Abstract
Previous studies have shown that the brain is a target of persistent infection with bovine viral diarrhea virus (BVDV) and have demonstrated viral tropism for neurons as well as other endogenous cell types in diverse brain areas. Apart from foci of mild residual inflammation in some postnatal calves, consistent brain lesions, per se, have not been reported. No similar comprehensive studies of the brain have been reported in bovine fetuses. In the current study, 12 BVDV-seronegative heifers were inoculated intranasally with a 2-ml 4.4 log10 TCID50/ml dose of noncytopathic type 2 BVDV at 75 and 175 days of gestation to create persistently and transiently infected fetuses, respectively. In only persistently infected fetuses, encephaloclastic lesions resulting in pseudocysts were observed in the subependymal zone in the region of the median eminence and adjacent corona radiata as well as in the region of the external capsule associated with lenticulostriate arteries. Additionally, areas of rarefaction in white matter were observed at the tips of cerebrocortical gyri and in the external capsule. The distribution of viral antigen was examined by immunohistochemical labeling using the 15C5 anti-BVDV monoclonal antibody. Viral antigen was detected only in calves inoculated at 75 days of gestation, i.e., persistently infected. The pattern of BVDV immunolabeling revealed both similarities and differences compared with previous studies in postnatal calves, suggesting that viral infection in the brain is a dynamic and progressive rather than static process.
Keywords
Introduction
Infection with bovine virus diarrhea virus (BVDV), a pestivirus in the
The ability of ncpBVDV to cause persistent infection in calves after intrauterine fetal exposure prior to 120–125 days of gestation has significant ramifications and is a major cause of economic loss in the industry. 3, 36 Persistently infected calves are typically normal at birth, and many remain relatively normal throughout life. Others fail to thrive, are stunted, may have reduced fertility, and are immunosuppressed. Resulting from the immunosuppression, persistently infected calves can succumb to infections during the first year of life, usually due to respiratory or gastrointestinal disease. Additionally, calves persistently infected with a homologous strain of cpBVDV invariably succumb to fatal mucosal disease. Calves persistently infected with BVDV fail to mount an immune response against the virus. Because of this immunotolerance, persistently infected calves remain serologically negative and shed infective virus throughout life. These calves serve as a major source of infection for other cattle. 11 Acute, transient BVDV infection in production animals from exposure to a persistently infected calf can cause mild or subclinical illness with embryonic death, abortion, infertility, and further perpetuation of the virus and the persistently infected state. 9, 17, 21, 22 Acute postnatal infection can also have immunosuppressive effects that render the animals more susceptible to secondary viral and bacterial infections. 9, 17, 21, 22 The brain is a primary target in persistent infection. 7, 14, 23, 39 Despite the absence of consistent lesions and clinical signs of neurological disease in these calves, immunohistochemical studies have indicated that BVDV antigen is widely distributed throughout the neuraxis. No similar comprehensive studies of the brain in persistently infected bovine fetuses have been reported. 37 It is undetermined whether the brain infection in these calves reflects a static or dynamic process or if the infection would have functional consequences. There is virtually no knowledge of the near- or long-term consequences of acute, transient fetal BVDV infection during the later stages of pregnancy.
The purposes of this study were to characterize the fetal brain lesions, if any, that occur after experimental BVDV infection at 75 and 175 days of gestation and to map the distribution of viral antigen in the brains of these fetuses collected at 190 days of gestation.
Materials and Methods
Experimental infection
Eighteen yearling Hereford heifers, seronegative for the virus and BVDV free using a serum neutralization assay and ear notch ELISA (IDEXX Laboratories, Westbrook, ME), were obtained from a ranch that did not routinely vaccinate for BVDV. 35 The heifers were artificially inseminated with BVDV-negative semen, confirmed pregnant by ultrasound examination, and divided into 3 groups, each with 6 animals. For the production of persistent and acute, transient infection, heifers were inoculated intranasally with a 2-ml 4.4 log10 TCID50/ml dose of noncytopathic type 2 BVDV, Wyoming isolate 96B2222, at 75 and 175 days of gestation, respectively. 35 Uninfected heifers (negative controls) were kept isolated from the infected animals. Blood samples from heifers in the 2 infected treatment groups and uninfected controls were collected for BVDV serum neutralization assays at 60, 90, 120, and 180 days of pregnancy to confirm infection status. 35
Necropsy, virology, and histology methods
Complete necropsies were performed, and blood and a comprehensive set of tissues were collected from cesarian section–derived fetuses on day 190 of gestation. The status of persistently infected fetuses inoculated at 75 days of gestation was validated by positive BVDV isolation, immunohistochemistry, and seronegative response for BVDV type 2. The virus was confirmed as the same as the infecting inoculum by polymerase chain reaction. 35 The status of acute, transiently infected fetuses inoculated at 175 days of gestation was validated by negative virus isolation, immunohistochemistry, and seropositive response for BVDV type 2. For negative control fetuses, absence of infection was validated by negative virus isolation and immunohistochemistry and seronegative status for BVDV type 2. Details of the virology methods have been published. 35
Brains were removed from the cesarian section–derived fetuses, the right frontal lobe was excised and frozen, and the remaining brain was immersed in 10% neutral buffered formalin, adding sufficient concentrated 37% formaldehyde to float the brain in fixative. Brains were trimmed for histological examination after a minimum of 7 days of fixation. Coronal sections, representing major neuroanatomical areas (see Table 1 for approximate planes of sectioning), were embedded in paraffin, sectioned, and stained with hematoxylin and eosin.
Intensity of bovine viral diarrhea virus antigen staining in major brain regions of persistently infected fetal brains.
