Abstract
A 13-year-old, obese, female cynomolgus monkey (
In humans, polycystic ovary syndrome (PCOS) is the most common form of chronic anovulation associated with androgen excess and is viewed as a heterogeneous disorder of multifactorial etiology. The currently recommended diagnostic criteria for PCOS are androgen excess and ovulatory dysfunction (amenorrhea, oligomenorrhea, or other forms of irregular bleeding). This syndrome is commonly associated with insulin resistance and associated hyperinsulinemia, luteinizing hormone (LH) hypersecretion, abdominal obesity, type 2 diabetes mellitus, atherosclerosis, and endometrial carcinoma; many patients with PCOS manifest only one or two of these components. 1– 5 In addition, current studies demonstrate that both leptin and adiponectin levels are altered in PCOS in association with the peripheral regulatory role in increasing tissue oxidation and thereby insulin sensitivity. Serum leptin concentrations are significantly elevated proportionally to the degree of adiposity, whereas adiponectin levels are paradoxically reduced in individuals with obesity and insulin resistance. 10 PCOS is well recognized in humans, with a prevalence rate between 5–10% in women of reproductive age. 1, 5
Unlike in humans, spontaneous PCOS is uncommon in animals, including nonhuman primates. In experiments, in utero androgen (testosterone) excess produces a syndrome in female rhesus monkeys (
A 13-year-old (estimated from dentition) female cynomolgus monkey (
Observational data on body mass index, fasting insulin, glucose, cycle status, ovulation, androstenedione, leptin and adiponectin level.∗
NM = not measured, n = number of animals; androstenedione AUC = androstenedione area under the curve in response to adrenocorticotropic hormone.
Total of 22 months of observation.
