Abstract
An ovarian mucinous cystadenoma was found in a 5-year-old female cynomolgus monkey (
Surface epithelial–stromal-tumor–containing mucinous cystadenoma is the most common ovarian tumor in humans.
3,
4
This tumor is characterized by proliferation of the epithelial component that forms glands or cysts with various amounts of the stromal component. The epithelial components are derived from the ovarian surface epithelium, and the tumor is regarded as mucinous (cyst)adenoma if the epithelial cells contain abundant intracytoplasmic mucin and resemble endocervix or intestine.
4
However, evidence concerning the origin of surface epithelium is less obvious for the mucinous type than for the other types of surface epithelial tumor, and mucinous metaplasia of the surface epithelium or mucinous surface epithelial inclusion cysts are encountered only rarely.
4
Spontaneous ovarian serous or mucinous cystadenoma in nonhuman primates have been reported in rhesus monkeys (
The present case was found in a 5-year-old female cynomolgus monkey, which was bred in and imported from Mauritius, and was being used for the control group in a toxicologic study, but did not show any abnormalities in clinical signs, hematological, or serum biochemical analyses. The animal was euthanized by exsanguination under pentobarbital anesthesia and necropsied. After the necropsy, all of the tissues were fixed in phosphate buffered 10% formalin solution and were processed using routine methods. The ovarian tumor was sectioned serially at 4 μm. Representative sections were stained with hematoxylin and eosin, alcian blue–periodic acid–schiff (AB-PAS), Masson's trichrome, silver stain, or Grimelius' stain. The other sections were allowed to react immunohistochemically with α-smooth muscle actin (α-SMA), proliferating cell nuclear antigen (PCNA), or cytokeratin (CK, AE1/AE3) using LSAB kit (DAKO Japan Co., Kyoto, Japan). Sections of the normal ovary and uterus from another animal were used for comparison. Small pieces of the formalin-fixed ovarian tissue were processed for the ultrastructure examination.
At the necropsy, the right ovary was swollen and measured 1 cm in diameter. The external ovarian surface was smooth with a milky-white color. There was no adhesion seen between the ovarian tumor and adjacent organs. The cut surface of the ovarian tumor revealed a multilocular cyst filled with mucinous fluid. The normal ovarian tissue was pushed aside by the multilocular cystic tumor that filled approximately 80% of the ovary. The left ovary and other organs of this animal were all normal.
Light-microscopic examination of the right ovary revealed an ovarian tumor, which was characterized by various sizes of multilocular cystic glands (Fig. 1). The cystic glands were lined by a single layer of flattened to tall columnar mucin filled epithelium with basal nuclei reminiscent of endocervical epithelium (Fig. 2). Alcian blue–positive material was present in the mucinous epithelium and lumen. The luminal surface was often ciliated. Grimelius' positive stained cells regarded as Paneth cells were not contained in the mucinous epithelium. Malignant features such as nuclear pleomorphism, mitotic figure, stratification, and papillary proliferation were not found in the glandular epithelium. The cystic glands were surrounded by a large amount of solid fibrous stroma consisting of interlacing fascicles of spindle cells with elongated nuclei. The fascicles of stromal cells paralleled to the linings of the cysts, especially in the vicinity of the tumor epithelium. These stromal elements resembled smooth muscle cells (Fig. 3). Masson's trichrome staining showed a small amount of collagen fiber and abundant muscle fiber in the stroma (Fig. 4). Argyrophilic fibers separated the individual stromal cells according to the silver staining. The results of the Masson's trichrome and silver stainings suggested that the stromal elements were smooth muscle cells. The smooth muscle cells in the stroma did not have any malignant features either. Because of the pressure of the epithelial and stromal tumor element, normal ovarian components such as ovarian follicles and corpus luteum remained only on the edge of the ovary. There was no clear boundary between the normal tissue and tumor. The ovarian surface epithelium consisted of a single layer of cuboidal to columnar cells with focal pseudoglandular appearances. The surface epithelium was partially invaginated into the ovarian cortex and led to the mucinous tumor cyst. The invaginated surface epithelial cells passed through the tunica albuginea and transformed into cells with mucin-filled cytoplasm at the outer ovarian cortex. There was an obvious region of transition from the invaginated surface epithelium into mucinous cyst-forming epithelium (Fig. 5). Immunohistochemically, the glandular epithelium of the tumor and the ovarian surface epithelium were positive for CK (AE1/AE3). Almost all stromal spindle cells were positive for α-SMA (Fig. 6), and some of them were positive for PCNA.