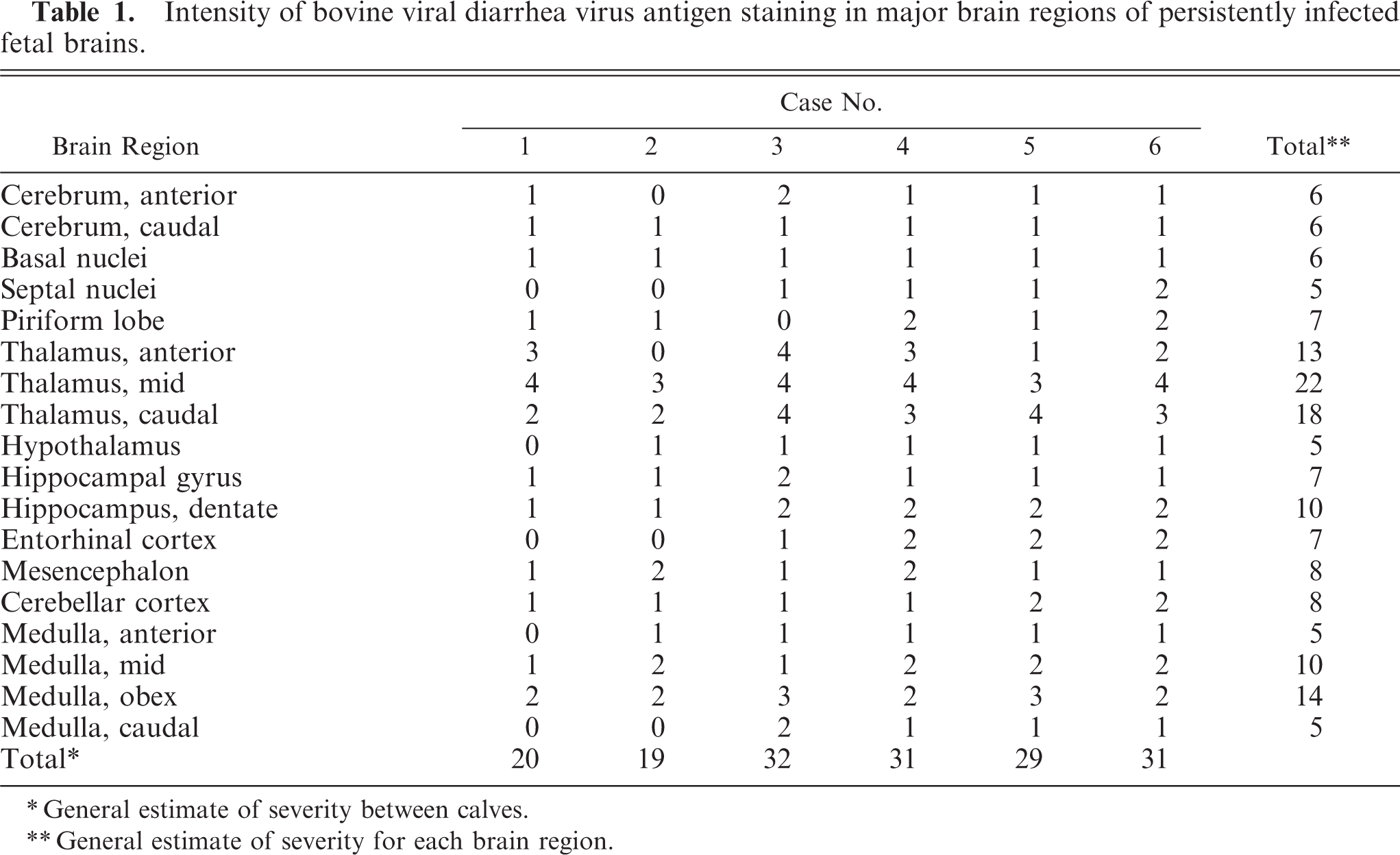
General estimate of severity between calves.
General estimate of severity for each brain region.
Immunohistochemical staining
For the detection of BVDV antigen, the primary monoclonal anti-BVDV antibody 15C5 (Syracuse Bioanalytical, Ithaca, NY) was used and labeling was conducted as per published procedure. 4, 23 Briefly, sections were deparaffinized and rehydrated. After pretreatment with proteinase K and an appropriate blocking step, sections were exposed to the primary antibody at a dilution of 1 : 1,000 using an automated immunostainer (DakoCytomation, Carpenteria, CA). Secondary detection used a commercial alkaline phosphatase system (DakoCytomation, Carpenteria, CA), fast red chromagen, and hematoxylin counterstain. Selected sections were immunolabeled for glial fibrillary acidic protein using an alkaline phosphatase detection method and fast red chromagen.
For the primary neuroanatomical reference, the Navigable Atlas of the Sheep Brain (http://www.msu.edu/brains/brains/sheep/index.html) was used. For the major neuroanatomic areas in each brain section, the distribution and intensity of neuronal BVDV antigen were graded (Tables 1, 2).
Grading criteria for intensity of neuronal staining for bovine viral diarrhea virus antigen in fetal brains infected on day 75 of gestation. 23

Results
Definitive brain lesions and BVDV antigen were demonstrated only in the persistently infected fetuses experimentally infected at 75 days of gestation but not in acute, transiently infected fetuses inoculated at 175 days of gestation or in the negative control fetuses.
Histologic lesions
The most common lesions observed in persistently infected fetuses were multiple small areas of cavitation. These pseudocysts, typically less than 1 mm in diameter, were most common in subependymal zones in the area of the germinal matrix located dorsolateral to the body of the caudate nucleus (Fig. 1). The cystic structures sometimes contained macrophages and/or faint-staining proteinaceous fluid. The cysts were devoid of an epithelial/ependymal lining; the lining was, however, composed of often compactly organized germinal cells with interspersed glial fibrillary acidic protein–positive astrocytes. Astrocytes, however, did not form an organized layer around these pseudocysts. In all persistently infected fetuses, additional changes occurred in white matter areas at the tips of cerebrocortical gyri, in areas of confluence of corpus callosum–internal capsule as well as internal-external-extreme capsules, and/or in the white matter surrounding the lenticulostriate areas. At these sites, the neuropile was rarefied and had a “lacy” appearance with a peripheral zone of hypercellularity (Figs. 2, 3). A pseudocyst was also located in the white matter dorsal and lateral to the lateral ventricle and germinal matrix of the frontal cortex in 1 fetus (Case 1; see also Table 2). Here, the wall of the cavity was hypocellular, lacking the germinal cells surrounding the pseudocysts in subependymal areas. In this same fetus, another pseudocyst containing foamy macrophages was located adjacent to the penetrating lenticulostriate arteries in the region of the external capsule (Figs. 4–6).
