Abstract
An acute to chronic idiopathic necrotizing meningoencephalitis was diagnosed in 5 Chihuahua dogs aged between 1.5 and 10 years. Presenting neurologic signs included seizures, blindness, mentation changes, and postural deficits occurring from 5 days to 5.5 months prior to presentation. Cerebrospinal fluid analyses from 2 of 3 dogs sampled were consistent with an inflammatory disease. Magnetic resonance imaging of the brain of 2 dogs demonstrated multifocal loss or collapse of cortical gray/white matter demarcation hypointense on T1-weighted images, with T2-weighted hyperintensity and slight postcontrast enhancement. Multifocal asymmetrical areas of necrosis or collapse in both gray and white matter of the cerebral hemispheres was seen grossly in 4 brains. Microscopically in all dogs, there was a severe, asymmetrical, intensely cellular, nonsuppurative meningoencephalitis usually with cystic necrosis in subcortical white matter. There were no lesions in the mesencephalon or metencephalon except in 1 dog. Immunophenotyping defined populations of CD3, CD11d, CD18, CD20, CD45, CD45 RA, and CD79a immunoreactive inflammatory cells varying in density and location but common to acute and chronic lesions. In fresh frozen lesions, both CD1b,c and CD11c immunoreactive dendritic antigen-presenting cells were also identified. Immunoreactivity for canine distemper viral (CDV) antigen was negative in all dogs. The clinical signs, distribution pattern, and histologic type of lesions bear close similarities to necrotizing meningoencephalitis as described in series of both Pug and Maltese breed dogs and less commonly in other breeds.
Keywords
Introduction
In the dog, meningoencephalitis has been associated with a variety of infectious agents including bacteria, fungi, protozoa, rickettsia, parasites, and viruses. 14 However, several other syndromes with a nonsuppurative idiopathic meningoencephalitis have been described, including necrotizing meningoencephalitis (NME) in a series of Pug and Maltese dogs and individually in other breeds including a Shih Tzu, a Chihuahua, a Papillon, and a Pekingese dog. 1, 2, 7, 13, 16, 19 NME is characterized by a multifocal, asymmetric, necrotizing, nonsuppurative meningoencephalitis of the cortex and subcortical white matter essentially restricted to the cerebral hemispheres. A possible variant of NME has been reported as a separate entity of necrotizing encephalitis (NE) in Yorkshire Terrier dogs, 4, 6, 8, 11, 19, 22 and a French Bulldog 18 and has been seen in a Spitz, a Miniature Pinscher, and a Pekingese dog (K. Matiasek, M. Vandevelde, personal communication). NE has necrotizing, intensely inflammatory, and malacic lesions restricted to subcortical white matter of cerebral hemispheres and usually inflammatory lesions in the brain stem and cerebellum. Here we report 5 Chihuahua dogs that had an idiopathic, nonsuppurative, necrotizing meningoencephalitis similar clinico-pathologically to NME described in the Pug and Maltese dog breeds.
Materials and Methods
Selection of dogs
Four Chihuahua dogs (dog Nos. 2–5) were identified retrospectively by evaluation of computerized medical records and necropsy pathology reports of all dogs examined at the Veterinary Medical Teaching Hospital (VMTH), University of California Davis, Davis, California, between January 1990 and December 2004. One Chihuahua dog (dog No. 1) was examined at Veterinary Consultant Associates, West Los Angeles Animal Hospital, Los Angeles California (VCA), and a full necropsy performed after referral to the Anatomic Pathology service of the VMTH.
Magnetic resonance (MR) imaging
MR imaging of the brain of dog Nos. 3 and 5 was done under general anesthesia using a 1.5 Tesla Sigma LX Scanner (GE Medical Systems, Waukesha, WI) and a 0.4 Tesla Resonex 5000 Scanner (Resonex Inc., Sunnyvale, CA), respectively. Contiguous 3-mm transverse images of the brain were generated with T1-weighted (T1W), T2-weighted (T2W), proton-weighted (PW) spin echo pulse sequences, and fluid attenuation inversion recovery (FLAIR) sequences. Additional T1W transverse images were acquired after administration of intravenous contrast medium (Magnevist, 100 mg/ml gadopentatate dimeglumine, Berlex Laboratories, Inc., Wayne, NJ).
Cerebrospinal fluid (CSF) collection and analysis
Samples of CSF, obtained from the atlanto-occipital cistern of 3 dogs, were analyzed in the VMTH Clinical Pathology laboratory within 30 minutes of collection for gross appearance, total protein level and total red blood (RBC), and total (TNCC) and differential (DNCC) nucleated cell counts and by aerobic culture on sheep blood agar. Cytological evaluation following cytocentrifugation was done, either on 100 nucleated cells or on all cells if fewer than 100 cells were recovered, with Wright's stain essentially as described (CSF analysis, Table 3). 3
Results of cisternal cerebrospinal fluid analysis in 3 dogs.
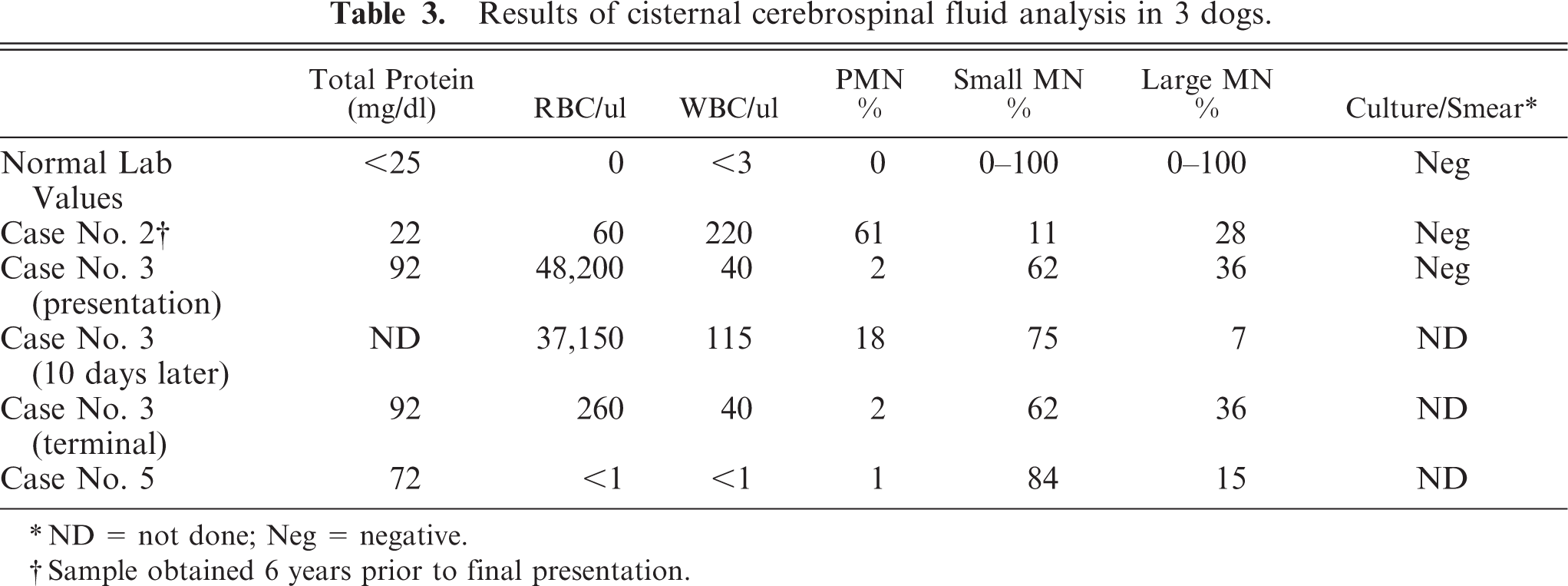
ND = not done; Neg = negative.
Sample obtained 6 years prior to final presentation.
Necropsy and pathology findings
All dogs had a complete necropsy. Tissue samples including the brain from all dogs were immersion-fixed in 10% neutral buffered formalin, and selected samples were subsequently routinely embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin (HE) for initial microscopic evaluation. Then selected sections were stained with HE-Luxol fast blue stain (HE-LFB). Other unfixed fresh samples of brain from 1 dog were embedded and frozen in TissueTek OCT compound (Sakura Inc., Torrance, CA) and subsequently stored at −80°C.
