Abstract
A herpesvirus infection affecting mini Rex and crossbred meat rabbits was identified in a rabbitry in Alaska. Illness affected over half of the 55 rabbits on the premises, and 16 rabbits died or were euthanatized because of illness. Disease affected all ages from adults to nursing young and occurred over an approximately 2-month period. Clinical signs included conjunctivitis and periocular swelling, ulcerative dermatitis, progressive weakness, anorexia, respiratory distress, and abortion. Hemorrhagic dermatitis and panniculitis were associated with epidermal microvesicular degeneration, dermal and subcutaneous vascular necrosis, and thrombosis. Eosinophilic intranuclear inclusions consistent with herpesvirus were found within the epidermis and superficial follicular epithelium and within mesenchymal cells within the dermis and subcutis. Syncytial cells containing viral inclusions occurred within the epidermal and superficial follicular epithelium. Other findings were hemorrhagic necrosis of the myocardium with rare intranuclear inclusions within stromal cells, multifocal pulmonary hemorrhage, hemorrhage with sinus erythrophagocytosis in lymph nodes, and massive necrosis and fibrin deposition within red pulp of the spleen. A virus isolated from the skin produced syncytia, intranuclear inclusions, and cell lysis typical of herpesvirus in rabbit kidney cells in vitro. The viral isolate was characterized ultrastructurally as an enveloped virus with icosahedral nucleocapsids 100 nm diameter, consistent with a herpesvirus.
Keywords
Herpesvirus disease in rabbits includes both experimental and natural infections. 2– 9, 11– 14, 16, 18– 22 Experimentally, domestic rabbits have been utilized for studies of human herpes simplex virus (HSV) latency reactivation. 3, 6, 9, 12, 16 Experimental HSV infection can cause neurologic disease and death in domestic rabbits. 3, 9 Naturally occurring HSV infection causing encephalitis in domestic rabbits is rare. 4, 21 The most common naturally occurring herpesvirus infections identified in rabbits are Leporid herpesvirus 2 (Herpesvirus cuniculi) and Leporid herpesvirus 3 (Herpesvirus sylvilagus). 5, 7, 8, 11, 13, 18, 19, 22 These naturally occurring viruses are in the subfamily Gammaherpesvirinae. The most characterized of these viruses, Leporid herpesvirus 3 (LHV-3), is indigenous in cottontail rabbits (Sylvilagus floridanus) and causes lymphoproliferative disease resulting in tumor-like lesions in lymph nodes, kidney, spleen, and liver. 5, 7, 8, 11, 18, 19 Leporid herpesvirus 3 does not cause infection or disease in domestic rabbits. 4, 5, 10, 11, 21 Domestic rabbits (Oryctolagus cuniculus) are the natural host of Leporid herpesvirus 2 (LHV-2), but infection is asymptomatic. 13, 22
Two outbreaks of an acute fatal herpesvirus infection in domestic rabbits in Canada have been previously described. 14, 20 This uncharacterized herpesvirus infection was associated with high morbidity and mortality. Lesions included hemorrhagic dermatitis on the face and back, interstitial pneumonia, and severe splenic necrosis. 14, 20 This report describes a similar outbreak of herpesvirus infection in domestic rabbits in Alaska. Initial viral characterization of this rabbit herpesvirus (RHV) suggests that this is a novel alphaherpesvirus with morphology similar to HSV.