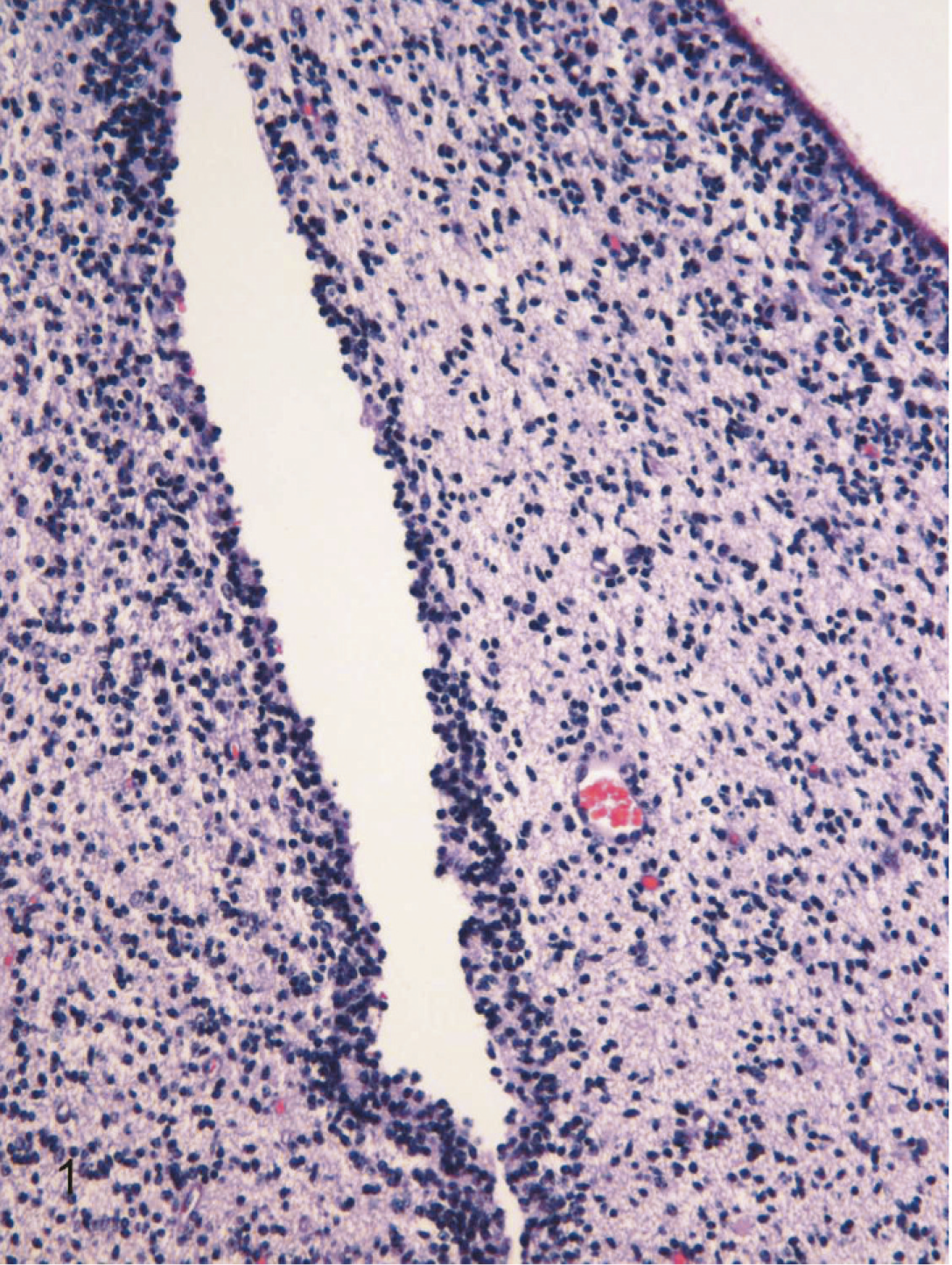
Brain, germinal eminence dorsal and lateral to the caudate nucleus, 190-day bovine fetus infected on gestational day 75 with noncytopathic BVDV. A pseudocyst, lacking an ependymal lining, is surrounded by a compact layer of germinal cells. Lateral ventricle lined by ciliated ependymal cells can be seen in the upper right. HE.
