Abstract
The pathogenesis of necrotizing meningoencephalitis (NME), necrotizing leukoencephalitis (NLE), and granulomatous meningoencephalomyelitis (GME) is still uncertain, although they are considered immune-mediated diseases. The purpose of the present study is to generate a rodent model(s) of these diseases. Rats were injected with rat cerebral or cerebellar homogenate. Rats injected with cerebral homogenate (Cbr) exhibited vacuolar or malacic changes mainly in the cerebral cortex. CD3-positive T cells and Iba-1–positive and CD163-negative microglia infiltrated and activated around the lesions. IgG deposited in the glial fibrillary acid protein (GFAP)–positive glia limitans from the early phase, and CD3-positive T cells attached to GFAP-positive astrocytes. Autoantibodies against GFAP were detected in the sera. These pathological features of Cbr rats were consistent with those of canine NME. In contrast, rats injected with cerebral homogenate (Cbe) exhibited demyelinating lesions with inflammatory reactions in the cerebellum, brainstem, and spinal cord. The presence of demyelination and autoantibodies against myelin proteins in Cbe rats was similar to murine experimental autoimmune encephalitis and differed from NME, NLE, and GME. All the present findings indicate that autoantibodies together with microglia and T cells may play a major role in the pathogenesis of idiopathic canine meningoencephalomyelitis.
Keywords
Idiopathic necrotizing meningoencephalitis (NE) and granulomatous meningoencephalomyelitis (GME) have been identified in dogs. NE is characterized by malacic changes, perivascular cuffs (PCs), and the infiltration of inflammatory cells in the neuroparenchyma (NP). NE is divided into 2 subtypes, necrotizing meningoencephalitis (NME) and necrotizing leukoencephalitis (NLE), according to the distribution of lesions. 24 NME and NLE may be breed specific.* In contrast, GME, characterized by granulomatous lesions in the cerebral white matter, cerebellum, brainstem, and spinal cord, 22 has been reported in various breeds.
Although the pathogeneses of these diseases have not been fully understood, they may be immune mediated because of (1) the identification of an autoantibody against glial fibrillary acid protein (GFAP) in NME and NLE, (2) the accumulation of autoreactive T cells and B cells against GFAP in the perivascular cuffs and neuroparenchyma, and (3) no detection of specific pathogens. 7,14,18,19,26,28–33 We evaluated the infiltration of inflammatory cells to confirm the differences among these diseases because it is difficult to differentiate these diseases histopathologically in some acute or subacute cases. 22 The proportion of CD20-positive B cells, IgG-positive plasma cells, and CD163-positive macrophages/microglia was not different in these diseases, while that of CD3-positive T cells was significantly high in GME. 22 Interferon (IFN)–γ and interleukin (IL)–17 are expressed in NME and GME cases, respectively. 23 However, whether these diseases are distinct from each other remains uncertain because different immune reactions may be produced by the same pathogen. 4,10 A disruption in the balance of Th1/Th2/Th17 immunity may account for the pathogenesis of these diseases.
Models for NE and GME are needed to confirm their pathogenesis. Murine experimental autoimmune encephalitis (EAE) is a model for human multiple sclerosis (MS). 2,17 The autoimmune response against myelin proteins, such as myelin basic protein (MBP), proteolipid protein (PLP), and myelin oligodendrocyte glycoprotein (MOG), was analyzed, and changes in various responses such as cytokine expression and the dynamics of inflammatory cells were examined using this model. 3,16,20,21,27 However, autoantibodies against myelin proteins, demyelination, and axonal damage associated with EAE have never been detected in canine NME, NLE, and GME. 17,19,20,32
In the present study, rats injected with allogeneic brain tissues were examined to develop a suitable model(s) for canine encephalitis.
Materials and Methods
Rat
Eight-week-old male LEW/SsN rats were purchased from SLC (Shizuoka, Japan). These animals were housed in isolator cages under specific pathogen-free conditions. The experimental procedures in the present study were approved by the Experimental Animal Committee of the University of Tokyo.
Immunization Using Rat Brain Homogenates
The rat cerebrum and cerebellum were homogenized in phosphate-buffered saline (PBS). Cerebral (500 μg/μl) and cerebellar (500 μg/μl) homogenates with heat-killed Mycobacterium tuberculosis H37RA (Difco Laboratories, Detroit, MI) were emulsified in complete Freund’s adjuvant (5 mg/ml; Sigma, St Louis, MO). The emulsion was injected subcutaneously (SC) into the lower flanks of rats. An outline of the experiment is summarized in Table 1. Body weights and clinical symptoms were recorded daily.
Summary of the Experimental Design.
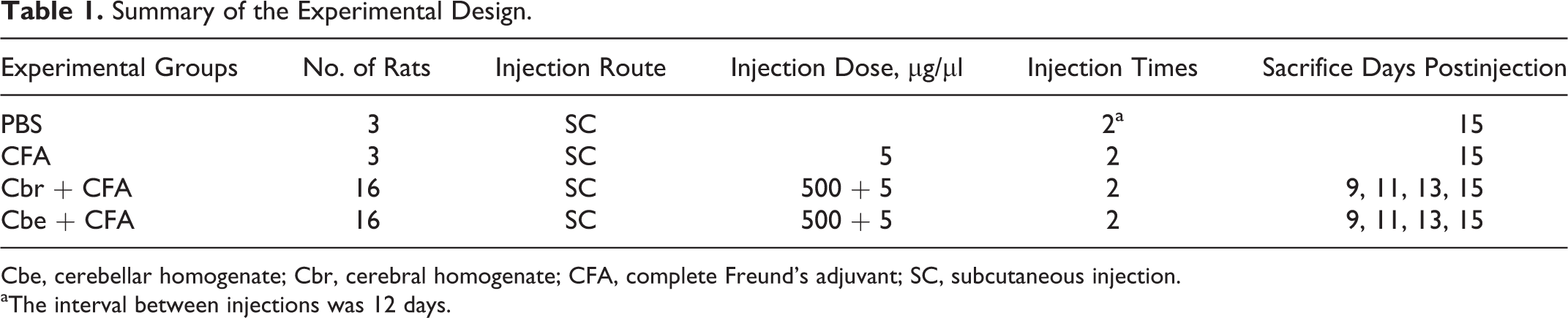
Cbe, cerebellar homogenate; Cbr, cerebral homogenate; CFA, complete Freund’s adjuvant; SC, subcutaneous injection.
aThe interval between injections was 12 days.
Pathological Analysis
The brain and spinal cord were removed from rats euthanized when the loss in body weight and clinical symptoms seemed significant. Brain and spinal cord tissues were fixed in 10% neutral buffered formalin and subsequently embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin (HE) or Luxol fast blue–HE (LFB-HE) for microscopic examinations. Some fresh brain tissues were frozen in Tissue-Tek O.C.T. Compound (Sakura Finetek Japan, Tokyo, Japan) and stored at –80°C.
Immunohistochemistry
Immunohistochemistry (IHC) was performed according to the method in our previous study. 22 The primary antibodies applied are listed in Table 2.
Primary Antibodies Used for Immunohistochemistry, Immunoblotting, and Immunofluorescence.

GFAP, glial fibrillary acid protein; IgG, immunoglobulin G; MBP, myelin basic protein.
Double-Labeling Immunofluorescence
In total, 6-μm-thick paraffin sections were autoclaved at 121°C for 15 minutes for antigen retrieval and treated with skimmed milk at 37°C for 40 minutes to block nonspecific reactions. The first primary antibodies listed in Table 2 were employed at 4°C overnight. Sections were then incubated with the second primary antibodies also listed in Table 1, at 37°C for 1 hour. The secondary antibody cocktail was employed at room temperature for 1 hour. The cocktail included fluorescein isothiocyanate–labeled goat anti–rabbit IgG (1:200; Vector Laboratories, Burlingame, CA), fluorescein isothiocyanate–labeled horse anti–mouse IgG (1:200; Vector Laboratories), Alexa Fluor 594–labeled goat anti–mouse IgG (1:200; Invitrogen, Eugene, OR), and Alexa Fluor 594–labeled donkey anti–rabbit IgG (1:200; Invitrogen). These sections were finally counterstained and mounted with Vectashield HardSet with DAPI (Vector Laboratories) and observed using a Leica DMI3000 B fluorescence microscope.
RNA Extraction and cDNA Synthesis
Total RNA was prepared by the guanidinium thiocyanate-phenol-chloroform method using the ISOGEN kit (Wako, Osaka, Japan) from brain and spinal cord tissues and reverse-transcribed with the PrimeScript RT-PCR kit (Takara, Otsu, Japan).
Real-Time PCR Quantification
Real-time PCR on the ABI PRISM (Applied Biosystems, Foster City, CA) was performed in a total volume of 25 μl of the SYBR Green Realtime PCR Master Mix (TOYOBO, Osaka, Japan). Primers used were CCR2, CCR4, CCR6, CXCR3, GAPDH, IFN-γ, IL-4, IL-10, IL-17, IL-23, transforming growth factor (TGF)–β, and tumor necrosis factor (TNF)–α (Table 3). The PCR reaction was performed on a MicroAmp Optical 96-well Reaction Plate (Applied Biosystems) with an initial degeneration step at 54°C for 30 seconds, at 95°C for 3 minutes, and following 10 cycles at 95°C for 30 seconds, as well as an annealing step at 68°C for 1 minute, at 72°C for 30 seconds, and following 30 cycles at 95°C for 30 seconds. At the end of each cycle, the intensity of the fluorescence emitted from SYBR Green was measured. After completion of the process, samples were subjected to dissociation curve analysis (from 5°C above annealing temperature to 95°C). Samples were then quantified automatically (7900HT version 2.3 Sequence Detection Systems; Applied Biosystems). A single narrow peak was observed by the dissociation curve analysis at the specific melting temperature, and a single band of the predicted size was observed by 2% agarose gel electrophoresis. The results of each case were compared with that of the control group using the ▵▵Ct method, and relative quantification was performed.
