Abstract
Epithelioid trophoblastic tumor (ETT) is an unusual type of trophoblastic tumor, with features resembling carcinoma. In this study, we describe a 4-year-old cynomolgus monkey (
The apparently low incidence of spontaneous tumors in nonhuman primates is probably because of the lack of studies involving large groups of aged monkeys. 8 Ovarian tumors are most frequently represented by teratomas and granulosa cell tumors in baboons and in macaques. 1, 2, 4, 9, 10 Ovarian carcinoma is rare, 8 and choriocarcinoma, a malignant tumor arising from germ cells, has been described only once. 14
The epithelioid trophoblastic tumor (ETT) is a relatively new entity in human pathology, first described by Mazur and Kurman in 1994. 7 It is a rare but distinctive tumor whose morphologic and immunohistochemical features suggest an origin from chorionic-type intermediate trophoblast.
In this study, we present a case of ETT arising in the ovary of a cynomolgus monkey (
The animal was a 4-year-old cynomolgus monkey, born in captivity, which had been treated intravenously for two cycles of 3 days, 1 week apart, with an antineoplastic drug (a kinase inhibitor) as part of a toxicologic study. No clinical signs or hematologic or blood chemistry abnormalities were recorded either before or during the study period. The animal was anesthetized intravenously with sodium thiopental and euthanatized by bleeding from the femoral vessels 41 days after the last treatment. A firm, grayish, slightly lobulated mass replacing the left ovary was found at necropsy. The mass measured 4 cm at the greatest diameter and weighed 28 g. One cyst about 8 mm in diameter was observed on the cut surface (Fig. 1). Numerous, firm, grayish nodular masses, 5–15 mm in diameter, were present bilaterally in all lobes of the lungs. The pelvic lymph nodes were enlarged, and the thymus was reduced in size. No changes were observed in the remaining organs. Specimens from all major organs and tissues were collected and fixed in 10% neutral buffered formalin. Sections 5 μm thick were obtained from paraffin-embedded tissue and stained with hematoxylin and eosin (HE). Additional sections of the tumor and affected lung were stained with an immunohistochemical technique (labeled streptavidin-biotin method), using primary antibodies directed against cytokeratins (AE1/AE3), human chorionic gonadotropin (hCG), placental alkaline phosphatase (PLAP) (Dako, Glostrup, Denmark) and -α (Serotech, Oxford, England) according to manufacturers' instructions.
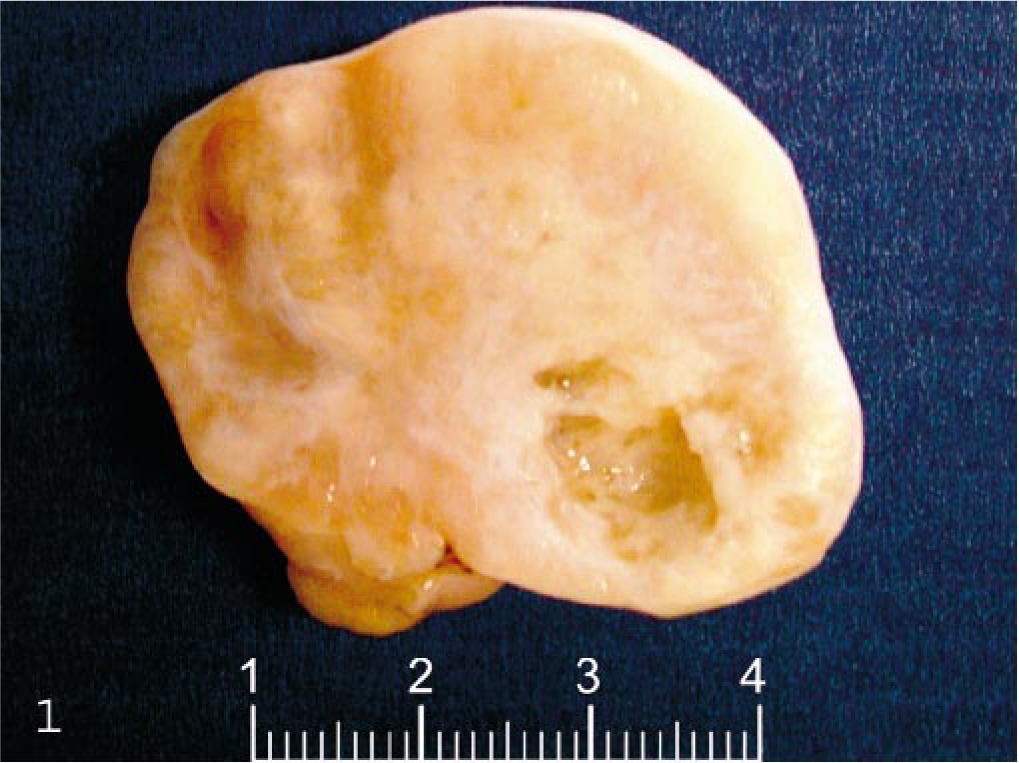
Ovary; cynomolgus monkey. Lobulated mass replacing the left ovary.