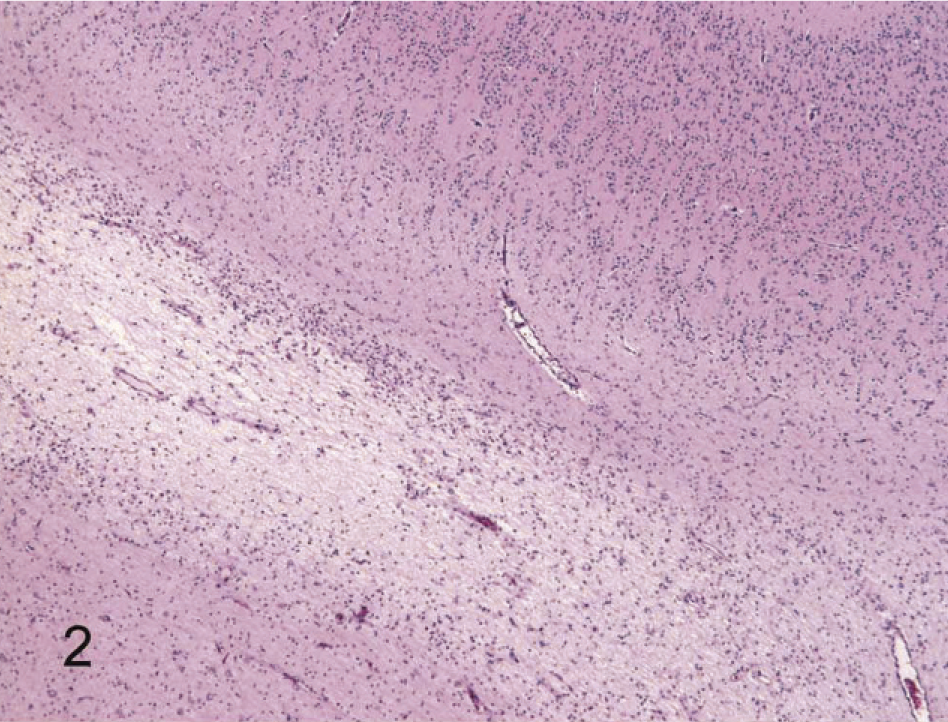
Brain, cerebral cortex near the tip of a gyrus, 190-day bovine fetus infected on gestational day 75 with noncytopathic BVDV. Rarefaction of the corona radiata imparts a pale, lacy appearance to the white matter. The area of rarefaction is bordered by a zone of hypercellularity. HE.
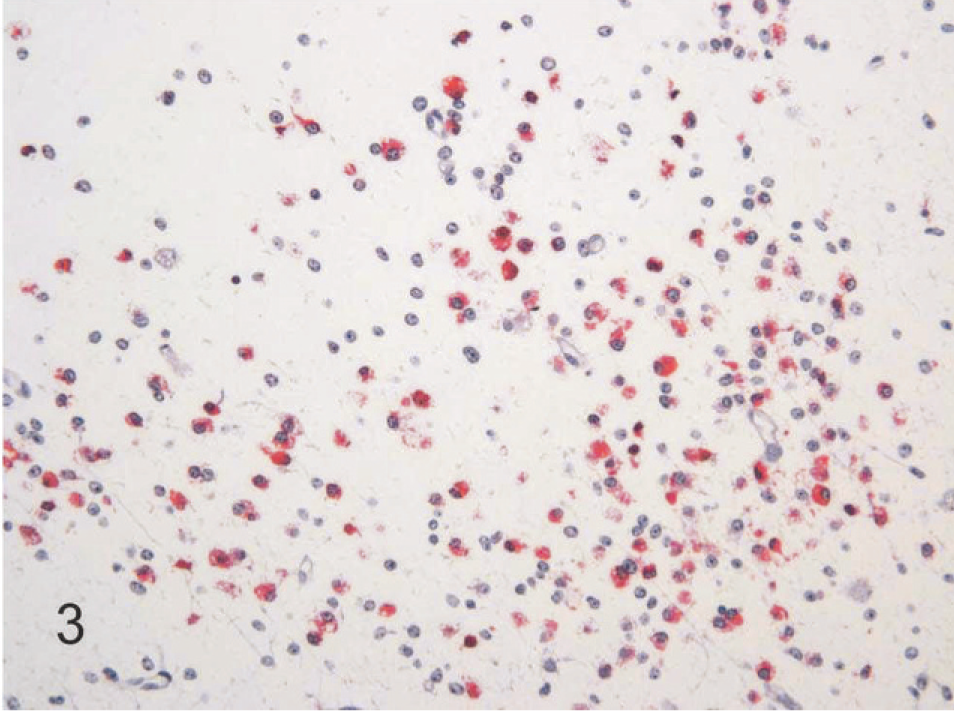
Brain, higher magnification of Fig. 2, 190-day bovine fetus infected on gestational day 75 with noncytopathic BVDV. The hypercellular zone surrounding the rarefied area contains BVDV-positive macrophages and process-bearing astrocytes. Alkaline phosphatase immunostaining method, fast red chromagen, hematoxylin counterstain.
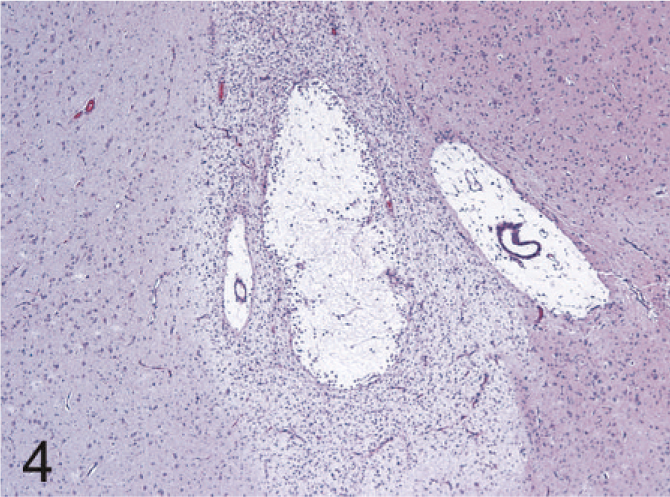
Brain, external capsule in the region of the lenticulostriate arteries, 190-day bovine fetus infected on gestational day 75 with noncytopathic BVDV. A pseudocyst with a peripheral zone of hypercellularity is present between 2 lenticulostriate arteries. HE.
