workers of the local slaughterhouse (Mercazaragoza) for their help in obtaining samples. We are indebted to Prof. Erik Gruys (University of Utrecht) for reviewing in depth the manuscript and for giving many useful suggestions. We are also very thankful to Dr. Lourdes Sánchez for assistance in preparing the polyclonal anti-AA antibody and to Santiago Becerra for technical help at the postmortem room. This work was supported in part by a grant (218-130) from the University of Zaragoza and USPHS grant CA10056 from the National Cancer Institute. A. Solomon is an American Cancer Society Clinical Research Professor.