Microscopic examination of HE-stained sections of the left ovary showed a partially encapsulated, poorly demarcated, nodular mass, which completely effaced the organ. It was composed of nests of large, polygonal cells separated by a moderate amount of fibrovascular stroma. The tumor cells had abundant, clear vacuolated or eosinophilic cytoplasm and distinct borders (Fig. 2). The nuclei were centrally located, with finely stippled chromatin and single, prominent, eosinophilic nucleolus. Occasional multinucleate tumor cells, consistent with syncytiotrophoblastic cells, were randomly distributed throughout the tumor. Dispersed among the neoplastic cells, there was a small amount of eosinophilic, hyaline-like material (Fig. 3). Mitotic figures were rare (three/10 high-power field). Large, multifocal to coalescent areas of coagulative necrosis were present in the central portion of the mass. The stroma supporting the tumor was infiltrated by a variable number of plasma cells, macrophages, and lymphocytes.
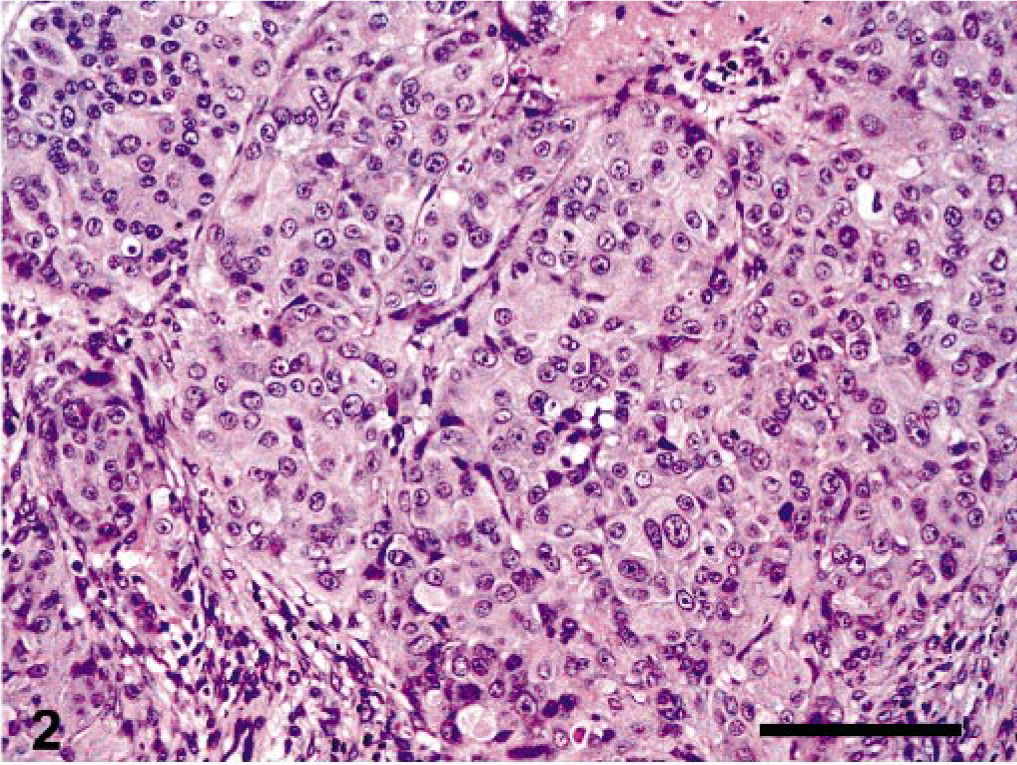
Ovary, epitheliod trophoblastic tumor; cynomolgus monkey. Epithelioid appearance of neoplastic cells arranged in a nodular pattern. HE. Bar = 140 μm.