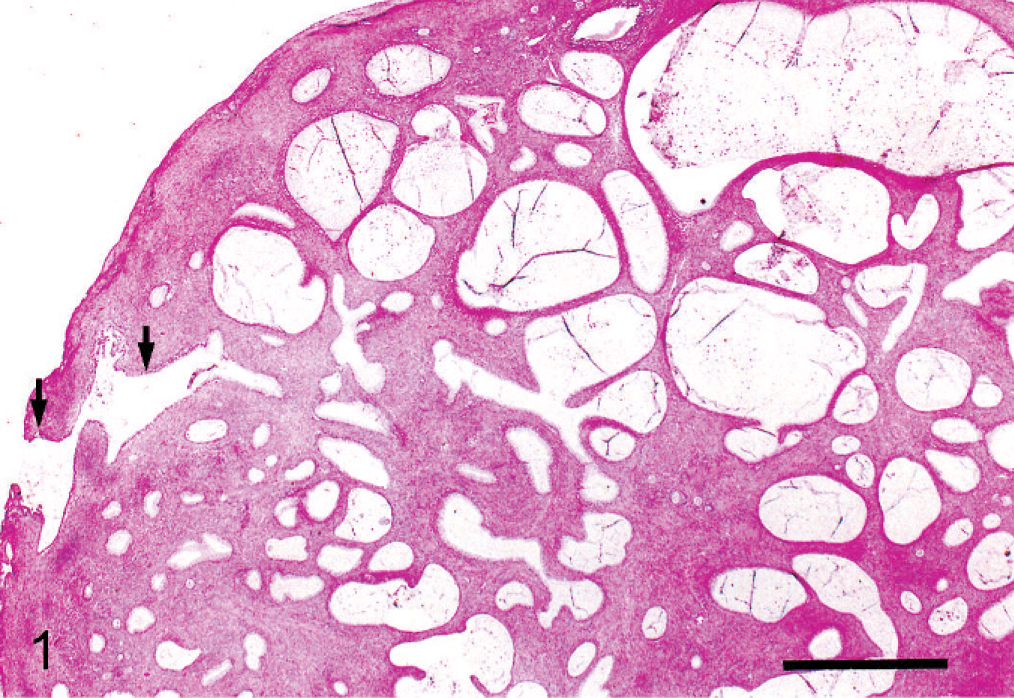
Mucinous cystadenoma, ovary; cynomolgus monkey. Tumor characterized by mucin-filled multilocular cystic glands surrounded by stroma. The invaginated ovarian surface epithelium leads to the mucinous tumor cyst (arrows). HE. Bar = 0.1 cm.
