Abstract
An outbreak of tracheitis, sinusitis, and conjunctivitis, originating in recently imported birds, caused high morbidity and mortality in a flock of finches in Central Illinois. Although several species were present, Gouldian finches (Erythrura [Chloebia] gouldiae) were most commonly and severely affected. Birds submitted for necropsy displayed microscopic lesions characteristic of herpesviral infection, including epithelial cytomegaly and karyomegaly with basophilic, intranuclear inclusion bodies in the nasopharynx, sinuses, trachea, parabronchi, conjunctiva, and occasionally the lacrimal gland or proximal proventricular glands. Viral particles consistent with herpesvirus were visualized within affected epithelial cells with electron microscopy. Based on a partial sequence of the viral DNA polymerase gene, this virus was found to be identical to a herpesvirus previously implicated in a similar outbreak in Canada and is most likely an alphaherpesvirus.
Herpesviruses are divided into three subfamilies: Alphaherpesvirinae, Betaherpesvirinae, and Gammaherpesvirinae, which have historically been categorized based on biologic properties, including speed of replication, cytolytic properties, and breadth of host range. 3, 6 With advances in genetic sequencing technology, however, the comparison of DNA sequences of highly conserved motifs within the DNA polymerase gene has resulted in new findings in the phylogenetic relationships between herpesviruses. 12 In mammalian herpesviruses, most of these molecular relationships have correlated well with the historical subfamily relationships, but those in avian herpesviruses have not.
Avian herpesviruses cause some of the more familiar diseases of birds, such as Marek's disease, Pacheco's disease, and infectious laryngotracheitis. Many additional herpesviruses have been identified in cases of avian disease, 6 but few have been fully characterized. Based on their clinical features and behavior in cell culture, Marek's disease virus and Pacheco's disease virus were originally placed into the gammaherpesvirus subfamily, 7 while the infectious laryngotracheitis virus fell into the alphaherpesvirus subfamily. 4, 9 However, recent sequencing of the herpesviral DNA polymerase gene has not supported the placement of Marek's or Pacheco's disease viruses in Gammaherpesvirinae. In fact, all avian herpesviruses analyzed to date are most closely related phylogenetically to the alphaherpesvirus subfamily. 1, 14, 15 As with mammalian alphaherpesviruses, some of these viruses have broad host ranges, and many cause lifelong, latent infections. Infected birds may shed viral particles with or without clinical signs. In most cases, herpesviruses cause mild, subclinical, latent infections in host-adapted species, but they can cause severe, life-threatening disease in other species. 3
This report details an outbreak of herpesviral conjunctivitis and respiratory disease resulting in the death of approximately 250 Gouldian finches (Erythrura [Chloebia] gouldiae) and 19 finches of other species in a private aviary housing approximately 700 finches. Sequencing of a variable region between highly conserved motifs of the DNA polymerase gene from a herpesvirus implicated in this outbreak indicated that it is identical to the passerid herpesvirus 1 (PHV-1), sequenced by Wellehan et al, 15 from a 1999 Canadian outbreak. Other similar outbreaks have previously been observed in Europe. 2, 11, 13 However, this is the first documentation of such an outbreak in the United States.
Materials and Methods
Clinical history
In the summer of 1998 in central Illinois an outbreak of respiratory disease occurred with high morbidity and mortality in a flock of approximately 700 finches of several African and Australian species. The outbreak started in a small group of approximately 20 Gouldian finches imported from Canada as part of a shipment of 70 birds of mixed species and subsequently spread to the main flock. The imported birds were housed in a quarantine room in a building separate from the main aviary. After 2 weeks of quarantine these isolated birds began showing clinical signs of weight loss, labored breathing, respiratory snicks, and unilateral or bilateral conjunctivitis, with red, swollen, crusty, adherent eyelids. Affected birds huddled near heat lamps and attempted to eat and drink with apparent difficulty. Death invariably ensued within 14 days of the first signs of illness, with some deaths occurring prior to the onset of clinical signs.
Bacterial infection of the respiratory tract was initially suspected due to the rapidity of disease spread, combined with the clinical presentation of conjunctivitis and respiratory distress. Affected birds were treated with oxytetracycline ophthalmic ointment and enrofloxacin in the drinking water at 200 mg/liter (Bayer, Shawnee Mission, KS) but showed no improvement in clinical signs or decrease in morbidity rate after 4 days of treatment. At this time 5 dead Gouldian finches were submitted to the Veterinary Diagnostic Laboratory at the University of Illinois for necropsy. Bacterial cultures of conjunctival swabs from live, affected birds in the quarantine room revealed Staphylococcus aureus and several environmental bacterial species. Based on the sensitivity pattern of the S. aureus isolate, antimicrobial therapy was changed to cephalexin at 53 mg/kg (Ranbaxy Laboratories Ltd, Jacksonville, FL) orally every 8 hours, again with no improvement in clinical signs or mortality rate. All birds in the quarantine group died within a few days of the initiation of cephalexin and within 14 days of the onset of the outbreak. Unfortunately, the species present in this group of birds was not documented.
