Abstract
Placentitis, premature birth, and perinatal death were associated with Encephalitozoon cuniculi infection in an alpaca. Histologically, chorionic trophoblasts contained many Gram-positive, period acid-Schiff positive, variably acid-fast spores. Multifocal necrosis and infiltration by lymphocytes, eosinophils, and neutrophils were scattered throughout the chorionic membrane. Spores in trophoblasts were approximately 1 μm X 2 μm, thick-walled, and contained polar filaments and polar vacuoles consistent with microsporidia. The presence of E. cuniculi DNA was confirmed by sequencing the polymerase chain reaction amplicon from frozen placental tissue. A few glial nodules were scattered throughout the cerebrum, and mild lymphocytic inflammation was present in the heart, liver, and lung. No organisms were detected in tissues other than the placenta. This is the first reported case of E. cuniculi infection in an alpaca.
Encephalitozoon cuniculi is an obligate intracellular parasite of the phylum Microspora, which has been associated with spontaneous infections in numerous species. 17, 20, 21 In most species, E. cuniculi infections are subclinical; however, clinical disease with nonsuppurative encephalitis, nephritis, vasculitis, pneumonia, hepatitis, and placentitis can occur in neonatal, immunosuppressed, and, less frequently, immunocompetent animals. 21 To the authors' knowledge, this is the first description of placentitis, premature birth, and perinatal death associated with E. cuniculi in an alpaca.
A reportedly 290-day-gestation female alpaca cria and placenta were submitted to the Animal Disease Diagnostic Laboratory at Purdue University for necropsy on January 13, 2007. Reportedly, the cria was dead when first seen the previous day. Its 3-year-old primiparous dam and other camelids on the farm were clinically normal.
The cria weighed 3.8 kg and was 65.5 cm long from crown to rump. Based on the gestation length (normal = 335 to 360 days), 10 the weight (normal = approximately 8 to 9 kg), 10 and unerupted teeth, the cria was considered premature. The cria was in good body condition with moderate autolysis. The subcutis of the head was edematous and mottled light to dark red. The pleural and peritoneal cavities each contained approximately 200 ml of red, watery fluid. The lungs were mottled pink to dark red and oozed blood on cut section. Sections of lung floated in formalin. The chorionic surface of the placenta, especially near the body, was mottled dark red to pink and covered with thick, 0.2-cm- to 0.4-cm-diameter, tan–gray, viscous, loosely adherent plaques (Fig. 1). Near the umbilical cord, the allantois was covered with similar exudate. No gross lesions were observed in other tissues. Samples of placenta, lung, heart, brain, sciatic nerve, skeletal muscle, spleen, thymus, lymph node, liver, stomach compartments, small intestine, colon, kidney, and adrenal gland were fixed in 10% neutral buffered formalin, routinely processed, paraffin embedded, and stained with hematoxylin and eosin for light microscopic examination.
Many chorionic trophoblasts were distended with 1 μm × 2 μm, lightly basophilic, refractile spores within parasitophorous vacuoles (Fig. 2). Necrotic foci were scattered throughout the edematous chorioallantois, which was infiltrated by scattered eosinophils, neutrophils, and lymphocytes. The chorionic surface was segmentally covered by amorphous eosinophilic material, keratin, cellular debris, erythrocytes, neutrophils, and many sloughed trophoblasts filled with spores. The cerebral cortex had scattered glial nodules with a few shrunken, hypereosinophilic neurons with pyknotic nuclei. In the lung, a few alveolar spaces contained sloughed epithelial cells, and there were scattered pleural hemorrhages. Few lymphocytes, plasma cells, and neutrophils were scattered throughout pulmonary interalveolar septa, hepatic portal tracts, the endocardium, and the myocardium. Pyknotic and karyorrhectic lymphocytes were scattered throughout splenic periarteriolar lymphoid sheaths.
Sections of placenta, cerebrum, and kidney were stained with Brown and Brenn modified Gram's stain; placental sections were also stained with Ziehl-Neelson acid fast and periodic acid–Schiff (PAS) stains. In the placenta, spores were strongly Gram-positive, PAS-positive, and variably acid fast–positive, consistent with microsporidia and distinct from protozoa, such as Toxoplasma gondii or Neospora caninum. No organisms were identified in other tissues.
One millimeter cubes of formalin-fixed placenta were fixed in 3% glutaraldehyde, postfixed in 1% osmium tetrachloride, and embedded in Epon. Seventy- to ninety-nanometer-thick sections were stained with lead citrate and uranyl acetate, and examined with a Phillips 201 electron microscope. The spores in trophoblasts had single central nuclei, coiled polar tubes, thick walls, and posterior vacuoles, consistent with microsporidia (Fig. 3).