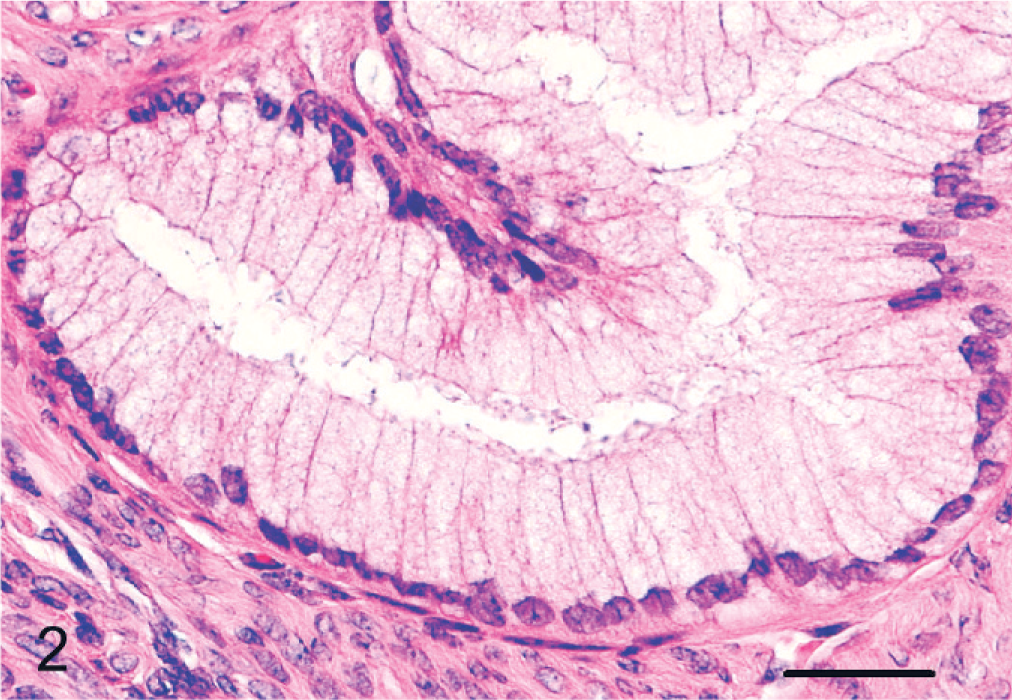
Mucinous cystadenoma, ovary; cynomolgus monkey. High magnification depicted in Fig. 1, showing a mucin-filled epithelium with basal nuclei, reminiscent of endocervical epithelium. HE. Bar = 50 μm.
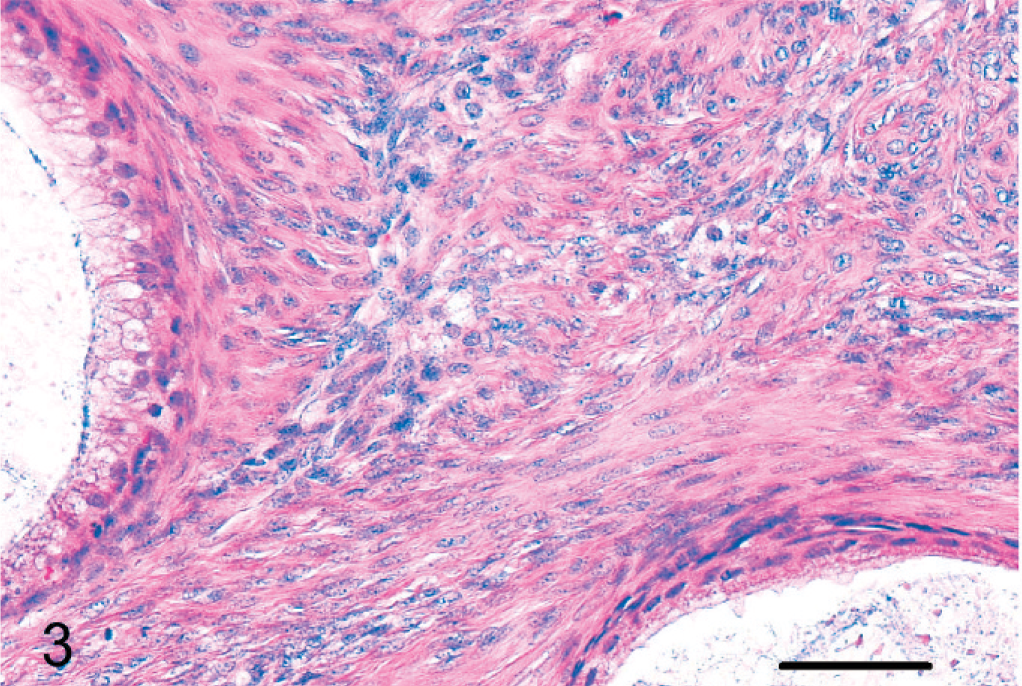
Mucinous cystadenoma, ovary; cynomolgus monkey. The tumor stroma consisted of interlacing fascicles of spindle cells. HE. Bar = 100 μm.