Two weeks after the initial outbreak in the quarantine facility scattered birds in the main aviary began showing similar clinical signs. Approximately 250 of the 700 birds in the main aviary were Gouldian finches of various color mutations; the remainder of the flock was composed of various African and Australian finch species (Table 1). The first birds to show clinical signs were a clutch of Gouldian chicks the owner had banded after visiting the quarantined birds. Clinical signs were similar to those described in the quarantined group and included respiratory distress, severe conjunctivitis (Fig. 1), and sinusitis, despite a good appetite. Also, occasional birds expired without premonitory clinical signs. As soon as histologic lesions consistent with herpesviral infection were identified in the submitted birds, acyclovir was administered via the water at 800 mg/liter to remaining birds, with no change in mortality. Ultimately, Gouldian finches within this flock experienced nearly 100% morbidity and mortality (only two survived, and these two never showed clinical signs). A few birds of other species within the flock died during the outbreak (Table 1), but none displayed conjunctivitis or respiratory distress prior to death. The youngest birds to die were approximately 10 days of age. In all, more than 40 birds that died during the outbreak were submitted to the Veterinary Diagnostic Laboratory.
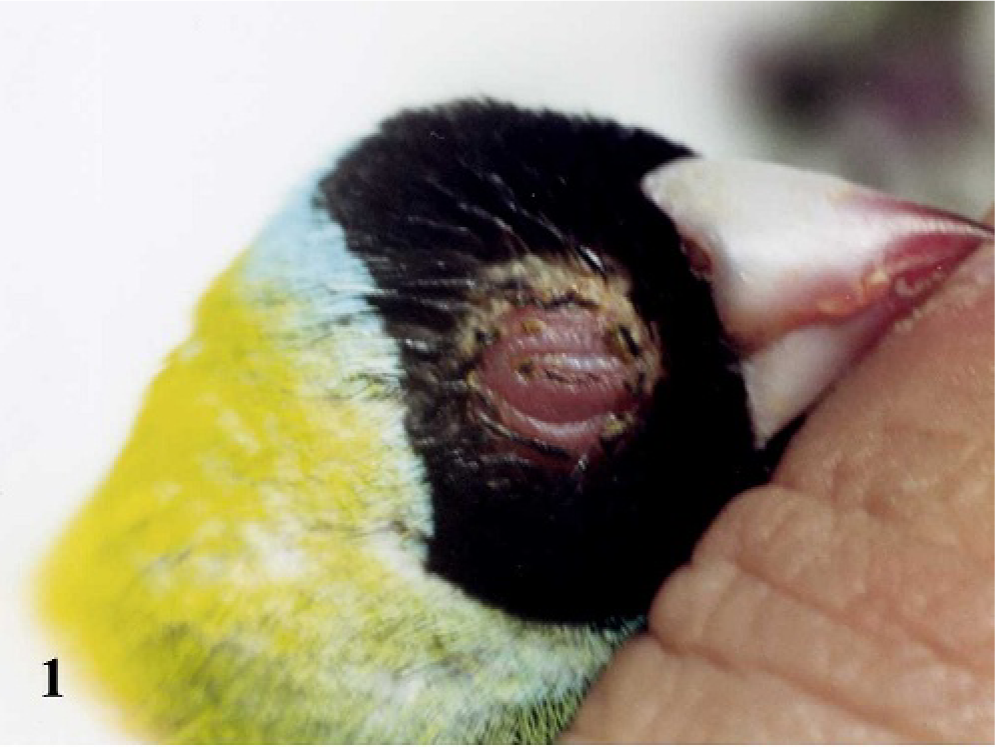
Head; Adult male Gouldian finch showing hyperemic, swollen eyelids with crusting of the surrounding feathers and moderate distension of the infraorbital sinus typical of affected birds during the outbreak.
Mortality by species during the outbreak.
Although society finches (Lonchura domestica) were used as foster birds, none developed the disease; however, the Gouldian finch chicks in their care all died with clinical signs of the disease.
Tissue processing and light microscopy
Initially, 5 freshly dead Gouldian finches were presented for necropsy. At necropsy, representative tissues from all major organs, including the entire head, were collected and fixed in 10% neutral buffered formalin. After formalin fixation, heads were placed in formic acid sodium citrate solution until decalcified. All tissues were embedded in paraffin blocks, sectioned at 3–4 μm, and stained with hematoxylin and eosin by routine techniques. At the peak of the outbreak approximately 35 additional dead birds were submitted. These carcasses were frozen whole and held at -80°C for further testing.
Ultrastructural analysis
At necropsy, 1-mm thick pieces of conjunctiva and trachea were fixed in Karnovsky's fixative in phosphate-buffered 2% glutaraldehyde and 2.5% paraformaldehyde. These were processed for transmission electron microscopy (TEM) using a standard microwave procedure at the University of Illinois at Urbana-Champaign Center for Microscopic Imaging (http://treefrog.cvm.uiuc.edu/meth_stdMW.html). Briefly, tissues were postfixed in 2% osmium tetroxide (Ted Pella, Inc, Redding, CA), treated with 3% potassium ferrocyanide, and washed. Tissues were then stained with saturated uranyl acetate, dehydrated in progressively increasing concentrations of ethanol, treated with acetonitrile as a transition fluid, and then embedded in Epon substitute Lx112 (Ladd Research Industries, Wiliston, VT). Ultra-thin sections were cut and stained with uranyl acetate and lead citrate. The tissue sections were examined and photographed with a 75KV Hitachi H600 transmission electron microscope.