Materials and Methods
In July and August of 2006 mini Rex and crossbred meat breed rabbits in a breeding facility near Wasilla, Alaska, developed systemic illness that often began with unilateral or bilateral conjunctivitis with marked periocular swelling (Fig. 1). The ocular discharge was initially serous and often became purulent with time. Within 24 hours affected rabbits developed depression and anorexia. Clinical signs often progressed to include weakness, difficulty breathing, torticollis, head tilt, ataxia, bruxism, weight loss, diarrhea, ulcerative lesions in the skin, abortion, and genital swelling. Severely affected animals died or were euthanatized within 3–7 days of onset of clinical disease. Some animals showed only signs of anorexia prior to death. Illness affected over half of the 55 rabbits on the premises (approximately 50% morbidity), including adults and nursing young, and approximately 16 affected rabbits died or were euthanatized because of severe illness (approximately 29% mortality). Animals were individually housed in outdoor open-sided hutches, and ill animals were immediately isolated. Asymptomatic animals were moved to clean cages in another section of the property. There had been no new animal introductions and there was no history of travel. Mosquito (Ochlerotatus spp., Aedes spp., Culex spp.) and biting fly (Culicoides spp., Simulium spp.) activity was high during this time. There were snowshoe hares (Lepus americanus) and feral domestic rabbits in the area, and two feral domestic rabbits had been observed on the premises near the rabbit hutches, one in May and one in June. Neither rabbit appeared ill, and both rabbits were shot and disposed of by the owner. Other animals on the premises included dogs, cats, poultry, and horses, and neither these animals nor the facility owners became ill during the time of this outbreak.
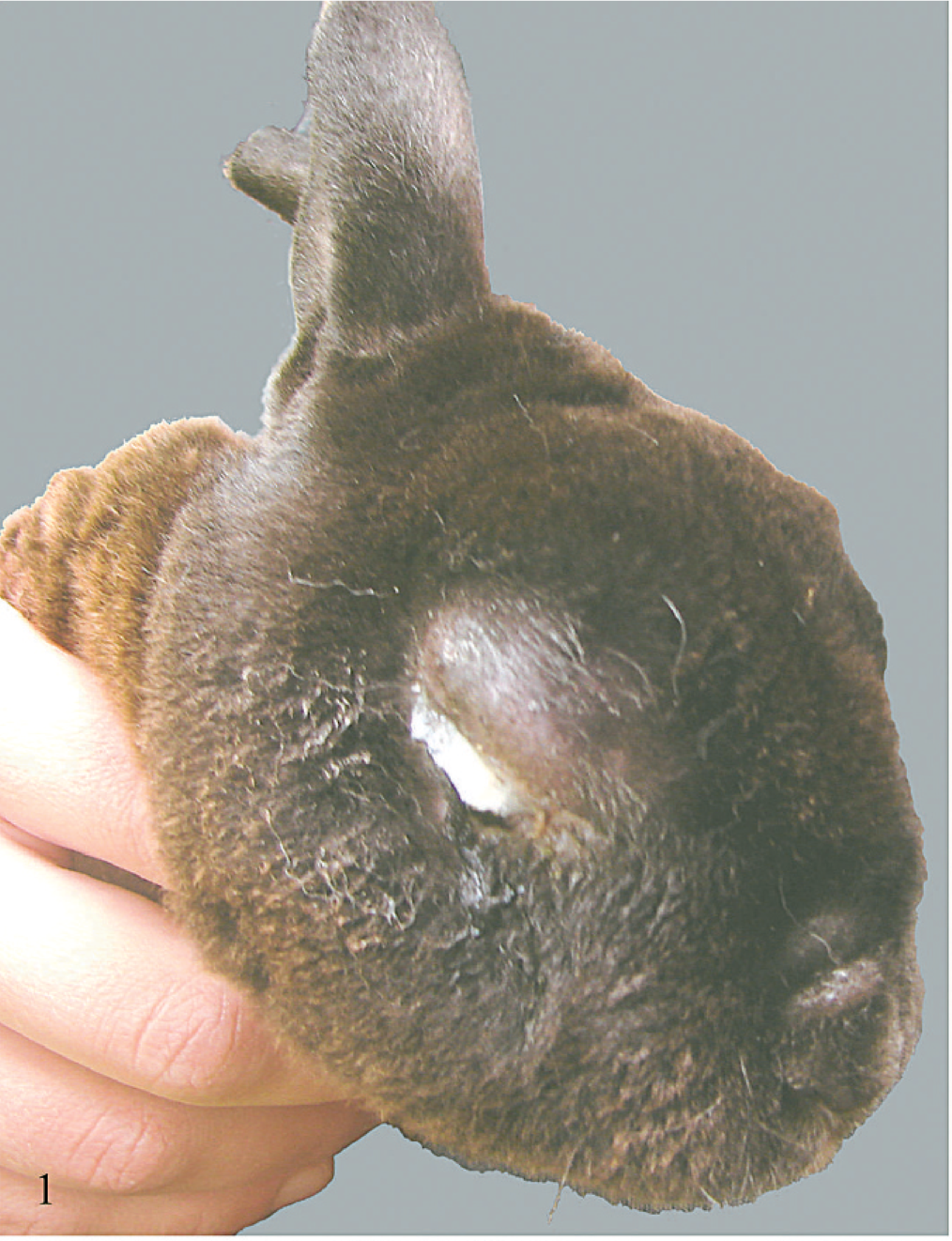
Affected rabbit with severe facial swelling and purulent conjunctivitis.
