Abstract
The purpose of this study was to investigate clinical and histomorphometric features of cat skin under long-term solar exposure. Ear skin of 34 Domestic Shorthair cats that were chronically exposed to sun was classified as follows: group 0, normal (n = 13); group 1, initial stage of photodamage (PD) (n = 10); group 2, advanced stage of PD (n = 11). Histologic sections were examined independently by 2 pathologists, and epidermal thickness, adnexal unit area, and dermal cellularity were assessed by morphometry. A positive correlation was obtained between age, degree of edema and sclerosis in the upper dermis, telangiectases, squamatization of basal keratinocytes, and epidermis thickness and the degree of PD. The area occupied by adnexal structures in the dermis diminished with increased PD. Dermal sclerosis and edema best separated the 3 groups. The results indicated a high level of skin hypersensitivity to sun rays in cats. The findings may be useful for clinical testing and in general veterinary pathology and dermatology.
Photodamage (heliodermatosis) (PD) describes clinical and histologic changes caused by long-term exposure to solar radiation. 2 In humans, although visible manifestations may take years before becoming clinically evident, the underlying damage occurs from the earliest exposures. 25 Photoaging is a synonymous term for PD, but many investigators limit its use to the more commonly recognized changes of wrinkling and dyspigmentation. 2 The dermatologic importance of photoaging lies in the considerable cosmetic and psychosocial distress it causes in older persons and in its strong, presumably etiologic relationship to skin cancer. 12 Heavy ultraviolet (UV) exposure is also harmful to animals. 18 Thus, global climate changes that result in increased UV radiation may be economically important when affecting cattle and farm-raised fish. 28 In animal models, most of our knowledge about the effects of UV rays is based on studies in mice. 19
Domestic cats usually share the same environment as their owners and may serve as sentinels for cancer development in humans. 27 In white- or orange-faced cats, solar keratosis (SK) is a well-known, naturally occurring dermatosis. Lesions (ill-defined keratotic papules and plaques with epidermal dysplasia) are often multiple and arise on a background of sun-damaged skin (PD). They frequently coexist with frank squamous-cell carcinoma (SCC). 7, 9, 40 SK occurs most commonly on the pinnae, nose, and eyelids, 9, 13, 16, 39, 42 and there are no predilections based on sex or breed. There are, however, predilections based on coat color. 38