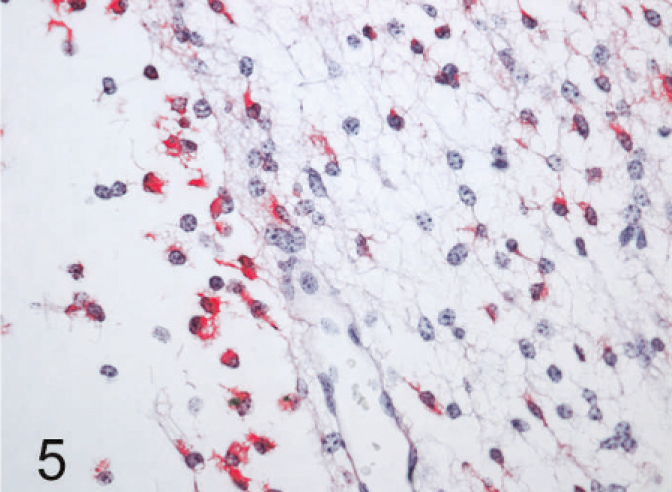
Brain, higher magnification of Fig. 4, 190-day bovine fetus infected on gestational day 75 with noncytopathic BVDV. Staining for BVDV antigen is present in macrophages in the lumen of the pseudocyst (left of photo), as well as in the surrounding hypercellular zone. Staining is also noted in process bearing cells, compatible with astrocytes, in the hypercellular zone. Alkaline phosphatase immunostaining method, fast red chromagen, hematoxylin counterstain.
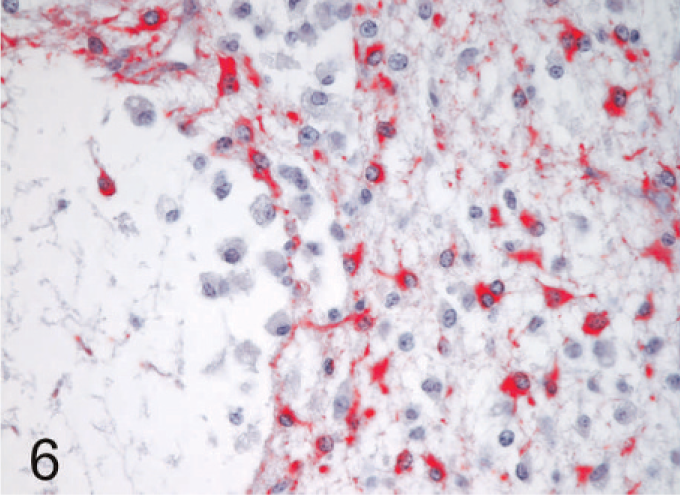
Brain, replicate section of Fig. 5, 190-day bovine fetus infected on gestational day 75 with noncytopathic BVDV. Process-bearing glial fibrillary acidic protein–positive astrocytes are prominent in the hypercellular zone surrounding the pseudocyst. Note the absence of staining in macrophages within the lumen of the pseudocyst. Astrocytes do not form an organized wall around the pseudocyst. Alkaline phosphatase immunostaining method, fast red chromagen, hematoxylin counterstain.
Septum pellucidum cavum, not a definitive lesion, per se, was identified in all persistently infected fetuses, 2 transiently infected fetuses, and 2 uninoculated control fetuses (Fig. 7). These cava lacked an ependymal lining; occasionally contained foamy macrophages; and were formed laterally by the 2 unfused leaves of the septum pellucidum, dorsally by the corpus callosum, and ventrally by the septal gray matter.
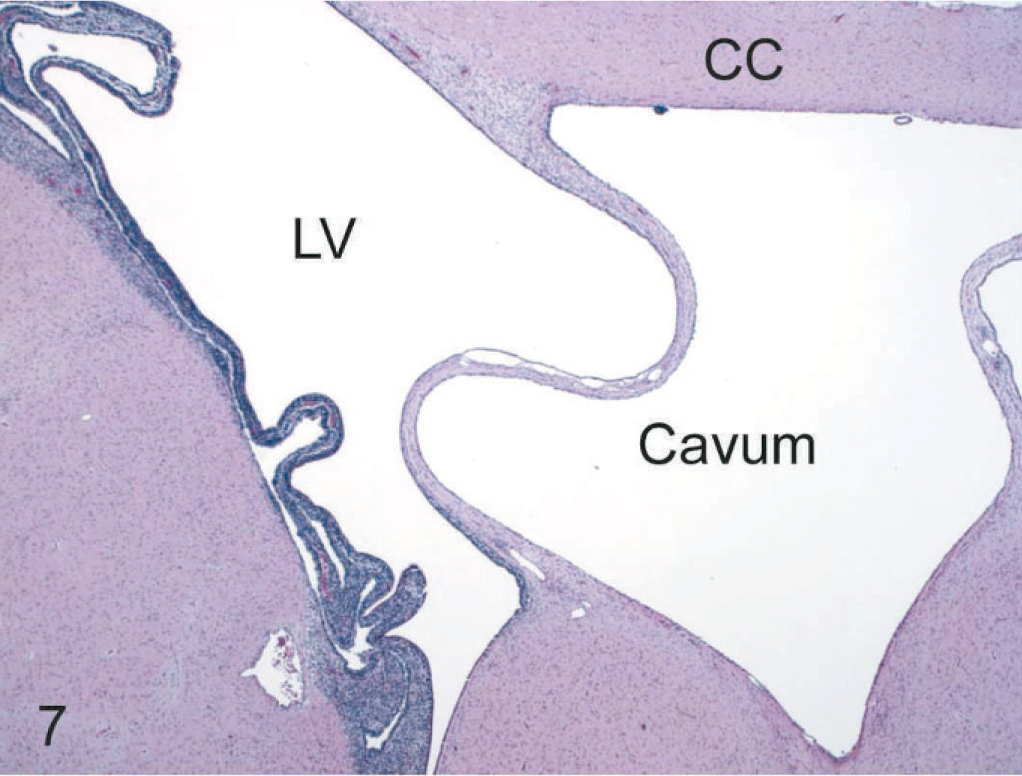
Brain, level of the caudate nucleus and interventricular septum, 190-day bovine fetus infected on gestational day 75 with noncytopathic BVDV. A septum pellucidum cavum (cavum) is bordered dorsally by the corpus callosum (CC), laterally by the unfused leaves of the interventricular septum, and ventrally by the septal nuclei. A large collapsed pseudocyst is also present in the germinal zone separating the lateral ventricle (LV) from the underlying caudate nucleus. HE.