No bacteria or viruses were isolated from fetal tissues. Bovine viral diarrhea viral antigens were not detected by fluorescent antibody testing, and anti-Neospora antibodies were not detected by ELISA.
DNA from the frozen, unfixed placenta was extracted using a commercial kit (DNeasy Tissue Kit, Qiagen, Valencia, CA) according to the manufacturer's suggested procedure. Approximately 1 g of tissue was homogenized in 5 ml of deoxyribonuclease (DNase)-free water using a Stomacher, and a 160-μl aliquot of the homogenate was used for DNA extraction. Extracted DNA was eluted in 200 μl of kit AE buffer. A polymerase chain reaction (PCR) with previously described primers 14 was performed on the extracted DNA to amplify the intergenic spacer region of genes encoding small-subunit and large-subunit rRNA from several microsporidian species. DNA extracted from cell culture–grown E. cuniculi spores (kindly provided by Dr. R. Livingston, Research Animal Diagnostic Laboratory, University of Missouri, Columbia, MO) was used as the positive control, and buffer was used as a negative control. The PCR was performed in a total volume of 25 μl using 5 μl of the extracted DNA as template and primers MSP-1 (5′-TGAATG(G/T)GTCCCTGT-3′), MSP-2A (5′-TCACTCGCC GCTACT-3′), and MSP-2B (5′-GTTCATTCGCAC TACT-3′). DNA amplification was performed in a thermocycler (Thermo Hybaid, Milford, MA) using the following protocol parameters: initial denaturing at 95°C for 5 minutes followed by 35 cycles of 94°C for 2 minutes, 55°C for 2 minutes, 72°C for 3 minutes, and a final extension at 72°C for 5 minutes. Electrophoretic analysis of 12 μl of the amplified products in 1.0% agarose gel revealed amplification of a ∼400 bp DNA fragment from the placenta and the positive control; no amplified products were detected in the negative control (Fig. 4). The amplified DNA fragment from the placenta was cloned into pCR2.1 vector (Invitrogen, Carlsbad, CA), and the nucleotide sequence of the cloned DNA fragment was determined at Purdue University Low Throughput Genome Sequencing facility. A basic local alignment search tool search of databases at the National Center for Biotechnology Information (http://www.ncbi.nlm.nih.gov) revealed that the amplified product was 98 to 100% identical to the corresponding sequences of E. cuniculi. The presence of 4 5′-GTTT-3′ repeats in the amplified product indicated that E. cuniculi in the placenta belonged to genotype III. 7 Extracted DNA tested negative for Toxoplasma gondii and Neospora caninum genomic sequences by real-time and nested PCR assays, respectively. 8, 9
Infectious causes of camelid abortions have recently been reviewed 19 ; however, few individual cases have been reported. Bovine viral diarrhea virus, Neospora caninum, and Arcanobacterium pyogenes have been directly implicated in alpaca abortions. 3, 12, 18, 19 Additionally, maternal eosinophilic myositis, attributed to Sarcocystis aucheniae infection, was associated with abortion with no fetal lesions. 15
Microsporidia are single cell, obligate intracellular, spore-forming parasites that are classified as fungi based on the presence of chitin in spore walls and phylogenetic analyses. 6 Microsporidia are characterized by the presence of a polar filament, or polar tube, which is important for intracellular infection. 6, 21
Microsporidia are important emerging human pathogens, mainly infecting immunocompromised patients. 5, 21 Enterocytozoon bieneusi and Encephalitozoon intestinalis are most commonly associated with human disease 5, 21 ; however, E. cuniculi has also been diagnosed in human immunodeficiency virus/acquired immunodeficiency disease patients. 4 Encephalitozoon sp. have similar morphologies, and PCR-based methods are necessary for species determinations. 5, 21, 22
E. cuniculi infections have been reported in rabbits, rats, mice, dogs, foxes, cats, squirrel monkeys, and horses. 17, 20, 21 Microsporidia have direct life cycles and are transmitted mainly by ingestion and inhalation; however, traumatic and transplacental transmission also have been reported. 6, 21 Transplacental E. cuniculi transmission has been proposed for several species, including rabbits, carnivores, guinea pigs, horses, and squirrel monkeys. 1, 2, 13, 16, 17, 20, 23
E. cuniculi–associated placentitis has been reported in squirrel monkeys, 23 horses 17 (Armed Forces Institute of Pathology [AFIP] Wednesday Slide Conference [WSC] 2004 #23 [AFIP #2890687] and AFIP WSC 1988 #13 [AFIP #2133004]), and blue foxes. 16 In blue foxes 16 and 1 equine infection (AFIP WSC 1988 #13 [AFIP #2133004]), the placenta contained organisms with few other histologic changes, whereas 2 other infected equine placentas 17 (AFIP WSC 2004 #23 [AFIP #2890687]) had multifocal necrosis with moderate nonsuppurative inflammation. Lesions were reportedly more severe in squirrel monkeys with granulomatous placentitis and vasculitis. 23 In this alpaca, placental lesions were moderate, with scattered necrotic foci and moderate infiltration by lymphocytes, neutrophils, and eosinophils. Despite the difference in severity of lesions, these reports suggest that E. cuniculi can infect the placenta of a variety of mammals and result in fetal loss.
