Abstract
This case report describes the clinical, magnetic resonance imaging (MRI)-related, and pathologic features of a nasal acinic cell carcinoma in a cat. A 16-year-old, castrated male, oriental shorthaired cat, weighing 3.8 kg, was presented with history of sneezing, coughing, and nasal discharge persisting several months. Evaluation by MRI revealed an heterogeneous, space-occupying lesion that filled the left nasal cavity and was diagnosed by histopathologic examination as an acinic cell carcinoma arising from a minor salivary gland of the nasal cavity. Acinic cell carcinoma is a rare tumor in veterinary medicine. The tumor is composed mainly of cells resembling serous cells of salivary glands and originates from major or minor salivary glands. Clinicians and pathologists should be aware of the occurrence of acinic cell carcinoma in the sinonasal tract and include the tumor in the differential diagnosis of feline nasal diseases.
Keywords
Primary tumors of the nasal and paranasal sinuses can arise from any of the cell types found in that region. Reported prevalence values for sinonasal tumors range between 1% and 8.4%, with the vast majority (approximately 90%) being malignant. 12 Of those, malignant lymphomas are the most common feline nasal and paranasal sinus tumors. 12 Among epithelial tumors, adenocarcinomas are the most common. 12 To the author's knowledge, nasal acinic carcinoma has not been previously reported in veterinary medicine.
A 16-year-old, castrated male, oriental shorthaired cat, weighing 3.8 kg, was referred to the Department of Clinical Veterinary Medicine, Division of Small Animal Internal Medicine, with history of sneezing, coughing, and nasal discharge persisting several months. The cat had been treated previously with antibiotics and corticosteroid drugs without any signs of clinical improvement. During the two weeks prior to referral, the cat had become more depressed and anorexic. Upon clinical presentation, the cat was bright, alert, and responsive. The cat was squinting. No ocular discharge was observed. An inspiratory and expiratory stridor could be auscultated, and in the left nostril a clear nasal discharge was observed with no airflow. Hematology and serum chemistry values were within normal reference ranges with the exception of an elevated albumin concentration and total solids, most likely consistent with slight dehydration.
Magnetic resonance imaging (MRI) was performed with a 0.3 Tesla open magnet (Airis II, Hitachi Medical Systems, Düsseldorf; Germany). Sequences included a transverse T2-weighting (repetition time [TR], 3540 ms; echo time [TE], 125 ms); a dorsal fat suppressing, short inversion time inversion recovery (STIR; TR, 4305 ms; TE, 25 ms; inversion time [TI], 110 ms), and a dorsal T1-weighted gradient echo (TR, 30 ms; TE, 12 ms). The T1-weighting was repeated after the intravenous application of contrast agent (Omniscan, GE Healthcare, Oseo, Norway) in a dosage of 0.15 mmol/kg body weight. The left nasal cavity was filled with a heterogeneous mass. In T2 and STIR evaluation sequences, the mass appeared moderately hyperintense to muscle; in plain T1, it was isointense, with mainly peripheral moderate contrast uptake. The borders of the lesion were irregular, resulting in a multilobular appearance. The conchal structure of the left nasal cavity was destroyed, and the lesion extended caudally to the nasopharynx, where it crossed the midline toward the right side. The left frontal sinus was filled with isointense fluid material in all sequences and showed mucosal contrast uptake. The bony borders of the nasal cavity were intact. The MRI diagnosis was neoplasia of the left nasal cavity with obstruction of the left frontal sinus opening. The differential diagnosis included adenocarcinoma, lymphosarcoma, or chondrosarcoma.