A 2-ml aliquot of each sample prepared for virus isolation attempts, as described below, was also prepared for TEM using a standard negative stain procedure at the University of Illinois at Urbana-Champaign Center for Microscopic Imaging (http://treefrog.cvm.uiuc.edu/meth_negstain.html). Briefly, the 2-ml aliquot was diluted to 3 ml total volume in water and ultracentrifuged for 60 minutes at 48,800 × g at room temperature. The pellet was resuspended in a small amount of water. A 15-μl drop of this suspension was placed on a Parafilm sheet, and a plastic-coated grid was floated facedown on the sample for 5–10 minutes. The grid was then removed from the suspension, gently blotted, and placed on a 15-μl drop of 2% ammonium molybdate (pH 6–6.5) in water, where it was incubated for 2 minutes at room temperature. After drying, the grid was examined and photographed on the electron microscope as described above.
Virus isolation attempts
Tracheas from thawed birds, previously stored at -80°C, were isolated, macerated, and suspended in 10 ml of minimum essential medium (ATCC, Manassas, VA) with 100 mg/ml gentamicin (Sigma, St. Louis, MO), 20,000 U/ml penicillin-streptomycin (Sigma), and 10 mg/ml amphotericin B (Sigma,). Tissues were then homogenized with a Stomacher 80 (Seward Ltd, Norfolk, UK) for 2 minutes at room temperature. The suspension was centrifuged at 500 × g for 20 minutes at 4°C. The supernatant was filtered through a 0.45-nm nylon membrane syringe filter, and a 2-ml aliquot of the filtrate was submitted for TEM with negative staining. Remaining filtrate was used for virus isolation attempts.
Continuous cell lines LMH (chicken hepatocytes) and QT (quail fibroblasts) were used for virus isolation attempts. LMH cells were maintained in Waymouth's medium MB 752/1 (Invitrogen, Carlsbad, CA) with 10% fetal bovine serum (Sigma), 100 mg gentamicin/liter, 5 mg amphotericin/liter, 200,000 U penicillin/liter, and 200 mg streptomycin/liter. QT cells were maintained in a mixture of M199 and F10 media consisting of a 141°109 (V°V) mixture of M199 medium and F-10 nutrient mixture (both from Invitrogen), supplemented with 10% fetal calf serum (Sigma), 10% tryptose phosphate, 0.1 M HEPES (pH 7.4; Sigma) and 10 μg/ml gentamicin.
Both cell lines were grown to 100% confluence in 25-ml cell culture flasks (Corning Inc., Corning, NY). The medium was removed from 2 flasks of each cell line and replaced with 0.5 ml of inoculum, which consisted of a 1°5 dilution of the tissue filtrate described above in fresh tissue culture medium. Flasks were rocked for 1 hour at room temperature, after which an additional 10 ml of culture medium was added to each flask. Flasks were then incubated at 37°C in 5% CO2. An additional flask of each cell line was maintained at 100% confluence under identical conditions to serve as uninfected controls. Flasks were observed daily under an inverted microscope for evidence of cytopathic effect for 8 days. At this time cells were detached from the flasks by removing the medium and rinsing the cells with 0.25% trypsin (Sigma), after which they were incubated for 5 minutes at 37°C until the cells detached with gentle tapping. One-half ml of the resultant suspension from each flask was then transferred to a new flask, and 8 ml of fresh culture medium was added. These flasks were incubated at 37°C in 5% CO2 and monitored daily for 8 days, after which cells were scraped from the flasks, fixed in glutaraldehyde, and the suspension subjected to negative stain TEM in a fashion similar to the 2-ml aliquot of filtrate as described above.
Four, specific pathogen–free (SPF), embryonated chicken eggs at 10 days of incubation were obtained (Sunrise Farms, Catskill, NY). The chorioallantoic membranes (CAMs) of these eggs were inoculated with 0.2 ml of tissue filtrate (described above) and incubated for 5 days at 42°C. Only 2 of the eggs remained viable after 5 days of incubation. The viable eggs were refrigerated overnight and opened the following day. No lesions were noted grossly so histopathology was performed on the CAMs and embryos.
Polymerase chain reaction amplification of the herpesviral DNA polymerase gene
DNA was isolated from the tracheal tissue of 2 carcasses held at -80°C, using a DNeasy tissue kit (Qiagen Inc, Valencia, CA). A region encoding highly conserved motifs (motifs A, B, and C) in the herpesviral DNA-dependent DNA polymerase gene was amplified using a degenerate forward (GAY TTY GCN AGY YTN TAY CC) primer, 12 a reverse primer (CGT TGA TAG ACC ACC CGA CGC TTT CTG C), 15 and Platinum High Fidelity Taq (Invitrogen). Polymerase chain reactions (PCR) were carried out in a Minicycler thermocycler with a heated lid (M.J. Research Inc, Watertown, MA) for a single denaturing cycle at 95°C for 60 seconds, followed by 45 cycles of denaturing at 95°C for 30 seconds, annealing at 46°C for 60 seconds, and extension at 72°C for 60 seconds, after which the sample was maintained at 4°C overnight. The size of the PCR product was verified at slightly less than 500 bp on a 0.7% agarose gel. The amplicons were A-tailed and cloned into the pGEM-T Easy vector (Promega, Madison, WI). Viral DNA from each bird was sequenced separately. Forward and reverse sequencing was completed by the W. M. Keck Center for Comparative and Functional Genomics, University of Illinois, using the standard 21-bp M-13 forward and reverse primers. Our sequence was compared to the PHV-1 sequence submitted to GenBank by Wellehan (accession number AF520812) using BLASTN (National Center of Biotechnology Information, Bethesda, MD). Phylogenetic analysis of predicted protein sequences was done by using the TREE-PUZZLE version 5.2 software package 10 (http://www.tree-puzzle.de). Sequences reported here were aligned with those reported by Wellehan et al and those of several representative mammalian and avian alphaherpesviruses using Dialign (Göttingen Bioinformatics Compute Server, Göttingen, Germany). 8 Representative alphaherpesviruses and the GenBank accession numbers of their DNA polymerases used were: bovine herpesviruses 1 (GenBank accession number X94677) and 2 (AF181249); columbid herpesvirus 1 (AF141890); equine herpesviruses 1 (AY665713) and 4 (AF030027); feline herpesvirus 1 (AJ224971); gallid herpesviruses 1 (AF168792), 2 (AF243438), and 3 (AB049735); human herpesviruses 1 (AB231457) and 3 (X04370); meleagrid herpesvirus 1 (AF282130); and psittacid herpesvirus 1 (AY372243).