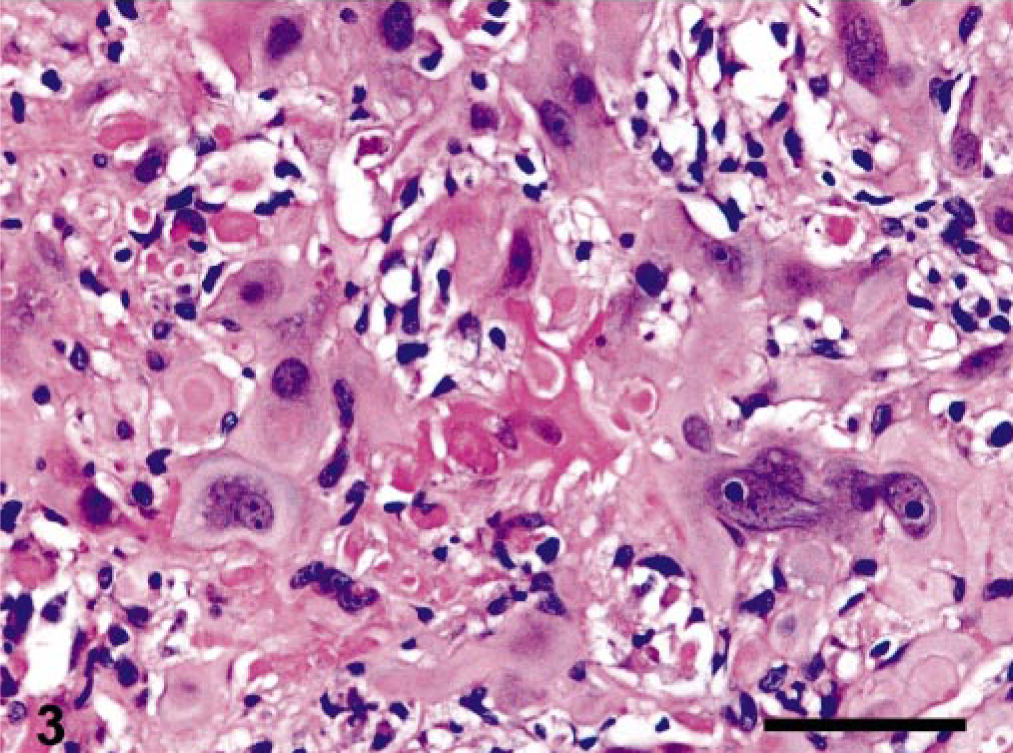
Ovary, epitheliod trophoblastic tumor; cynomolgus monkey. Dense, eosinophilic, hyaline-like material intimately associated with trophoblastic cells. HE. Bar = 70 μm.
The lung contained multiple, unencapsulated, poorly demarcated nodules, composed of nests of cells morphologically similar to those observed in the ovary and containing multifocal to coalescing areas of necrosis. The uterus showed features characteristic of early endometrial proliferative stage. Contralateral ovary was unremarkable. Pelvic lymph nodes showed marked plasmacytosis. Other organs and tissues examined histologically were normal.
Immunohistochemically, the tumor cells were diffusely positive for cytokeratins (Fig. 4)) in both the ovarian tumor and the lung metastases, with focal positivity for α-inhibin in the ovary (Fig. 5) and rare positive cells in the lung. Occasionally, small groups of neoplastic cells showed strong cytoplasmic positivity for hCG (Fig. 6) in both the ovary and the lung, whereas PLAP staining was always negative.