Due to poor prognosis, the cat was humanely euthanatized, and a complete necropsy was performed. The cadaver was moderately autolytic. Transverse section of the nasal cavity revealed a whitish-to-tan, heterogeneous, soft, and infiltrative mass that occupied the left nasal cavity, replacing the conchae. In the caudal portion, the mass invaded focally through the nasal septum into the right cavity (Fig. 1). The right nasal conchae were severely congested. Cross sections of the nasal cavity, including the mass, were fixed in 100% neutral buffered formalin, processed, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin (HE), periodic acid Schiff (PAS) with and without diastase digestion, alcian blue, and mucicarmine. Additional sections were stained by the LSAB (peroxidase/AEC) method for cytokeratin (polyclonal rabbit anti-bovine cytokeratin; Z0622) and S-100 (polyclonal rabbit anti-bovine S100; Z0311) antigen (DAKO, Glostrup, Denmark). Histologic examination revealed an exophytic, poorly circumscribed, nonencapsulated, and infiltrative mass adjacent to a minor intranasal salivary gland. The mass had replaced most conchal tissue and consisted of neoplastic epithelial cells resembling serous cells of salivary glands. Neoplastic cells were arranged in two patterns. In one area, they formed numerous lobules of epithelial cells arranged in acini that were surrounded by a delicate fibrovascular stroma. Numerous acini were filled with a homogeneous, amphophilic-to-basophilic secretion and were lined by a single layer of tall, columnar, epithelial cells with basilar nuclei, resulting in a follicular appearance (Fig. 2). Neoplastic cells were well differentiated and had distinct cell borders, a moderate-to-abundant and clear-to-granular basophilic cytoplasm, and eccentrically or centrally located hyperchromatic nuclei. Anisokaryosis and anisocytosis were low. Mitotic counts were less than 1 per 400×field. The second pattern consisted of neoplastic cells with a more solid growth pattern and arrangement in small acini embedded in a delicate stroma. These were lined by one layer of well-differentiated cells, containing prominent basophilic cytoplasmic granules. The granules and secretory products within acini were PAS-positive and diastase resistant (Fig. 3). Furthermore, both were stained with alcian blue and mucicarmine (Fig. 4), indicating the presence of acid mucins. Immunohistochemically, approximately 40% and 70% of neoplastic cells showed cytoplasmic positivity for S-100 and cytokeratin, respectively. Additionally, very few neoplastic cells (approximately 1%) showed nuclear staining