Immunohistochemistry
Viral antigen was prominent in blood vessels throughout the neuraxis in all persistently infected fetuses. Labeling was confined to capillaries, venules, and arterioles but not in larger veins or arteries. Where a distinction could be made, viral antigen was observed in cells in the blood vessel walls interpreted as pericytes, distinct from the unlabeled endothelial cells lining the vascular lumens. Viral antigen was also observed in perivascular macrophages.
In glial cells, viral antigen was conclusively detected only in astrocytes (Figs. 3, 5). Here, the labeling often revealed the stellate, process-bearing nature of these cells. Immunolabeling for glial fibrillary acidic protein in replicate sections helped confirm the astrocytic identity of the cells (Fig 6). Astrocytes positive for BVDV antigen were most notable in areas with rarefied neuropile as indicated in hematoxylin and eosin–stained sections, i.e., in lenticulostriate areas, at the confluence of corpus callosum-internal-external-extreme capsules, and at the tips of cerebrocortical gyri. Viral antigen was not detected in other glia but was found in macrophages accumulating in areas of rarefaction and in the pseudocystic cavities (Figs. 3, 5). Viral antigen was also associated with scattered immature cells in the germinal matrix but was not concentrated around the subependymal pseudocystic lesions (vide supra) and also occurred in a more prominent, patchy laminar pattern external to the layer of germinal cells bordering the lateral ventricles.
In the leptomeninges of all persistently infected fetuses, BVDV antigen was present in round cells (probably macrophages) and also followed the pattern observed for blood vessels in the brain parenchyma.
Neuronal cytoplasmic immunolabeling for BVDV antigen was present throughout the neuraxis in all persistently infected fetal brains (Figs. 8–10). The distribution and relative intensity of BVDV antigen in neurons are summarized in Table 1. Viral antigen was detected in cell bodies as well as their processes, the latter most notable in the cerebral cortex. The most intense, uniform labeling for viral antigen was in the thalamus (Fig. 8), where it was prominent in most nuclear groups except for those in the dorsal and midline areas.
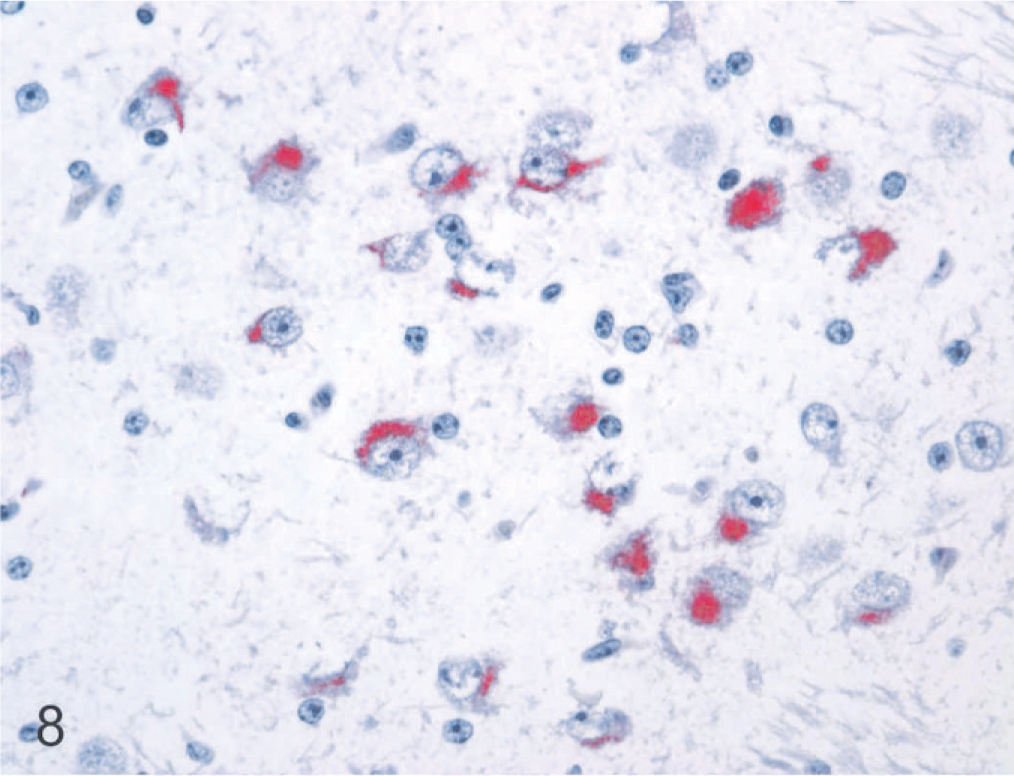
Brain, anterodorsal thalamic nucleus, 190-day bovine fetus infected on gestational day 75 with noncytopathic BVDV. Staining for BVDV antigen is prominent in the cytoplasm of neurons. Alkaline phosphatase immunostaining method, fast red chromagen, hematoxylin counterstain.
In the remainder of the brain stem, BVDV antigen in specific nuclear groups was variable and generally minimal to mild. In the hippocampal formation, viral antigen was present in all 6 persistently infected fetal brains and was slightly more intense and uniform in the dentate compared to the hippocampal gyrus (Fig. 9). In the hypothalamus, neuronal labeling was detected in 5 of the 6 persistently infected fetuses. Specific nuclei with the number of cases having detectable viral antigen in parentheses were supraoptic (1), periventricular (2), paraventricular (1), lateral hypothalamic area (2), arcuate (1), and suprachiasmatic (2). In the mesencephalon, BVDV antigen was detected in all 6 fetal brains infected at day 75. Affected nuclei included substantia nigra (3), red (2), ventral periaqueductal gray (3), oculomotor (2), raphe (3), rostral colliculus (3), caudal colliculus (2), and reticular (3). In the medulla oblongata, labeling was most consistent and intense in hypoglossal nuclei and dorsal motor nuclei of the vagus nerve, observed in all 6 persistently infected fetal brains. Other nuclei positive for viral antigen included pontine (1); dorsal cochlear (4); dorsal trapezoid (3); parabrachial (3); motor (3) and spinal (4) trigeminal; ambiguus (3); facial (5); medial (3), lateral (2), and spinal (1) vestibular; lateral reticular (2); raphe (5); reticular (5); caudal olivary (3); cuneate (1); and ventral horn motor (4) in terminal areas of the brain stem. Immunolabeling in the cerebellum was confined to Purkinje cells, was minimal to mild, and occurred in a patchy distribution in both coronal and sagittal sections in all 6 persistently infected fetuses (Fig. 10).