Primers Used for Real-Time Reverse Transcription Polymerase Chain Reaction.
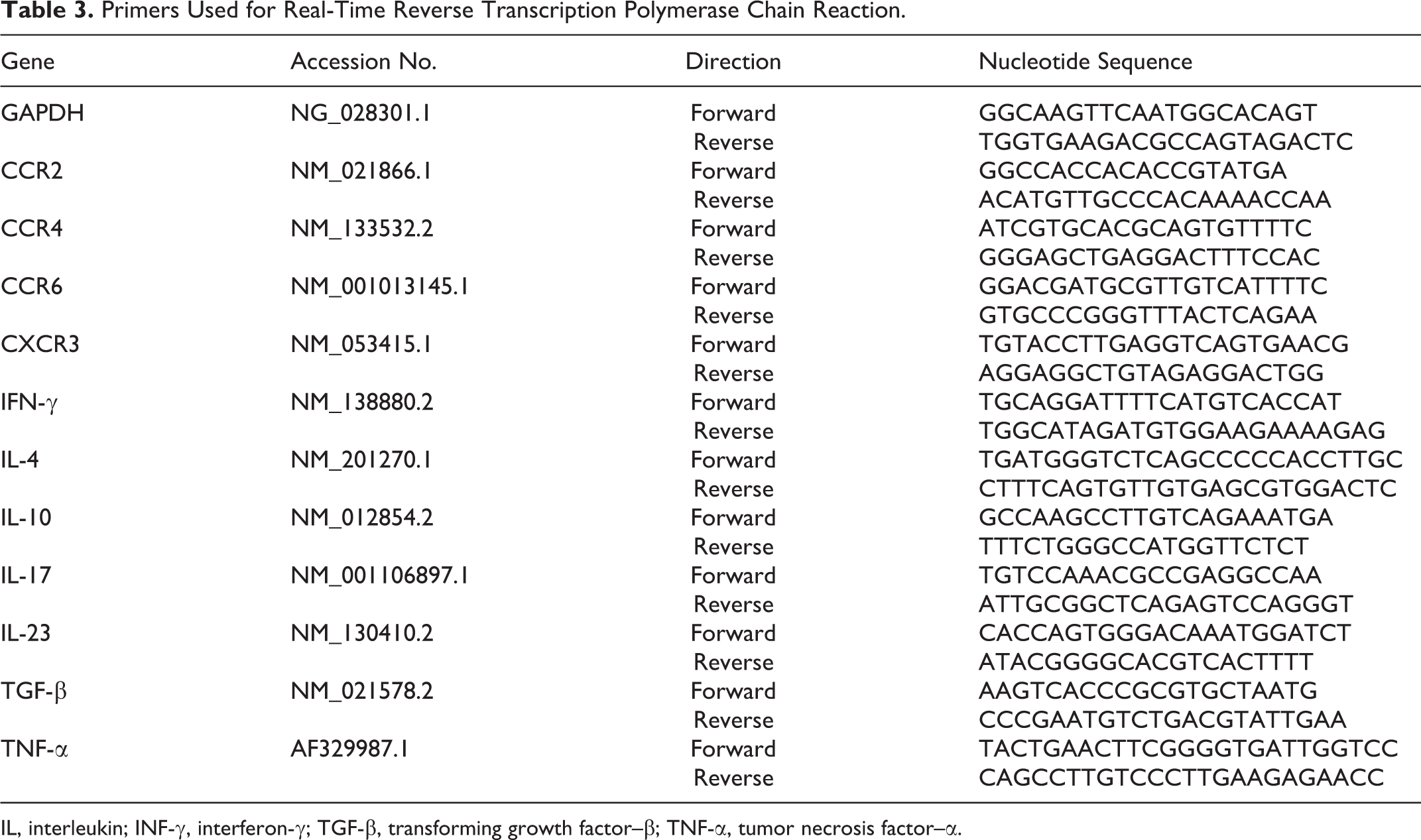
IL, interleukin; INF-γ, interferon-γ; TGF-β, transforming growth factor–β; TNF-α, tumor necrosis factor–α.
Immunoblot
Normal rat brain and spinal cord tissues were homogenized with the TissueRuptor (QIAGEN, Homebrechtikon, Switzerland) and TissueRuptor disposable probes (QIAGEN) in a buffer containing 10 mM Tris-HCl (pH 7.8), 1% NP-40, 0.1% sodium dodecyl sulfate (SDS), 150 mM NaCl, 1 mM EDTA (pH 8.0), proteinase inhibitor cocktail, 2 mM Na3VO4, 10 mM NaF, and distilled water (DW). Lysates were incubated on ice for 30 minutes and centrifuged at 12 000 g at 4°C for 20 minutes. Proteins in supernatants were separated by SDS–polyacrylamide gel electrophoresis (PAGE) and transferred to Immun-Blot PVDF Membranes (Bio-Rad, Hercules, CA). Membranes were incubated with one of the primary antibodies at 4°C overnight and then with appropriate secondary antibodies at room temperature for 1 hour. The primary antibodies used are listed in Table 2, and the secondary antibodies were horseradish (HRP)–conjugated goat anti–rat IgG (1:5000; GE Healthcare, Buckinghamshire, UK) and HRP-conjugated donkey anti–rabbit IgG (1:5000; GE Healthcare). Protein bands were visualized with the Amersham ECL Prime Western blotting detection reagent (GE Healthcare, Uppsala, Sweden) using the Molecular Imager ChemiDox XRS+ with Image Lab software (Bio-Rad, Tokyo, Japan).
Autoantibody Detection
Sera were collected from rats injected with cerebral or cerebellar homogenate for the detection of autoantibodies. The 10-μm-thick cryostat sections of the rat brain were reacted with the sera at 4°C overnight. A secondary antibody against rat IgG (Dako, Kyoto, Japan) was employed at room temperature for 40 minutes. Then, fluorescein streptavidin (1:200; Vector Laboratories) was employed at room temperature for 1 hour. The sections were then incubated with a second primary antibody, mouse monoclonal against human glial filament protein (GFP, prediluted; PROGEN, Heidelberg, Germany) or rabbit polyclonal against human MBP (Dako) at 37°C for 1 hour. The secondary antibodies were then employed at room temperature for 1 hour. The secondary antibodies were Alexa Fluor 594–labeled goat anti–mouse IgG (1:200; Invitrogen) or Alexa Fluor 594–labeled donkey anti–rabbit IgG (1:200; Invitrogen). The sections were finally counterstained and mounted with the Vectashield HardSet with DAPI (Vector Laboratories) and observed using a Leica DMI3000 B fluorescence microscope.
Results
Clinical Signs
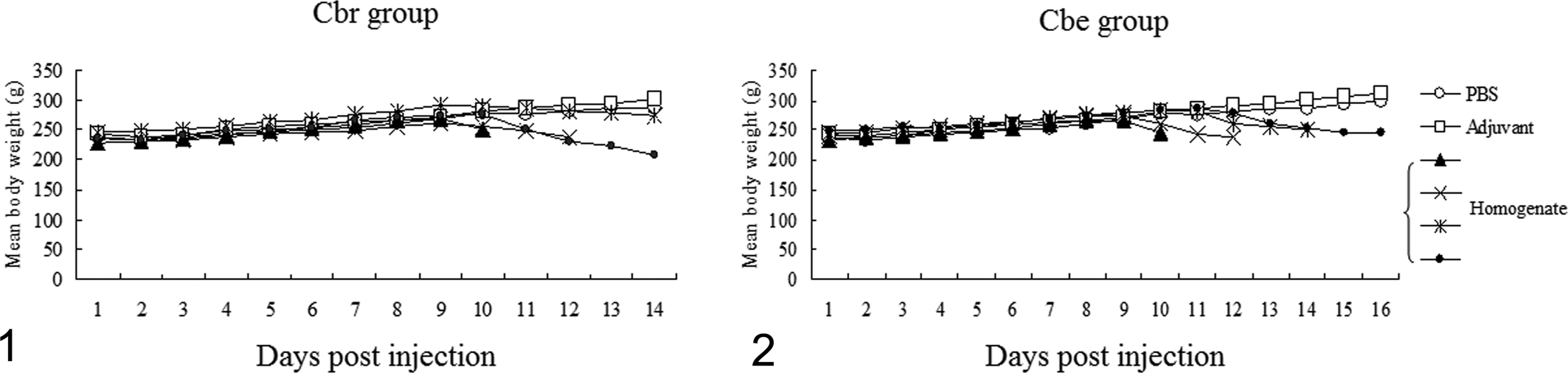
The body weights of the cerebral homogenate-injected (Cbr) rats and cerebellar homogenate-injected (Cbe) rats from 8 or 9 days postinjection (dpi) were lower than those of control rats (PBS-injected or adjuvant-injected rats) (Figs. 1, 2). Cbr rats exhibited mild tremors, head tilting, and circling without paralysis. However, Cbe rats exhibited posterior paresis or paralysis and tremors. No clinical signs appeared in the control group.