for S-100. Multifocal infiltration of lymphocytes, plasma cells, and few histiocytes and a focal area of necrosis and mineralization were observed. The remaining conchae showed multifocal Howship's lacunae, indicating bone resorption. The mucosa that covered the nasal septum was hyperplastic. The nasal mucosa was infiltrated with neutrophils and covered with fibrinous exudate admixed with neutrophils. No evidence of neoplasia was found in the regional lymph nodes or any other organ. Based on histopathologic, histochemical, and immunohistochemical features, a nasal acinic cell carcinoma was diagnosed.
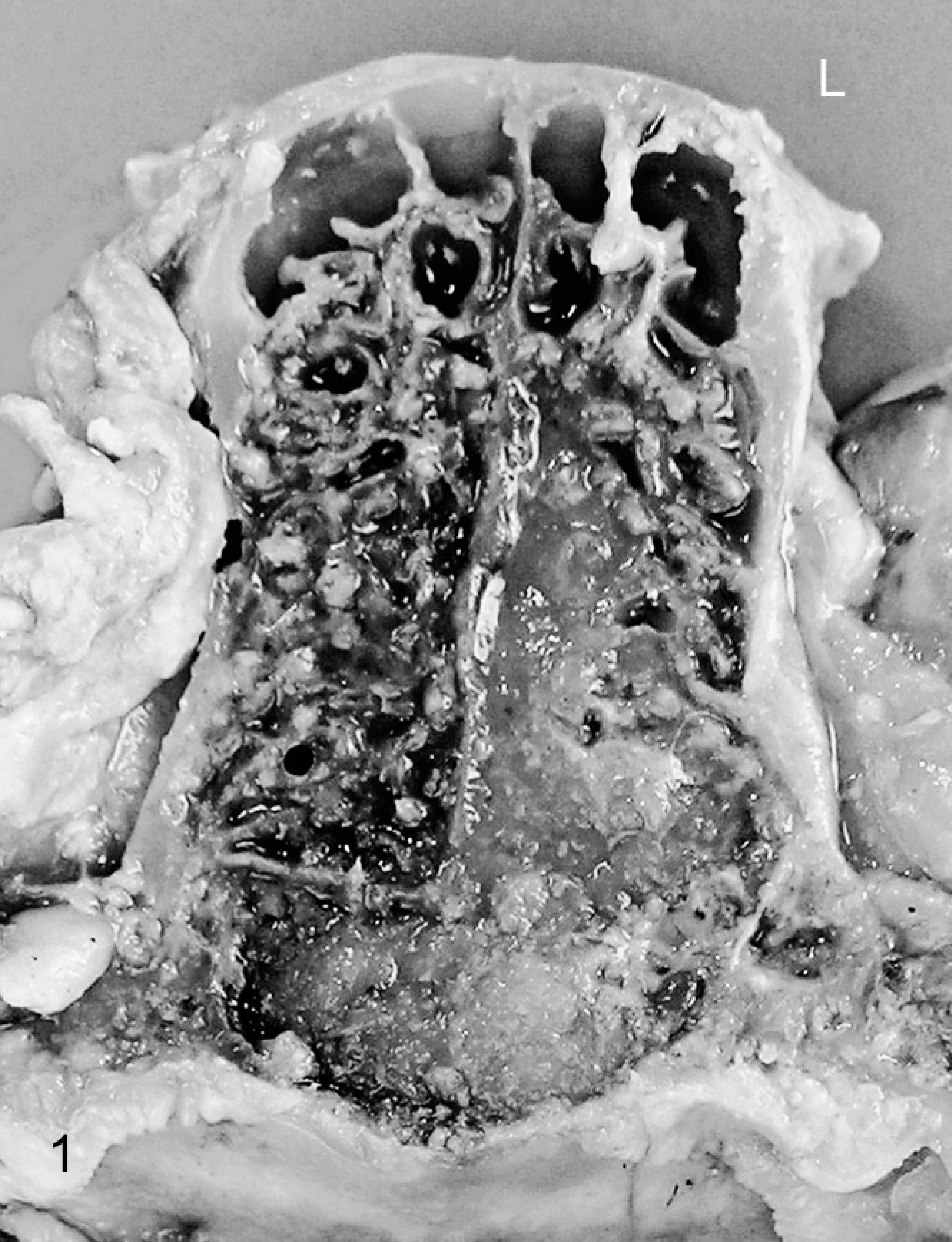
Acinic cell carcinoma; cat. Gross photograph of formalin-fixed specimen. The left nasal cavity is occupied by a tan mass that focally invades through the nasal septum into the right cavity. L = Left side.
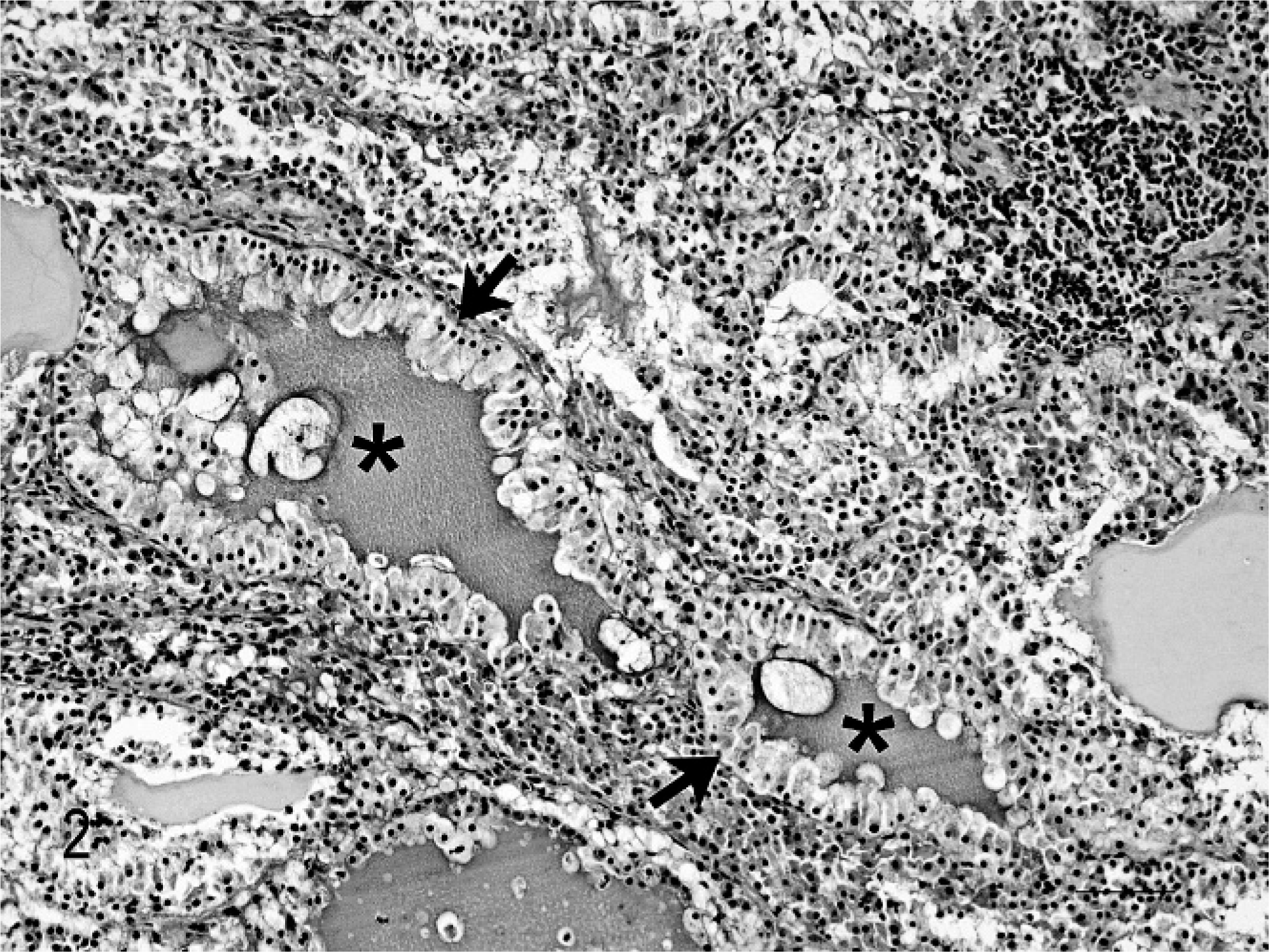
Acinic cell carcinoma; cat. Follicular pattern: Acini are filled with homogeneous secretion (asterisks) and lined by a single layer of tall columnar epithelial cells (arrows) with basilar nuclei. HE stain. Bar = 100 μm.
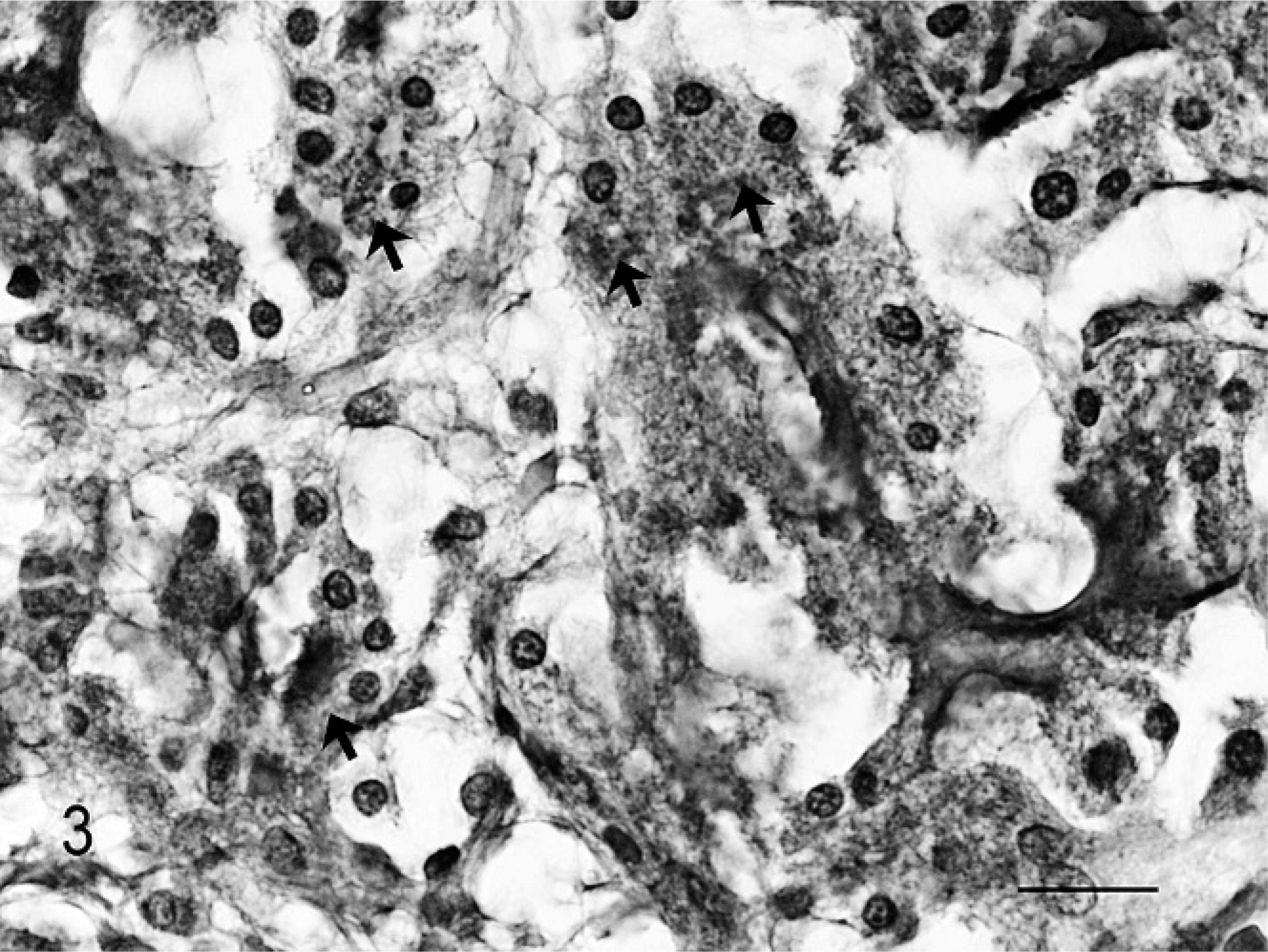
Acinic cell carcinoma; cat. Granules of neoplastic cells are PAS-positive and diastase resistant (arrows). PAS and PAS-diastase digestion. Bar = 20 μm.