Immunocytochemistry
Immunocytochemical staining was done on selected sections with primary mono- or polyclonal antibodies, used as recommended by the manufacturer or by the Leucocyte Antigen Biology Laboratory (LABL), Davis, California, for antigen visualization in either formalin-fixed, paraffin-embedded tissue sections or from fresh frozen brain sections embedded in OCT medium and employing an unlabeled streptavidin–biotin immunocytochemical procedure as previously described with poly- or monoclonal antibodies to canine distemper virus (CDV) antigen; glial fibrillary acidic protein (GFAP); CD1 b and c; CD11a, b, c, and d; CD18; CD20; CD45; CD45RA; CD79a; canine IgA and IgG as previously described (Table 1). 5, 9 Appropriate negative and control tissues from our laboratory for both the frozen and paraffin-embedded brain tissue were used when positive endogenous antigen immunoreactivity was not available. The functional cell specificities of the antibodies (Table 1) were interpreted as previously described. 9, 10
Details of antibodies for immunocytochemistry.∗
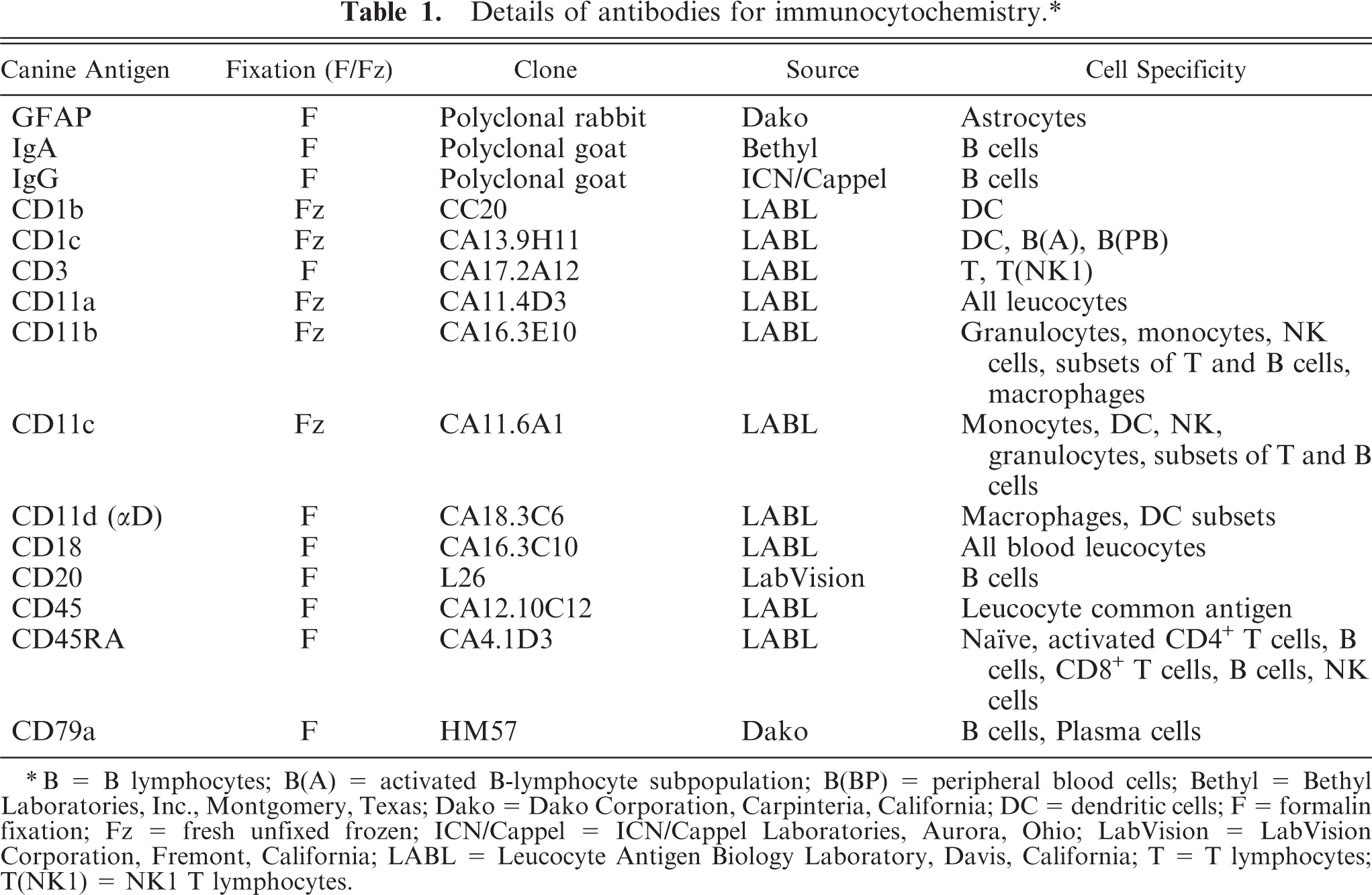
B = B lymphocytes; B(A) = activated B-lymphocyte subpopulation; B(BP) = peripheral blood cells; Bethyl = Bethyl Laboratories, Inc., Montgomery, Texas; Dako = Dako Corporation, Carpinteria, California; DC = dendritic cells; F = formalin fixation; Fz = fresh unfixed frozen; ICN/Cappel = ICN/Cappel Laboratories, Aurora, Ohio; LabVision = LabVision Corporation, Fremont, California; LABL = Leucocyte Antigen Biology Laboratory, Davis, California; T = T lymphocytes; T(NK1) = NK1 T lymphocytes.
Results
Clinical examination (summary in Table 2)
Summary of signalment and clinical neurologic findings in 5 Chihuahua dogs.

F = female; MC = male castrate; FS = female neutered.
Dog No. 1, a 3-year-old intact female Chihuahua weighing 2 kg, presented to VCA with a history of several seizures over the last 5 days. The owners noted behavioral changes during this period consisting of sudden fear and aggression. The animal was recumbent and obtunded but responsive at presentation. Neurological abnormalities included a head aversion to the left, decreased menace response on the right, and decreased postural reactions in the right pelvic and thoracic limbs. The dog died that evening after cardiopulmonary arrest.
Dog No. 2, a 10-year-old intact female Chihuahua weighing 1.5 kg, was presented to the VMTH with a history of an acute onset of several seizures during the previous 2 hours. Neurological abnormalities included obtundation, a left-sided leaning gait, and decreased facial sensation on the left side. After 1 week free of seizures, poor owner compliance with medication instructions then resulted in re-presentation of the dog 3 days later for status epilepticus, at which time the owner elected euthanasia. The dog had presented to the VMTH 6 years previously for anorexia, cervical pain, and anterior uveitis (CSF analysis, Table 3). Those clinical signs resolved following a 4-week course of prednisone.
Dog No. 3, a 1.5-year-old intact female Chihuahua weighing 2 kg, presented to the VMTH with a history of circling for approximately 3 months. Neurological abnormalities included tetraparesis with generalized ataxia, circling to the left and right, left head tilt, and decreased postural reactions predominantly on the left side (CSF analysis, Table 3). At this time 6 weeks before necropsy, MR imaging revealed a poorly defined area of heterogeneously increased signal intensity in the left dorsal medulla, caudal to the pons on T2W and FLAIR images. The lesion was iso- to hypointense on T1W images with no contrast enhancement (Fig. 1). Clinical improvement was seen for approximately 6 weeks following corticosteroids and phenobarbital, with resolution of all neurological deficits other than mild hypermetria on the left side. Euthanasia was requested because of the sudden rapid deterioration ending in status epilepticus.
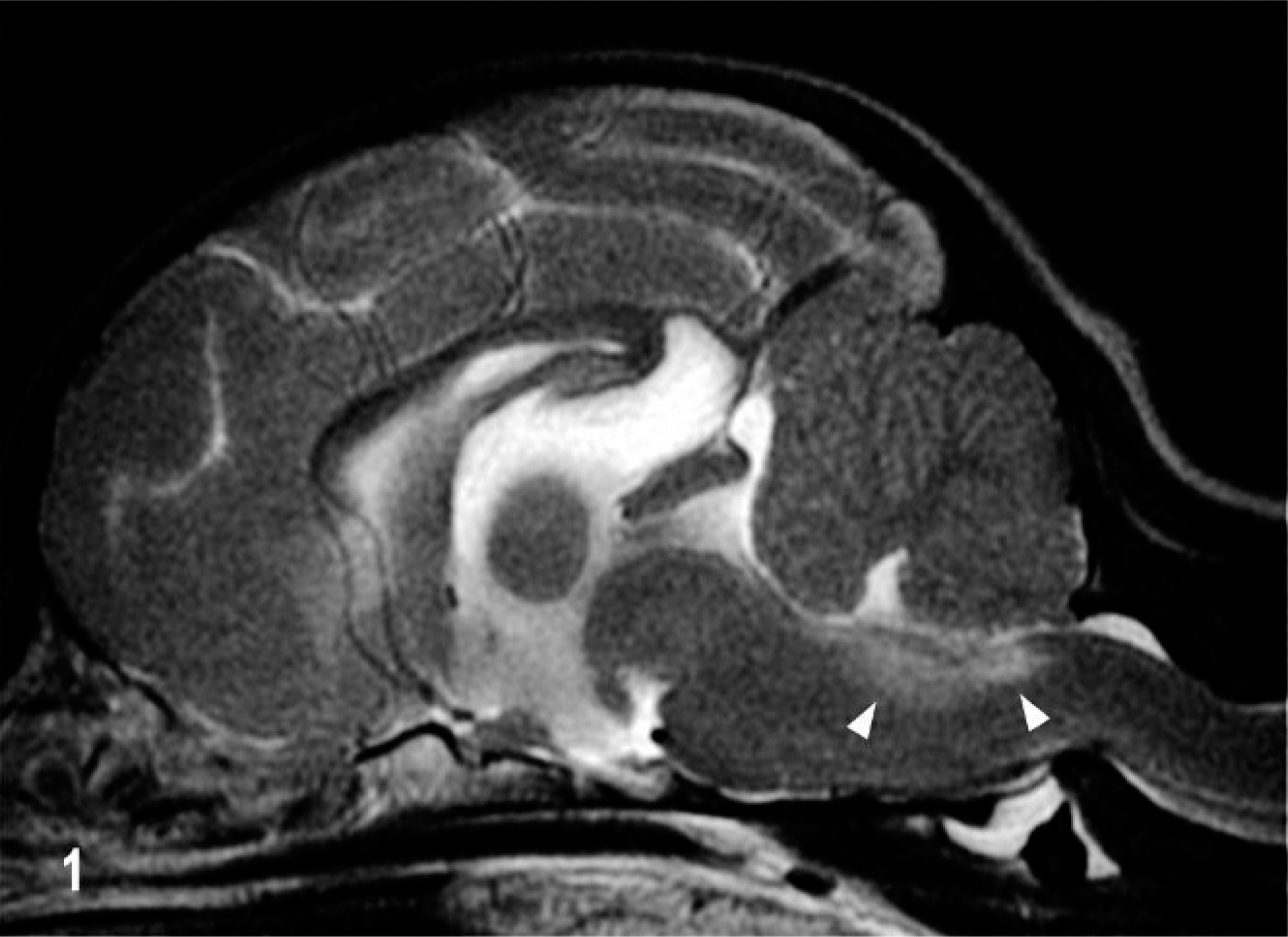
Brain; dog No. 3. Sagittal T2W MR image. A poorly defined area of hyperintensity in the subventricular dorsal medulla (arrows).