Pathological Changes
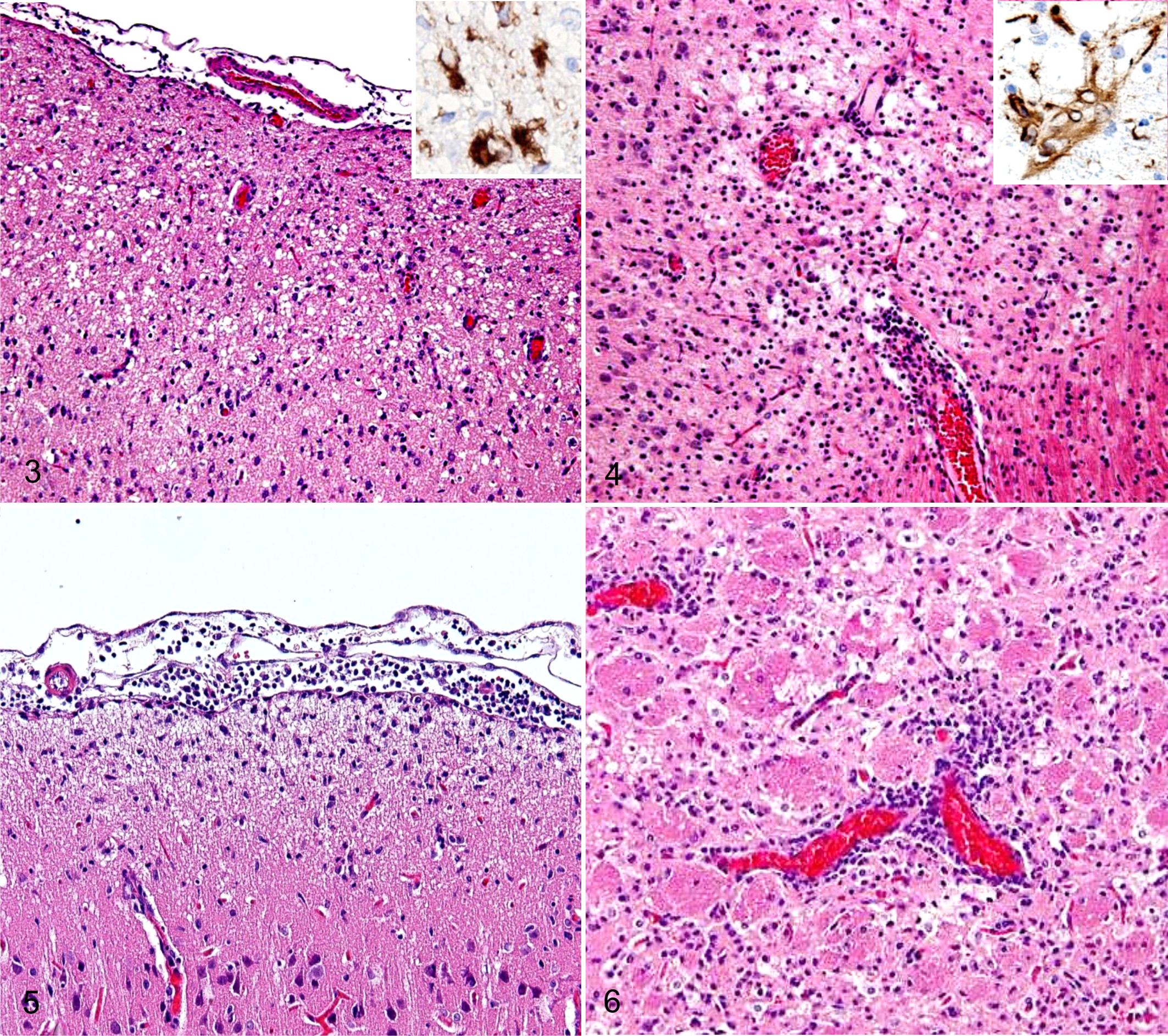
In Cbr rats, vacuolar and malacic changes and the appearance of gemistocytes were observed particularly in the cerebral cortex (Figs. 3, 4). A large number of inflammatory cells infiltrated around the lesions. In addition, perivascular cuffs, the infiltration of inflammatory cells in the leptomeninges and neuroparenchyma, microgliosis, the accumulation of gitter cells, and endothelial hyperplasia were mainly distributed in the cerebrum (Figs. 5, 6). Inflammatory cell infiltration in the leptomeninges was marked in the frontal lobe of the cerebrum from the early phase (Fig. 5). These inflammatory lesions were also observed in the hypothalamus, midbrain, and basal ganglia (Table 4).
Distribution of Lesions in Rats Injected With Brain Homogenate.
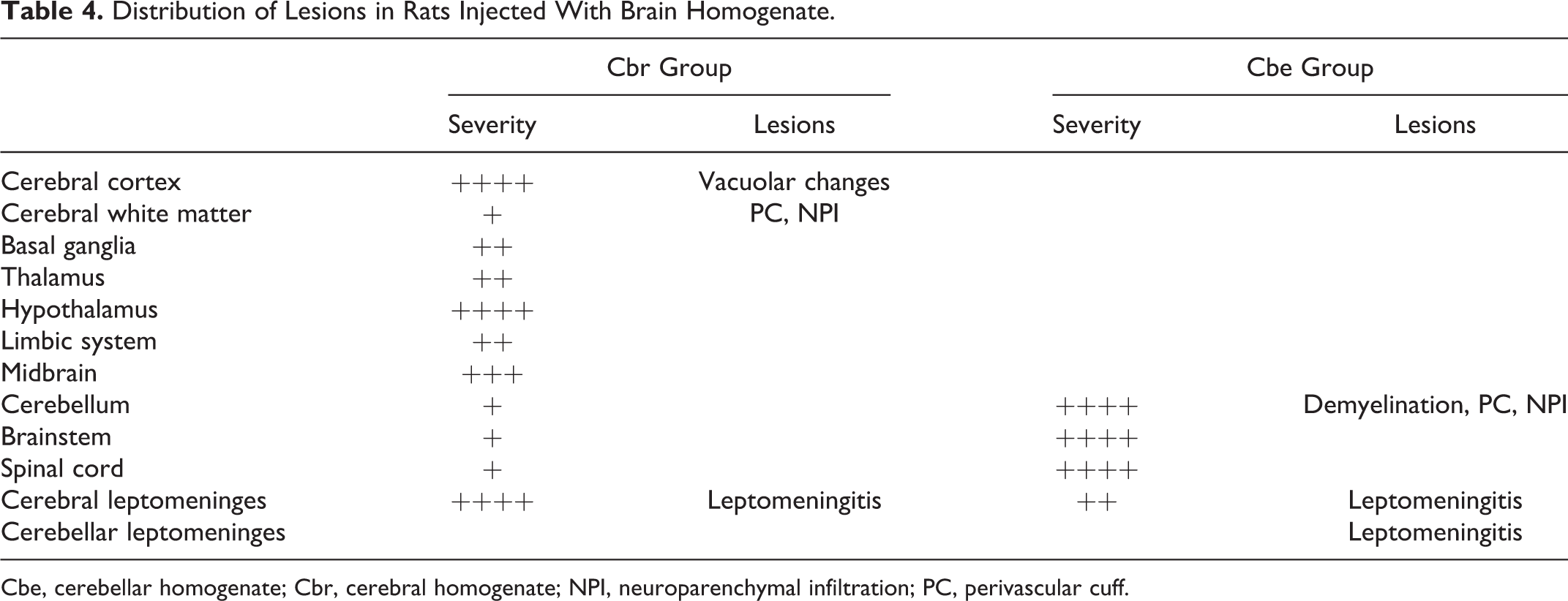
Cbe, cerebellar homogenate; Cbr, cerebral homogenate; NPI, neuroparenchymal infiltration; PC, perivascular cuff.