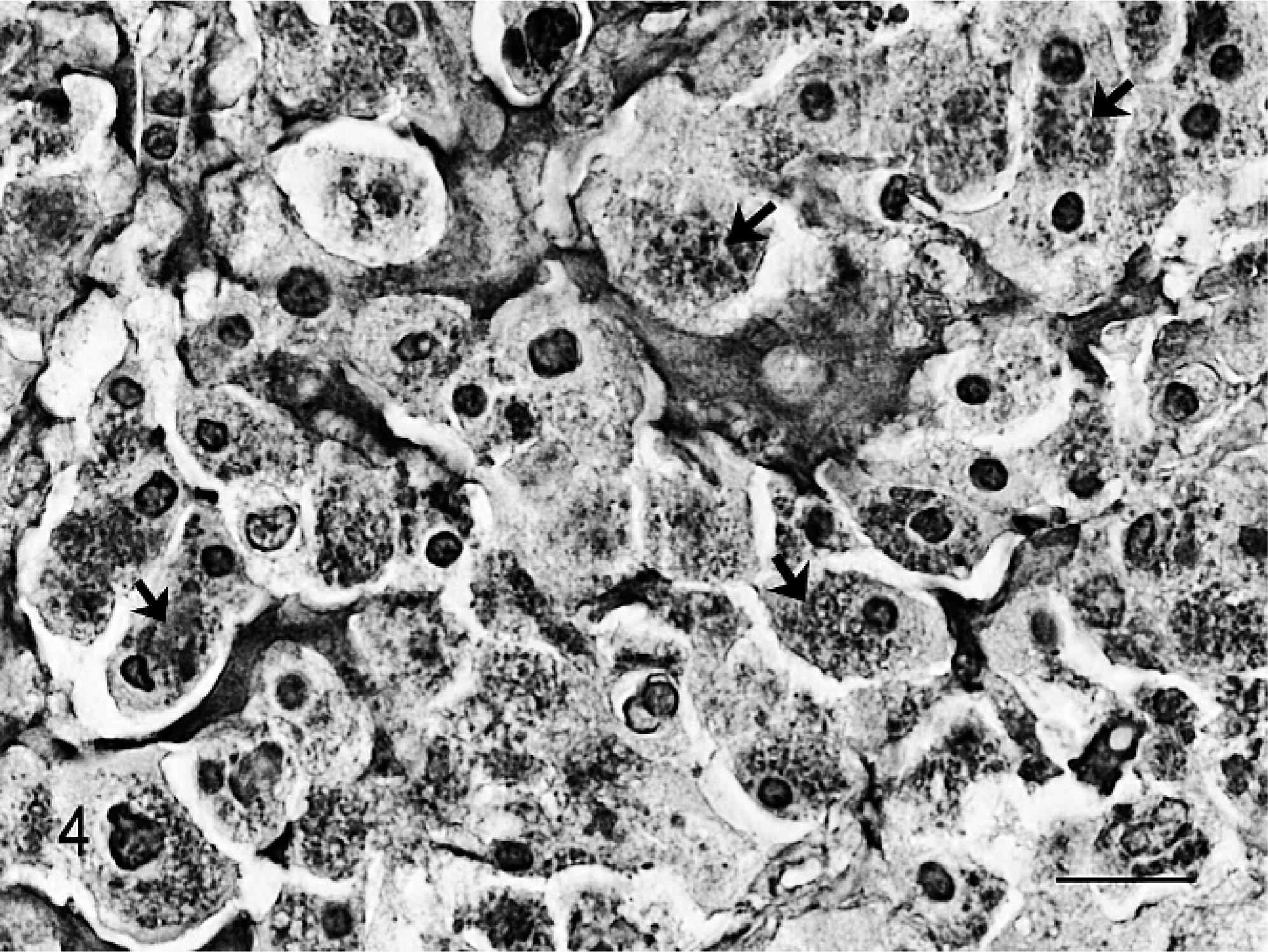
Acinic cell carcinoma; cat. Granules of neoplastic cells are stained with mucicarmine (arrows). Mucicarmine stain. Bar = 20 μm.