A phylogenetic tree was generated using a quartet puzzling algorithm implemented by TREE-PUZZLE. 10 First, unrooted trees were generated by maximum likelihood analysis using the Jones, Taylor, and Thornton evolutionary model for protein sequences. 5 Then 1,000 quartet puzzling steps were implemented to obtain intermediate trees, and finally the majority rule consensus tree was determined and reported. 10
Results
Gross, microscopic, and bacterial culture data
At necropsy, most birds were moderately emaciated and had swollen eyelids with matted periocular feathering. Occasional birds had yellow-white, caseous exudate in the trachea, sinuses, or both. Some also had lesions consistent with septicemia, such as mild enlargement of the liver and/or spleen, or pinpoint, depressed, white foci on these organs.
Microscopic examination of the respiratory tracts and conjunctivae revealed markedly enlarged epithelial cells (cytomegaly) with basophilic intranuclear inclusion bodies throughout the respiratory tract and conjunctiva (Figs. 2 and 3). Usually the cytomegalic cells formed large, cohesive rafts that sometimes sloughed into the lumen. Affected epithelial cells frequently exceeded 40 μm in diameter with abundant, fibrillar, basophilic cytoplasm. Nuclei were also severely enlarged, measuring up to 30 μm in diameter (karyomegaly). Affected nuclei were pleomorphic and usually had an irregularly shaped, 10- to 20-μm diameter, centrally located, basophilic inclusion. Variable amounts of necrotic cellular debris were present in airway lumens, and in some birds there were large colonies of cocci in the lungs and sinuses. In affected areas, the submucosa was infiltrated by scattered lymphocytes, plasma cells, macrophages, and rare heterophils. Proventricular and lacrimal glands also displayed variable involvement with karyomegaly and cytomegaly of epithelial cells, as well as single-cell necrosis. The grossly visible white foci on the livers and spleens of some birds were histologically confirmed to represent small foci of necrosis. The microscopic findings in the respiratory tracts and conjunctivae were consistent with a herpesviral infection, 14 while those in the spleen and liver were likely due to secondary bacteremia.
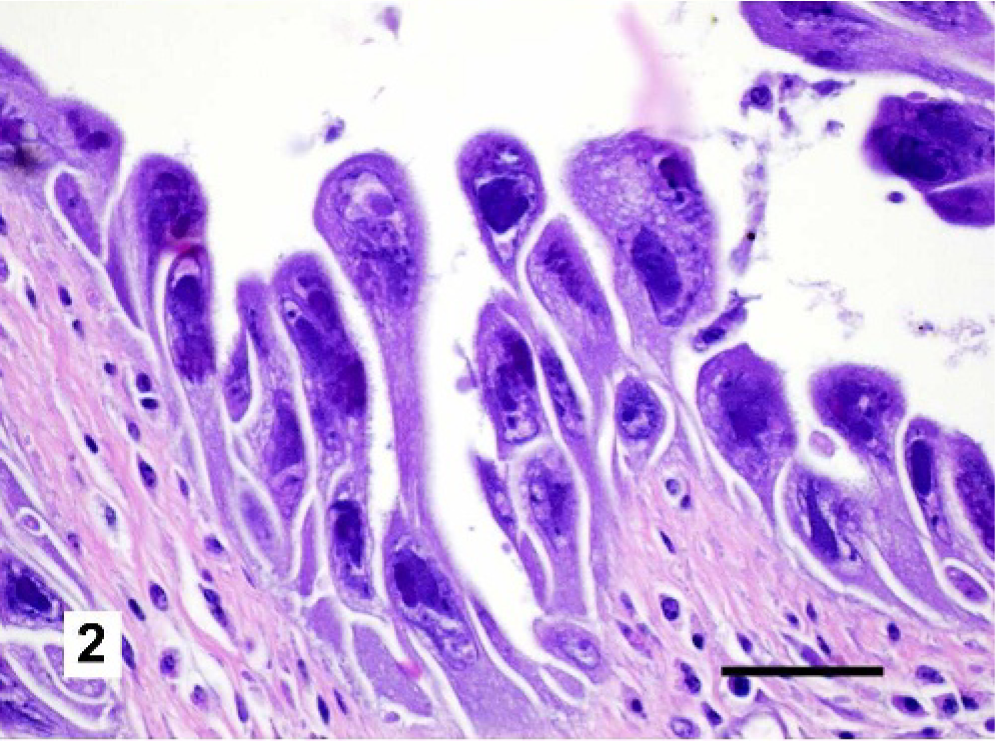
Trachea, Gouldian finch. Respiratory epithelial cells are markedly enlarged with cytokaryomegaly and large basophilic intranuclear inclusion bodies. These cells pile upon one another and slough into the tracheal lumen. Scattered lymphocytes and plasma cells are present within the submucosa. HE. Bar = 50 μm.