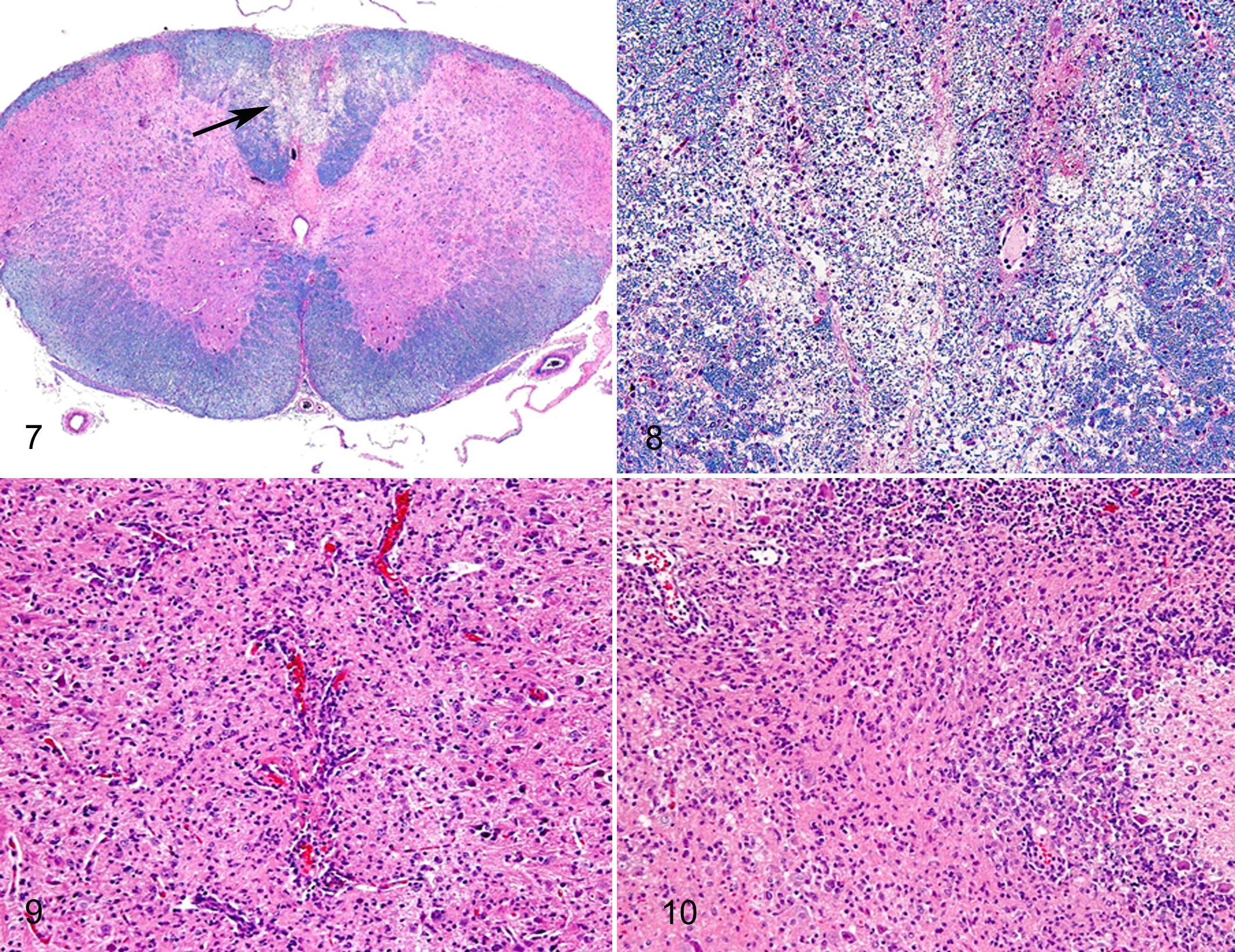
In Cbe rats, lesions were demyelination, microgliosis, perivascular cuffs, inflammatory cell infiltration in the leptomeninges and neuroparenchyma, and endothelial hyperplasia (Figs. 7–10). These lesions were restricted to the cerebellum, brainstem, and spinal cord (Table 4). Demyelination was marked from the brainstem to the spinal cord (Figs. 7, 8).
Immunohistochemical Analysis
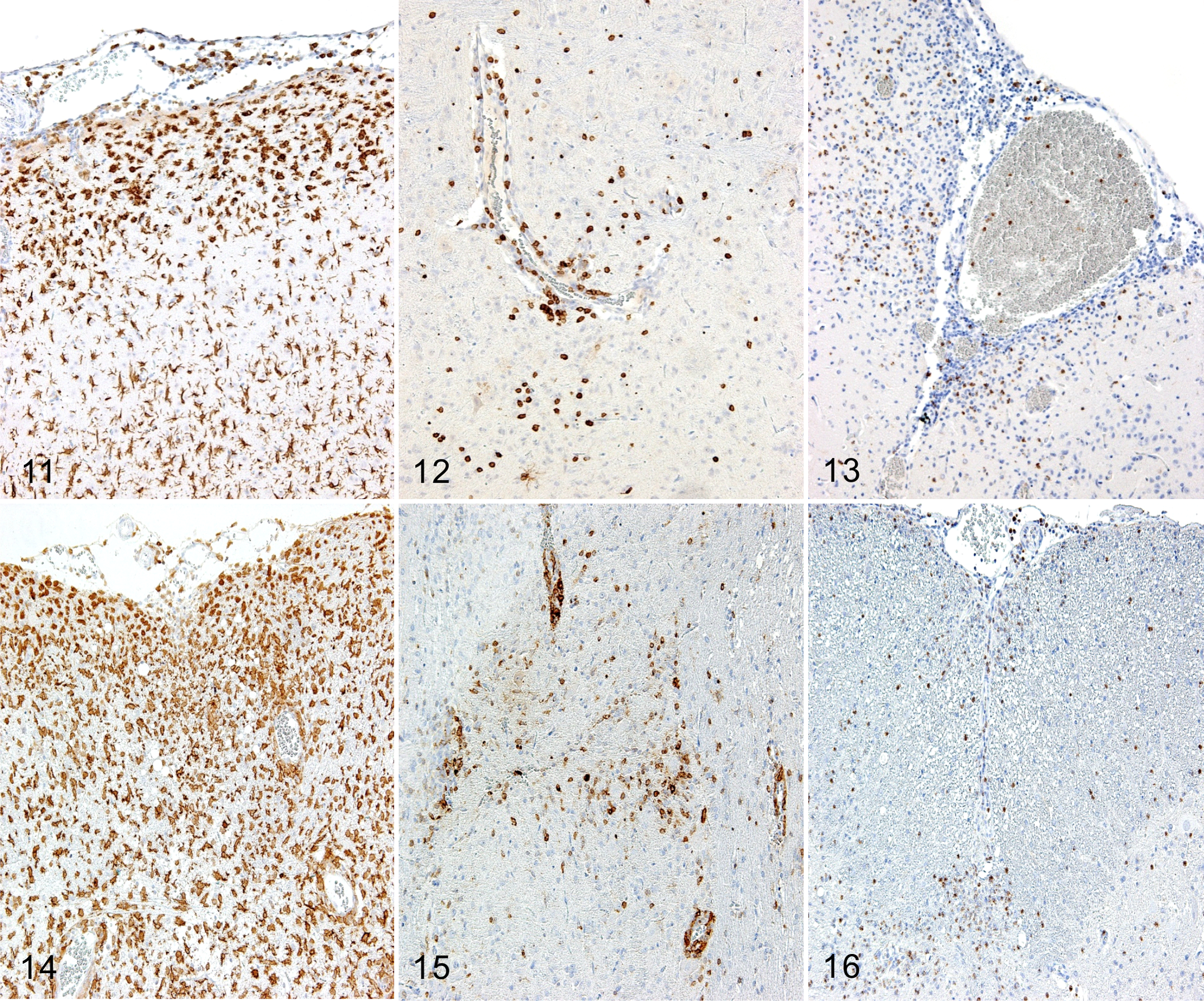
In Cbr rats, CD3-positive T cells, Iba-1–positive macrophages/microglia, and myeloperoxidase-positive neutrophils initially appeared in the cerebral leptomeninges. The infiltration of these cells was marked around the vacuolar or malacic lesions in the cerebral cortex (Figs. 11–13 and Table 4). In Cbe rats, CD3-positive T cells, Iba-1–positive macrophages/microglia, and myeloperoxidase-positive neutrophils appeared in the cerebellar leptomeninges and spinal cord in the early phase and then in the cerebral white matter, midbrain, cerebellum, and brainstem (Figs. 14–16 and Table 4).