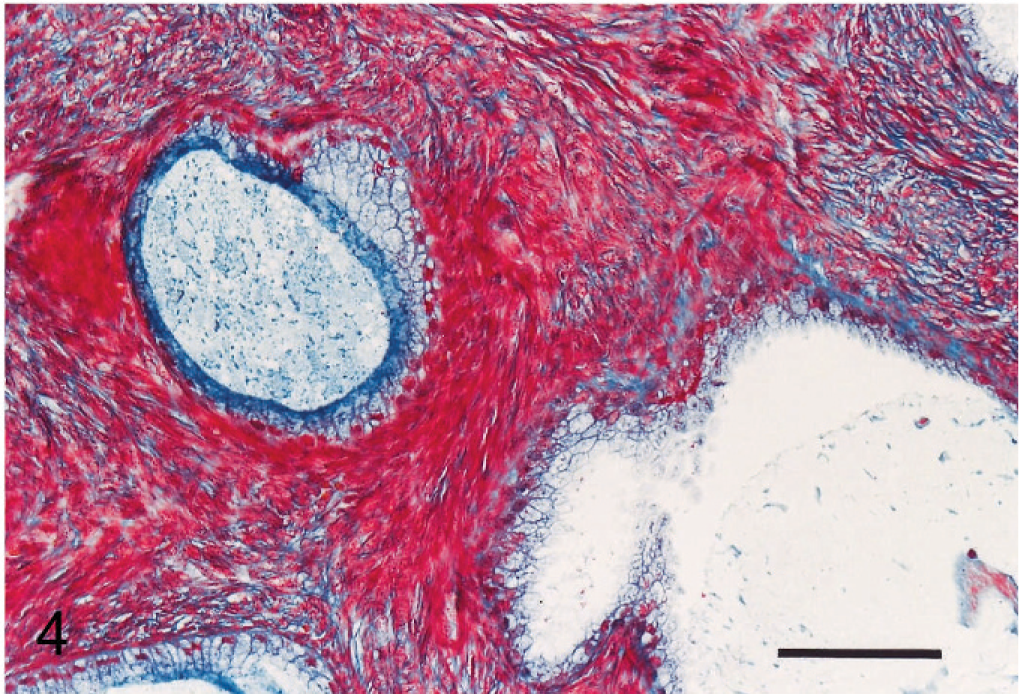
Mucinous cystadenoma, ovary; cynomolgus monkey. The interlacing fascicular stroma shows red staining by Masson's trichrome stain, suggesting muscle fiber. Masson's trichrome stain. Bar = 100 μm.