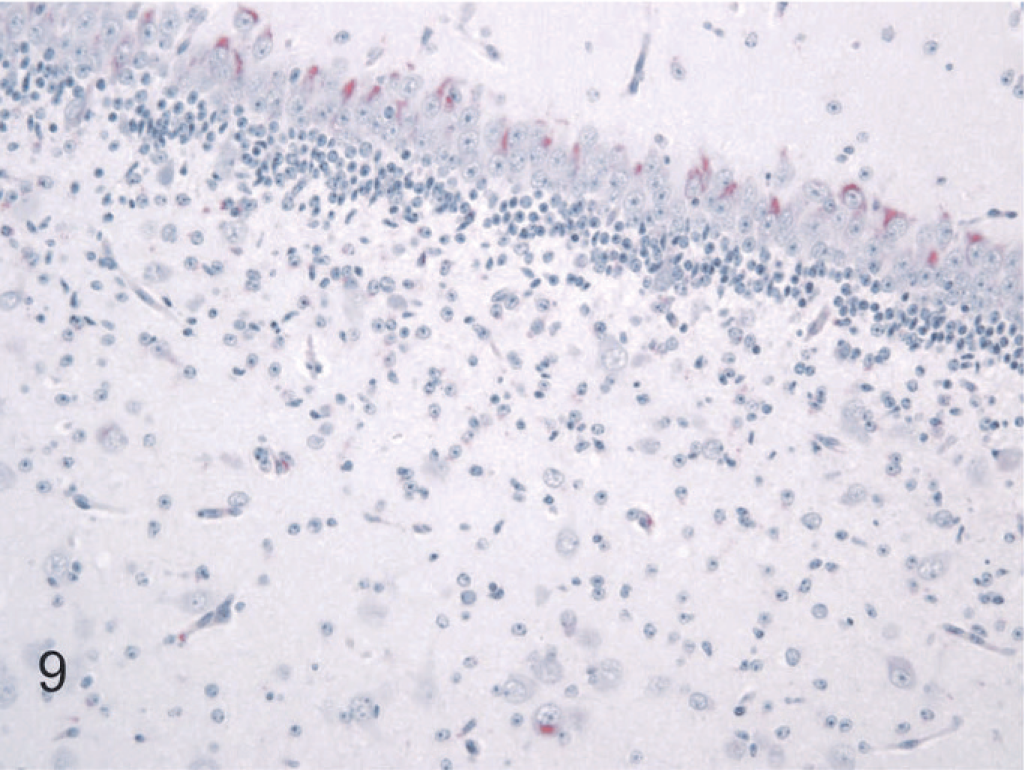
Brain, hippocampal formation, 190-day bovine fetus infected on gestational day 75 with noncytopathic BVDV. Cytoplasmic staining for BVDV antigen is present in neurons of the endfolium (lower part of photo) and in the dentate gyrus. Alkaline phosphatase immunostaining method, fast red chromagen, hematoxylin counterstain.
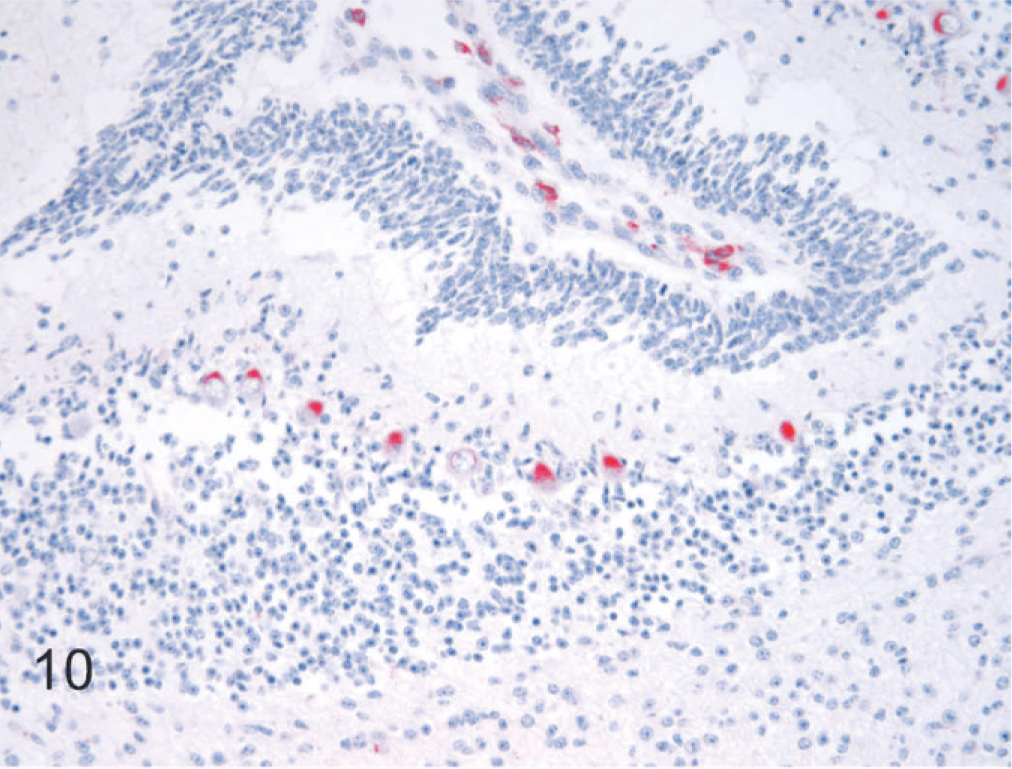
Brain, cerebellar cortex, 190-day bovine fetus infected on gestational day 75 with noncytopathic BVDV. Staining for BVDV antigen is limited to Purkinje cells and macrophages in the leptomeninges. Alkaline phosphatase immunostaining method, fast red chromagen, hematoxylin counterstain.