Dog No. 4, a 5-year-old spayed female Chihuahua weighing 2.7 kg, was presented to the VMTH with a 5.5-month history of generalized seizures, and altered behavior with signs of fear and aggression. The dog had a cluster of seizures 1 day prior to presentation. The dog was nonambulatory and recumbent at presentation. Neurologic abnormalities included obtundation, head aversion to the left, decreased menace response bilaterally but most prominent on the left, anisocoria (left pupil smaller), a transient positional vertical nystagmus bilaterally and a mild bilateral ventral strabismus. Postural reactions were decreased in the right pelvic and thoracic limbs. The owners declined further diagnostics and treatment, and the animal was euthanized.
Dog No. 5, a 10-year-old castrated male Chihuahua weighing 4.5 kg, was presented with a 2.5-year history of generalized seizures. Four days prior to presentation, the dog had an acute onset of blindness and 4 generalized seizures. At presentation the dog was agitated and had partial seizures restricted to the face (CSF analysis, Table 3). MR imaging 2 hours before euthanasia revealed marked asymmetrical ventriculomegaly of the lateral ventricles (the right larger than the left). Normal gray/white matter demarcation was lost throughout most of the cerebral cortex on both sides, with marked thinning and collapse of the right parietal, temporal, and occipital cortices on the right side (Fig. 2). Several relatively well-demarcated areas of decreased signal intensity were present throughout the cortex, predominantly on the right side, on T1W images. Left and right cortices appeared diffusely hyperintense on T2W images. Postcontrast studies showed diffuse, patchy, poorly demarcated areas of contrast enhancement involving the left cerebral cortex (Fig. 2). The owners declined further diagnostics, and the dog was euthanized.
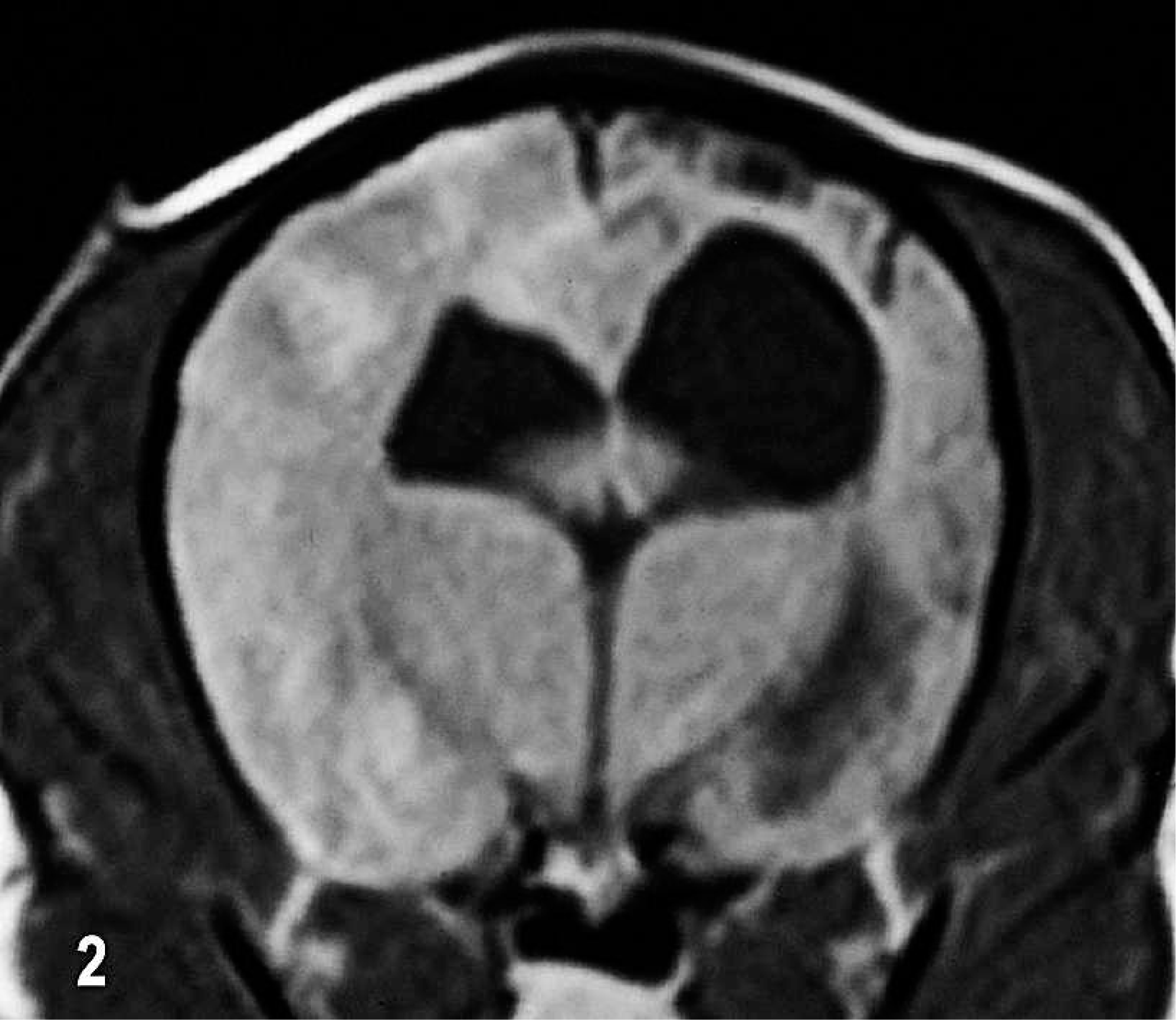
Brain; dog No. 5. Transverse T1W postcontrast MR image. Ventricular asymmetry and right sided ventriculomegaly with poorly defined areas of contrast enhancement in the swollen left parietal and temporal lobes. In the right parietal lobe there is focal loss of cortex and underlying white matter.
Pathology findings
Significant gross and histologic lesions were limited to the central nervous system (CNS) in each dog.
Macroscopic lesions
In dog No. 1 in transverse sections there was marked focal thickening and blurring of the white and gray matter junction and yellow tan discoloration of the left parietal cortex. The left lateral ventricle was reduced in size.
In dog No. 2 there were no external gross lesions of the brain, but on transverse sections there was asymmetric cortical thinning of the left parietal lobe with an enlarged left ventricle.
In dog No. 3 there was a marked asymmetric dilation of both lateral ventricles with focal thinning of subependymal white matter and of the overlying cortex of the temporal lobe.
In the brain of dog No. 4, there were areas of multifocal bilateral asymmetrical gyral collapse over the parietal and temporal lobes with brownish, cystic malacic lesions in both the white matter and overlying cortex (Fig. 3). There was marked dilation of both lateral ventricles. There was also mild cerebellar vermal herniation caudally.