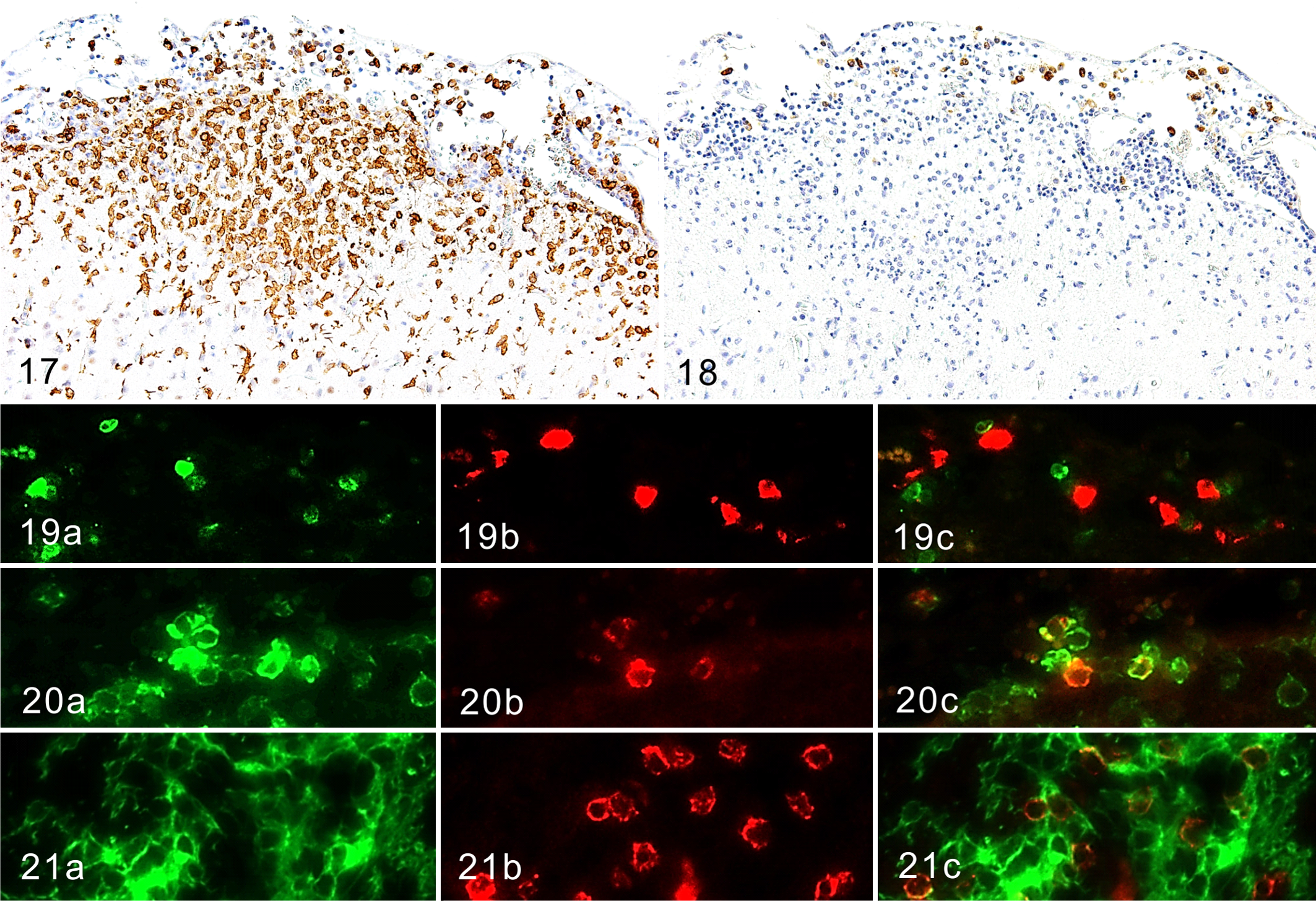
In both rats, the proportion of CD163-positive macrophages was subtle, and their distribution was restricted to the leptomeninges and perivascular Virchow-Robin spaces but not in the neuroparenchyma (Figs. 17, 18). CD163, recognizable by the ED2 antibody, is a member of the scavenger receptor cysteine-rich group B (SRCR-B) family and functions as a scavenger receptor. 9,25 CD163 is expressed in approximately 50% of peritoneal macrophages, a subset of splenic macrophages, and macrophages in other tissues but not in monocytes, alveolar macrophages, or microglia. 9,25 CD3-positive T cells, Iba-1–positive and CD163-negative microglia, and Iba-1–positive and CD163-positive macrophages were in close proximity to each other in the leptomeninges and Virchow-Robin space (Figs. 19–21).
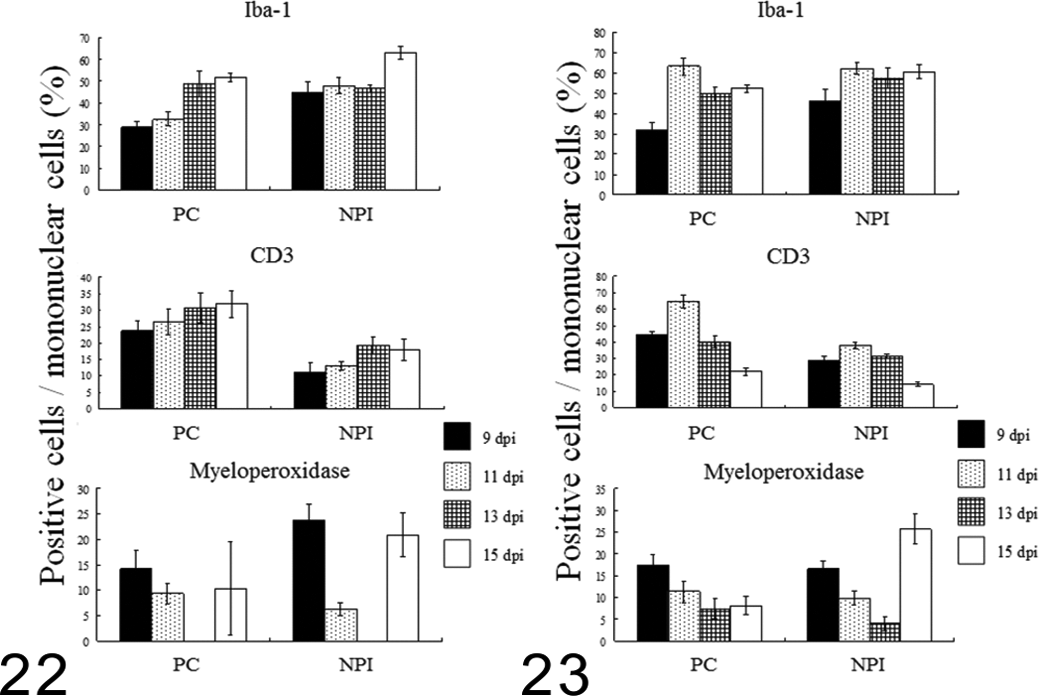
In Cbr rats (Fig. 22), the proportion of Iba-1–positive macrophages/microglia and CD3-positive T cells increased in the perivascular cuffs and neuroparenchyma from 9 to 15 dpi. In contrast, the proportion of myeloperoxidase-positive neutrophils decreased from 9 to 13 dpi in both the perivascular cuffs and the neuroparenchyma but increased again at 15 dpi. The proportion of Iba-1–positive macrophages/microglia in Cbe rats (Fig. 23) slightly increased from 11 to 15 dpi in both the perivascular cuffs and the neuroparenchyma. The proportion of CD3-positive T cells increased once at 11 dpi, both in the perivascular cuffs and neuroparenchyma, but decreased at 13 and 15 dpi. The proportion of myeloperoxidase-positive neutrophils decreased from 9 to 13 dpi, although the infiltration of these cells was marked in the neuroparenchyma at 15 dpi. CD20-positive B cells and IgG-positive plasma cells were not observed in either Cbr or Cbe rats (data not shown).
Expression of Cytokines and Chemokine Receptors
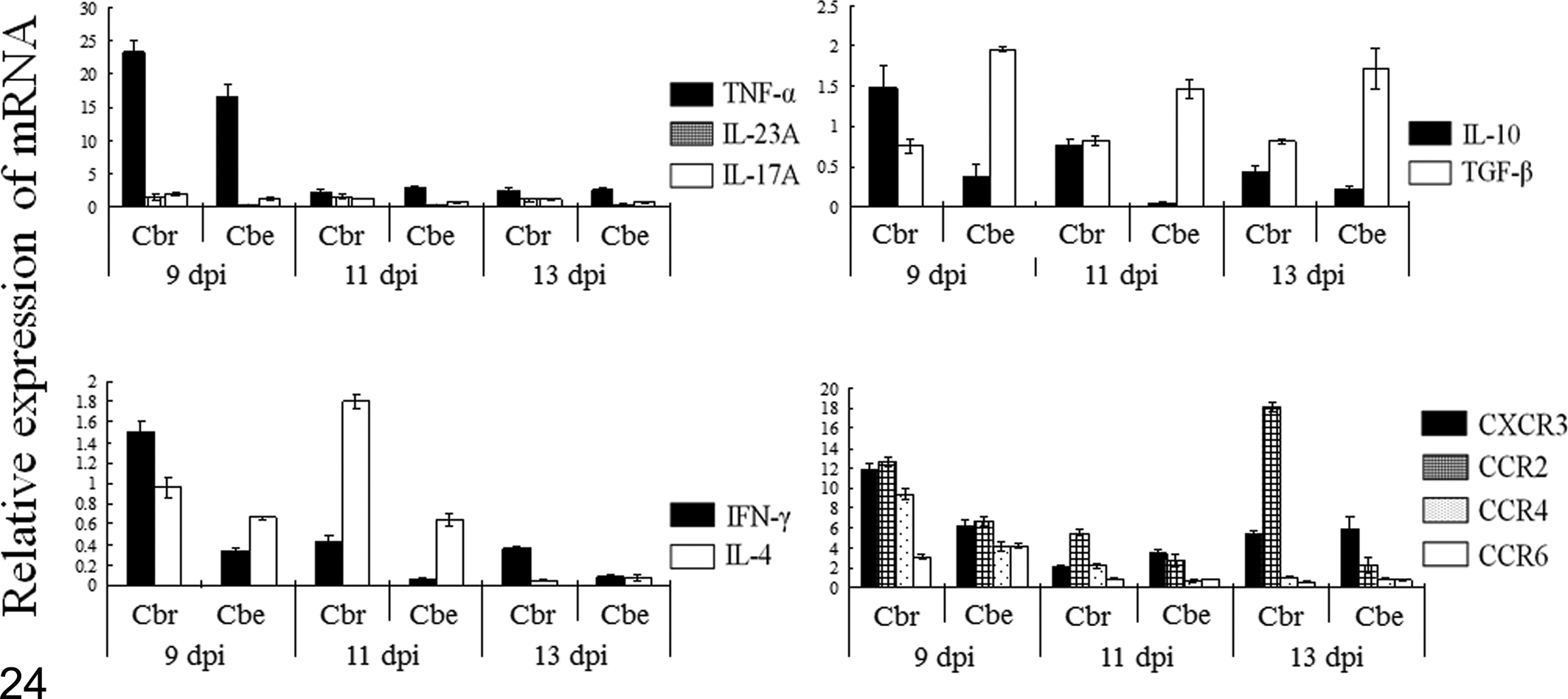
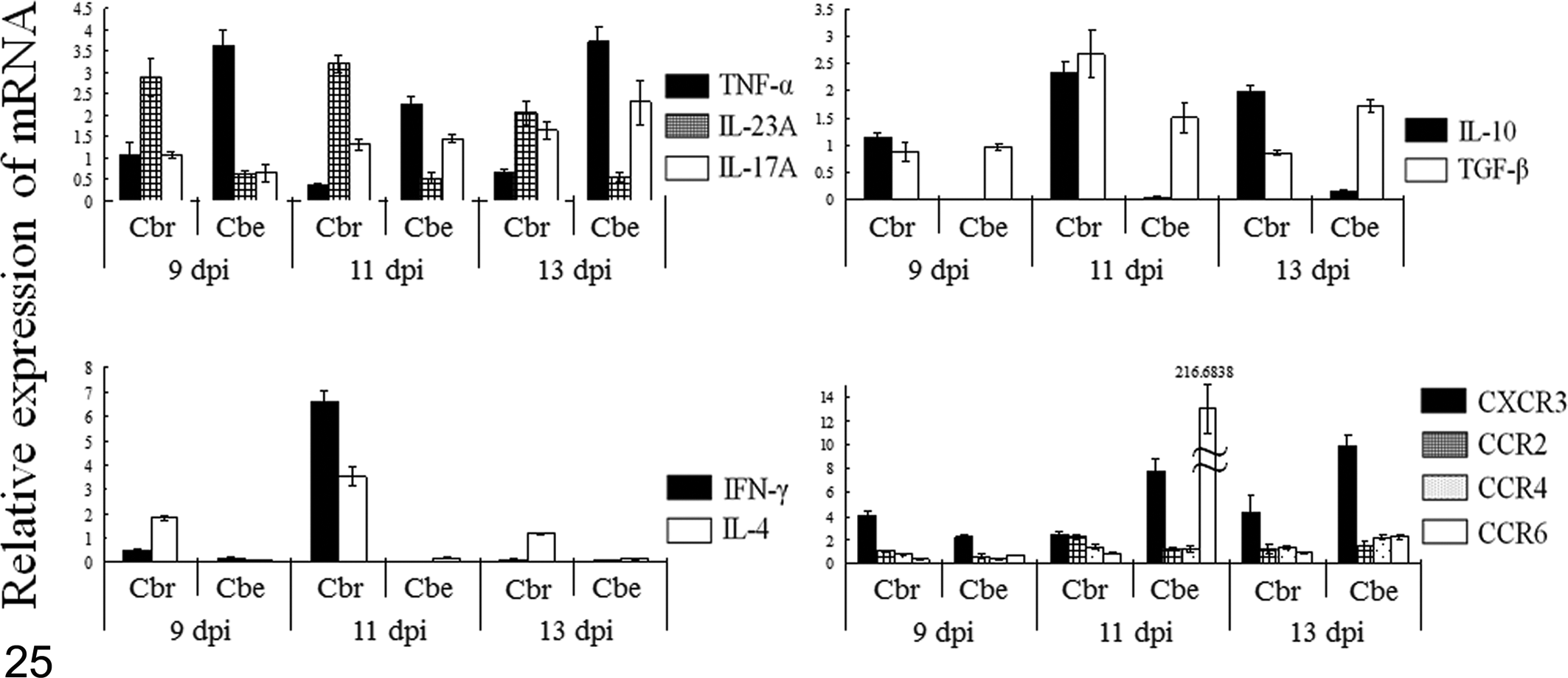
The messenger RNA (mRNA) levels of cytokines and chemokine receptors in Cbr and Cbe rats were measured using real-time reverse transcription polymerase chain reaction (RT-PCR). The results are shown in Figs. 24 and 25. In Cbr rats (Fig. 24), the mRNA level of TNF-α was very high in the cerebrum at 9 dpi. However, there was a marked decrease in the TNF-α level thereafter. The mRNA levels of IL-23 and IL-17 were low on all days in Cbr and Cbe rats. The mRNA levels of IFN-γ and IL-4 were also low. The mRNA levels of IL-10 and TGF-β were moderate, although the level of TGF-β in the cerebellum was slightly higher than that in the cerebrum. The mRNA level of CCR2 was the highest in the cerebrum, followed by CXCR3 at 9 to 13 dpi. The mRNA level of CCR4 was high at 9 dpi and decreased thereafter. The mRNA level of CCR6 was low on all days.