During the past few decades, cats have grown in popularity as pets. As a result of increased ownership of cats and the demand for veterinary services, knowledge about skin diseases in cats has rapidly expanded. 32, 38 The relatively high incidence of spontaneously occurring feline SK and the significant association between SK development and white fur make the cat a good model for PD research in natural populations. The purpose of this study was to examine the clinical and histomorphometric features of cat skin under long-term solar exposure. In particular, the study investigated the relationship between morphometric characteristics, age, sex, and clinical features according to a modified Evans classification. 7
Material and Methods
Animals
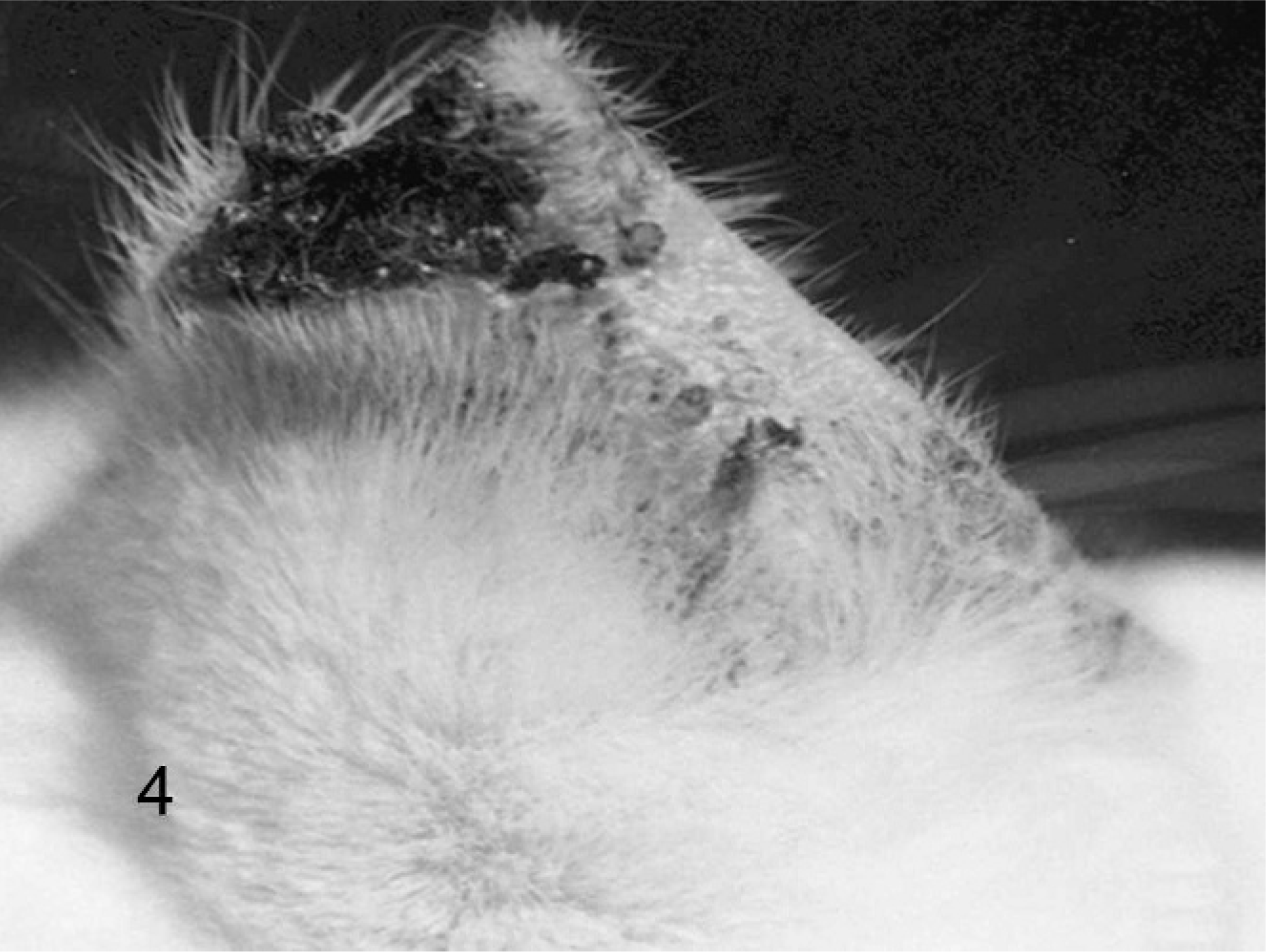
The study was based on 34 cats from the Zoonoses Control Centre (ZCC) of Taubaté, in the state of São Paulo, Brazil, a region of intense solar radiation. The animals were fed commercial dry food and water ad libitum and were housed in a partially enclosed environment. The cats spent most of their time outdoors, especially from 10:00 a.m. to 4:00 p.m., when UV radiation is most intense. The cats developed bilateral ear lesions; incipient lesions with erythematous and scaling patches; and late lesions that consisted of erosions, ulcerations, crusting, plaques, and alopecia. The skin of the ear chosen for the biopsy was clinically classified according to a modified Evans system 7 in the following way: group 0 (n = 13), normal (Fig. 1); group 1 (n = 10), initial PD, with erythematous and/or scaling skin, yet without infiltration, crust, or erosion (Fig. 2); group 2 (n = 11), advanced PD, with at least one of the following signs: erosion, ulceration, plaque, and alopecia (Figs. 3, 4). The control group (group 0) was obtained either from adult animals housed under the same conditions previously described, which had been euthanatized because of problems unrelated to their health status, or from anesthetized animals during ovariosalpingohysterectomy or orchiectomy. All the animals were independently evaluated by 2 observers (EMPA and MLC). The weighted kappa coefficient was calculated as a measure of interobserver agreement. Only chronically sun-exposed, tumor-free skin was used for this study. Areas of possible neoplasia were also biopsied and separately recorded.

Skin, ear; cat No. 11. Normal.
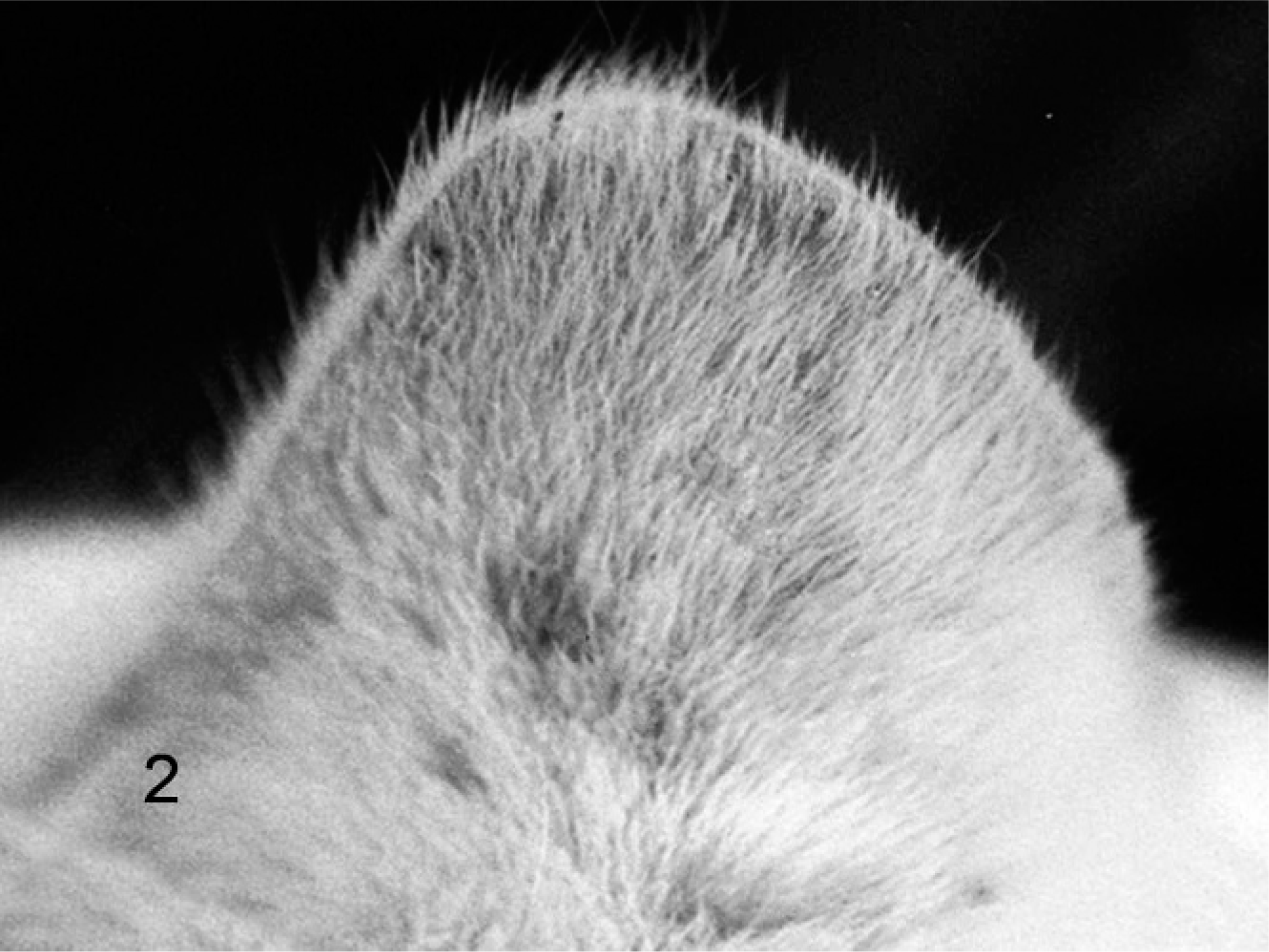
Skin, ear; cat No. 16. Initial PD, with erythema and scaling.

Skin, ear; cat No. 24. Advanced PD, with erythema, scaling, erosion, ulceration, and plaques.
Skin, ear; cat No. 34. Advanced PD and SCC.
Sex, age, breed, skin color at the biopsy site, and the modified Evans clinical classification 7 were recorded. Photographic documentation was performed in parallel. This study was approved by the Committee of Ethics in Biological Research in the State University of Campinas (492-2/2003).