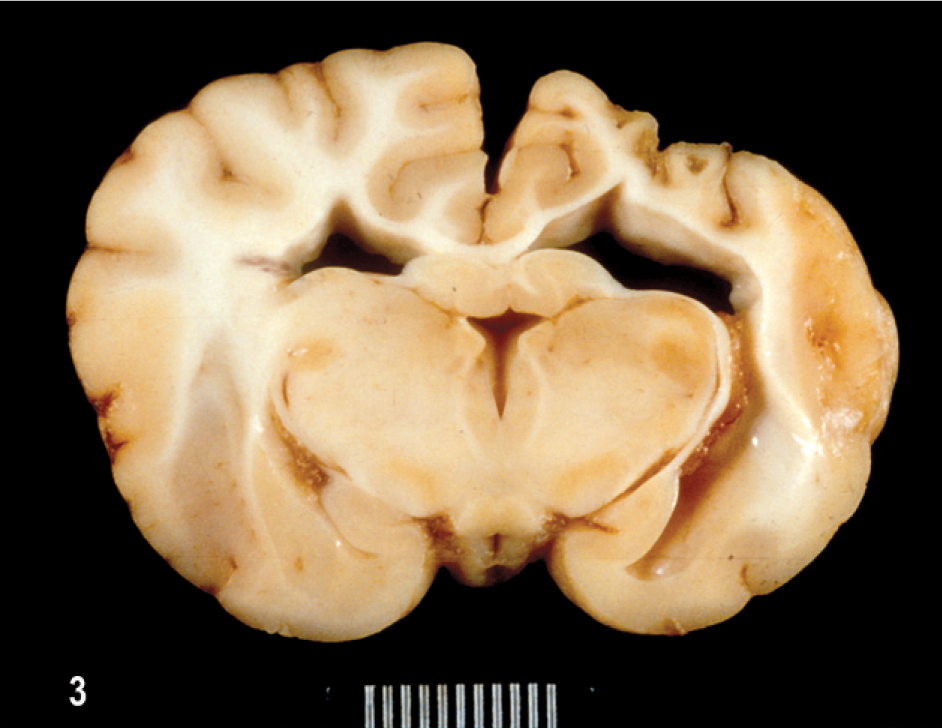
Brain; dog No. 4. Focal brownish areas of gyral cortical collapse and white matter loss in right parietal lobe; asymmetric ventricular dilation.
In the brain of dog No. 5, there was some collapse and partial loss of the gyral pattern in the right parietal and temporal cortex. On transverse sections of brain multifocal malacic areas were apparent in the cortex and underlying white matter on the left side, with right-sided cortical gyral collapse and white matter loss and asymmetrical enlargement of the right lateral ventricle, best demonstrated after LFB-HE staining (Fig. 4).
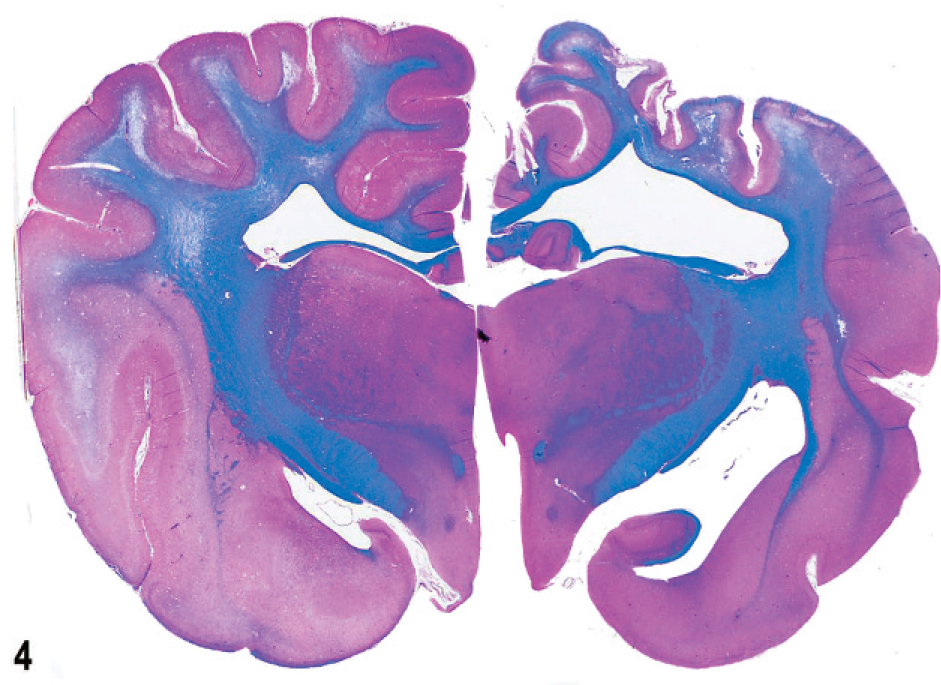
Brain; dog No. 5. Transverse section with focal loss of white matter and cortex and collapse on right parietal lobe; on left side multifocal areas of necrosis in corona radiata and centrum ovale extending into the overlying cortex; asymmetrical ventricular enlargement. HE-LFB.
Histologic lesions and immunophenotyping findings
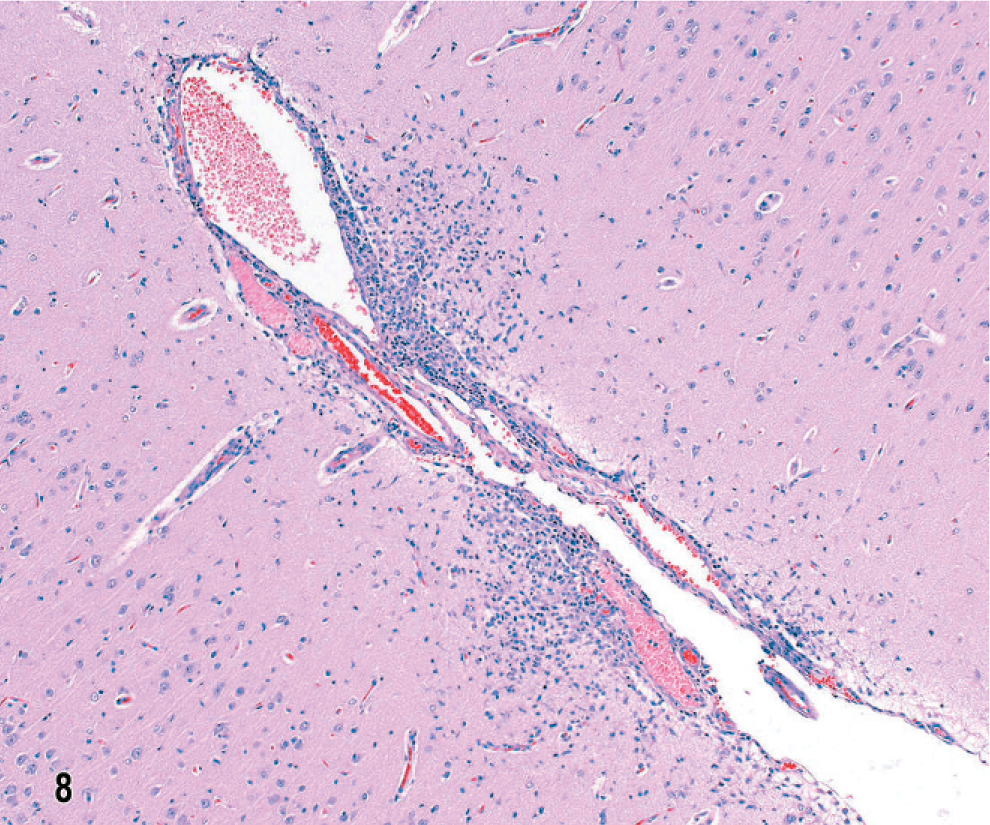
In dog No. 1 the most striking features of this untreated NME of 5 days' clinical duration, with lesions restricted within the left parietal and temporal lobes, were focal to confluent intense inflammatory cell infiltrates in cortex and underlying white matter (Fig. 5). The overlying meninges were focally thickened by infiltrates of lymphocytes, macrophages, and plasma cells (Fig. 6). In the cortex were predominantly mononuclear cells, some activated microglia and neutrophils, and also prominent perivascular lymphocytic cuffing. Ischemic necrosis affected many neurons in this inflammation. In the subjacent corona radiata there was extensive edema and necrosis with mononuclear cell infiltrates, gitter cells, and marked lymphocytic perivascular cuffing (Figs. 5, 7). There was also a characteristic predilection for focal cellular infiltrates in the cerebral hemispheric meninges and underlying cortex but sparing the underlying white matter (Fig. 8). Lesions decreased in intensity and incidence from rostral to caudal within the cerebral hemispheres. There were no lesions in the mesencephalon or metencephalon. With immunophenotyping, the inflammatory cell infiltrates consisted of CD20+ and CD79a+ B lymphocytes accumulated both diffusely and perivascularly in the meninges (Fig. 9). CD3+ T lymphocytes were plentiful in the meninges, scattered throughout the parenchymal lesions and within perivascular cuffs (Fig. 10). However, mononuclear CD18+ cells dominated the cell infiltrates in both the meninges and brain with fewer CD45R+ and CD45RA+ cells. Gitter cells in cavitary lesions were also identified with strong CD18 but minimal CD11d immunoreactivity. There was both IgA and IgG immunoreactivity within plasma cells restricted mainly to the meninges.