Discussion
This study reaffirms that widespread BVDV infection occurs in the brain after early in utero exposure leading to persistent infection. 7, 14, 23, 39 In contrast, no viral antigen or lesions were detected in fetuses exposed to the virus at 175 days of gestation.
Encephaloclastic lesions, cavitation, and formation of pseudocysts, as well as rarefaction, occurred afet infection at 75 days of gestation with BVDV in this study. There was no active cellular necrosis at the stage the persistently infected fetal brains were examined, and, except for prominent viral antigen in astrocytes surrounding these lesions and in infiltrating macrophages, there was little to directly associate these changes with the viral infection. These lesions undoubtedly occurred after infection at 75 days but prior to 190 days of gestation and can be attributed to a direct effect of the virus. 15, 16, 39 These findings are somewhat surprising because similar lesions have not been consistently recorded in postnatal studies of the brain in persistently infected calves. If these lesions persist into postnatal life, it would seem to be an uncommon event, and it is possible that these lesions might resolve during later periods of brain development and maturation 29 (see also the following discussion of septum pellucidum cavum). Alternatively, this might be a lesion specific for infection in highly vulnerable areas of the brain at or around 75 days of gestation and/or of this particular strain of virus.
Septum pellucidum cavum is a normal finding during fetal brain development. 13, 18, 25, 27 As the brain matures, these cava gradually close; in humans, this occurs at approximately 39 weeks of gestation. 27 Closure is initially passive, a loose apposition of the 2 leaves forming the lateral walls of the cava mediated by pressure and increasing volume from expansion of the lateral ventricles as the brain develops and matures. Permanent fusion of the 2 leaves may not occur until some time after birth. 25 In humans, persistence of the septum pellucidum cavum can occur in approximately 0.73% of adults, but reported incidence varies from 0.14 to 20%. 13, 32 Septum pellucidum cavum was observed in all 6 persistently infected and in 2 acute, transiently infected and 2 noninfected control fetuses, respectively, suggesting that closure was delayed in the persistently infected fetal brains. There was no association between the location of viral antigen and delayed closure of the cava. It seems possible or even likely, however, that delayed closure is related to encephaloclastic events occurring in periventricular brain areas during the earlier fetal stages of viral infection. 2, 18 Persistence of the septum pellucidum cavum, however, was not reported in 3 more comprehensive previous studies in postnatal calves. 7, 14, 39 This may be due to an interpretation that this finding is normal in a percentage of otherwise normal calves analogous to the situation in humans.
Similar to previous studies in persistently infected postnatal calves, immunolabeling for BVDV antigen indicated infection of neuronal populations in widespread areas of the brain. 7, 14, 23, 39 Involvement of nuclei throughout most areas of the thalamus was pronounced and sharply contrasted with sections taken at the level of the basal nuclei, mesencephalon, and medulla oblongata. This pattern of relatively intense labeling in the thalamus is analogous to a recently reported study in postnatal persistently infected calves 23 but differs from earlier comprehensive reports. 7, 14, 39 However, one notable difference is the presence of BVDV antigen in Purkinje cells in these fetal cerebella, which was previously observed in 2 calves at 1 day of age 39 but not later in the postnatal period. 23 Other differences include a more widespread but generally less intense pattern of immunolabeling in fetal brain stem nuclei caudal to the thalamus and less intense labeling in other brain regions, including the basal nuclei, hippocampus, hypothalamus, and entorhinal cortex, compared with postnatal calves. 23 The similarities between fetal and postnatal brains would suggest that there are sites of predilection, such as the thalamus and hippocampus. Differences, illustrated by infection of Purkinje cells in persistently infected fetal but not postnatal brains, suggest that the pattern of infection changes during brain maturation. Infection of the brain seems to represent a dynamic rather than static process.
Encephaloclastic events and viral persistence during critical periods of neural development and maturation could have long-term effects on neural function. In a previous study, it was suggested that persistent infection of the brain could provide a common mechanism for disturbances in growth, reproduction, and immune function that are protean manifestations of BVDV-induced clinical disease. 23 Studies have indicated that even minor perinatal perturbations can have potentially serious effects on the multidirectional communications that occur between the brain, endocrine, and immune organs. 19, 20, 33, 34, 38 This study was not, however, designed to specifically assess altered brain function after viral infection. Even if the encephaloclastic lesions, often considered benign, 29 were to be absent in BVDV infection, viral infection may alter normal gene expression and transcriptional pathways, leading to subtle but significant changes in neuronal function in the absence of overt lesions. This may be especially true of viruses capable of causing persistent infections. 1, 5, 6, 8, 10, 12, 24, 26, 28, 30, 31, 40 Additionally, although BVDV infection at 175 days of gestation resulted in only transient infection with no appreciable lesions and no detectable viral antigen, a subtle but significant effect on brain function related to this transient viral infection during critical periods of brain development could not be ruled out. 1, 26, 31
Footnotes
Acknowledgements
The project was supported by the National Research Initiative (NRI) of the USDA Cooperative State Research, Education, and Extension Service (CSREES), grant number 2004-35204-17005 (TH, AVO) and a faculty grant-in-aid, UWGIA49315, from the University of Wyoming (DLM). The authors acknowledge the expert technical support of histologists Paula Jaeger and Mercedes Thelen, Department of Veterinary Sciences, University of Wyoming. The authors also thank Ms. Kathy Austin and Dr. Hyungchul Han, Department of Animal Science, University of Wyoming, for assistance in managing heifers, collecting blood samples, and assisting with necropsies.