Histologic examination
Skin biopsy specimens of approximately 1 cm2 were obtained, immediately fixed in phosphate-buffered formalin (pH 7.0) solution for 24 hours, and routinely dehydrated and paraffin embedded; 5-μm sections were stained with hematoxylin and eosin. All specimens were independently evaluated by 2 investigators (EMPA and MLC) blinded to the clinical data. Areas with epidermal dysplasia were excluded from further morphometric analysis. In particular, the observers examined the epidermis for changes in melanocytes, irregular melanin pigmentation, squamatization of epidermal basal cells (degenerative process in the basal-layer during which basal cells are replaced by eosinophilic keratinocytes, similar to that of the spinous layer 21 ), and solar lentigo (small pigmented buds at the basal cell layer that protrude slightly into the upper papillary dermis). In the dermis, special attention was given to edema, atrophy (degradation and loss of mature collagen), sclerosis (closely packed collagen fibers with scant space between them), elastosis, and telangiectases (empty spaces lined by flattened endothelial cells). Squamatization of epidermal basal cells, edema, sclerosis, and telangiectasias were quantified by analyzing 10 randomly chosen high power fields (HPF) in the epidermis (first variable) or dermis. For each HPF, the 3 characteristics were separately recorded (1 = present or 0 = absent), so that each variable had a final score between 0 and 1.
Dermal cellularity, defined as the sum of inflammatory, endothelial, and mesenchymal cells within the subepidermal zone, was quantified in 10 randomly chosen HPFs with the help of an eyepiece graticule with cycloides. 11, 20 In every field, the percentage of cycloids that touched or crossed any nucleus was recorded. Adnexal epithelial cells were not recorded.
For the measurement of epidermal thickness and the relative area occupied by adnexae, 5 photographs per biopsy of the epidermis and the adjacent dermis (Axiophot photomicroscope, Carl Zeiss) at a magnification of 20× were digitized, with the stratum corneum placed at the top of the pictures. Separate outlines of the epidermis and the adnexal structures were interactively traced by an observer blinded to the diagnosis (RAC) by using the KS300 software. The acral portion of the adnexal structures was recorded as the epidermal area, and the remainder was recorded as the dermal adnexal area. Epidermal thickness and area, total adnexal area, and the ratio between adnexal and dermal areas were calculated.
Because the 3 clinical groups constituted a biological continuum (from normal animals in group 0 to animals with advanced PD in group 2), we compared the morphometric variables by using Spearman rank order correlations. To evaluate which morphologic changes best identify the clinically defined groups of PD, a discriminant analysis was run in which the 3 clinical groups represented the dependent variable, and the histopathologic or morphometric characteristics represented the independent variables.