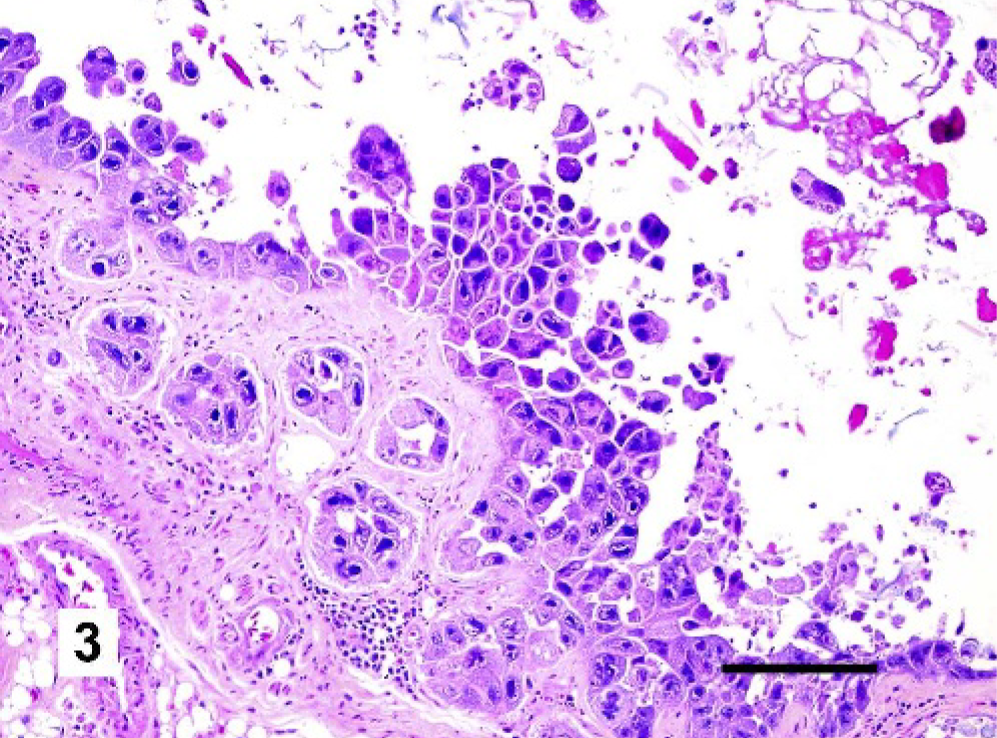
Nasal cavity, Gouldian finch. Epithelial cells within the nasal cavity and submucosal glands are markedly enlarged with cytokaryomegaly and large basophilic intranuclear inclusion bodies. Necrotic cellular debris and plant material are present in the lumen. Accumulations of lymphocytes and plasma cells are present between submucosal glands. HE. Bar = 100 μm.
Bacterial culture of the conjunctiva of live, affected birds in the quarantine room yielded heavy growth of S. aureus and several environmental bacterial species. Similar results were obtained from cultures of livers, air sacs, conjunctiva and sinuses of the dead birds at necropsy.
The only microscopic change in the CAMs or embryos from the virus-inoculated SPF eggs was a small focus of fibrosis on each of the CAMs. This fibrosis was interpreted as mild scarring secondary to exposure to the inoculum. However, no evidence of viral or bacterial infection was present.
Electron microscopy
Ultrastructural evaluation of affected cells revealed the intranuclear inclusions to contain scattered viral particles with the morphologic characteristics of a herpesvirus (Figs. 4 and 5). Cellular changes included large numbers of cytoplasmic vesicles containing nucleocapsids. The nucleocapsids showed icosohedral symmetry, measured 150 nm in diameter, and were often enveloped. Additionally, infected epithelial cells showed loss of cellular cilia and tight junctions. Electron microscopy of aliquots prepared for virus isolation attempts also showed numerous viral particles with features consistent with a herpesvirus.