In the spring and summer of the following year, the owner reported several rabbits with severe conjunctivitis, cutaneous lesions, depression, and anorexia. In addition, stillbirths and neonatal deaths occurred in a litter from a doe that had recovered following clinical illness the prior year. The owner elected to depopulate the rabbitry.
Postmortem evaluation was performed on 6 rabbits from the initial outbreak. Samples of spleen, liver, and lung were submitted for aerobic bacterial culture, and no Francisella tularensis, Pasteurella multocida, or other pathogens were isolated. Samples of heart, lung, lymph node, spleen, kidney, small intestine, pancreas, liver, skeletal muscle, large intestine, and sacculus rotundus, and multiple samples of abnormal skin were obtained from an affected 6-week-old male mini Rex rabbit, fixed in 10% buffered formalin, and processed routinely for light microscopic evaluation. Ultrastructural examination of tissue was not performed. Samples of unfixed lung, liver, skin, and gastrointestinal tract from this rabbit were frozen at −80°C prior to study. Histopathology was not performed on rabbits exhibiting clinical disease the following year.
Frozen skin samples from the 6-week-old mini Rex rabbit were homogenized in 1× Dulbecco's modified Eagle's medium (DMEM; 1:5 ratio [w/v]) clarified by centrifugation and inoculated onto rabbit kidney cells (RK13; American Type Culture Collection, Rockville, MD) maintained in DMEM supplemented with 1% fetal bovine serum (Invitrogen, Carlsbad, CA), penicillin (100 U/ml), and streptomycin (100 μg/ml) (Sigma-Aldrich, Inc., St. Louis, MO). Cultures were maintained at 37°C with 5% CO2 in a humidified incubator and monitored daily for cytopathic effect (CPE).
The isolated virus was propagated in RK-13 B cells maintained in DMEM supplemented with 5% calf serum and antibiotics as above. Confluent cell monolayers were infected with plaque-purified virus at a 0.1 multiplicity of infection and were harvested when more than 90% of the cells showed visible CPE. Virus suspensions were freeze thawed twice, clarified by centrifugation, and density gradient purified on sucrose gradients. The purified virus was suspended in phosphate buffered saline and frozen at −70°C until further investigation.
Purified virions were adsorbed to formvar coated, carbon stabilized copper grids by floating grids on droplets of the sample spotted on parafilm. The grids were then blotted dry with filter paper rinsed with distilled water and stained with 1.5% phosphotungstic acid. Images were obtained with a Phillips EM 300 electron microscope.
The nucleocapsid and virions were isolated from cells harvested when 70–90% of the monolayer showed viral CPE. Nucleic acid was extracted from virions and nucleocapsids isolated from the nucleus as previously described. 10 Viral DNA from herpes simplex virus 1 (HSV-1; McKrae strain) and equine herpesvirus 1 (EHV-1; NVSL strain) were prepared similarly.