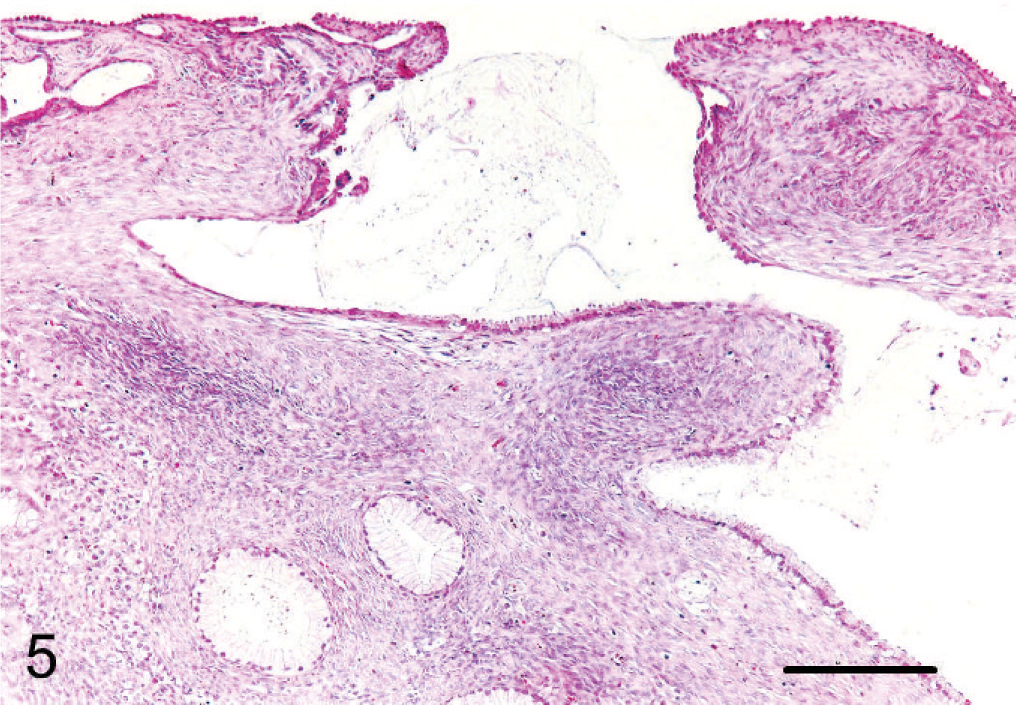
Mucinous cystadenoma, ovary; cynomolgus monkey. Notice transition of surface epithelium from a normal simple cuboidal epithelium to simple columnar mucinous epithelium. HE. Bar = 200 μm.
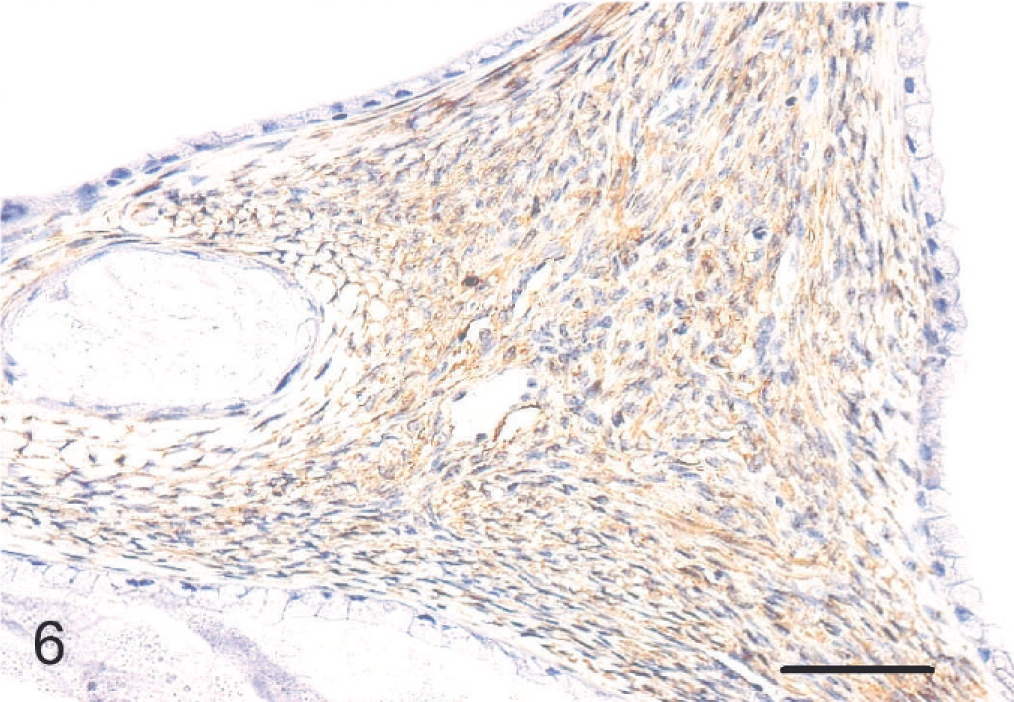
Mucinous cystadenoma, ovary; cynomolgus monkey. The stromal area showing positive reaction for α-SMA. Immunoperoxidase stain, counterstain with Mayer's hematoxylin. Bar = 100 μm.
