Abstract
Mucoepidermoid carcinoma is a malignant tumor that arises from the salivary glands. The recommended treatment strategy typically involves surgical intervention, sometimes complemented by radiotherapy, depending on the histological grade of the tumor. A case of a 22-year-old female patient without medical history was described. The clinical examination revealed a bluish lesion located on the hard palate. The histological examination confirmed the diagnosis of a low-grade mucoepidermoid carcinoma. Resection of the lesion was performed and oro-nasal communication was immediately closed by a prosthetic obturator and later on by a rotational palate flap. The patient was followed up for 12 months, and there was no evidence of any recurrence. This article highlights the importance of prompt clinical diagnosis of such lesions and provides an opportunity to review these cancer therapeutic measures to reduce postoperative morbidity.
Keywords
Introduction
Tumors affecting the major and minor salivary glands are rare and have a variety of histological, clinical, epidemiological, and developmental features. Due to the low incidence of these tumors, physicians and histopathologists face significant challenges in their treatment. Among salivary gland tumors, pleomorphic adenoma is the most common benign tumor, while mucoepidermoid carcinoma (MEC) is the most common malignant one. 1
These tumors exhibit different biological behavior, with high-grade tumors being aggressive presenting poor prognosis, while low-grade tumors are often benign and have acceptable survival rates.
Treatment of salivary gland cancer depends on tumor location, clinical features, and histopathological grade. Typically, standard treatment involves surgical resection and adjuvant radiation therapy to reduce the risk of treatment failure. Although the effectiveness of adjuvant chemotherapy is still uncertain, it may be used for distantly metastatic, unresectable tumors and can help reducing the risk of recurrence.
The purpose of this article is to emphasize the importance of intraoral examination and complete medical history for early detection of potentially dangerous diseases.
Observation
A 22-year-old female patient was referred to our oral surgery department for a painful swelling on the hard palate that has been developing for 8 months. The patient was in good health with no significant family or medical history. The extraoral examination yielded no notable findings. No palpable lymph nodes were detected.
During the clinical examination, a well-defined bluish collection, approximately 2 × 2.5 cm in size was noticed at the posterolateral region of the hard palate in relation to the second and third right molars. The lesion was well defined with normal overlying mucosa, showing no ulceration or other surface changes (Figure 1). Upon palpation, the swelling was tender and fluctuant with regular smooth borders, and no discharge was observed upon compression.
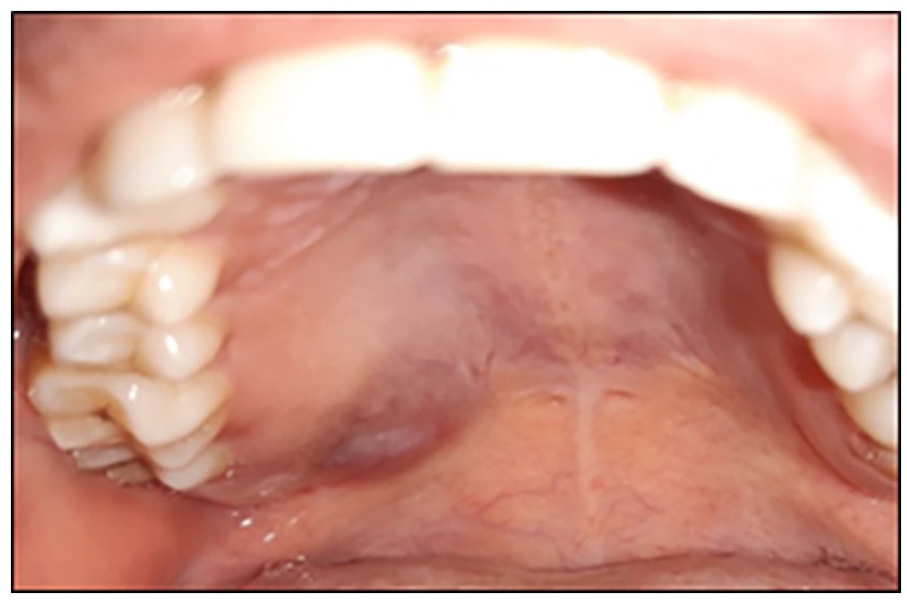
Intraoral view showing pale bluish-purple lump.
On initial examination, the diagnosis of an acute dental abscess related to the third molar was considered due to the decay and negative vitality test.
A fine needle aspiration was performed to investigate the presence of purulent content but instead it returned a yellowish translucent liquid (Figure 2). As a result, the diagnosis of dental abscess was rejected and revised to salivary gland tumors based on the findings.

Needle aspiration yielded a yellow liquid.