To obtain a more realistic classification matrix, the leave-one-out (jack-knife) method was applied. For this procedure, new data sets were constructed on n – 1 subjects, where the missing case was classified according to the remaining n – 1 cases. This technique permitted an estimate of the stability of the created model. For stable models, the results obtained after the jack-knife procedure are very close to that of the overall classification matrix. 3, 23 Finally, the morphometric variables of the skin in cats presenting in situ or invasive carcinomas were compared with those of tumor-free animals by applying the Mann-Whitney rank sum test. For all calculations, the Winstat 3.0 statistical program and SPSS 10.0 software were used.
Results
Animals
There were no significant differences between male and female animals of differing ages, clinical group, or morphometric parameter. In nearly all animals, the area that showed PD was a white color (95.2%). When looking at the 3 groups as a continuum, from normal animals in group 0 to animals with advanced PD (group 2), a positive correlation between the age and the degree of the PD (r = 0.424; P = .0125) was found 1 (Fig. 5a). The interobserver agreement for the clinical classification of the PD was very good, with a weighted kappa value of 0.87.
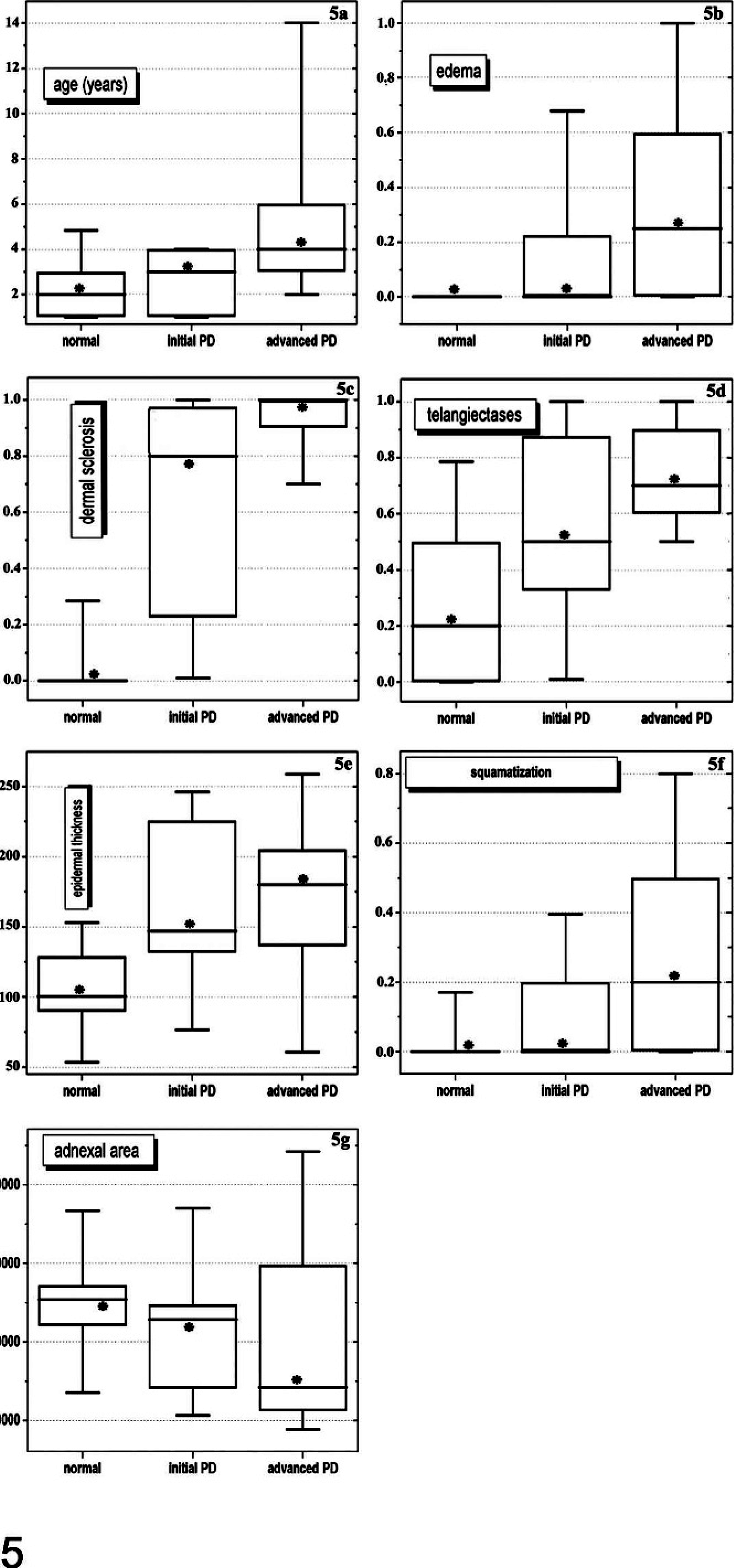
Box plot design of data by using the Spearman rank/order correlations between clinical photo damage (PH) groups (X-axis) and morphometric variables (Y-axis). Fig. 5a to 5f show positive correlations between the variables and the degree of the PD (median values∗ of each variable increased gradually with PD). Fig. 5g shows, to the contrary, that the area occupied by adnexal structures in the dermis diminished when PD grew. Whiskers represent 5 and 95 percentiles. Y-axis values in Figs. 5b to 5d, and 5f represent mean values in 10 HPF, with zero being absent and 1 being present. A field is considered positive when the variable is at least visibly present. Epidermal thickness values are expressed in μm; adnexal area values are expressed in μm2.
Histologic examination
The following histologic and morphometric variables revealed continuous changes with increasing PD:
Subepidermal edema was absent in the control group (Fig. 6), minimally present in group 1, and more intensively present in group 2, thus revealing a positive correlation with the degree of PD. (r = 0.604, P < .001) (Figs. 5b, 7). In a similar way, the degree of sclerosis in the upper dermis increased gradually with PD (Spearman correlation coefficient r = 0.803, P < .0001) (Figs. 5c, 7). Furthermore, telangiectases were more frequent, with increasing degrees of clinical PD (r = 0.542, P < .001) (Figs. 5d, 8). Parallel to this, the epidermis was thicker (r = 0.526, P = .0014) (Figs. 5e, 7, 8) and more irregular. The latter was estimated by variance of epidermal thickness, which reflected the variability of the thickness within a case, which increased with the degree of PD: r = 0.354, P = .040. Moreover, areas with squamatization of basal keratinocytes were more frequent (r = 0.564, P < .001) (Figs. 5f, 8). To the contrary, the area occupied by adnexal structures in the dermis diminished (r = −0.347, P = .044) when PD increased (Fig. 5g). Animals in group 2 exhibited the smallest adnexal area, the histologic substrate of the alopecia. Dermal cellularity, however, was not related to PD (r = 0.13, P = .45). Melanocyte hyperplasia and elastosis were not seen.
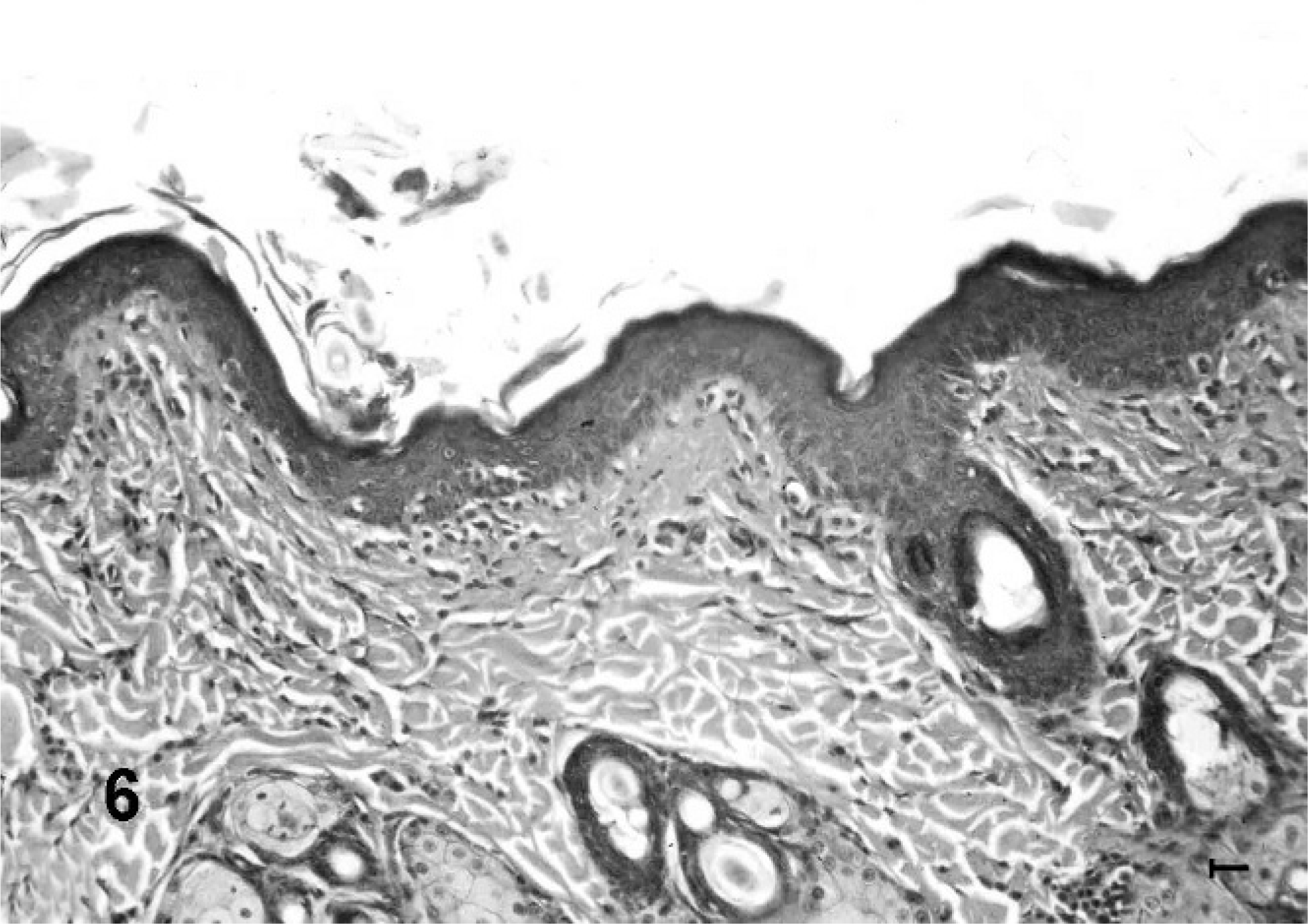
Skin; cat No. 10. Normal, wavy configuration of the epidermis, laminated cornified layer, and dermis with interlaced collagen bundles (Cat no. 10). HE. Bar = 0.01 mm.