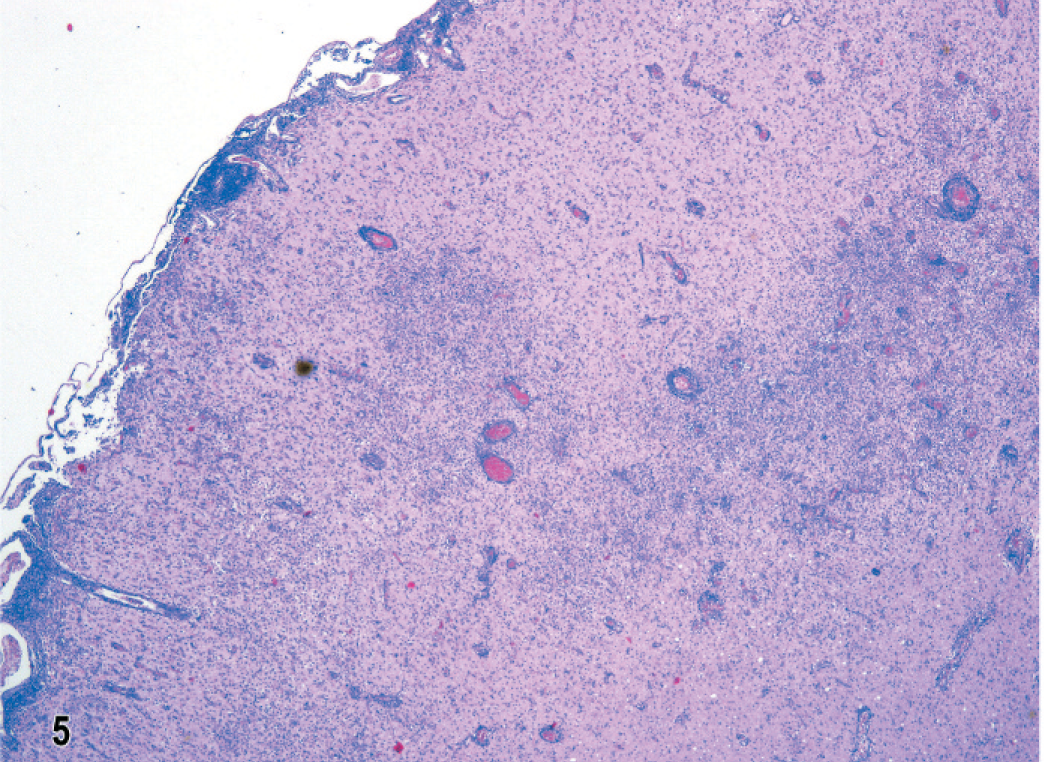
Brain; dog No. 1. Temporal lobe with acute, nonsuppurative multifocal meningitis and encephalitis in cortex and underlying white matter. HE.
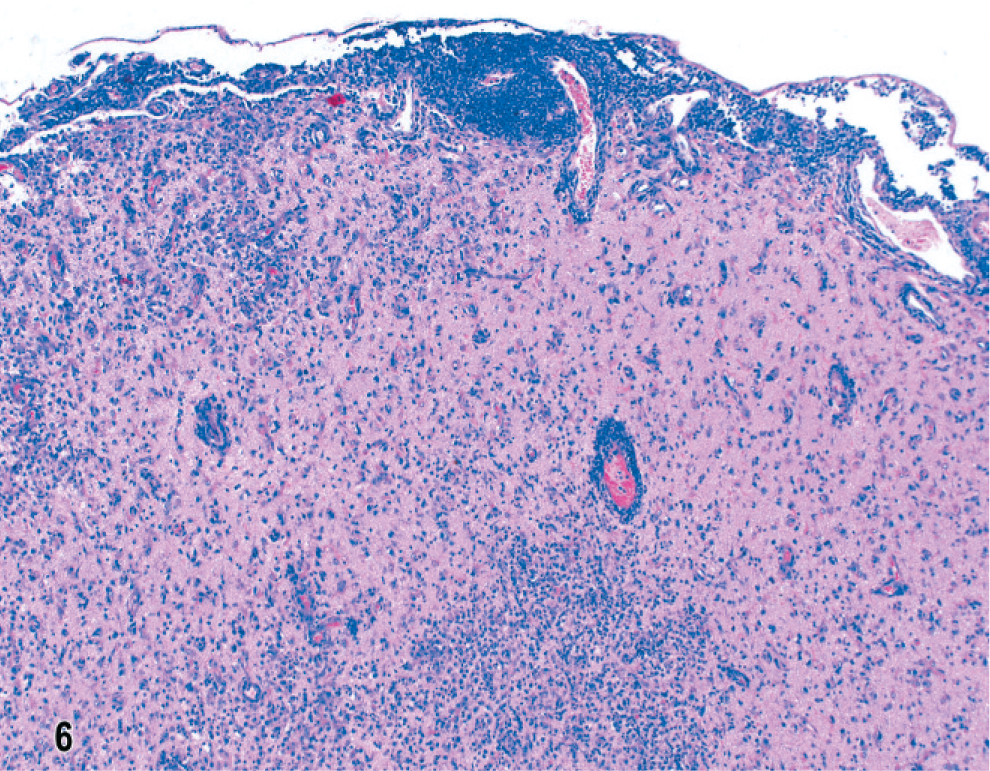
Brain; dog No. 1. Temporal lobe with dense multifocal mononuclear cell infiltrates in meninges and underlying cortex with perivascular cuffing. HE.
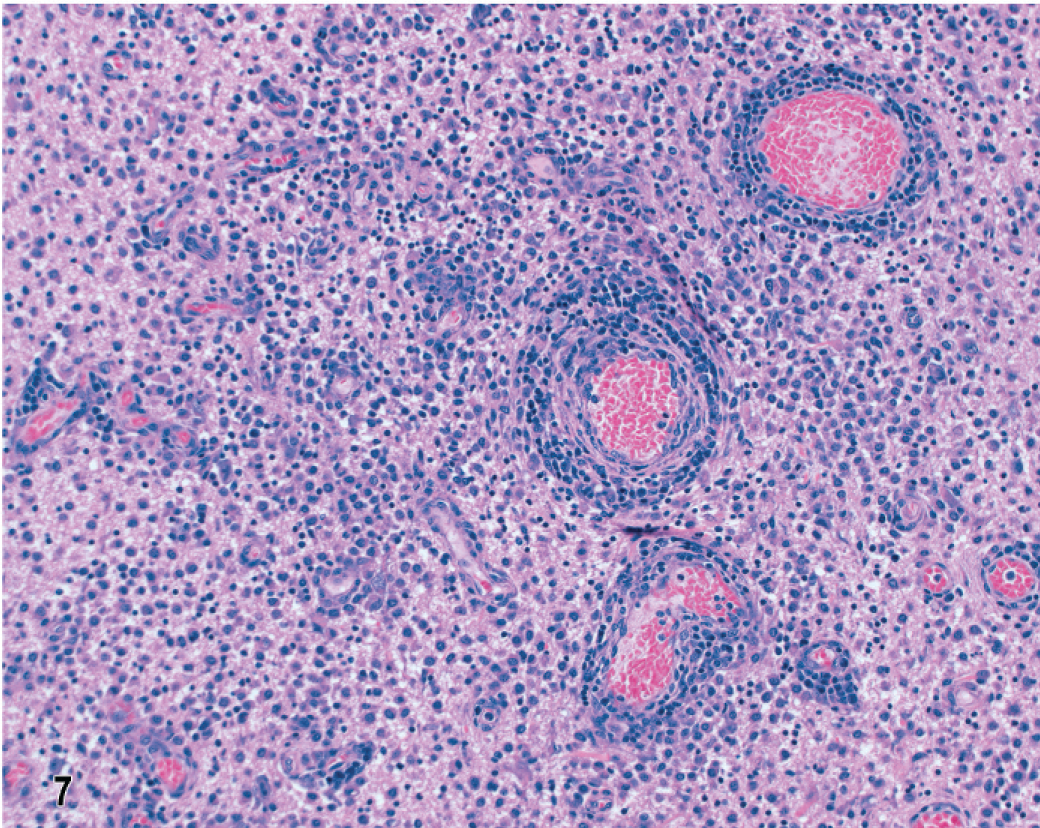
Brain; dog No. 1. Frontal lobe with multifocal nonsuppurative meningitis and underlying polioencephalitis. HE.