The relative cytokine and chemokine receptor messenger RNA level in the cerebral tissues (Cbr) and cerebellar tissues (Cbe) in rats injected with cerebral homogenate and examined at 9–13 days postinjection (dpi). IFN-γ, interferon-γ; IL, interleukin; TGF-β, transforming growth factor–β; TNF-α, tumor necrosis factor–α.
The relative cytokine and chemokine receptor messenger RNA level in the cerebral tissues (Cbr) and cerebellar tissues (Cbe) in rats injected with cerebellar homogenate and examined at 9–13 days postinjection (dpi). IFN-γ, interferon-γ; IL, interleukin; TGF-β, transforming growth factor–β; TNF-α, tumor necrosis factor–α.
In Cbe rats (Fig. 25), the mRNA level of TNF-α was high in the cerebellum. The mRNA level of IL-23 was slightly higher in the cerebrum than in the cerebellum. The mRNA level of IL-17 increased from 11 dpi and was higher in the cerebellum than in the cerebrum. The mRNA levels of IFN-γ and IL-4 were low, although these were high in the cerebrum at 11 dpi. The mRNA levels of IL-10 and TGF-β were moderate and increased from 11 dpi. The mRNA level of CXCR3 increased in the cerebellum with time. The mRNA level of CCR6 was prominent in the cerebellum at 11 dpi only. The mRNA levels of other chemokine receptors were moderate.
Detection of Autoantibodies
Sera from rats were obtained from 9 to 15 dpi. Sera from Cbr rats reacted with 40- to 50-kDa proteins extracted from the cerebrum, cerebellum, and spinal cord, which corresponded to GFAP. Bands corresponding to MBP of 20 to 30 kDa were also detected (Fig. 26). Sera from Cbe rats reacted with both 20- to 30-kDa and 40- to 50-kDa proteins (Fig. 26). The period when autoantibodies in sera first appeared was different between the Cbr and Cbe groups. Autoantibodies against GFAP in the Cbr group appeared approximately from 11 dpi and the most distinct in 15 dpi. Autoantibodies against myelin protein in the Cbe group appeared prominently from 9 to 15 dpi. However, autoantibodies could not be detected in the sera of rats exhibiting few symptoms or lesions in both Cbr and Cbe groups and the control group (data not shown). Thus, autoantibodies against either GFAP or MBP could be generated in both rats and may be involved in the formation of lesions.
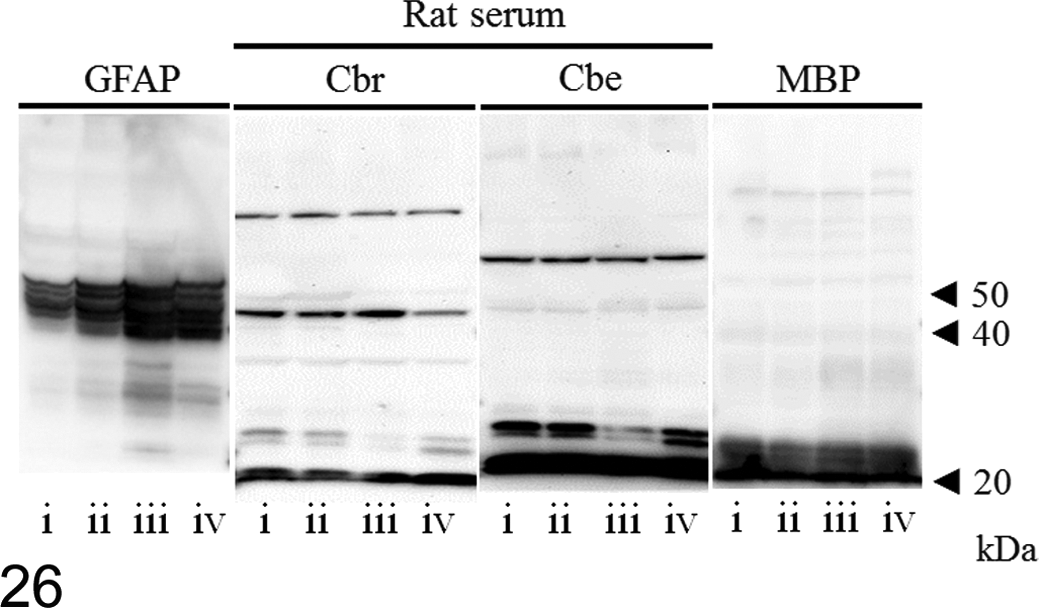
Detection of autoantibodies in sera from cerebral homogenate-injected (Cbr) and cerebellar homogenate-injected (Cbe) rats; immunoblot. Proteins of the cerebrum (i and ii), cerebellum (iii), and cervical spinal cord (iv) from normal rats are extracted and separated by electrophoresis. Anti–glial fibrillary acid protein (GFAP), serum from a Cbr rat (15 days postinjection [dpi]), serum from a Cbe rat (15 dpi), and anti–myelin basic protein (MBP) are used as the primary antibodies. In Cbr rats, strong positive bands are detected between 40 and 50 kDa, corresponding to GFAP. Also, there are weak positive bands between 20 and 30 kDa, corresponding to MBP. In Cbe rats, there are positive bands around 20 kDa, corresponding to MBP.