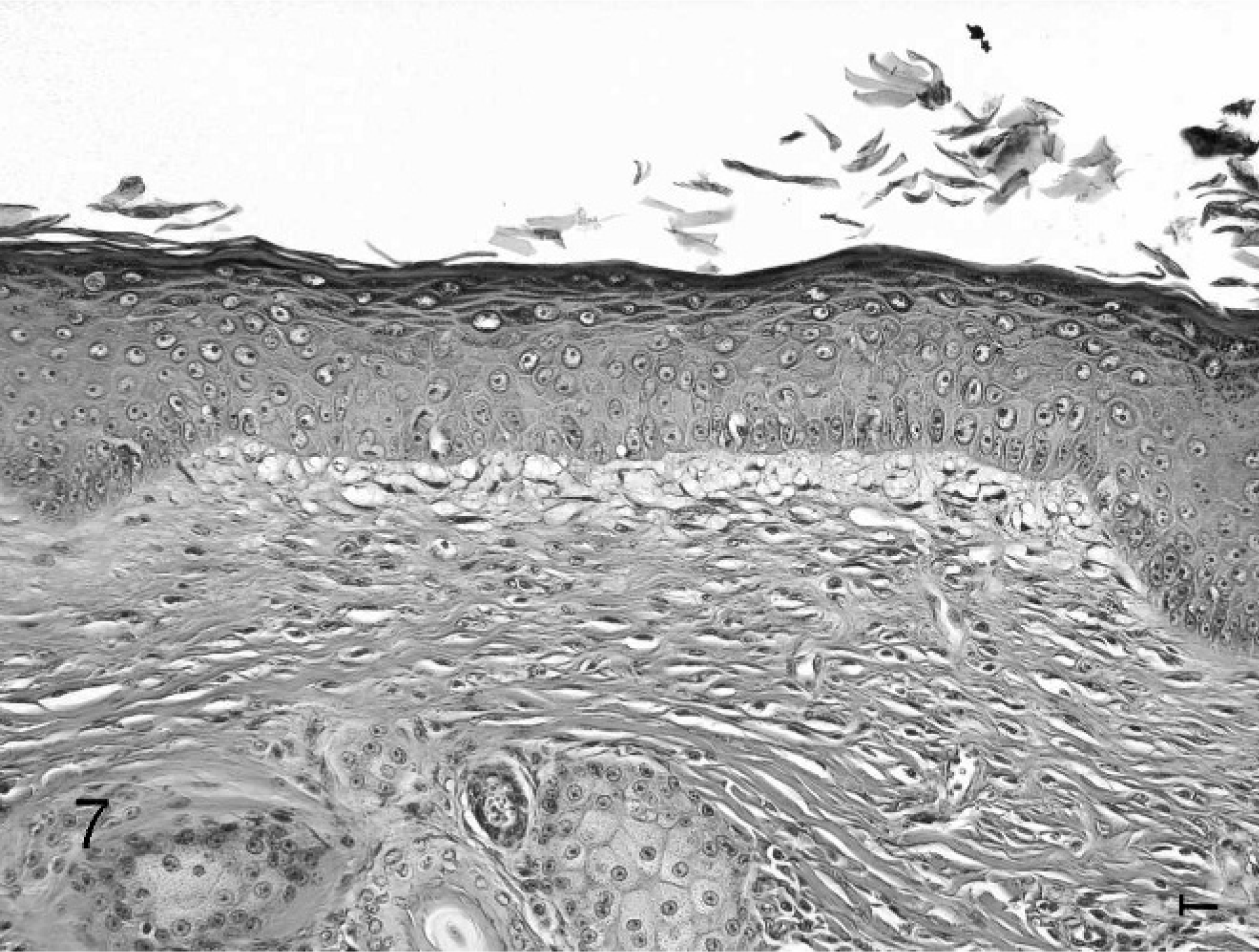
Skin; cat No. 29 (advanced PD group). Thickened epidermis, increased dermal collagen in a horizontal compact arrangement and subepidermal edema. HE. Bar = 0.01 mm.