Acinic cell carcinomas are low-grade carcinomas consisting mainly of cells closely resembling serous cells of salivary glands. 5 Cytoplasmic basophilia is a characteristic of this neoplasm, and the PAS-positive cytoplasmic granules are typically resistant to diastase digestion, which makes differentiation from other epithelial neoplasms relatively easy. 13 These tumors arise from the major salivary glands (parotid and submandibular salivary glands) or minor salivary glands that are located throughout the upper aerodigestive tract. In human medicine, acinic cell carcinomas are rare, accounting for approximately 1% of salivary gland tumors. 13 These tumors occur most frequently in the parotid gland and less often in the submandibular and minor intraoral salivary glands. 6 The occurrence in the sinonasal cavities is extremely rare. Recurrence is described in 35% of the patients, and death due to metastastic disease occurs in 16%. 13 In veterinary medicine, acinic cell carcinomas of the salivary glands have been reported only sporadically in the dog, originating from the major or minor salivary glands. 2, 10 Moreover, acinic cell carcinoma has been described once in the horse, in the African pygmy hedgehog, and in the cat, respectively. 8, 9, 10 In dogs, the majority of acinic cell carcinomas occur in the parotid gland and are of low malignancy with local invasion but no metastasis. 2, 10 However, they tend to recur after excision. 5 Primary neoplasms of major and minor salivary glands are rarely described in cats. In a retrospective study of 30 feline salivary gland tumors, Siamese and Siamese-cross cats were overrepresented, and most tumors occurred in the submandibular glands. 9 The large majority of these tumors are adenocarcinomas that readily metastasize into regional lymph nodes and distant organs. 9, 11 In contrast, the tumor presented here showed only local invasion and no metastasis.
Acinic cell carcinomas of the salivary glands apparently arise from the intercalated duct reserve cells as a result of the neoplastic proliferation and aberrant cytodifferentiation of pluripotent stem cells that reside at the acinar-intercalated duct junction and in the intercalated duct of mature salivary ducts. 1 Regarding the growth pattern, acinic cell carcinomas are categorized as solid, microcystic, papillary-cystic, lobular, and follicular. The categorization according to cellular features includes acinic, intercalated ductal, vacuolated, clear, and nonspecific glandular patterns. 6 Although an individual tumor usually has a predominance of one pattern or cell type, many tumors represent a mixture of these features. 13 In the present case, a mixed pattern was present with lobular, solid, and follicular growth and predominance of an acinic cell type. The diagnosis of acinic cell carcinoma is supported by routine histochemistry demonstrating the characteristic PAS-positive and diastase-resistant cytoplasmic granules. Furthermore, cytoplasmic granules stained with mucicarmine and alcian blue indicate that some tumor cells showed cytodifferentiation into mucous acinar cells and that the tumor arose from pluripotential duct cells rather than from fully differentiated serous cells. In human medicine, variable staining of acinic cell carcinomas for mucicarmine has been described. 3, 7 The expression of immunohistochemical markers in acinic cell carcinomas is less consistent. They may be positive for S-100 and cytokeratin, as in the present case. 4 Additionally, positivity for α-amylase and α-antitrypsin has been described, but none of these markers have been proved specific. 4 Furthermore, there is a high variation in intensity and distribution of positive staining, not only between tumors but also within the same tumor.
To the author's knowledge, this is the first report of a nasal acinic cell carcinoma in a cat. Veterinarians and veterinary pathologists should be aware of acinic cell carcinoma, which is readily distinguished from other neoplasms by histopathology and histochemistry, and include this rare tumor in the differential diagnosis of feline nasal diseases.
Footnotes
Acknowledgements
We thank Valérie Bornand and Peter Girling for the critical reading of the manuscript.