Ultrastructurally, the glandular epithelial cells contained oval basal nuclei, numerous dark secretory granules in the cytoplasm, and microvilli. They were separated from the underlying stroma by basal lamina and collagen fibrils. The stromal cells were characterized by elongated spindle-shaped cytoplasm with invaginated nuclei and were separated by adjacent collagen fibrils. The cytoplasm of the stromal cell was composed of some rough endoplasmic reticulum, mitochondria, and numerous parallel microfibrils with focal density. These stromal cells resembled myofibroblasts (Fig. 7).
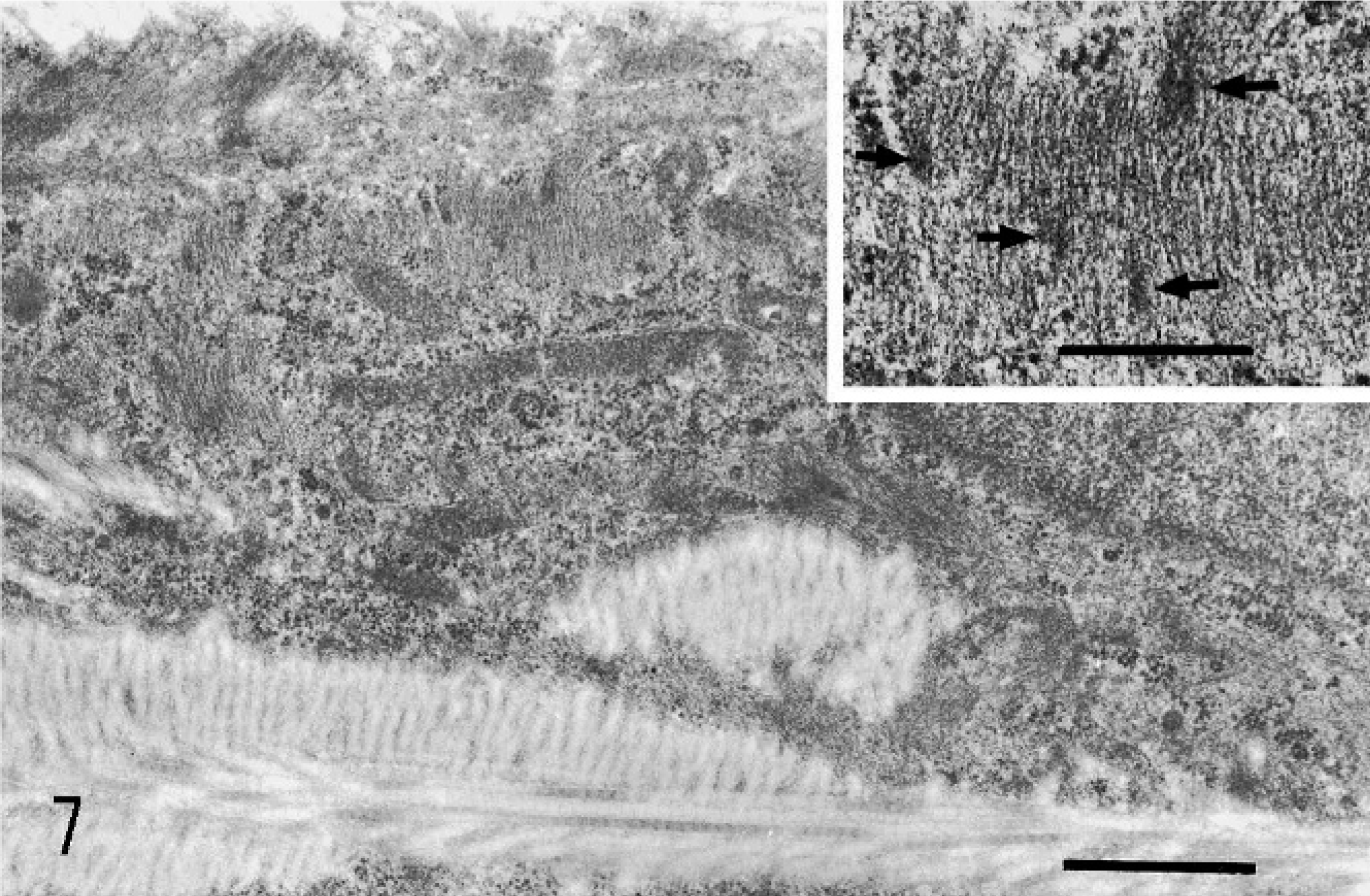
Mucinous cystadenoma, ovary; cynomolgus monkey. Electron micrograph of stromal cells, characterized by numerous parallel microfilaments with focal density. Bar = 2.5 μm.