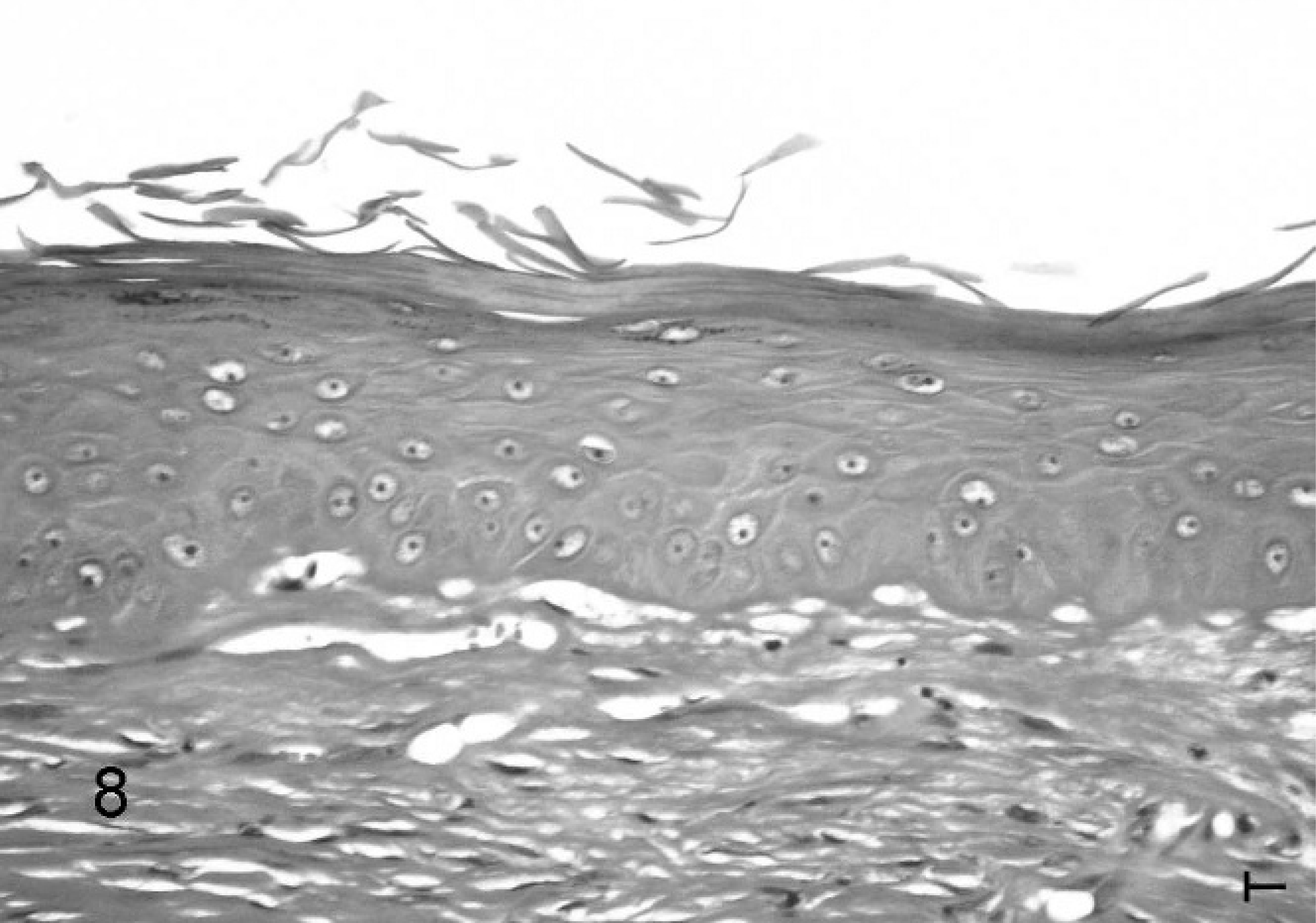
Skin; cat No. 31 (advanced PD group). Compact orthokeratosis, epidermal hyperplasia, squamatization of basal cells, sclerosis, and telangiectases throughout the upper part of the dermis; HE. Bar = 0.01 mm.
Three of the 34 cats (nos. 18, 19, and 29) developed foci of in situ, Bowen-like carcinoma characterized by irregular dysplastic epidermal thickening, with disorientation and pleomorphism of the keratinocytes (Fig. 9). Another 3 cats (nos. 22, 32, and 34) had invasive SCC (Fig. 4). These tumors appeared in areas not included in the morphometric study.
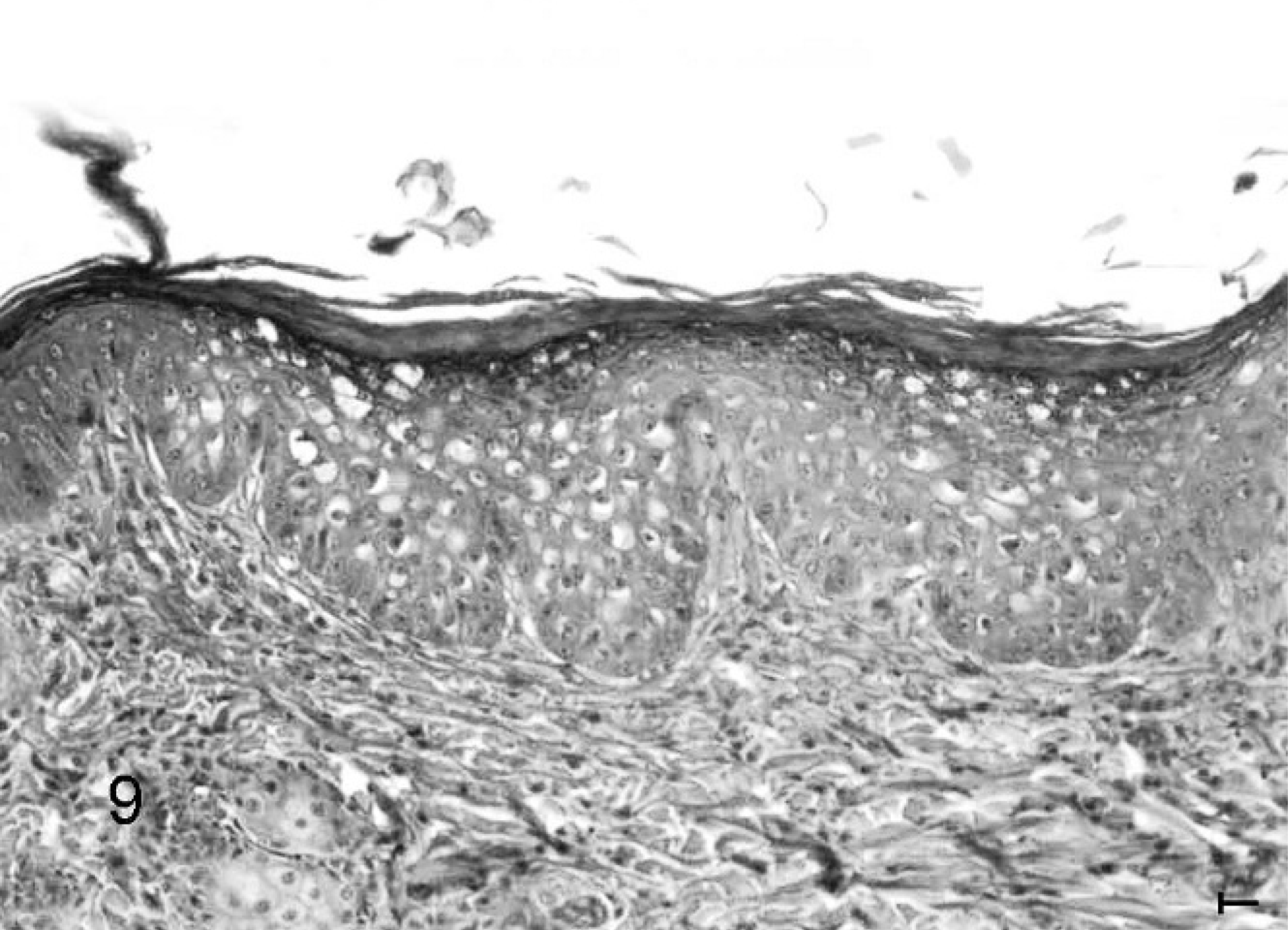
Cat skin, in situ (Bowen-like) carcinoma: irregular dysplastic epidermal thickening, with disorientation and pleomorphism of the keratinocytes (Cat no. 19, initial PD group). HE. Bar = 0.02 mm.