Infected animals commonly shed E. cuniculi in urine, and the chitinous wall probably provides spores with the ability to persist in the environment. 6 Recently, anti–E. cuniculi antibodies were detected in 14.1% of Brazilian horses, suggesting exposure to E. cuniculi. 11 Because of its wide host spectrum and ability to survive in the environment, E. cuniculi should be considered as a potential cause of sporadic abortion and perinatal death in multiple species, including camelids and horses. However, experimental studies fulfilling Koch's postulates are needed to confirm E. cuniculi's role in abortion and perinatal death.
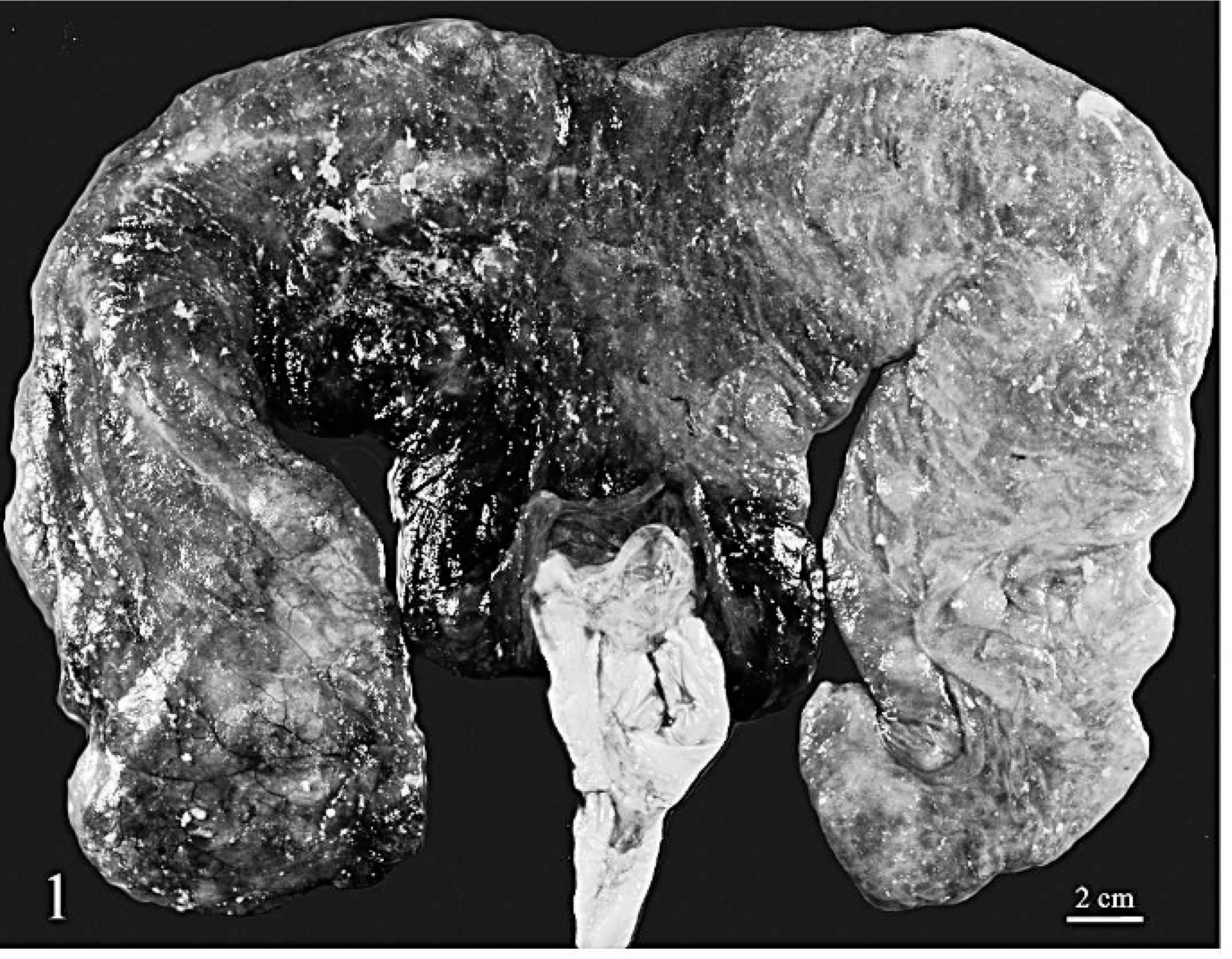
Placenta; alpaca. The chorion is mottled dark red to pink and covered with thick, loosely adherent plaques. Bar = 2 cm.