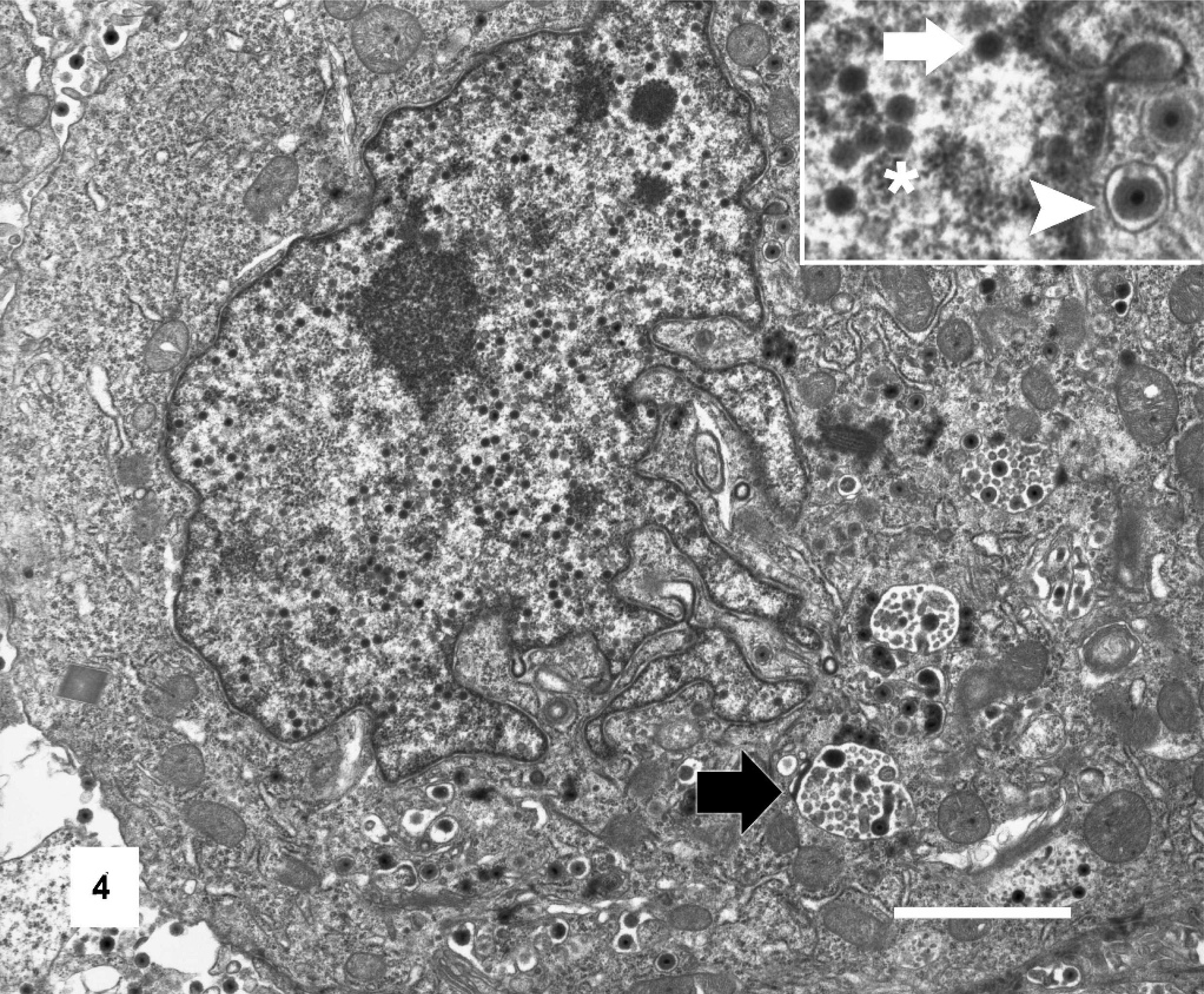
Tracheal epithelial cell, Gouldian finch. Affected tracheal epithelial cell displaying karyomegaly and cytomegaly with loss of cilia. Viral particles are present within cytoplasmic vesicles (black arrow). Inset: Detail of nuclear envelope with non-enveloped viral particles (white arrow) within the nucleus (∗) and enveloped viral particles (arrowhead) within the cytoplasm of cell. Transmission electron micrograph. Bar = 2 μm.
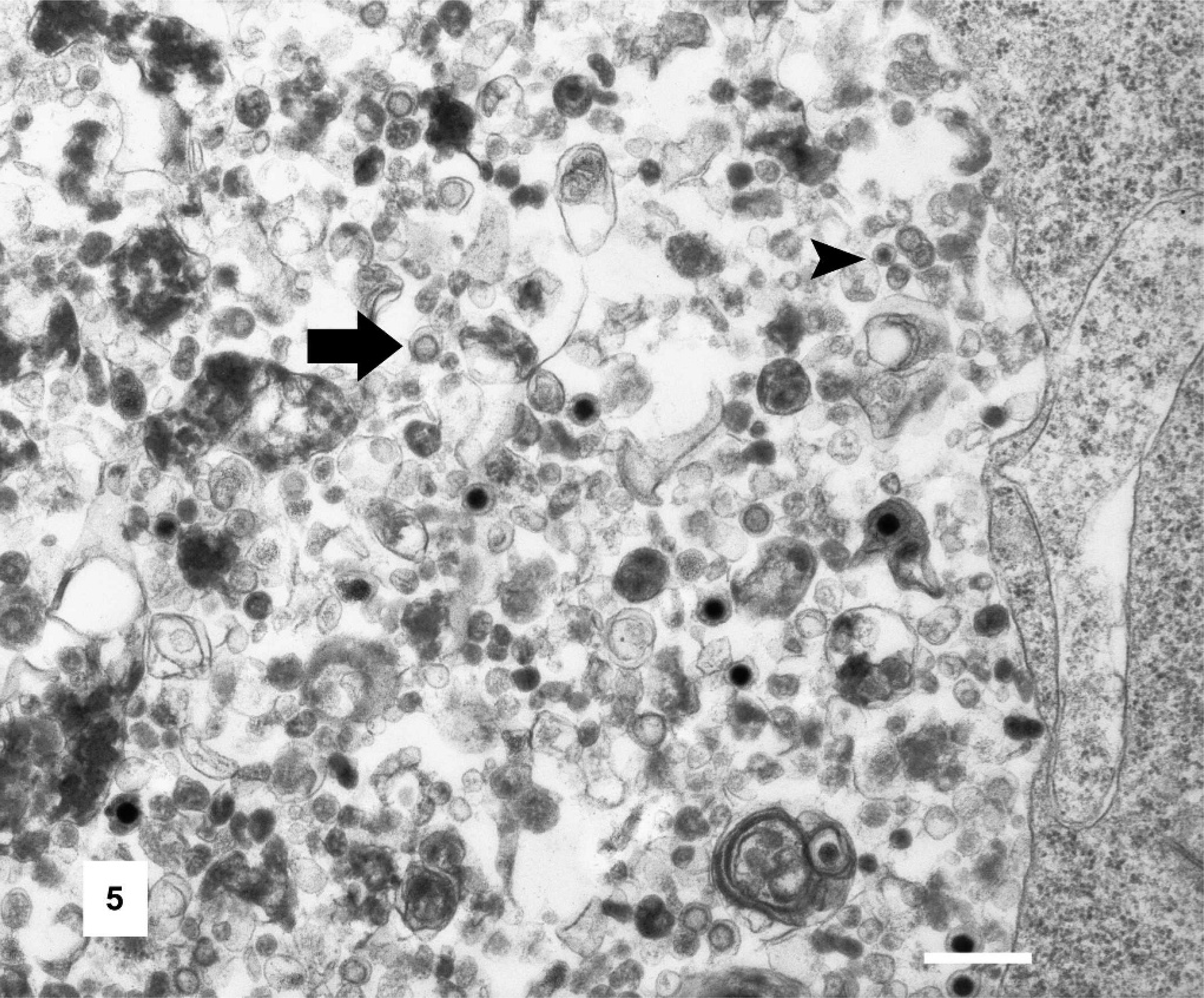
Tracheal epithelial cell, Gouldian finch. Enveloped (arrows) and non-enveloped (arrowhead) viral particles are identified within cytoplasmic vesicles. Transmission electron micrograph. Bar = 500 nm.
PCR amplification of the herpesviral polymerase gene
A 470-bp amplicon was generated from the viral DNA using the viral DNA polymerase primers described above. As expected, 175 bp at the 3′ end were 100% homologous to the fragment sequenced by Wellehan et al. 15 This particular region of the herpesviral DNA polymerase gene appears to be diverse among herpesviruses, allowing distinction between species. 12 This finding confirms that the herpesvirus implicated in our outbreak is indeed the same as that implicated in the Canadian outbreak (PHV-1). The remaining 295 bp represent new data for this virus. This segment was identical in the 2 birds from our study with the exception of 1 bp. The difference occurs at the 71st bp and results in a difference in the predicted amino acid sequence, coding either a proline or serine. Both complete sequences have been submitted to GenBank under accession numbers DQ287312 and DQ287313, respectively.
Phylogenetic analysis of sequences
The predicted amino acid sequences obtained were compared to the sequences of DNA polymerases of other herpesviruses to determine their phylogenetic relationships. The single amino acid difference between the 2 predicted sequences did not affect these results. Both sequences clustered with psittacid herpesvirus 1 (Pacheco's disease virus) and gallid herpesvirus 1 (infectious laryngotracheitis virus) (Fig. 6) in the constructed phylogenetic tree, as did the 175-bp segment previously submitted by Wellehan et al. 15 Psittacid herpesvirus 1 and gallid herpesvirus 1 have been fully characterized and found to be alphaherpesviruses within the genus Iltoherpesvirus. This clustering supports the classification of PHV-1 as an Iltoherpesvirus in the family Alphaherpesvirinae.