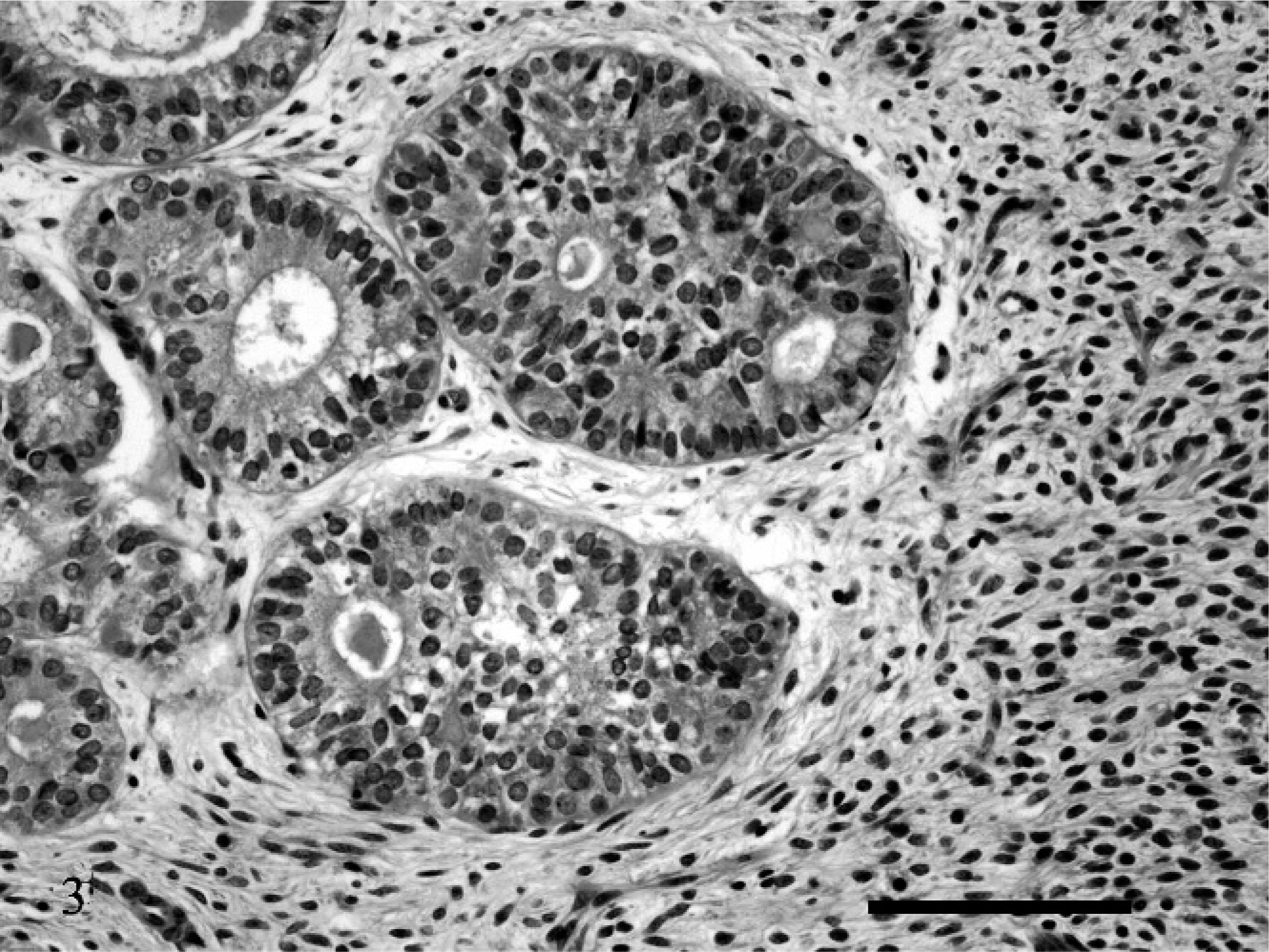
At the postmortem, the animal had excessive body fat (obesity), which was more prominent in the central abdomen. The uterus was enlarged, about 2 cm in diameter, with an eccentric uterine lumen and a broad-based endometrial polyp composed of irregularly sized cystic structures. On histopathologic examination, both ovaries contained many 2- to 3-mm follicles, without any corpora lutea (Fig. 1). The lumen of the uterus was displaced by an asymmetrical expansion of the endometrium by a sessile endometrial polyp. The polyp was composed of irregular glands within an edematous, highly vascular stroma that contained moderate numbers of endometrial granulocytes and stromal mitoses. The glands within the polyp had variable morphology: some resembled normal glands of the adjacent endometrium; others formed complex, closely packed clusters; and some were dilated by partially inspissated proteinaceous fluid into cystlike structures (Fig. 2). The complex structures were the most noteworthy and consisted of closely clustered glands lined by simple columnar-to-pseudostratified epithelial cells with abundant eosinophilic-to-clear cytoplasm, and many mitotic and apoptotic figures. In most glands, the epithelial nuclei were relatively uniform, oval, and vesicular, with marginated chromatin, although there were multifocal regions of disorganization and atypia (Fig. 3). Within some glands, there were low numbers of foamy macrophages. Accumulation of amorphous, eosinophilic material (collagen) was present multifocally in the stroma, with random distribution. Adjacent to the polyp, the more normal endometrial glands were pseudostratified, with low numbers of mitoses, subnuclear and supranuclear vacuoles, and stromal edema. Myometrial veins were surrounded by a moderate amount of