The viral genome was digested with BamHI restriction endonuclease (Invitrogen) and compared with HSV-1 and EHV-1 DNA. BamHI restriction endonuclease (Invitrogen) was performed at 37°C for 2 hours in the buffer system supplied by the manufacturer. BamHI digests of DNA from the rabbit virus, HSV-1, and EHV-1 were compared by electrophoresis on 0.8% agarose gel. The 1 kb Plus DNA ladder (Invitrogen) was used as a DNA size marker.
Results
External findings in the 6-week-old rabbit were slight swelling and ulceration of the anogenital skin, multiple 0.3- to 1.0-cm-diameter nodular thickenings within the skin of the back, ventral abdomen, and pelvic limbs with severe hemorrhage within the subcutis on section, swelling and subcutaneous edema of the muzzle, mucoid red tinged nasal discharge, and enlarged peripheral lymph nodes with hemorrhage on section. Internal findings were swollen, dark purple–red nodular swellings within the spleen, mucoid content within the small intestine, pulmonary hemorrhage, serosanguinous pericardial effusion, and multifocal to coalescing approximately 0.2-cm-diameter pale zones within the myocardium.
Necrotizing and hemorrhagic lesions were found within sections of nodular skin lesions, facial and eyelid skin, heart, spleen, and lung. Within affected skin there was frequent microvesicular degeneration of the epidermis and follicular epithelium. Individual cells and large syncytial cells within the epidermis and superficial follicular epithelium contained many prominent intranuclear eosinophilic inclusions (Fig. 2). Scattered epidermal and superficial follicular epithelial cells were undergoing single cell necrosis/apoptosis, and follicular bulb cells often exhibited nuclear condensation and fragmentation consistent with apoptosis. There was severe dermal and subcutaneous hemorrhage and necrosis, with vascular necrosis and thrombosis and moderate infiltration by a mixture of macrophages, often exhibiting erythrophagocytosis, lymphocytes, plasma cells, and a lesser number of heterophils. Similar intranuclear inclusions were found within scattered spindle to dendritic cells within affected dermis and subcutis (Fig. 3). Multiple large foci of hemorrhagic necrosis with a small number of associated lymphocytes and heterophils were present in both the right and left ventricular walls, with rare intranuclear inclusions within stromal cells. Scattered necrotic myocardiocytes were underlying mineralization. Large zones of necrosis, hemorrhage, and fibrin deposition were present within the spleen. Multifocal small zones of alveolar necrosis with fibrin exudation and prominent hemorrhage were present within the lung. Peripheral lymph nodes exhibited severe sinus hemorrhage and erythrophagocytosis. Lymphoid tissue within the peripheral lymph nodes, viable areas of the spleen, and the sacculus rotundus was normal. No convincing viral inclusions were seen within the spleen, lung, or lymph nodes. No lesions were found within the intestines, liver, or kidney.