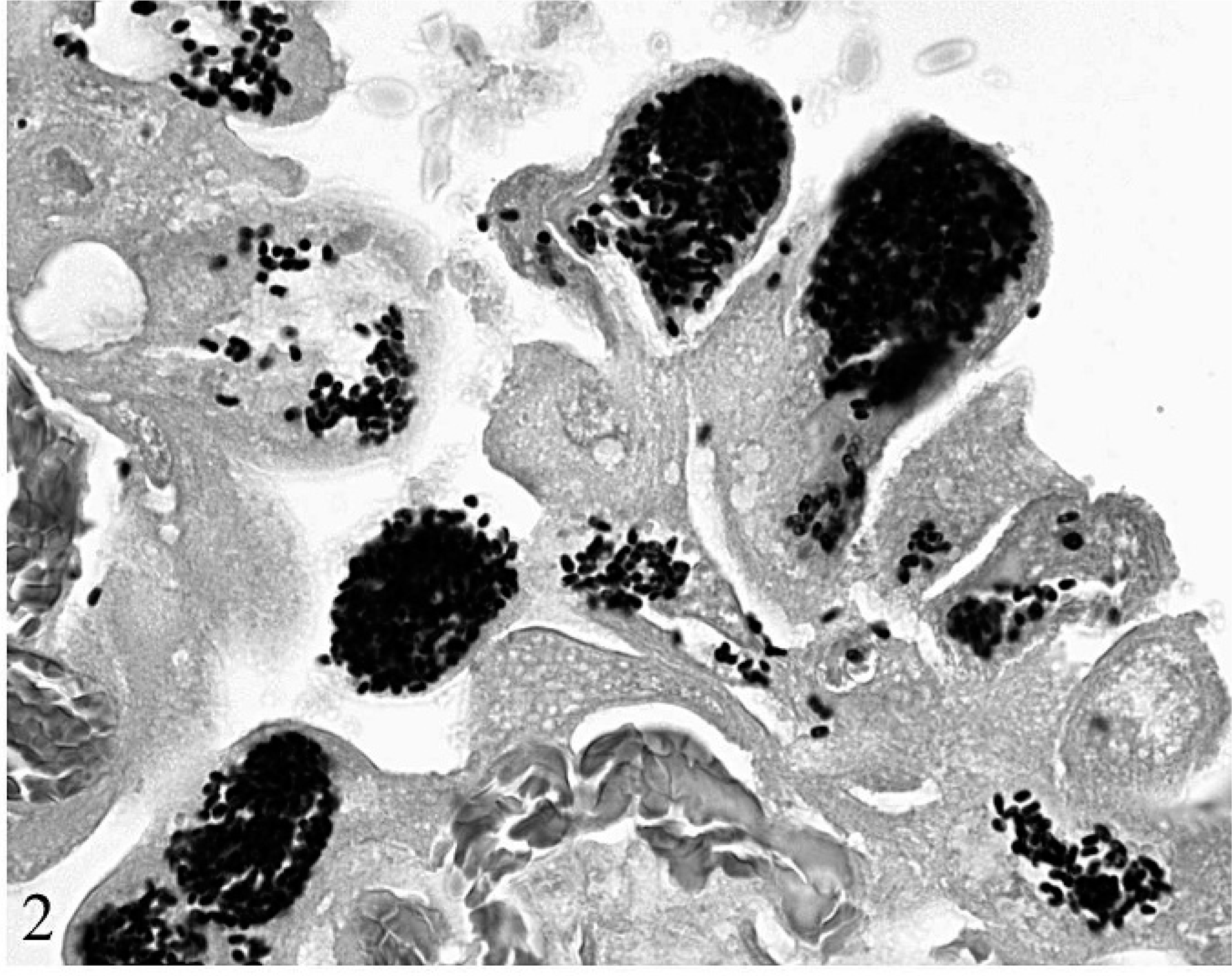
Placenta; alpaca. Chorionic trophoblasts contain many 1 μm × 2 μm Gram-positive spores consistent with E. cuniculi. Brown and Brenn modified Gram's stain.