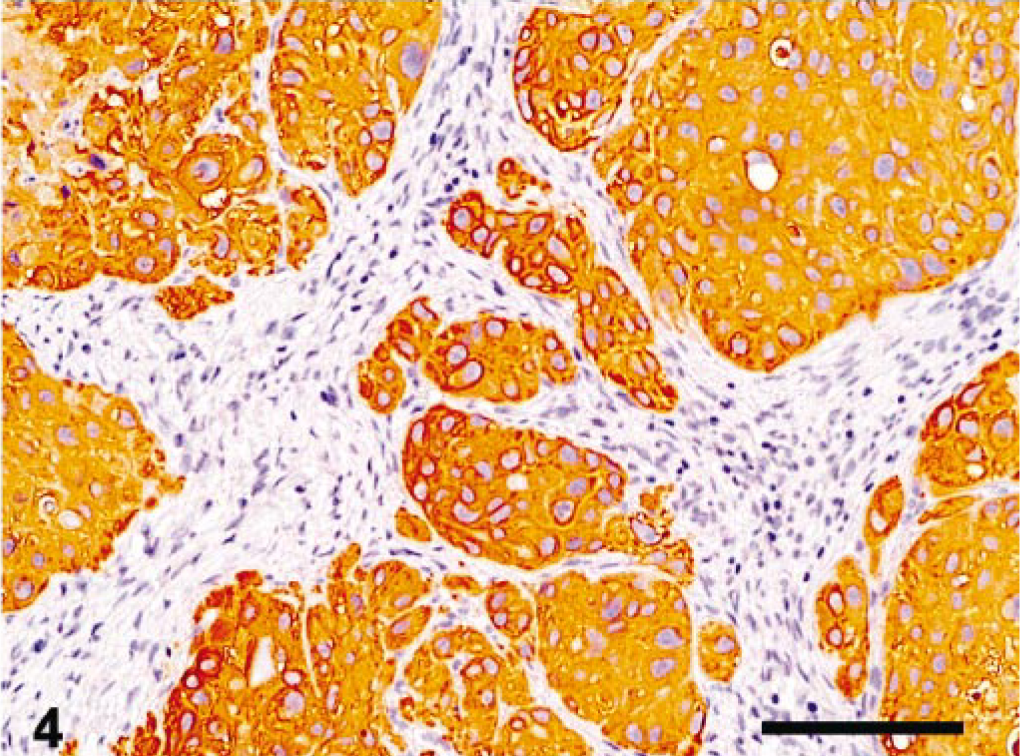
Ovary, epitheliod trophoblastic tumor; cynomolgus monkey. Neoplastic cells were diffusely immunoreactive for cytokeratin AE1/AE3. Streptavidinbiotin method, Mayer's hematoxylin counterstain. Bar = 280 μm.
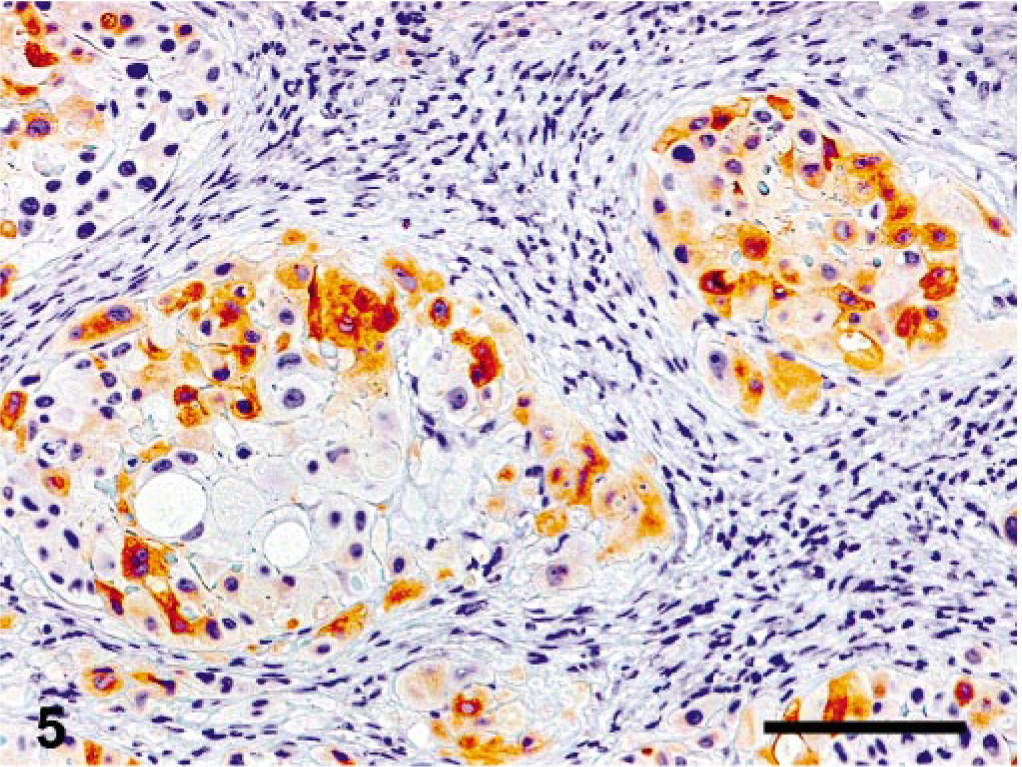
Ovary, epitheliod trophoblastic tumor; cynomolgus monkey. Multifocal immunoreactivity for inhibin-α. Streptavidin-biotin method, Mayer's hematoxylin counterstain. Bar = 140 μm.