Sera from Cbr rats reacted to immunohistochemically stained GFAP-positive glia limitans of the normal rat cerebrum (Fig. 27). Also, sera from Cbe rats reacted to MBP-positive neuronal fibers in the white matter of the cerebellum and spinal cord (Fig. 28). Overall, autoantibodies against GFAP and MBP were detected in the sera of Cbr and Cbe rats, respectively.
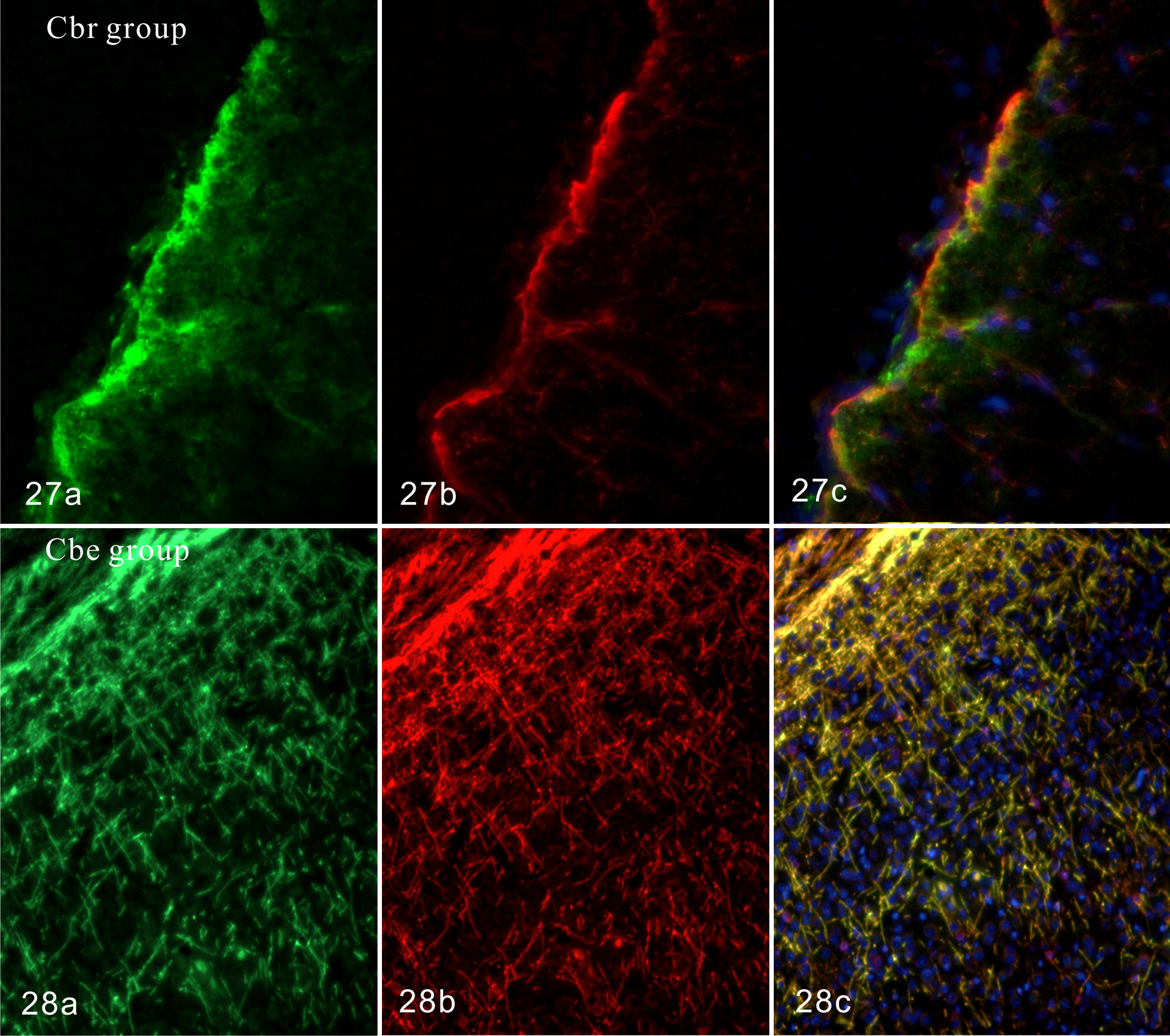
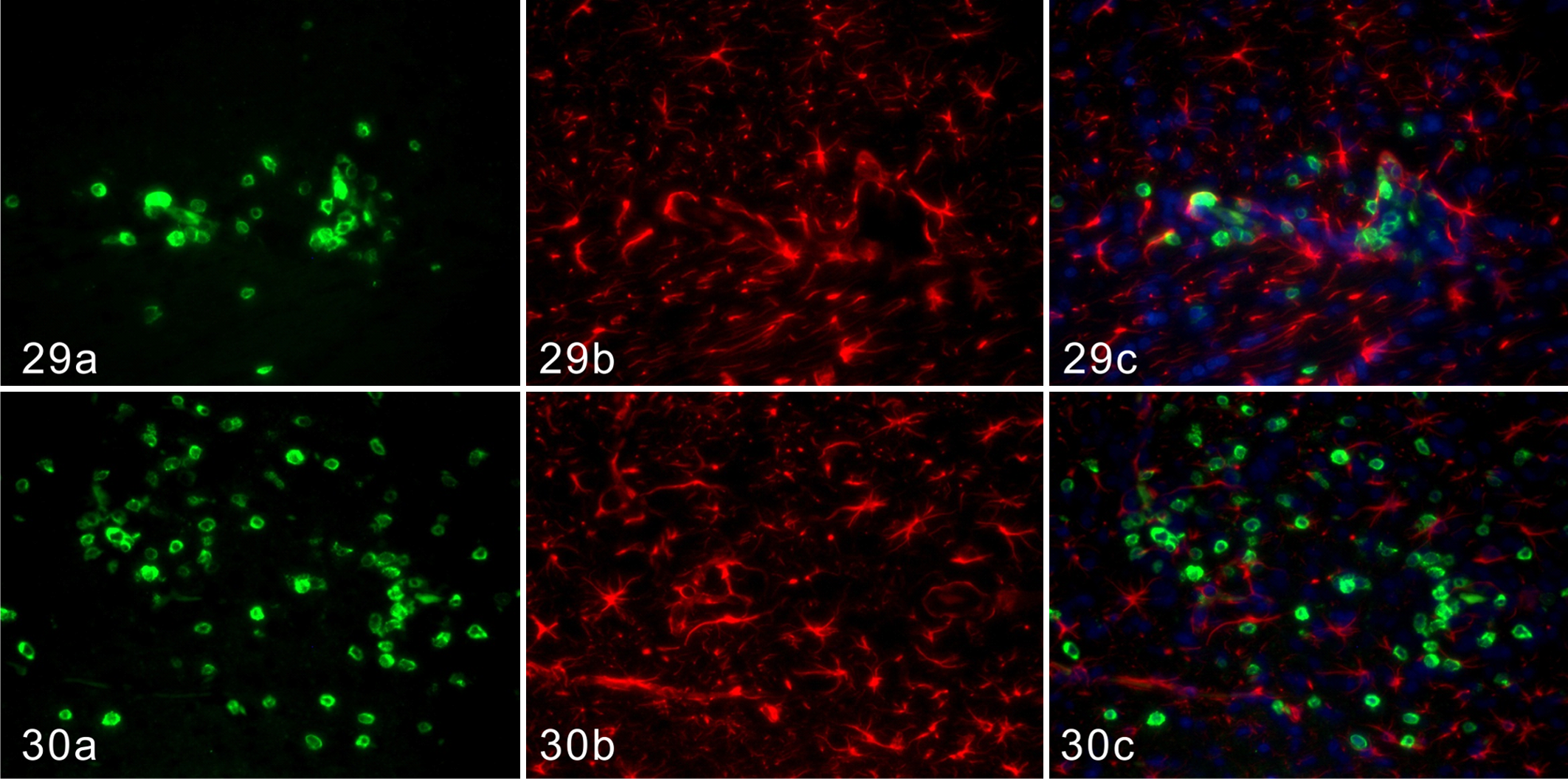
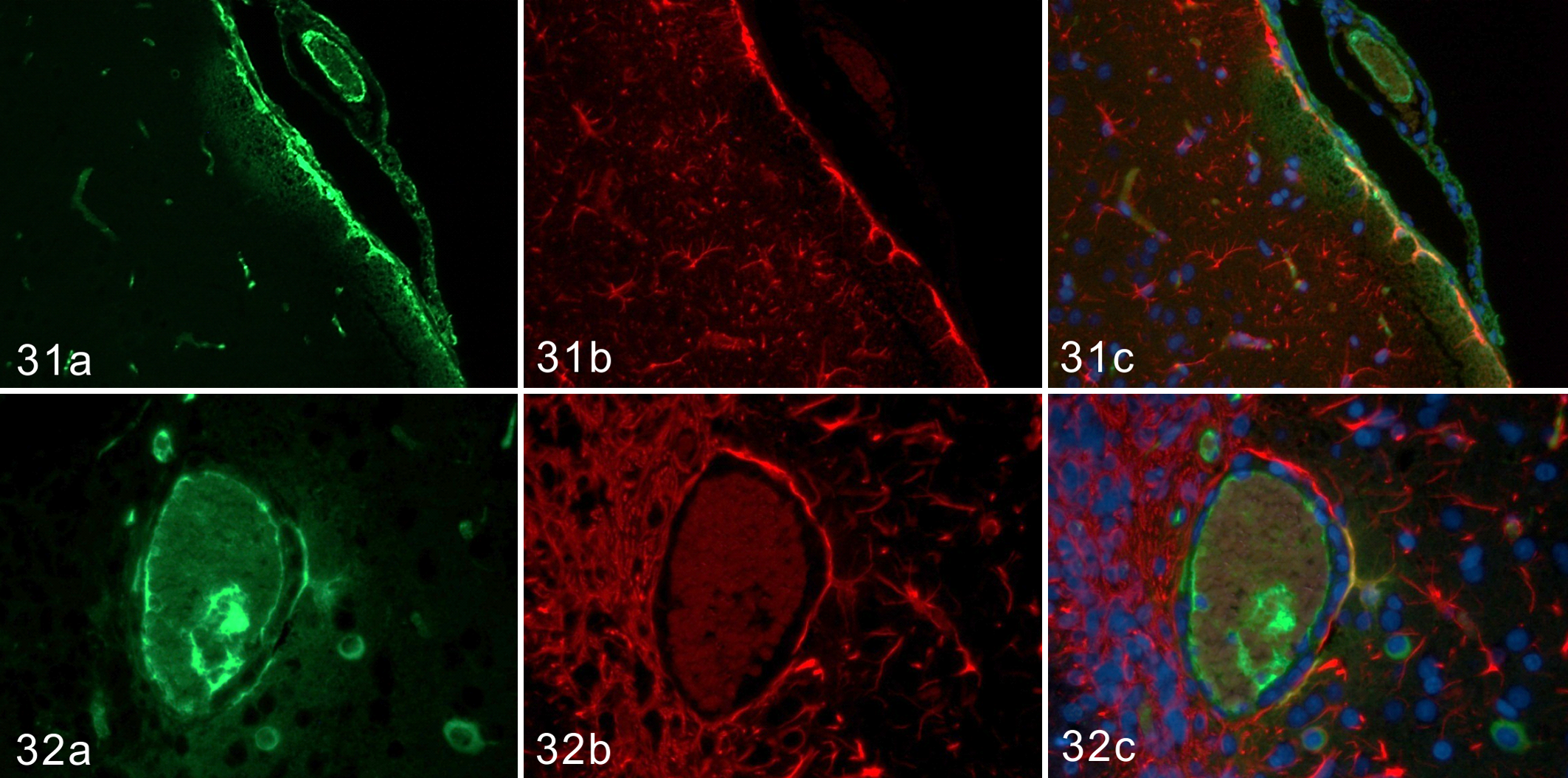
CD3-positive T cells infiltrated in close proximity to GFAP-positive astrocytes in the lesions and increased with time in Cbr rats (Figs. 29, 30). Some parts of the GFAP-positive glia limitans were IgG positive from the early phase to 15 dpi (Figs. 31, 32). A few IgG-positive plasma cells were observed in close proximity to GFAP-positive astrocytes at 15 dpi (Fig. 32). In contrast, such distributions of CD3-positive T cells and IgG were not observed in Cbe rats (data not shown).
Discussion
In the present study, models for canine idiopathic meningoencephalitis, NME, NLE, and GME were generated using rats. Since autoantibodies against GFAP were detected in NME and NLE, and the distributions of the lesions were similar between canine encephalitis and EAE, rats were injected with purified GFAP, MBP, and MOG proteins in our preliminary studies (data not shown). Rats injected with MBP and MOG showed demyelinating lesions with inflammatory reactions but not malacic changes, which were consistent with those of EAE and not those of NLE or GME (data not shown). Also, rats injected with GFAP did not exhibit any clinical symptoms or histopathological changes (data not shown). GFAP is an intracellular protein of astrocytes and is not exposed to inflammatory cells under normal conditions. The injection with GFAP alone, therefore, would not be enough to provoke diseases. Thus, the rat cerebral or cerebellar homogenate was used as a crude allogeneic antigen in the present study.
In Cbe rats, leptomeningitis was observed in the cerebral cortex at the early phase, followed by vacuolar or malacic changes restricted to the cerebral cortex. Other lesions, such as perivascular cuffs and the infiltration of inflammatory cells into the neuroparenchyma, were located in similar regions to those of NME. 22 The percentages of CD3-positive T cells and Iba-1–positive CD163-negative microglia increased according to time in Cbr rats. The infiltration of these T cells and microglia was marked around the vacuolar or malacic lesions of the cerebral cortex. A large amount of CD3-positive T cells and CD163-positive macrophages were observed around the malacic lesions of NME and NLE in our previous study. 22 In contrast, CD163-positive macrophages were restricted to the subleptomeningeal space or the Virchow-Robin space and did not infiltrate into the neuroparenchyma in Cbr rats. Iba-1–positive, CD163-negative microglia were in close proximity to Iba-1–positive, CD163-positive macrophages or CD3-positive T cells in these spaces. Thus, microglia could be activated by macrophages or T cells. 1,11,12 Considering that the lesions in the Cbr group were acute and those of NME and NLE were chronic, microglia may play a key role in the early phase, with macrophages infiltrating into the neuroparenchyma having a role in the late or chronic phase.
The mRNA expression of TNF-α and CXCR3 was high in the cerebrum of Cbr rats. In canine NME cases, the mRNAs of IFN-γ and CXCR3 were highly expressed, indicating the differentiation into the Th1 immune response. 23 In contrast, the expression of IFN-γ and IL-4 mRNAs was moderate, and the differentiation to Th1 and Th2 immune responses was subtle in the Cbr group. These results could account for the acute disease progression in this group. As CXCR3 has been shown to be expressed in the microglia as well as activated T cells such as Th1 cells, the importance of microglia has been implied in this group. 2,5,24 Also, since these phenomena were more prominent in the cerebrum than in the cerebellum, the disease that occurred in Cbr rats may specifically affect the cerebrum.