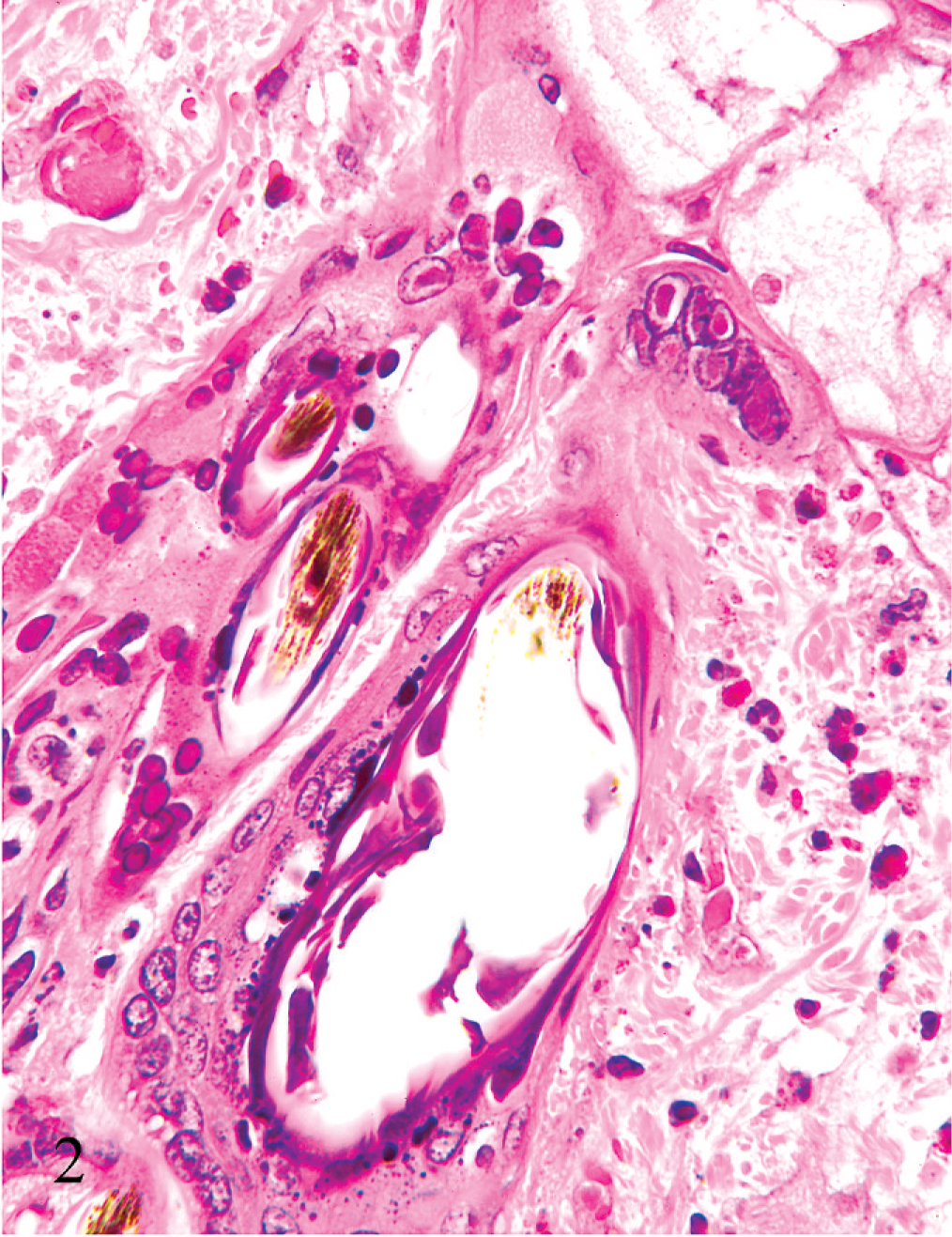
Skin; rabbit. The epidermis exhibits microvesicular degeneration with large syncytial cells. Syncytial cells and numerous epidermal and superficial follicular epithelial cells contain prominent eosinophilic intranuclear inclusions. There is also dermal edema, hemorrhage, and capillary fibrin thrombosis. HE.