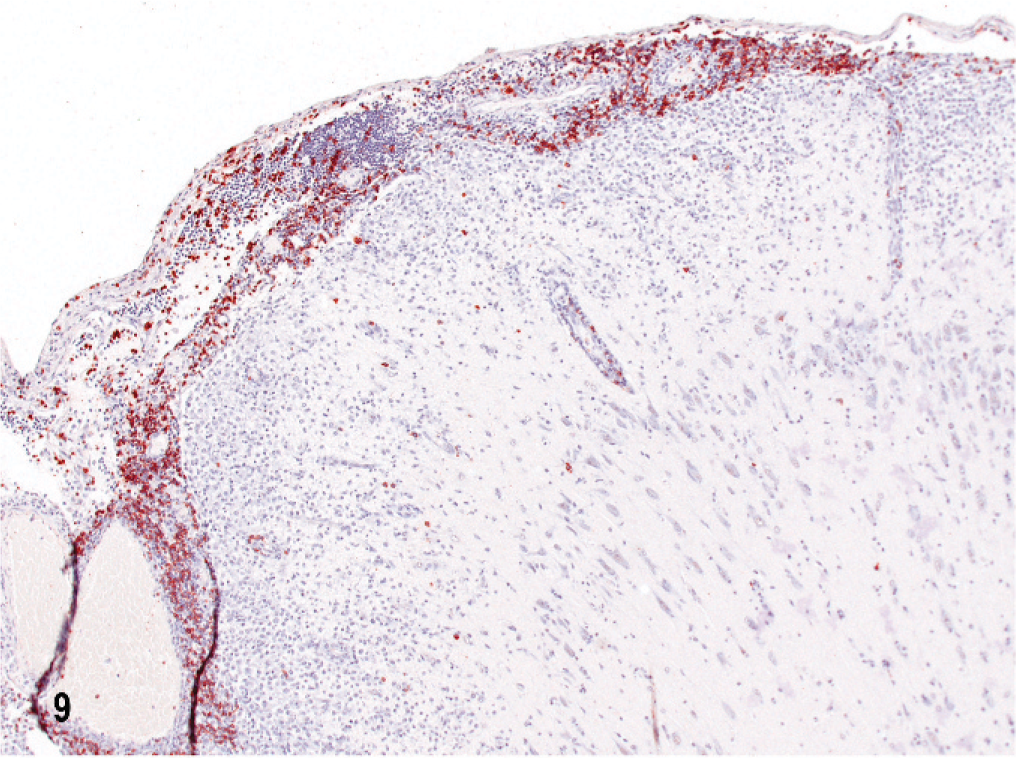
Brain; dog No. 1. Parietal lobe cortex with CD 79a immunoreactive B lymphocytes confined mainly to the meninges. Streptavidin–biotin complex method and hematoxylin.
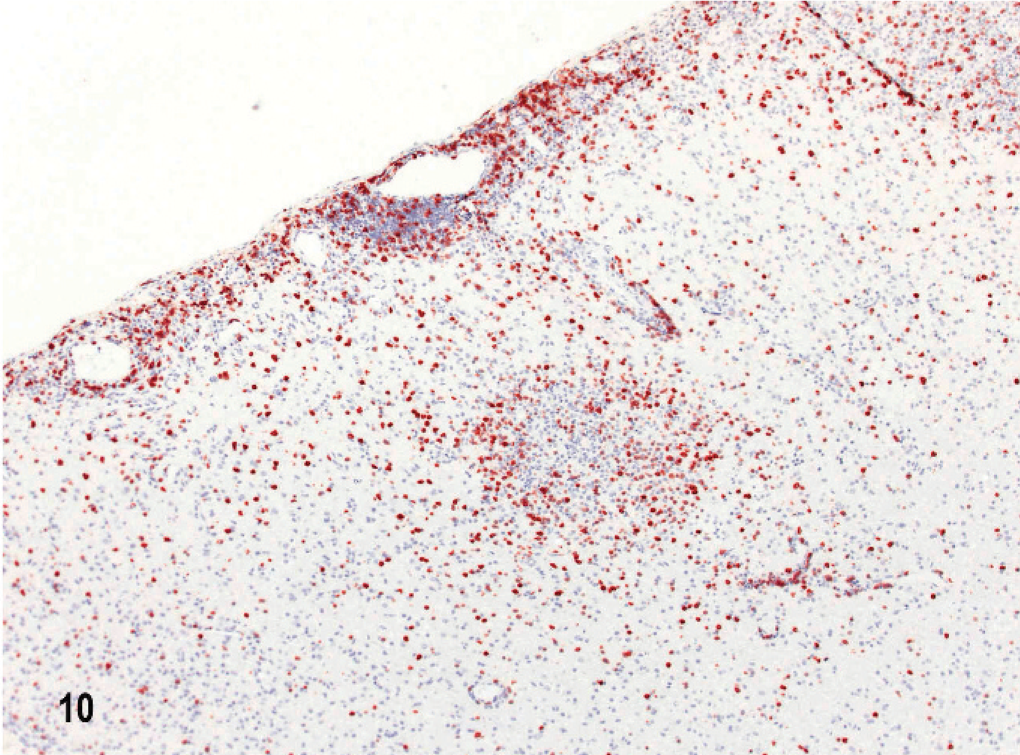
Brain; dog No. 1. Temporal lobe with CD3 immunoreactive T lymphocytes scattered throughout the meninges, underlying cortex and perivascularly. Streptavidin–biotin–peroxidase complex method and hematoxylin.
In dog No. 2, with a 1-week clinical history, microscopic lesions occurred unilaterally in the hemispheric meninges, and in white and gray matter of the right parietal and temporal lobes. Much of the swollen cortex was obliterated by multifocal densely cellular infiltrates of macrophages and an intense GFAP immunoreactive astrogliosis (Fig. 11). Subjacent white matter was focally edematous and necrotic, containing gitter cells, other macrophages, prominent neovascularization, and peripheral gemistocytic astrogliosis (Fig. 12). There were no lesions in the diencephalon, mesencephalon, or metencephalon. There were prominent CD20+ B cell and CD3+ T lymphocytes in meningeal infiltrates, massive infiltration of CD18+ large mononuclear cells in cortical and white matter lesions, and scattered CD3+ T lymphocytes throughout the lesions. There was some IgA and IgG immunoreactivity confined to the meninges.
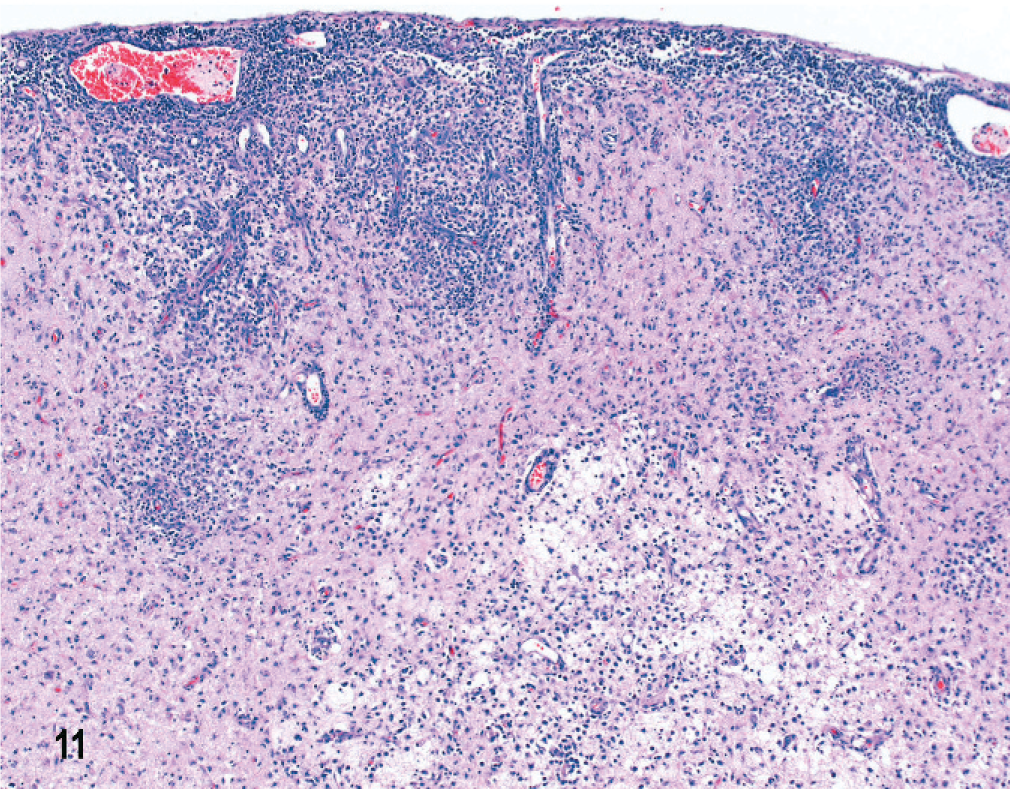
Brain; dog No. 2. Temporal lobe with chronic, severe, necrotizing meningoencephalitis with focal edema and necrosis in underlying white matter. HE.