Autoantibodies against GFAP were detected in the sera from Cbr rats from the early phase, and IgG was deposited in the GFAP-positive glia limitans. This result was consistent with our observation in dogs of the deposition of IgG in the glia limitans in acute NME cases. 22 However, few IgG-positive plasma cells or B cells were observed in rats. Therefore, in the early phase, the association of antibodies against GFAP with lesions has been indicated. Also, CD3-positive T cells were in close proximity to GFAP-positive astrocytes from the comparatively early phase and increased with time after the injection. As T cells were in close proximity to reactive or damaged astrocytes around the malacic lesions or reactive inflammatory lesions in canine NME and NLE cases, T cells have been considered self-reactive. 22 Consequently, the pathogenesis of the experimental disease by the injection of cerebral homogenate may be similar to that of canine NME in the acute phase, and this disease in rats could be a model for NME. Considering the rat model for NME, we can conclude that vacuolar or malacic lesions are induced by T cells and microglia in the comparatively early phase, with these lesions becoming more severe with IFN-γ or IL-4 secreted from activated T cells.
In Cbe rats, lesions such as perivascular cuffs, leptomeningitis, microgliosis, and inflammatory cell infiltration into the neuroparenchyma were observed in the cerebral white matter, cerebellum, brainstem, and spinal cord. These lesions mainly appeared in the leptomeninges of the cerebellum and spinal cord in the early phase and later in the cerebellar neuroparenchyma and cerebral white matter. Such a distribution pattern was consistent with that of NLE and GME. 8,22,28 Moreover, Iba-1–positive, CD163-negative microglia were observed the most, followed by CD3-positive T cells. Similar to Cbr rats, Iba-1–positive and CD163-positive macrophages were restricted to the subleptomeningeal space or Virchow-Robin space. However, in these rats, demyelination lesions with inflammatory responses were observed in the brainstem and spinal cord, and autoantibodies against myelin proteins were detected in sera. However, demyelination lesions and autoantibodies against myelin proteins were never detected in canine NLE and GME. 8,19,22,28–31 Consequently, the pathogenesis of Cbe-injected rats was similar to that of EAE but not to that of GME.
The pathogenesis of NME, NLE, and GME remains elusive, although it has been considered immune mediated. 7,14,18,19,26,29–33 Also, it is not certain whether autoantibodies against GFAP detected in the cerebrospinal fluid or serum are the cause or a secondary by-product. Although the present rat models for canine diseases are, unfortunately, incomplete, some knowledge has been obtained from these models: (1) the importance of autoantibodies against GFAP, T cells, and microglia in the early phase of the diseases; (2) the appearance of specific vacuolar or malacic lesions in the comparatively early phase; (3) the possibility of NME as an autoimmune disease; and (4) the less likely possibility of GME as an autoimmune response against cerebellum proteins. To our knowledge, the construction of a model for NME has not been reported until now. The present results may contribute to the further development of research on canine brain diseases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