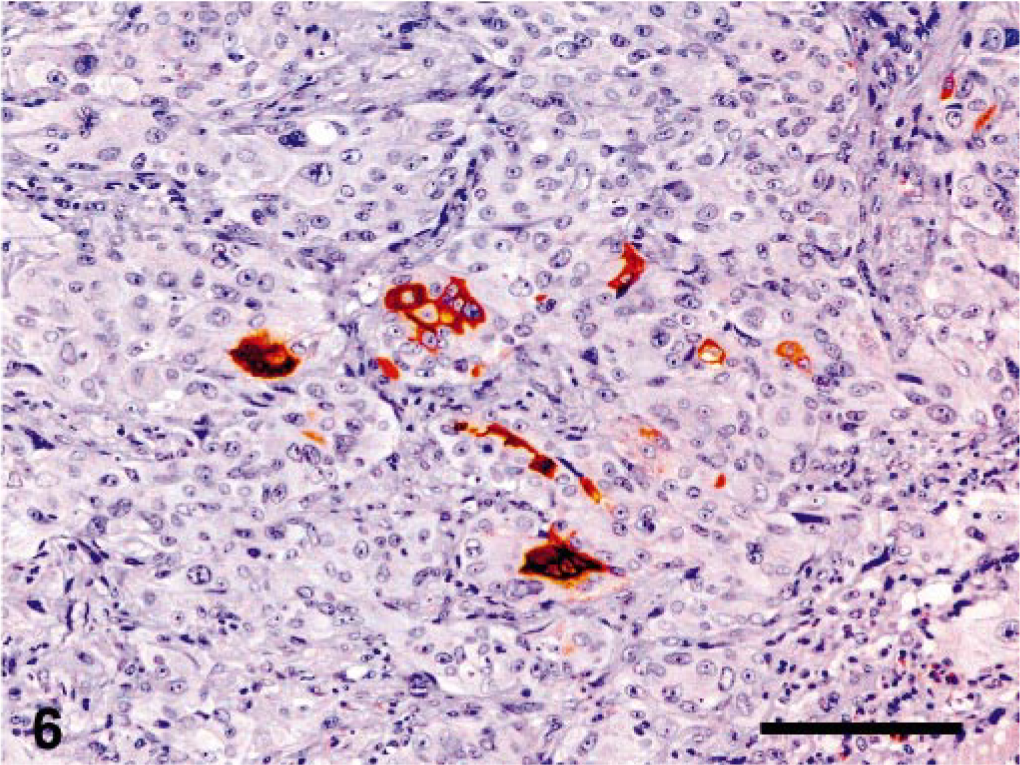
Lung, epitheliod trophoblastic tumor; cynomolgus monkey. Focally within the tumor, rare cells were positive for hCG. Streptavidin-biotin method, Mayer's hematoxylin counterstain. Bar = 70 μm.
Ovarian tumors in nonhuman primates usually occur after 10 years of age. In the genus
Recently, Toyosawa et al. described an ovarian choriocarcinoma in a 13-year-old cynomolgus monkey. 14 This tumor was characterized by a dimorphic population of cells, morphologically resembling cytotrophoblastic and syncytiotrophoblastic cells, the latter being positive for anti-hCG.