pale, eosinophilic, loose, extracellular matrix (indicative of previous pregnancy). Abundant 2- to 3-mm-diameter follicular cysts were present throughout the parenchyma of ovary, few contained macrophages and rounded cells with clumped, pyknotic nuclei. Corpora lutea were not observed in either ovary.
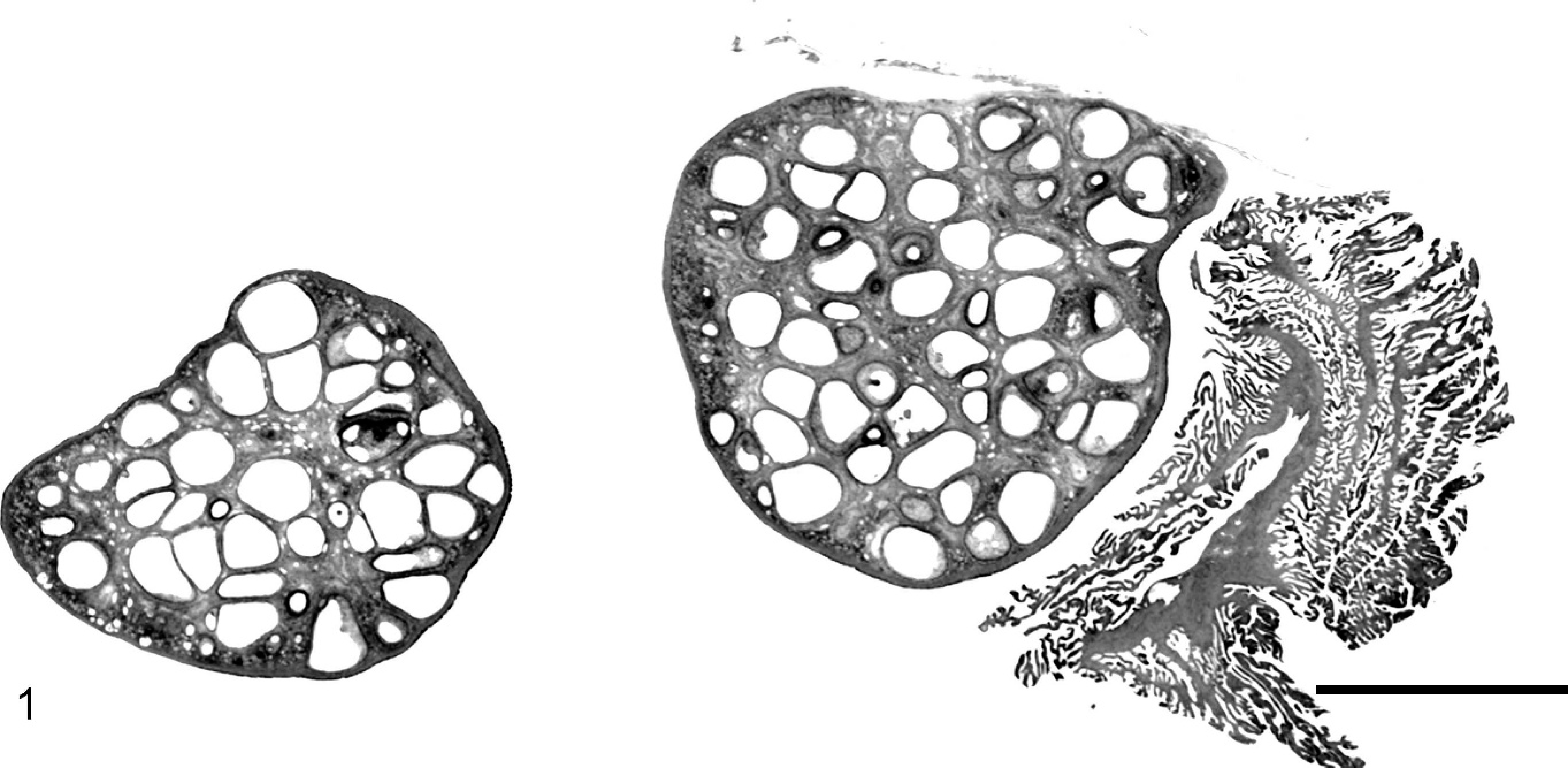
Subgross photomicrograph of both ovaries. Abundant 2- to 3-mm follicles throughout the parenchyma, without any corpora lutea. A section of normal fimbria is included at the right side of the image. HE. Bar = 5 mm.
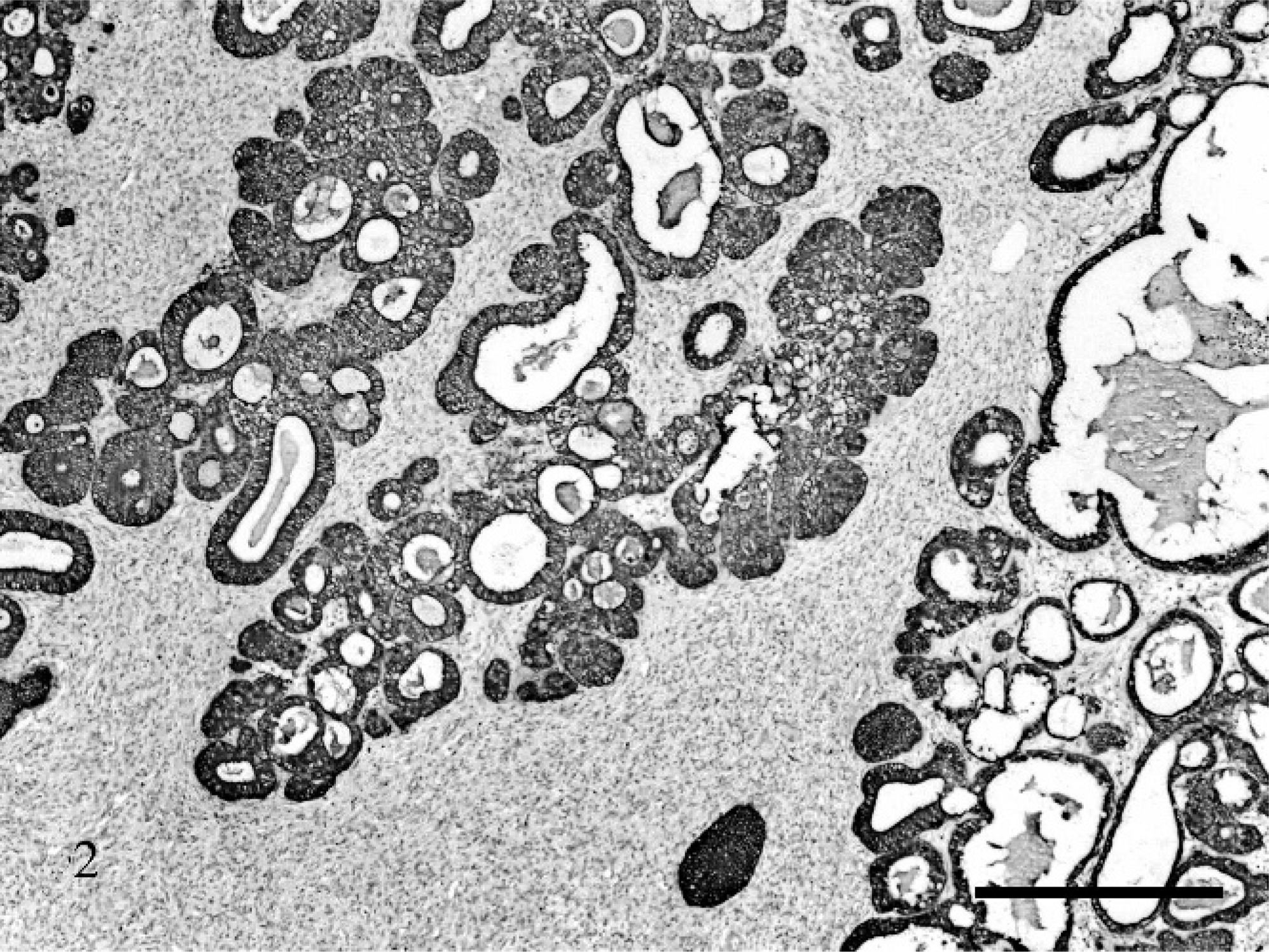
Uterus. Hyperplastic endometrial glands with simple columnar to pseudostratified epithelium with abundant proteinaceous secretion. Pan cytokeratin. Bar = 500 µm.
Uterus. Glandular hyperplasia with cellular atypia. HE. Bar = 100 µm.
The atypical hyperplastic epithelial cells in the uterus were strongly stained with pan cytokeratin (Pan CK-II, Clone B311.1, Biomeda, Foster City, CA, USA), and only small numbers were positively stained with Ki-67 (Clone MIB-1, DakoCytomation, Glostrup, Denmark).
On the day the animal was euthanatized, the plasma testosterone level was 21.2 ng/dl (premenopausal cynomolgus monkey reference, mean ± SD: 9.3 ± 0.6 ng/dl), 9 estradiol was 8.7 pg/ml (follicular phase reference, mean ± SD: 62.3 ± 8.49 pg/ml), 8 progesterone was 0.9 ng/ml (normal same follicular phase, mean ± SD: 1.1 ± 0.1 ng/ml), 8 leptin was 13.5 µg/L (group [n = 28] mean ± SD: 5.0 ± 4.0 µg/L), and adiponectin was 3.8 mg/L (group [n = 28] mean ± SD: 16.0 ± 9.0 mg/L) (Table 1).