To evaluate which morphologic changes best identified the clinically defined groups of PD, we ran a discriminate analysis, where the 3 clinical groups represented the dependent variable and the histopathologic or morphometric characteristics were included as independent variables. In a linear stepwise discriminate analysis, the final model contained only the variables “dermal sclerosis” and “edema.” Together, these variables best identified the 3 groups, correctly classifying 73.5% of the cases. All the controls (n = 13) were correctly recognized, as well as 5 of the 10 animals with initial PD and 7 of the 11 animals with advanced PD. After the application of the jack-knife procedure, this percentage was 70.6%, thus revealing relative stability of the model. All other variables were excluded from the final model.
When comparing the morphometric and clinical characteristics of the skin between the animals that had developed in situ or invasive carcinoma and those without neoplasia, we found increased epidermal thickness (P = .004) and more severe compaction of dermal collagen (P = .025; Mann-Whitney test) in tumor-bearing cats.
Discussion
Ample epidemiologic and experimental evidence indicates that chronic solar irradiation in humans induces a continuum of changes in the epidermis and the dermis. 6 Clinical manifestations include roughness, laxity, mottled pigmentation, scaling, telangiectasia, and purpura. By histologic examination, human epidermis that has been injured by exposure to UV light may appear either hypertrophic or atrophic. There is striking variability in thickness, pigmentation, and the degree of nuclear atypia for both keratinocytes and melanocytes. 12 Eventually, SK and malignant tumors may develop.
The histologic changes of the dermis in dermatoheliosis in humans were described by Unna in 1896, and by a great many researchers since then. 30 Changes in the dermis include solar elastosis (a hallmark of PD), degradation, and loss of mature collagen and telangiectasis, as well as mixed chronic inflammatory infiltrate. 26, 29 Photo-aged human skin shares decreased dermal collagen and a decline in Langerhans cells with intrinsically (chronologically) aged skin (IA). Other histologic alterations in IA are reduced elastin content and a general decline in a variety of cell populations, including melanocytes 1 and the number of hair follicles. 31 Repeated exposure to the sun accelerates the intrinsic aging process. Elastosis, inflammation, and dyspigmentation, however, are almost never found in unexposed skin, even in the aged. 12 Chronic UV radiation is also potentially harmful to dogs, cats, cattle, sheep, horses, and farm-raised fish. 40 In this study, we considered clinical and morphometric features of PD in cats.
Samples were collected by means of scalpel excision, to avoid rotation and scraping such as occurs with a punch biopsy. 32 The ear was the chosen site, because it is the area most frequently affected by solar rays. In contrast with dogs, 18 cats spend relatively little time in dorsal recumbence, and because their ventral hair is thicker, feline SK occur almost exclusively on the head. 40 In this study, no difference was found between groups distinguished by sex, as has already been observed by others. 38 In contrast, SK is more prevalent in male subjects, especially in the early decades of life, 24, 36 because of behavior patterns. 15, 33, 43 Our sample consisted exclusively of Shorthair cats, because this was the only type available at the facility. According to Sherding, in SK there is no breed predilection, except for coat color. 38
Chronic solar-induced lesions occur most commonly in white-faced, blue-eyed 37 cats exposed to excessive UV light. 9, 13, 16, 38, 39, 40, 48 Among our cases, the color most frequently found in the area affected by PD was white (95.2%), which corresponded to what was already observed in SK. Only 3 cats in the normal group had white skin, because, when animals were being selected, white-eared cats already presented sun-damaged skin. Among the almost 100 cats that lived in the ZCC, 23 white-eared cats were found and 20 had ear PD.