A Cone Beam Computed Tomography (CBCT) was recommended to evaluate potential underlying bone damage.
The radiological examination revealed a well-defined osteolytic bone lesion with a polycyclic pattern on the palate adjacent to the first and second upper right molars (16 and 17), without extension to the sinus or nasal cavity (Figure 3).
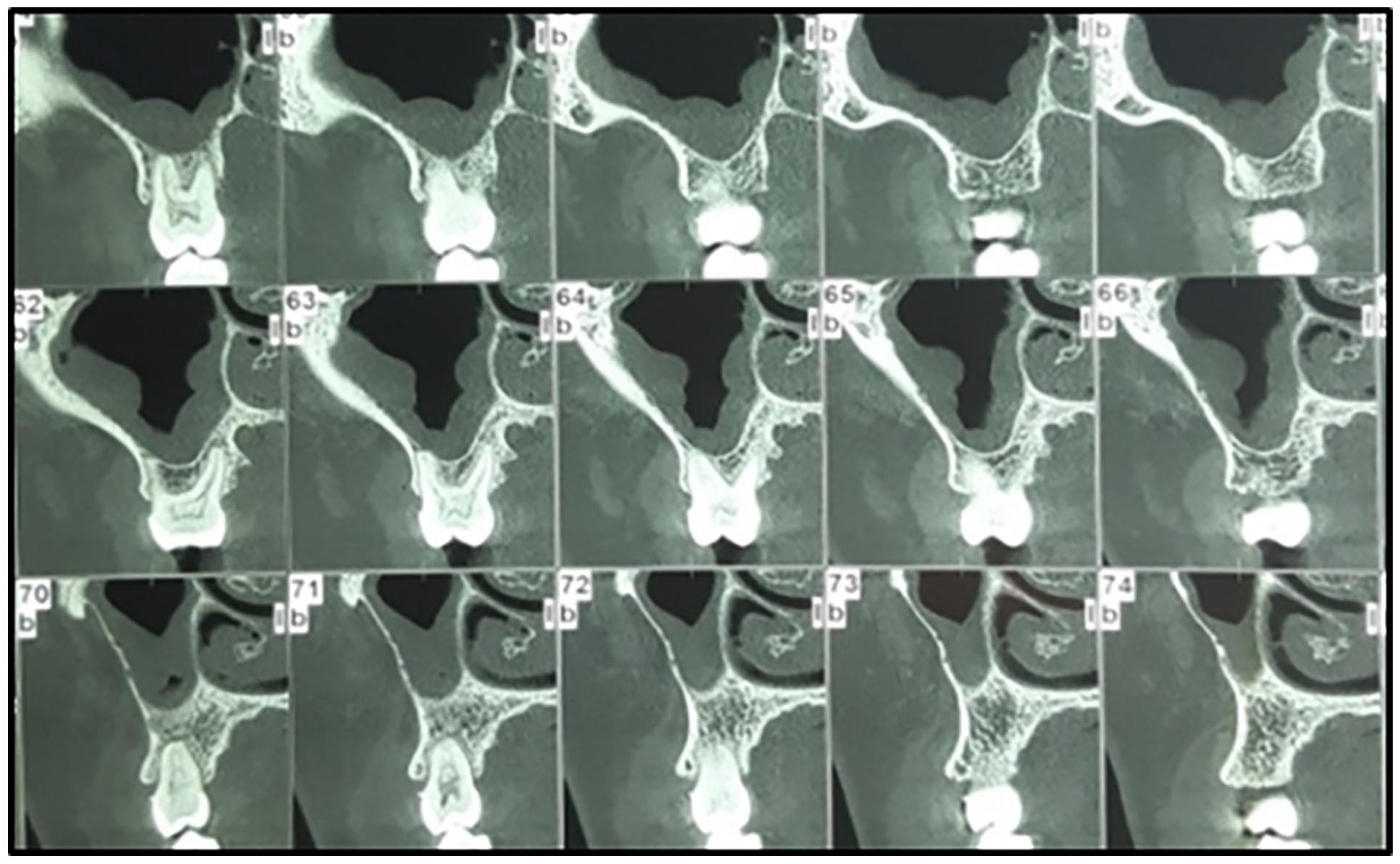
Cone Beam Computed Tomography sectional images showing osteolytic bone lesions with definite corticated borders.
An incisional biopsy was performed in the perilesional area under local anesthesia, and the tissue sample was submitted for histopathological evaluation.
Hematoxylin- and eosin-stained microscopic tissue section showed proliferation of nests of tumor cells containing intermediate and cylindrical cells with clear cytoplasm that are periodic acid-Schiff positive in the connective tissue (Figures 4 and 5).