Human choriocarcinoma involving the ovary may result from three different mechanisms: as a primary choriocarcinoma associated with ovarian pregnancy (gestational choriocarcinoma); as a metastasis from a tumor arising in another organ (mainly the uterus); or as a primary ovarian germ cell tumor with differentiation toward trophoblastic structures (nongestational choriocarcinoma). The latter generally develops in girls or young adults, in whom pregnancy needs to be excluded. 15
In this study, we report a tumor occurring in the ovary of a cynomolgus monkey, composed of a relatively uniform population of tumor cells, occasionally multinucleated, with prominent cell membranes, abundant eosinophilic cytoplasm, and large pleomorphic nuclei, accompanied by a multifocal accumulation of homogeneous eosinophilic material and confluent areas of necrosis. The tumor cells were identified as intermediate trophoblast on the basis of morphology and immunohistochemical reactivity. Tumors with similar features have been recently identified in humans and named epithelioid trophoblastic tumors. ETT is currently regarded as an unusual type of trophoblastic tumor, distinct from choriocarcinoma and placental site trophoblastic tumor. Its most striking feature is the fact that the growth pattern mimics that of a carcinoma. 12
The relationship between ETT and choriocarcinoma is still unclear. Tumor components of ETT have distinctive, morphologic and immunohistochemical features, which differ from those of choriocarcinoma. Generally, the tumor cells in ETT are positive for cytokeratins and inhibin-α and only occasionally for beta human chorionic guadotropin (bHCG). Conversely, in choriocarcinoma, there is frequently a diffuse positivity for cytokeratins and bHCG, but only syncytiotrophoblastic tumor cells are positive for inhibin-α. 11 In this tumor, there was strong and diffuse positivity for cytokeratins and, with lower intensity, for inhibin-α in all neoplastic cells. Furthermore, hCG was strongly positive only in rare cells, in both the main tumor and the lung metastases. This pattern of staining is consistent with ETT. 3, 6, 11 In particular, hCG is only focally positive in ETT, whereas immunostaining for hCG is diffusely positive in choriocarcinoma. 12 Eosinophilic, hyaline-like material, similar to that observed in this tumor, is also reported to be typically deposited around normal implantation site intermediate trophoblast, 13 as well as in ETTs. 3, 5, 6, 11
In this study, no gross or microscopic abnormalities were observed in the uterus. Although ETT generally arises within the uterus, extrauterine occurrences without an identifiable uterine lesion have been described. 5, 12 Uterine ETT is usually associated with a previous gestational event. Although it cannot be ruled out completely, the possibility of a previous undiagnosed pregnancy in the animal reported in this study is remote. In fact, this animal was born in captivity and, to the best of our knowledge, never mated, since it arrived at our facility when it was only 2 years of age and had always been kept separate from males in the breeding facility of origin.
Because most humans reported with ETT have had a history of chemotherapy for previous choriocarcinoma, it has been suggested that development of ETT could be caused by an inadequate response of choriocarcinoma to chemotherapy. According to this theory, some chemotherapeutic agents might produce a selection in the dedifferentiation process, with ETT emerging from the choriocarcinomatous component. 5, 11 Interestingly, this animal had received two cycles of treatment with a novel chemotherapeutic drug, 6 weeks before the diagnosis of the tumor. Thus, the possibility that the treatment could have modified a preexistent ovarian choriocarcinoma cannot be excluded. The large areas of necrosis within the tumor could be also related to the anti-tumoral treatment the animal had received.
Similar to the findings in this study, the lung seems to be the preferential site for metastases of this tumor type. 5
Several ovarian carcinomas have been reported in non-human primates. 8 It is possible that some of these tumors may have been ETT or choriocarcinomas that have been misdiagnosed as carcinomas because of their epithelioid pattern and lack of immunohistochemical characterization. Therefore, we strongly suggest immunohistochemical staining to better investigate the nature of anaplastic ovarian tumors found in nonhuman primates.
Footnotes
Acknowledgements
We thank Dr. Jan C. Klapwijk for critical review of the manuscript and Mr. Marco Colombo for technical assistance.