The mean age of affected animals was 3.28 years (1–14 years). According to the literature, in the most susceptible cats, the first lesions may occur as soon as 3 months of age, and they become progressively more severe with each passing summer. 37 Gomes et al. 14 described 7 Domestic Shorthair white cats that lived in an area of high-intensity solar rays (Rio de Janeiro, Brazil) affected with SK associated with SCC. The animals were 6 to 8 years old, 2.2 years younger than the average age reported in the literature. Also, although lesions occurred in older cats in our study, affected animals were younger than those cited in the literature. This would indicate more intense exposure to the sun. In humans, one of the most important factors for SK is age. Among Caucasians, it was found in less than 10% of people in their 30s, but in more than 80% of people in their 70s. 36
On clinical examination, feline skin chronically exposed to the sun presents mild erythema in affected areas, particularly the ear margins, preauricular areas, periocular areas, nose, and lips. 38 It is at these sites, particularly in white cats with blue eyes, that preneoplastic and early neoplastic changes may be seen, as was found in 6 of the cats. As the lesions worsened, scaling, crusting, and pruritus developed. Advanced lesions were accompanied by severe erythema of the ears, scaling, and marginal crusting formation. 37 In the late stages of the disease, there was extensive ulceration and destruction of the affected areas. 13
The histologic parameters selected for a preliminary analysis included the classical aspects of PD human skin, 12, 35 namely, melanocyte hyperplasia/atypia, squamatization of epidermal basal cells, solar lentigo, dermal collagen atrophy, elastosis, and telangiectases. Signs of solar lentigo, as often seen in human skin, were not observed. Nor were melanocytic hyperplasia/atypia and melanin hyperpigmentation of keratinocytes found. With regard to squamatization of basilar cells, there were differences between the groups. In humans, basal layer cell squamatization is seen in SK, 5, 10, 43 and in lupus erythematosus. 21 Abnormal light sensitivity (“photosensitivity”) is known to be a major feature of lupus erythematosus. 21
With regard to upper dermal edema in humans, it is characteristically seen in the papillary dermis in diseases associated with photosensibility, such as polymorphous light eruption (early phase) and lupus erythematosus, 21 as well as radiodermatitis. 22 In humans, dermal sclerosis is also a typical feature of radiodermatitis. 22 The results of this study identified that the presence of dermal edema and sclerosis differentiated normal cats from affected cats. According to Nikula et al., 33 in feline SK, the papillary and reticular dermis layers are thickened by collagen, which, in long-standing cases, becomes dense and poorly vascularized. In human PD, however, degraded collagen can be recognized. 30 After an initial increase in collagen synthesis, mature collagen becomes degraded and is lost. In hairless mice, as a result of UV-B radiation, the skin has higher numbers of fibroblasts. These appear to be metabolically hyperactive, producing more collagen early on, which results in a thickening of the dermis, 26 as was also observed in affected cats in this study. It was concluded that, in both cats and hairless mice, fibrosis is a reparative response to PD. This may well be adaptive, enabling the integument to survive under adverse conditions.
The perception of age as well as beauty in humans is largely dependent on the appearance of exposed skin, and its condition is dictated, in part, by environmental effects, especially UV light. More and more individuals are seeking treatment for reversal of age/environmental-associated changes in skin. Collagen production is decreased and degradation is enhanced in human skin after UV radiation. The use of resurfacing and laser techniques results in increased dermal thickness (as occurs in PD cat skin), giving the appearance of rejuvenation. 35 Perhaps further studies in skin biology will elucidate the mechanisms by which such different PD responses occur, when comparing humans and felines.
No solar elastosis was found in any of the animals. Solar elastosis is a rare manifestation of cutaneous lesion induced by UV rays in cats, 16, 37, 40 because elastic fibers in cats are only present in small numbers, compared with humans. 13 In humans, solar elastosis is commonly found, because elastic fibers are abundant, change color frequently, and become fragmented when there is excessive exposure to UV rays. 5, 10, 41, 43 The degree of elastosis in humans was shown to correlate with the relative amount of UV rays to which the skin was exposed. The elastotic material replaces atrophied collagen fibers. 2 In cats, on the contrary, sclerosis of the dermis occurs. The differences between humans and cats are great and may account for the different reactions of human and cat skin to solar exposure. The analysis of the telangiectatic areas in cats used in this study showed differences between the groups. In humans, blood vessels in areas of dermatoheliosis are frequently ectatic. 30 Neovascularization was reported 13 in SK of cats and is probably related to the repair phenomenon.
Normal human dermis is fairly acellular, 30 but, in PD, chronic inflammatory infiltrate composed of lymphocytes and histiocytes is seen in the upper dermis. 24 Overall, the cell population of the photo-aged human dermis increases. 35 In SK of dogs, the dermis often contains a dense lichenoid band of interface inflammation. 16 Normal feline skin presents only occasional perivascular lymphocytes and plasma cells, 34 but, in feline SK, mild and perivascular inflammatory infiltrate was reported. 16, 33 No differences were found in dermal cellularity between the PD groups in this study. Chronic inflammatory infiltrate could be associated with dysplastic epidermis. Areas with epidermal dysplasia, however, were excluded from further dermal quantitative analysis.
In this study, a negative correlation was observed between the degree of PD and the adnexal area present in the dermis. Indeed, there was clinical evidence of alopecia in the affected areas, more intense in the advanced PD group, as already described. 7 As a result of chronologic aging in cats, the epidermis and adnexal structures became atrophied, which was reflected in the reduction of the number of hairs. 32 Similarly, in intrinsically aged skin in humans, skin gradually experiences a reduction of the quantity of collagen fibers, as well as the number of hair follicles and epidermal thickness. 8 Continuous solar irradiation increases the atrophic process. 1
Epidermal thickness was found to increase in the affected groups, when compared with the control group (Fig. 6). In addition, the 6 tumor-bearing cats were found to have significantly thicker epidermis and more severe compaction of dermal collagen when compared with tumor-free animals. Half of the cats (nos. 18, 19, and 22) were still classified within the initial PD group. Hargis 17 observed that, in dogs and cats, acanthosis of the epidermis occurs within 72 hours after the aggression induced by UV rays. The amount of UV radiation that reaches feline skin is reduced by hair and depends on the proportion of daylight hours spent outdoors, hair color, and differences in epidermal thickness. 33 UV radiation, upon the dosage received, can function either as a complete carcinogen, a co-carcinogen agent or an immunologic modulator. UV radiation, specifically UV-B, causes thymidine dimer formation in deoxyribonucleic acid and ribonucleic acid, resulting in mutations that cause neoplastic transformation in keratinocytes. 5 In humans, the development of SCC that occurs as a result of sun exposure is characterized by a progression of clinical, histologic, and molecular changes. Sun-damaged skin may exhibit only minor abnormalities or epidermal thickening. As sun damage progresses, dysplasia, carcinoma in situ, and SCC develop. 4
In summary, the results of this study indicate a high level of skin hypersensitivity to sun rays in cats. Knowledge of the changes in PD is essential to general veterinary pathologists and dermatologists. These findings have the potential to provide additional information about the biology of PD in cats and may be useful for clinical tests in therapeutic research and in general veterinary pathology and dermatology.
Footnotes
Acknowledgements
We thank Drs. Maria Luisa C. R. Valladão and Karime C. Scarpelli from ZCC for their help in biopsy specimens collection, Adilson A. Piaza for assistance in photographic documentation, and Dr. and Mrs. Mark Ellis for their aid with English grammar usage. Dr. Konradin Metze receives a grant as researcher of CNPq. Dr. Ellen MP Almeida received a grant from CAPES.