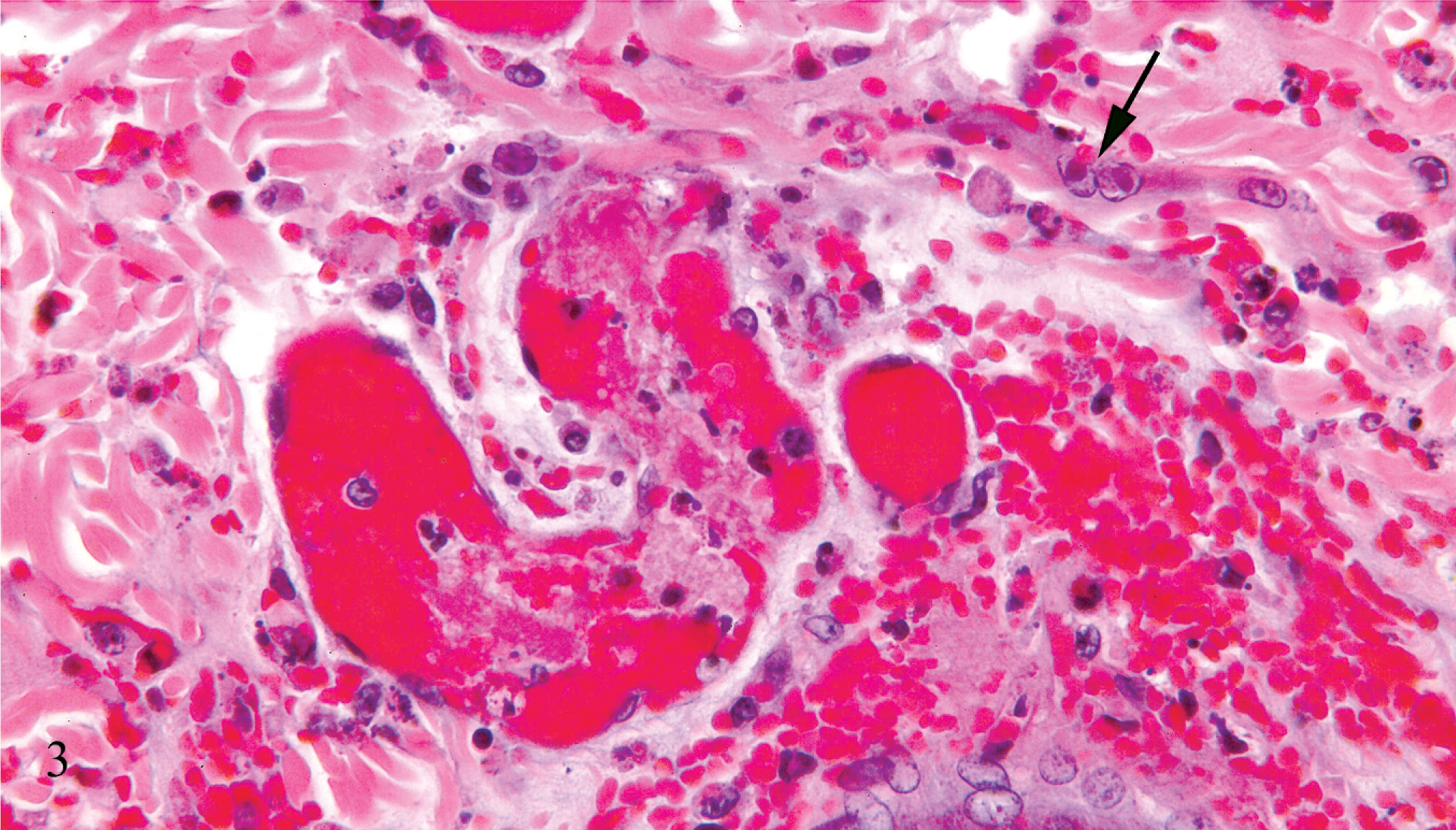
Skin; rabbit. There is dermal vascular necrosis and thrombosis with associated hemorrhage and edema. Scattered mesenchymal cells contain prominent eosinophilic intranuclear inclusions (arrow). HE.
RK-13B cells infected with the virus isolated from the rabbit developed cytopathic effects (syncytia, cytomegaly, cell lysis, and intranuclear inclusions) within 48 hours postinfection. Ultrastructural examination revealed virions with symmetric icosahedral morphology (Fig. 4) with an average core diameter of 100 nm and an envelope-like structure, consistent with herpesviridae. The rabbit herpesvirus DNA genome had a BamHI restriction digestion pattern different from HSV-1 and EHV-1, with fewer fragments than HSV-1 DNA, but was more similar to HSV-1 than to EHV-1 (Fig. 5).