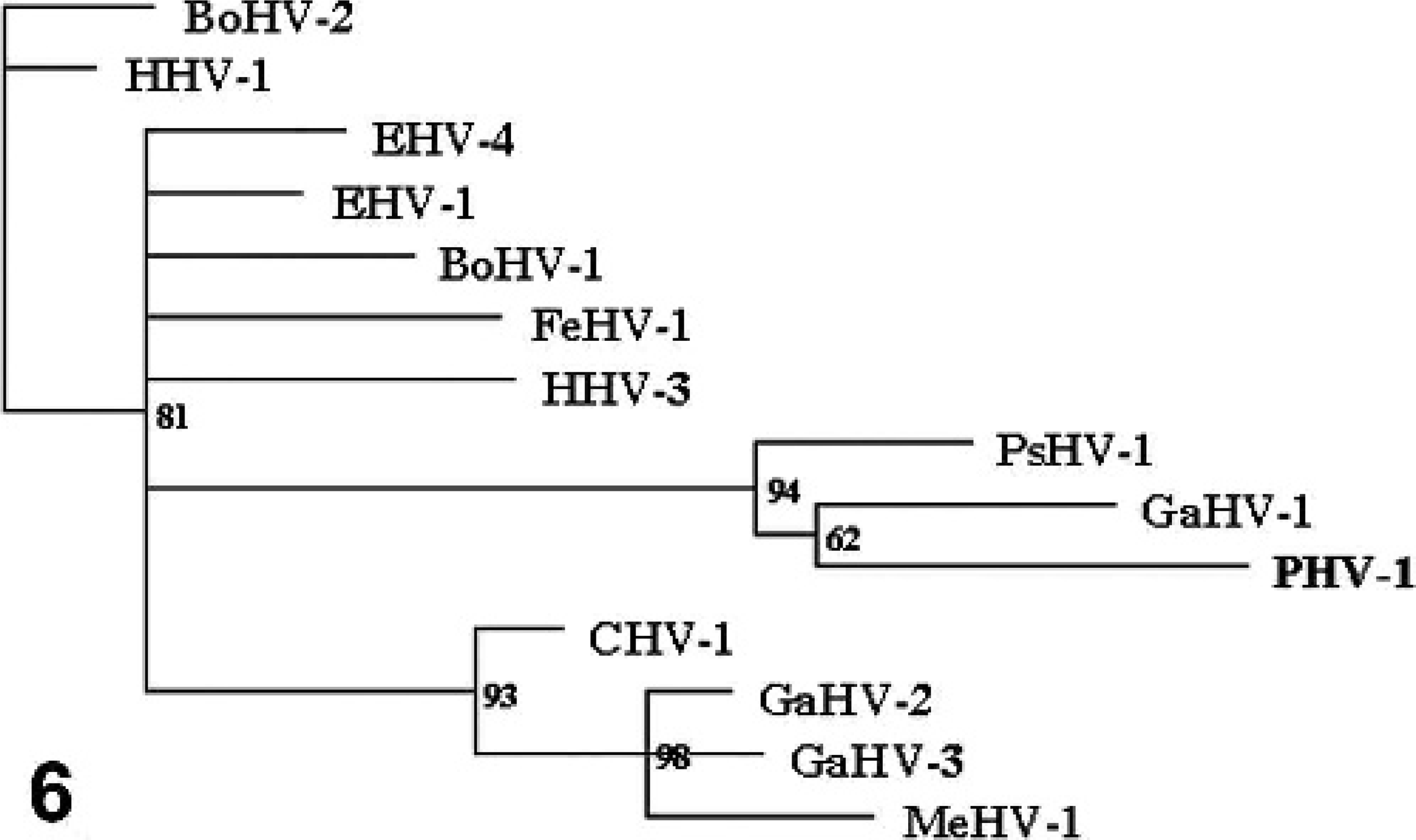
Unrooted phylogenetic tree of homologous predicted protein sequence of select alpha herpesviral DNA polymerases. Passerid herpesvirus 1 (PHV-1) clusters with gallid herpesvirus 1 (GHV-1) and psittacid herpesvirus 1 (PsHV-1). Bootstrap values are shown. The bar represents estimated distances.
Discussion
Similar clinical signs, morbidity, mortality, microscopic lesions, and viral morphology have been described in 3 reported outbreaks in European, mixed-species finch aviaries. These outbreaks occurred in Austria, Switzerland, and Belgium. 2, 11, 13 In all outbreaks, Gouldian finches were the primary species affected, although other species were always present. After a recent Canadian outbreak of the disease, a segment of the viral DNA polymerase gene was sequenced from finch tissues and found to cluster with gallid herpesvirus 1 (infectious laryngotracheitis), an alphaherpesvirus. 15 Additional DNA polymerase gene sequence obtained by our laboratory supports this conclusion.
Successful isolation of the virus implicated in these outbreaks has not been documented. Virus isolation attempts in our laboratory yielded no cytopathic effect, as well as no detectable virions in inoculated tissues or CAMs by negative staining TEM. It is likely that these results are related to the fact that available culture systems were of galliform origin, and herpesviruses tend to show marked species and tissue specificity. 3 Unfortunately, no Gouldian finch, fringillid, or even passerine cell culture lines have been developed. Further possible explanations for the difficulty of isolating this virus include that it may have been inactivated by freezing, the inoculum may have been inadequate for the available culture systems, the timing of inoculations and observations may not have been ideal, or the virus may have been sensitive to any of the solutions, temperatures, or other conditions to which it was exposed.