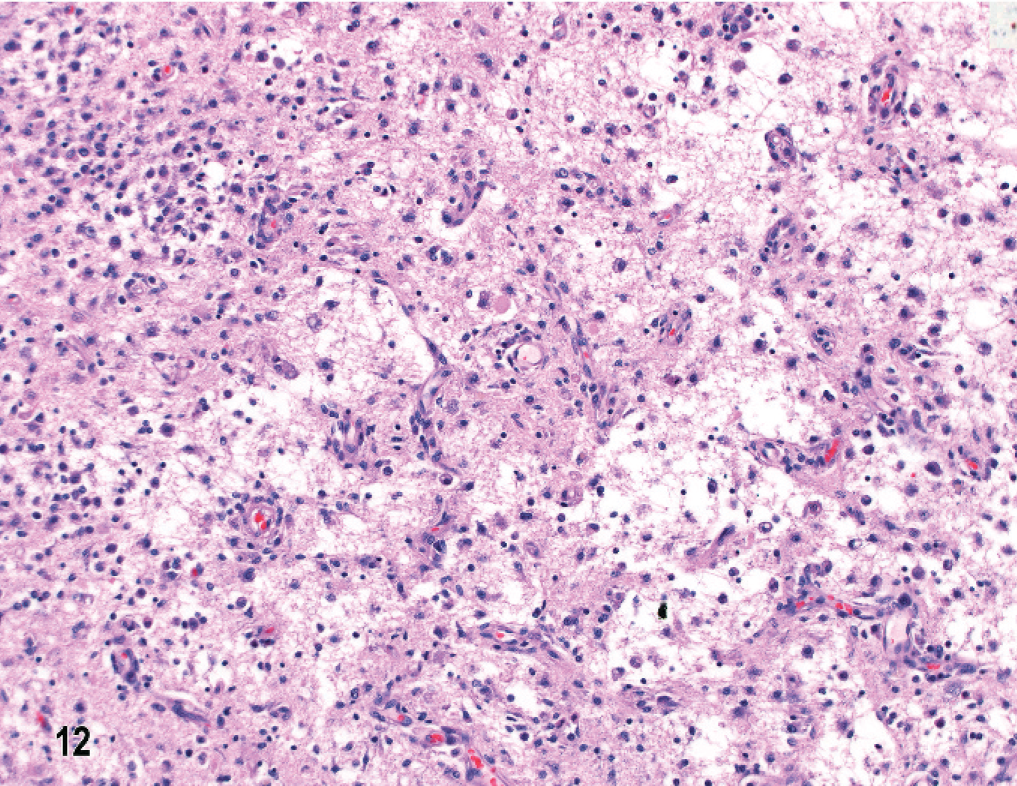
Brain; dog No. 2. Temporal lobe with inflammatory cell infiltrates, malacia, edema, and neovascularization in white matter. HE.
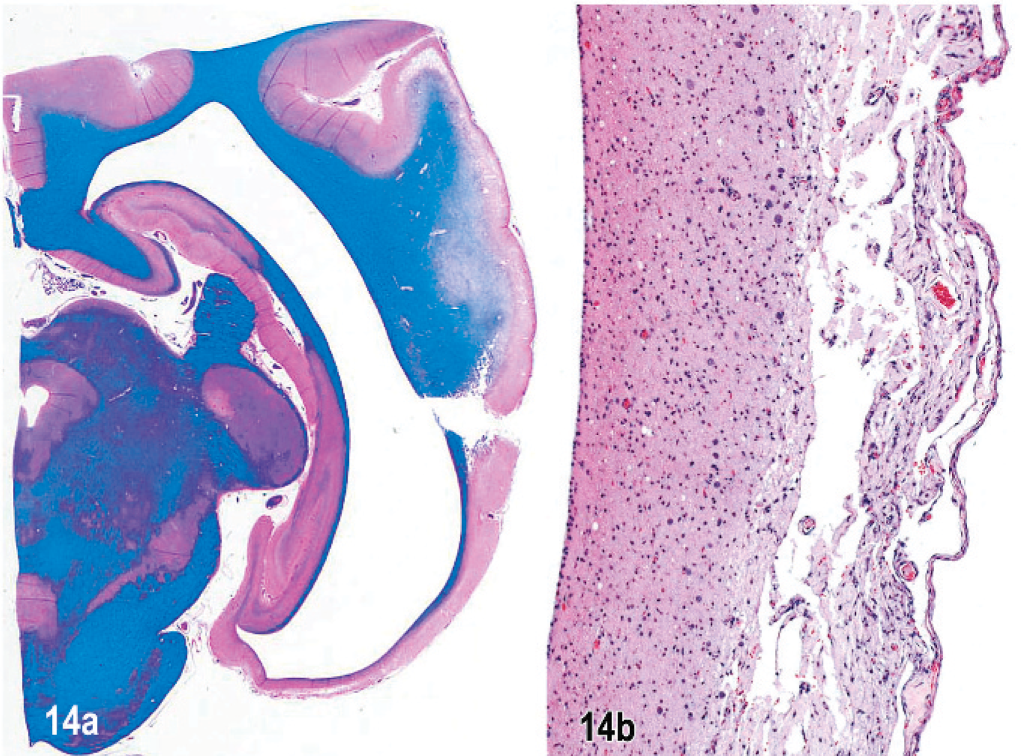
In dog No. 3, with a 3-month clinical history of circling and medication with prednisone and cytosine arabinoside, lesions were very similar in pattern, distribution, and severity to those in dog No. 1. Also there was acute multifocal meningoencephalitis in both septal nuclei. At the level of the dorsal cochlear nucleus, around the fourth ventricle, there were 2 small subependymal areas of cystic chronic malacia with a minimal inflammatory cell response (Fig. 13) corresponding to the MR imaging lesions detected 6 weeks before necropsy. Extensive cystic laminar cortical necrosis in both temporal lobes with minimal inflammatory response led to extreme thinning of the cortex and hydrocephalus (Fig. 14 a, b). In fresh frozen tissue from the parietal lobe, in both gray and white matter there was a predominant infiltration of both CD1b and CD11c immunoreactive dendritic antigen-presenting cells in the necrotizing lesions. There were fewer numbers of CD1c+ and CD11a, b+ cells. There were dense infiltrates of CD18 strongly immunoreactive cells in the necrotizing lesions and also similar populations of CD3+ T lymphocytes, and few CD11d+ macrophages as in dog Nos. 1 and 2. CD20/CD79a+ B cells were restricted to the meninges. There were IgA and IgG immunoreactive plasma cells in the meninges.
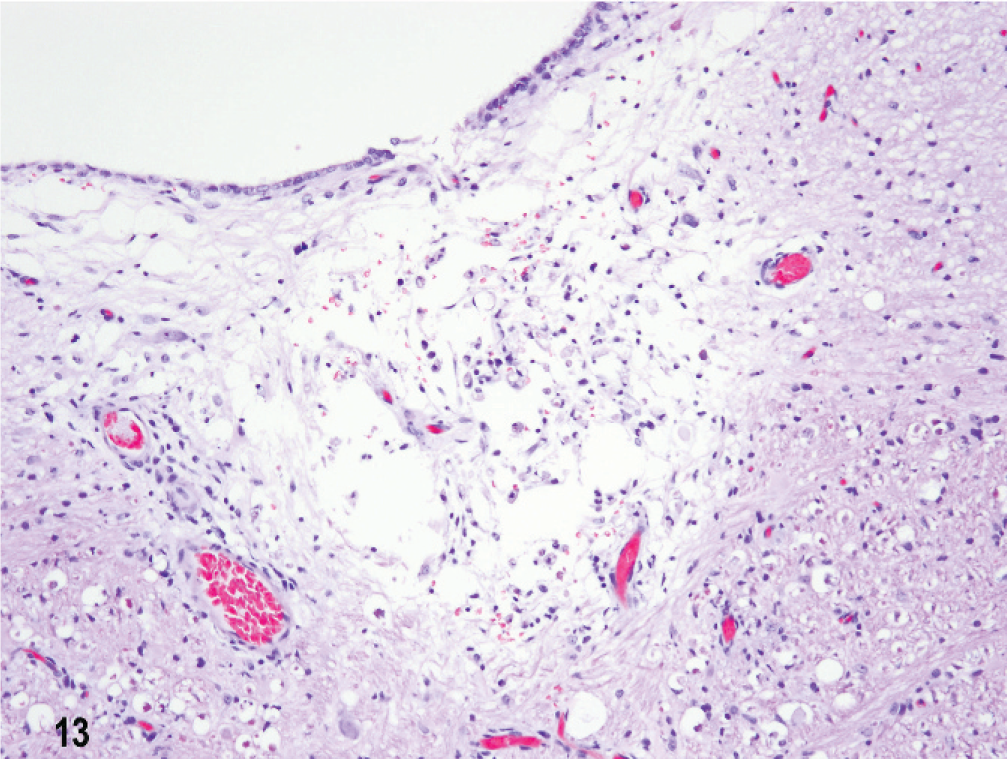
Brain; dog No. 3. Fourth ventricle subependymal focal malacia with minimal inflammatory cell infiltrates. HE.
In dog No. 4 of 5 months' clinical duration without anti-inflammatory medication, there was multifocal bilateral chronic NME, sometimes with gyral collapse, asymmetrically restricted to the parietal and temporal lobes of the cerebral hemispheres (Fig. 3) with other sites of chronic cystic leucoencephalomalacia relatively devoid of inflammatory cells. In contrast, in the thalamus and caudate nuclei there was acute, intense, random, multifocal encephalitis with prominent perivascular lymphocytic cuffing. In these lesions were dense infiltrations of CD11d+ macrophages and CD18+ mononuclear cells, with CD3+ T lymphocytes both in the lesions and perivascular cuffs. CD20/CD79a+ B lymphocytes were rare. In the ventral temporal cortex and cingulate gyrus bilaterally there was acute focal laminar cortical edema and necrosis, neovascularization and ischemic neuronal necrosis without inflammation. In these lesions there were only a few CD3+ T lymphocytes in the overlying meninges.