Photomicrograph (H and E stained sections).

Photomicrographs (periodic acid-Schiff stained sections).
The tumor corresponded to a MEC without cystic component. No necrosis, mitotic activity, or neuroinvasion were detectable. Taken together, these findings confirmed the diagnosis of low-grade MEC following the histopathological grading system described by Goode et al.
The patient was referred to a head and neck surgery department where they conducted a magnetic resonance imaging (MRI) confirming that the lesion was entirely confined to the bony area of the right posterior third of the maxillary bone. The 16, 17, and 18 teeth were removed, and the tumor was excised with safe surgical margins. At the end of the procedure, the dental team placed a temporary prosthetic obturator to close the bucco-nasal communication.
Three months later, the patient underwent a second surgical intervention to ensure the closure of the bucco-nasal communication which was achieved using a palatal rotation flap (Figure 6) which is a pedicled flap with a posterior base, in relation to the vascularization of the palatine artery. The palatal rotation technique involves moving the palatal mucosa in the overlapping area, ensuring there is enough laxity to achieve a tight closure. The palatal donor site will then undergo second-intention healing.
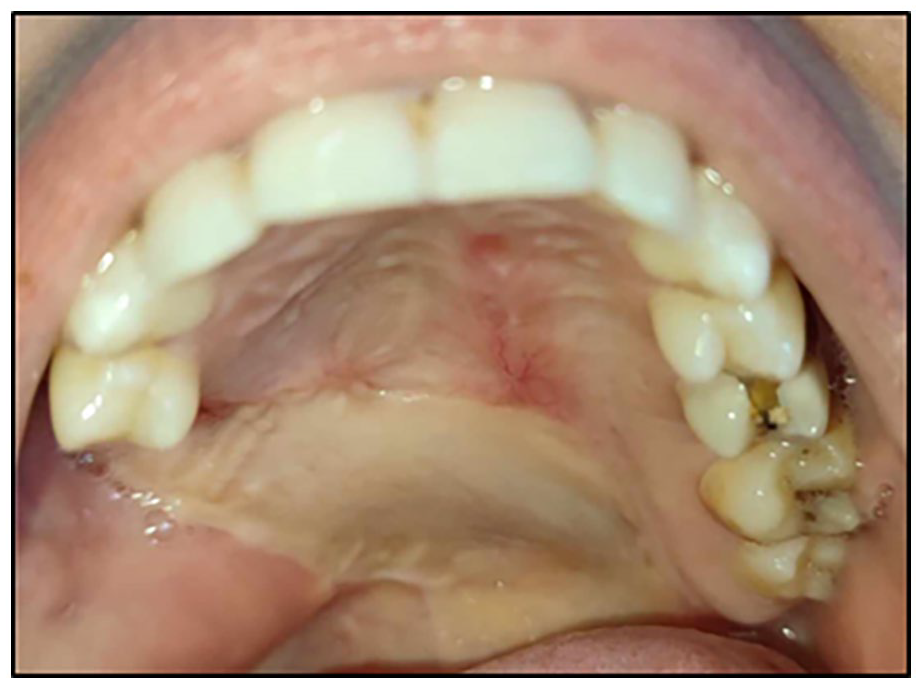
Clinical view at 2 months postoperatively.
The patient was followed up for 12 months, and there was no evidence of any recurrence.
Discussion
Salivary gland malignancies make up 0.5%–1.2% of all human cancers and 5% of head and neck cancers. 2
MEC is the most common salivary gland malignancy, occurring in both adults and children. The parotid gland is the most commonly affected site, followed by the minor salivary glands. 3 Among the various sites of MEC, the palate is the most common site, accounting for 28% of cases. Other common locations include the retromolar area (23%), floor of the mouth (14%), oral mucosa (11%), and lower lip (9%). 3
MEC arise from the epithelial cells that line the interlobular and intralobular salivary ducts. Risk factors include a history of radiation exposure and the translocation
MEC presents as an unencapsulated, ill-defined mass with a cystic component and often poses a diagnostic challenge due to the potential confusion with mucoceles. MECs are composed of six cell types, including progenitor cells with small, round nuclei and basophilic cytoplasm. Intermediate cells that can differentiate into glandular cells or epidermoid cells. Epidermoid cells have a homogeneous cytoplasm that occasionally contains keratin pearls. Clear cells have watery cytoplasm and a central nucleus, while columnar cells resemble the secretory duct cells of salivary glands and can transform into mucous cells. Mucous cells have small nuclei on the periphery, foamy reticular cytoplasm, and occasionally a signet ring appearance. 7
Tumors identified as MEC display varied biological aggressiveness, leading to the development of various rating systems. However, a universally acknowledged disease grading system is still lacking. Currently, the Armed Force Institute of Pathology (AFIP) and Brandwein grading systems are among the most commonly used rating systems.