Endometrial hyperplasia occurs in 35.7% of women with PCOS, and multiple mechanisms may be responsible for the increased risk of endometrial hyperplasia or cancer, without necessarily being accompanied with a high estrogen level. 3– 6 Results of some previous studies indicate that an increased risk of endometrial hyperplasia and carcinoma may be attributed to the persistent stimulation of endometrial tissue by estrogen (mainly estrone), without adequate progesterone-induced inhibition of proliferation and differentiation to secretory endometrium. Hyperinsulinemia may contribute by enhancing endogenous endometrial estrogen production, because insulin stimulates aromatase activity in both endometrial glands and stroma. 6 Overall, moderately elevated levels of estrogen are encountered because of increased peripheral conversion of androstenedione to estrone in adipose tissue, and free estrogen, as well as testosterone, is elevated in the circulation in the setting of hyperinsulinemia, also because, in part, of insulin downregulation of sex-hormone binding globulin. In this case, insulin resistance, along with low serum adiponectin and high serum leptin compared with the group average, supported the diagnosis of PCOS, as well as cystic endometrial hyperplasia, similar to changes that were described in humans. 10
This case is remarkable in that endometrial hyperplasia in association with spontaneous PCOS has not previously been diagnosed in nonhuman primates. In this case, a diagnosis of PCOS was made based on the clinical history of the animal, including suppression of ovulation with a prolonged menstrual cycle length, hyperinsulinemia, hyperandrogenism, and obesity, as well as multiple cystic follicles in both ovaries and endometrial complex hyperplasia. This clinical presentation was similar to that reported in humans.2
Leptin, adiponectin, and hormones levels at postmortem (year 5).
NM = not measured.
Footnotes
Acknowledgements
We thank Ms. Hermina Borgerink and Ms. Lisa O'Donnell for their assistance with histologic and immunohistochemical techniques.