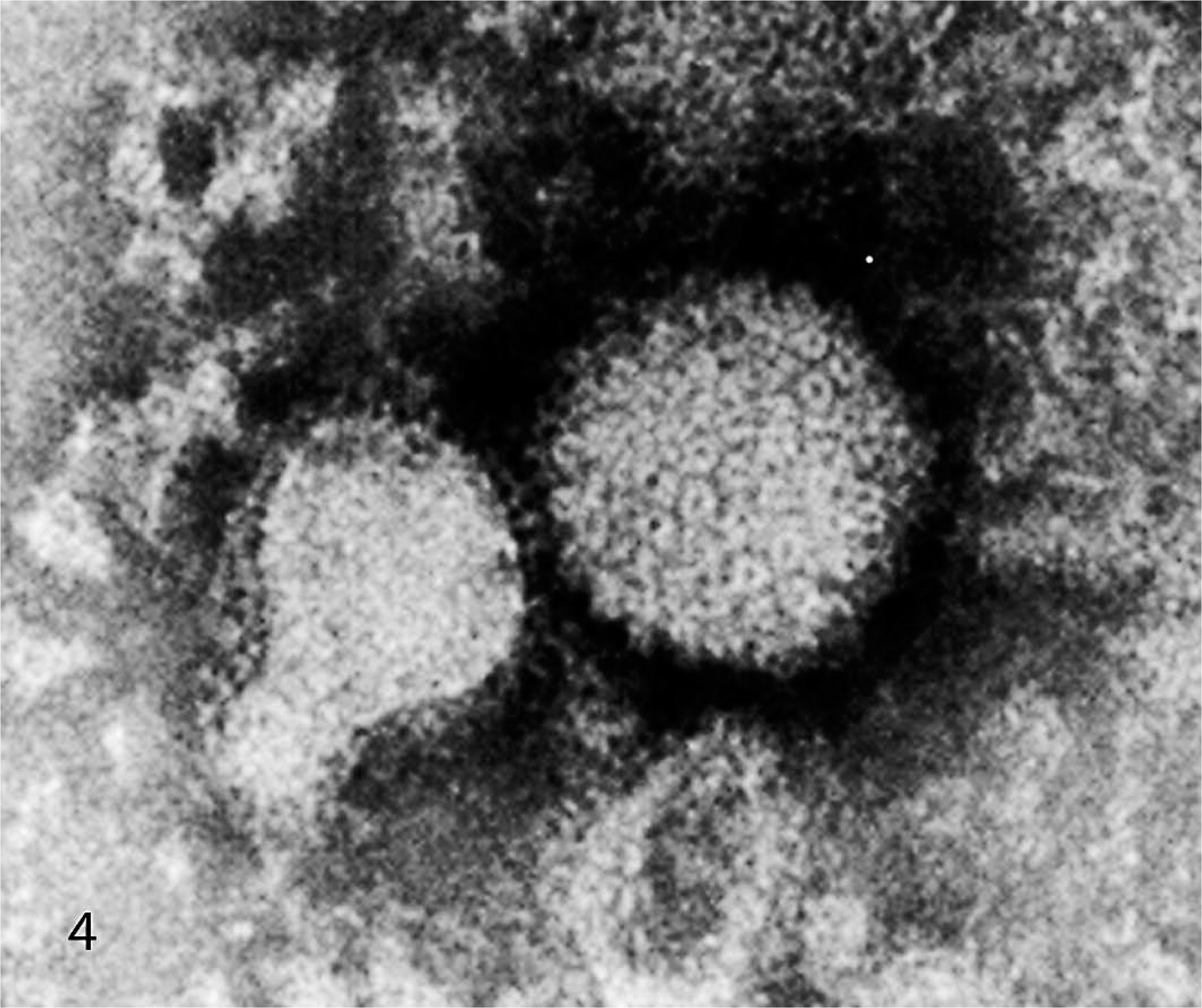
Viral particles harvested from infected RK-13B cells. The virus exhibits symmetrical icosahedral morphology with a core diameter of approximately 100 nm and a surrounding capsule. Transmission electron micrograph, negative staining with 1.5% phosphotungstic acid.