The AFIP criteria proposed by Goode et al. evaluates tumors histologically based on specific parameters, such as <20% intracystic component, neural invasion, necrotic foci, a mean of ⩾4 mitoses per 10 high-power fields and anaplasia, and then graded them accordingly using a score ranging from 0 to 14 as follows: 0–4 for low-grade, 5–6 for intermediate-grade, and ⩾7 for high-grade. 8
Low-grade MEC is characterized by the gradual appearance of a small swelling or mass, typically less than 3 cm in size, that is, bluish in color due to the presence of mucin-filled spaces visible through the overlying mucosa. Histologically, they are partially encapsulated and contain well-structured cystic spaces and a high glandular component, and they show minimal cytologic atypia or mitoses and lack perineural invasion. Intermediate grades tend to have more solid epidermoid nests and intermediate cells with less evident cystic spaces. Clinically, it presents as an ulcerated or solid mass, which is firm in consistency with normal colored overlying mucosa. These tumors grow faster and exhibit infiltrative behavior compared to low-grade tumors with a tendency to ulcerate in early stages. High-grade MEC has limited mucous cells with large amounts of solid, squamous cells that can often be misdiagnosed as squamous cell carcinoma; they show more cytologic atypia, necrosis, and perineural or lymphovascular invasion, along with mitotic figures. These tumors are the fastest growing of the different types and often show local tissue invasion at an early stage. At later stages, they can penetrate the underlying cortical plate and invade critical structures of the maxillary sinus and nasal cavity. They can spread rapidly toward the pterygoid body and eventually reach the infratemporal space and even the base of the skull. 9
Extensive studies have examined the use of immunohistochemistry (IHC) in MEC, but it is rarely necessary for diagnosis. As an epithelial neoplasm, the neoplastic cells typically exhibit reactivity with cytokeratin, EMA, and p63, except for mucous and certain clear cells. In general, IHC characterization plays no role in grading. In a retrospective analysis of 31 MEC cases, the use of S-100 to identify perineural invasion was described and found to be an important factor in the survival outcomes of MEC cases. In difficult cases, additional immunohistochemical staining for low molecular weight (CK7) or high molecular weight (CK5) cytokeratins is used to identify mucinous, intermediate, and epidermoid differentiation. 10
The size of the lesion and its invasion into the bone can be determined with the help of computed tomography, CBCT, and MRI. The appearance of MEC on imaging depends strongly on tumor grade. Usually, MEC appears as a cystic or tumor-like lesion on routine radiography. If larger in size, they may cause migration of the adjoining teeth. It may perforate the adjacent bone and can present as a well-defined radiolucency with definite corticated borders. Low-grade lesion can appear as lobulated or irregularly circumscribed cystic areas with intact underlying periosteum. Intermediate tumors may exhibit bony invasion in the form of erosion of the palatal bone. High-grade lesions will always perforate the underlying bone and reach the maxillary sinus and nasal cavity. High-grade MEC may have stages I to III lymph node metastasis (34.0%). 9
The MEC treatment is mainly surgical, depending on two major features: the extent of its spread and its histological grade. In cases where the tumor is confined to the palatal mucosa and the periosteum remains intact, a recommended approach is wide excision of the lesion, including the underlying periosteum. However, when the tumor infiltrates the periosteum, causing erosion of the underlying bone, it is advisable to perform excision of the lesion along with the affected underlying bone. The prevailing belief is that employing radical excision with ample safety margins is effective in treating low-grade malignant tumors. Conversely, high-grade tumors typically require radical excision, cervical lymph node dissection, and postoperative radiotherapy. 11 Chemotherapy is not recommended for this type of cancer. 12
The prognosis of MEC depends on factors such as tumor grade, stage at diagnosis, and treatment success. Generally, low-grade MECs have a positive prognosis, whereas high-grade tumors may be more aggressive nature, leading to a less-favorable outcome. Early detection and effective treatment are crucial for better prognosis, emphasizing the importance of personalized approaches and regular follow-up for individuals diagnosed with MEC. 13
Conclusion
MEC is a rare tumor that should be considered when encountering with a painless, progressively expanding, pale, bluish-purple mass, particularly in the palate. The following clinical case underlines the importance of early detection and immediate intervention to reduce the necessity of extensive surgery and improve the prognosis. Continuous clinical and radiological monitoring is essential for these lesions; whose origin is presently unidentified.
Footnotes
Acknowledgements
No additional acknowledgments are required.
Contributors statement
All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