The present ovarian tumor was characterized by proliferation of a multilocular cyst-forming mucinous epithelium reminiscent of the endocervical epithelium with abundant stroma containing smooth muscle fiber. According to the World Health Organization classification, this tumor was diagnosed as a mucinous cystadenoma in accordance with its morphologic characteristics. Mucinous cystadenoma is classified into the “surface epithelial tumor group,” originating from the transitioned coelomic mesothelial cell covering the ovary. However, evidence of a surface epithelial origin is less obvious for the mucinous type than for other types of surface epithelial tumors (such as serous or endometrioid tumors), and mucinous metaplasia of the surface epithelium or mucinous surface epithelial inclusion cysts are encountered only rarely. 4 In the present case, we were able to obtain evidence that proves the transition from the ovarian surface epithelium to the tumor epithelium of mucinous cystadenoma in the process of invaginating into the ovarian parenchyma by processing serial sections of the tumor. Although we examined all serial sections of the tumor, only one part of the invaginated surface epithelium leading to the tumor epithelium was observed. This tumor was strongly considered to have extended into the ovarian parenchyma from the invaginated area of surface epithelium only and then developed into the cystadenoma with multiple branches.
One of the main points of this case is the proliferation of smooth muscle in the stroma. There are abundant stromal elements stained for α-SMA and PCNA in this tumor. In a normal ovary, only the blood vessel, some stromal elements of the inner cortex, and the theca externa surrounding the larger follicles (Graafian follicles) are stained positively for α-SMA. 1, 5 In the present case, it can be hardly considered that the normal ovarian smooth muscle fibers were involved in the tumor tissue, because of the large volume, its distribution surrounding the tumor cysts, and PCNA-positive reaction of the stromal smooth muscle fibers. It has been reported that the stromal cells underneath the epithelial elements were stained positively for α-SMA in serous and mucinous cystadenomas. These α-SMA positive stromal cells are considered to be myofibroblasts that are modulated fibroblasts because of the pressure of the neoplastic epithelium. 1, 5 However, in the present case, the smooth muscle area was observed not only underneath the epithelial elements but also all over the stromal components. There are some reports referring to the transition of the ovarian stromal cells into smooth muscle cells along with the neoplastic epithelial proliferation. Moreover, it was suggested that there may be various histogenesis of ovarian smooth muscle proliferation, for example, endometriotic stroma, metaplastic or neoplastic potential of subcoelomic mesenchyme, in addition to ovarian stroma. 1, 2, 5 In the present case, from the close relationship between the cyst and the distribution of the stromal cells, it can be considered the ovarian stromal cells with potential of differentiation have transformed into proliferative smooth muscle cells through some kind of neoplastic epithelium stimulation.
There have never, to our knowledge, been previous reports concerning spontaneous mucinous cystadenoma in cynomolgus monkeys.
Footnotes
Acknowledgements
We thank Mr. Takayoshi Ito for his excellent technical assistance. We also thank Mr. Steve Yamakami and Mrs. Azami Takahashi for language editing.