Endonuclease restriction digestion of purified viral DNA with BamHI. Lane 1 contains molecular size markers; lane 2 is rabbit viral nucleocapsid; lane 3 is EHV-1; lane 4 = rabbit virus virions; lane 5 is HSV-1. The restriction digestion pattern of the rabbit herpesvirus differs from that of HSV-1 and EHV-1.
Discussion
Viral diseases causing high morbidity and mortality in rabbits include rabbit hemorrhagic disease (RHD), 1 myxomatosis virus, 15 and the previously uncharacterized herpesvirus. 14, 20 Morbidity and mortality of RHD (up to 90–100%) is higher than in this Alaskan outbreak. 1 Mucocutaneous lesions are not a feature of RHD, and RHD only affects rabbits greater than 2 months of age. 1 Mucocutaneous lesions and a mortality rate of 20–50% are typical of the California strain of myxomatosis virus, 15 which was the primary clinical differential diagnosis in this Alaskan outbreak. Myxomatosis is characterized by typical intracytoplasmic poxvirus inclusions, especially in ocular and anogenital skin, and severe lymphoid necrosis, 15 none of which were present in the rabbits studied in this outbreak.
Pathologic, tissue culture, and ultrastructural features of this virus are characteristic of herpesvirus. 17 Classification of a herpesvirus as an alphaherpesvirus relies on biologic behavior and on viral genome studies. Alphaherpesviruses have a variable host range, a relatively short reproductive cycle, exhibit rapid spread in tissue culture, and cause necrosis of infected cells. 17 This rabbit herpesvirus has a rapid in vitro replication cycle of less than 48 hours in rabbit kidney cells. It produces syncytia with intranuclear viral inclusions in infected skin and in vitro, typical of alphaherpesvirinae. Experimental or naturally occurring infection of rabbits by the alphaherpesvirus HSV-1 causes encephalitis and keratitis. 3, 4, 6, 9, 12, 16, 21 Brain tissue was not obtained from affected rabbits in this outbreak, but results of genomic endonuclease restriction digestion indicate that this rabbit herpesvirus is different from HSV-1.
The virus in this outbreak is considered to be the same as the previously reported uncharacterized herpesvirus causing outbreaks of disease in domestic rabbits in Canada. 14, 20 Infection of cutaneous epithelium is prominent. Vascular necrosis and profound hemorrhage suggest endothelial infection, but the identity of the mesenchymal cells containing viral inclusions in affected tissues is not clear. The natural host and route of transmission of this virus are not known. This and the prior outbreaks occurred during summer months. Previously reported outbreaks were preceded by introduction of a new animal, 14 but that was not the case in this Alaskan outbreak. Given that reported outbreaks have been confined to Canada and Alaska, it is tempting to speculate that a wild lagomorph native to these areas, such as the snowshoe hare, may serve as a reservoir. Clinical disease occurring in this rabbitry the following year suggests viral reactivation from latent infection, but unfortunately tissues were not available for study. Serologic studies are needed, but the sera available from this outbreak were not sufficient for adequate testing. Further studies of this virus to include viral sequencing and experimental infection are in progress.
Based on pathologic findings and biologic character, we conclude that this virus is a novel herpesvirus, most likely an alphaherpesvirus. It is a naturally occurring virus that is highly pathogenic in mini Rex and crossbred meat breed rabbits and may prove useful as an animal model of herpesvirus infection in humans.