In dog No. 5 necrotizing chronic inflammatory lesions were restricted to the right frontal and temporal lobes. There were no lesions in the mesencephalon or metencephalon. Segmentally in the meninges in these areas were intense perivascular inflammatory cell infiltrates of predominantly CD3+ T lymphocytes and CD20/CD79a+ B lymphocytes. In cystic malacic areas were accumulations of CD18+ macrophages and reactive astrogliosis. There were a few IgA-immunoreactive cells in meninges. In the left parietal cortex was extensive acute laminar polioencephalomalacia with ischemic neuronal necrosis and neovascularization but minimal inflammatory cell infiltrates. All brains were immunonegative for CDV antigen.
Discussion
The clinical signs, pattern of distribution, and the histologic type of lesions in these Chihuahuas bear striking similarities to those in each of 4 series of cases of idiopathic NME in Maltese and Pug breed dogs. 2, 7, 14, 17 They are also very similar to the clinical, radiographic, and histologic findings in individual cases in other breeds, including a Pekingese, a Papillon, a Chihuahua, and a Shih Tzu dog. 1, 14, 17, 21 The cause of NME remains unknown, but immunoreactivity for CDV antigen has been consistently negative as has been PCR-based screening for canine herpes, and adeno- and parvoviruses in formalin-fixed, paraffin-embedded brain tissue from cases of NME irrespective of the breed. 12
Despite thorough clinical examination, advanced MR imaging, CSF analysis, and molecular diagnostic techniques, a specific diagnosis of NME still depends on histologic evaluation from surgical biopsy tissue or at necropsy. Four of the Chihuahua dogs were female, but any trend toward either a gender or age predilection remains uncertain as it does in the even larger series of Pug and Maltese dogs. 2, 7, 14, 17 The clinical onset varied from less than 1 day to 5.5 months' duration. Clinical findings were suggestive of cerebral disease in 4 cases, with seizures as the major presenting sign similar to previous reports. One dog (No. 3), however, had atypical neurologic signs of central vestibular disease 6 weeks prior to subsequent readmission in status epilepticus. Whether the subependymal chronic lesions were antecedent to the intense coexisting inflammatory lesions in the cerebral hemispheres is unknown. Also it is unclear whether the inflammatory CNS disease diagnosed 6 years previously in dog No. 2 or the 2.5-year seizure history in dog No. 5 were related to the final histologic diagnosis or represented previous unrelated conditions. Furthermore, the diagnostic utility of both CSF analysis and MR imaging can be unreliable because there was a lack of either a CSF pleocytosis (dog No. 5) or of cerebral MR imaging abnormalities (dog No. 3) prior to the severe inflammatory NME seen 6 weeks later at necropsy. However, the MR imaging lesions in dog No. 5 and mononuclear pleocytosis in the CSF from dog No. 3 are more typical of findings in NME in other breeds. 1, 2, 14, 17
Microscopically one of the more intriguing findings was the coexistence of chronic cavitary lesions and minimal inflammation with separate sites of acute meningoencephalitis in several dogs. In 2 dogs with a 3-month and a 5.5-month history of seizures, respectively, and chronic malacic lesions, other sites of acute inflammatory lesions in the thalamus/caudate nucleus and septal nuclei were indicative of actively progressive inflammatory lesions. That 1 of these 2 dogs had no potentially modifying glucocorticoid or nonsteroidal anti-inflammatory medication suggests that the inflammatory process can continue despite concurrent older lesions in apparent remission. Such a scenario could result theoretically from either a persistent infectious agent, a dysregulated inflammatory response, or an autoimmune pathogenesis. 15 Also in some dogs there was severe multifocal, acute, laminar or diffuse cortical necrosis with ischemic neuronal necrosis but without any associated inflammatory cell response. These are presumed to be secondary lesions induced by primary seizure activity from the NME. The distinctive segmental, multifocal pattern of the intense meningitis and encephalitis was a consistent finding in all Chihuahuas as in the series of NME in Maltese and Pug dogs. 2, 7, 14, 17 The targeting mechanism resulting in the distinctive asymmetrical acute necrotizing inflammatory lesions in both cerebral gray and subcortical white matter remains unknown. Intriguingly, high levels of an auto-antibody against GFAP in the CSF and serum of Pug dogs with NME have been quantitated, but what role they may play in the lesions remains to be determined. 13, 21 Similar antibody levels occur in the CSF of dogs with brain tumors, and high levels of GFAP in the CSF are demonstrated in clinically normal dogs. 13 Curiously, NME has only been recognized in small breed dogs, which is suggestive of some genetic linkage with possibly aberrant immune response regulating genes. 15 It has been demonstrated that rat strains with differing MHC haplotypes respond with different but reproducible patterns of asymmetrical inflammation to a myelin-oligodendrocyte-glycoprotein–induced auto-immune encephalomyelitis. 15 Similar MHC haplotype differences between dogs within this breed might explain the apparent upregulated but variable inflammatory response in NME.
The NE originally described in 6 Yorkshire Terriers and a French Bulldog and seen in a Shih Tzu, Spitz, and a Miniature Pinscher (K. Matiasek, M. Vandevelde, personal communication) has morphologic features that differ from those in Maltese, Chihuahuas, and Pug dogs. 4, 6, 8, 18, 19, 22 In the cerebral hemispheres there is an asymmetrical bilateral intensely inflammatory leucoencephalitis with subsequent cavitary necrosis and prominent reactive gemistocytic astrogliosis. In the brain stem and cerebellum are similar focal intense inflammatory infiltrates but without cavitation. Clinically, most Yorkshire Terrier dogs have presented with signs of brain stem involvement from severe lesions in the metencephalon. However exceptions occur, in which lesions are confined to the cerebral hemispheres. 22 Whether these morphologic differences in distribution patterns and histologic lesions between NME and NE truly reflect a difference in the etiologic mechanism or just a small breed–specific variation in a variable immunologic response to the same disease remains conjectural.
Immunophenotyping of the inflammatory cells using a standard panel of antibodies effective in formalin-fixed, paraffin-embedded tissue was chosen as an initial strategy to establish a profile for comparison with other idiopathic or known inflammatory encephalitides. In normal formalin-fixed, paraffin-embedded canine brain tissue there are lightly scattered CD3+T cells, whereas CD11d/CD18+ macrophages are found perivascularly. Endogenous macrophage/microglial populations predominantly express CD11b/CD18 and to a lesser extent CD11c/CD18. 20 Additionally, there is marked downregulation of CD45 expression, which is unique to the CNS. In both the acute and chronic lesions clusters of CD20/CD79a+ B lymphocytes were mostly restricted to the meninges, in contrast to CD3+ T lymphocytes, which were disseminated in the meninges, in perivascular cuffs and scattered within areas of encephalitis. These findings contrast with the pattern of viral encephalomyelitis, e.g., CDV encephalitis, in which diffuse T-cell invasion is characteristic. 20 In protozoal and bacterial infections, B cells tend to predominate within lesions and in perivascular cuffs, which was not the pattern seen in either acute or chronic lesions. 15, 16, 20 In meningeal and parenchymal lesions, by far the most dominant infiltrative cells were CD18+ macrophages, decreasing in intensity away from the lesion. Since CD18 does not discriminate between microglia and blood monocyte-derived macrophages, the lineage of these cells is uncertain. However, CD45 expression was limited to <10% of these cells in both active and chronic lesions, which matches the pattern of CD45 expression exhibited by endogenous microglia. 20 There was almost no increase in CD11d/CD18+ macrophages, which were restricted within lesions to single cells in the meninges. In the malacic lesions in fresh frozen tissue examined in 1 dog there was a dominant infiltration of CD1 and CD 11c immunoreactive dendritic antigen-presenting cells, which were not identifiable in formalin-fixed tissue except as a subset of the CD18-expressing cells. The presence of CD1+ dendritic cells is putative evidence of an unidentified antigen-driven inflammatory process. Of course the duration of clinical onset, the sites and severity of lesions, and the type of anti-inflammatory drugs are all factors that may still modify such profiles. However, no major differences in immunophenotypic profiles or patterns were seen in acute or chronic lesions between untreated dogs and those on anti-inflammatory medication. Correctly interpreting any specific primary or secondary role that these identified cell populations play in evolution of lesions must await experimental reproduction of the disease.
Whether the morphologic criteria currently used for distinguishing between NME and NE actually reflect either etiologic, pathogenic, or merely artificial differences remains conjectural. However, with both of these once-claimed breed-specific patterns of lesions in Pug dogs and Yorkshire Terrier dogs, respectively, now being recognized in other small dog breeds, it is possible that such distinctions may be a result of variations in individual immunologic responses to one disease process.
Footnotes
Acknowledgements
We would like to thank Ms. D. K. Naydan for her excellent support for the immunocytochemistry and to Dr. T. W. Affolter for help with pathology data collection.