It is interesting to note that the observed characteristics of this virus are not necessarily those that would be predicted based on generalizations that arise from observations of mammalian alphaherpesviruses and betaherpesviruses. For example, all mammalian herpesviruses causing cytomegaly and karyomegaly that have been characterized to date have been categorized as betaherpesviruses (porcine and human cytomegaloviruses), while the herpesvirus implicated in this outbreak caused similar lesions and yet is likely an alphaherpesvirus. On the other hand, the apparent ability of the virus described here to infect multiple species of finches is consistent with the generalization that alphaherpesviruses tend to have variable host ranges, while betaherpesviruses tend to have more restricted host ranges. 3 Therefore, it may be inappropriate to predict such properties as the rate of replication, cytolytic potential, or pattern of viral shedding (periodic reactivation vs. continual shedding) based on generalizations made regarding data obtained from mammalian herpesviruses.
The biologic behavior of this particular herpesvirus should be experimentally determined in a controlled environment, but some properties may be inferred by the pattern of spread through the aviary. For example, the facts that the youngest birds affected were 10 days and that there was a 2-week interval between the outbreaks in the quarantine flock and the main flock suggest that the incubation period is 10 days to 2 weeks. It is possible, if not likely, that the virus identified in this aviary was introduced into the quarantine facility in a latently infected bird of one of the less severely affected species, after which it spread to the Gouldian finches, in which it was uniformly deadly. Further work is warranted to more definitively establish the phylogenetic relationship of this virus to other avian herpesviruses, to isolate and further characterize the virus, and to identify other avian species that may harbor the virus.
Footnotes
Acknowledgements
We sincerely appreciate the invaluable consultation and laboratory assistance of Gail Scherba and Katherine Kalivoda for cloning expertise, William Schnitzlein for cell lines, Gustavo Gelhon for phylogenetic analysis, and Lou Ann Miller for TEM.