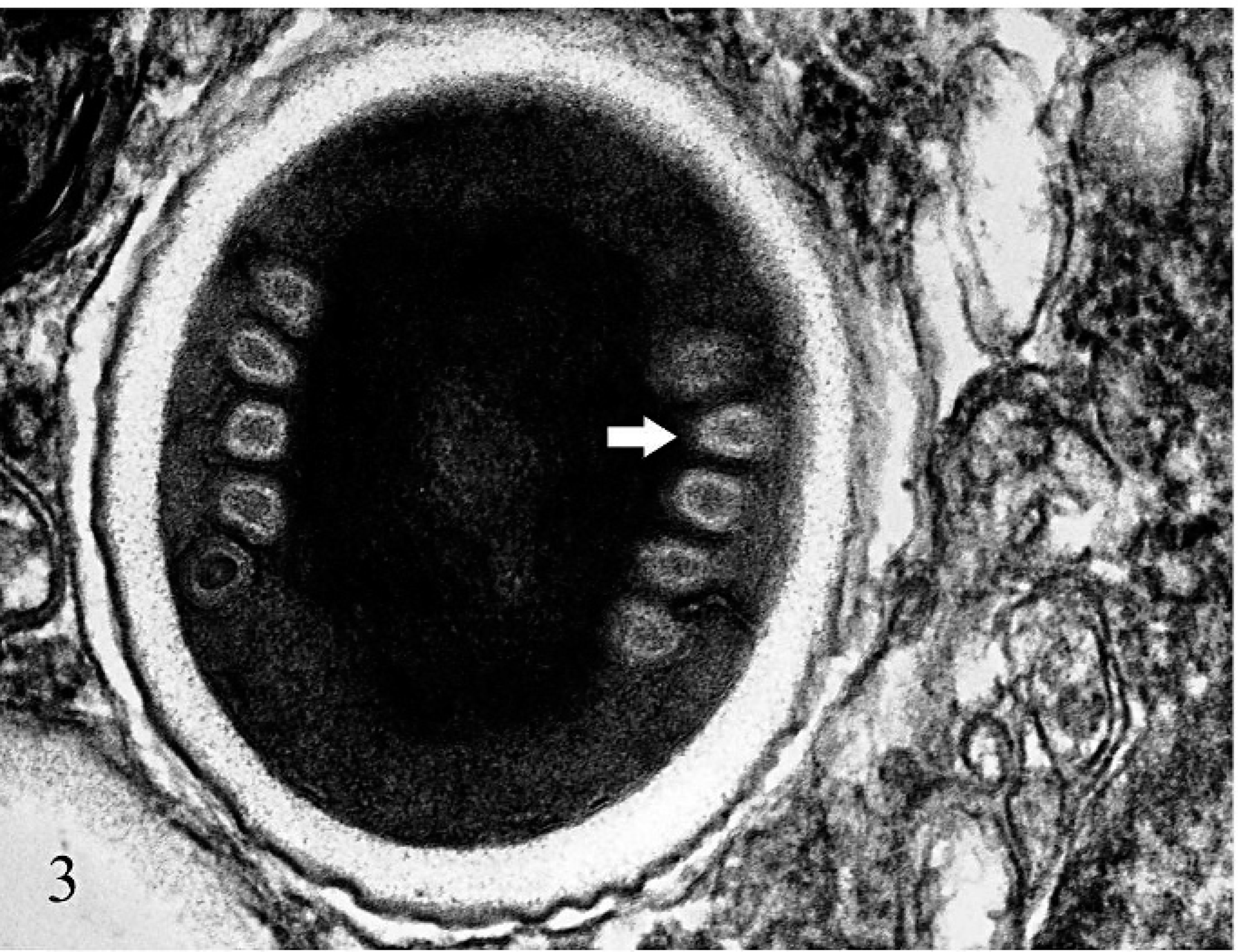
Placenta; alpaca. A trophoblast contains a 1 μm × 2 μm microsporidian spore with a coiled polar filament (arrow). Lead citrate and uranyl acetate.
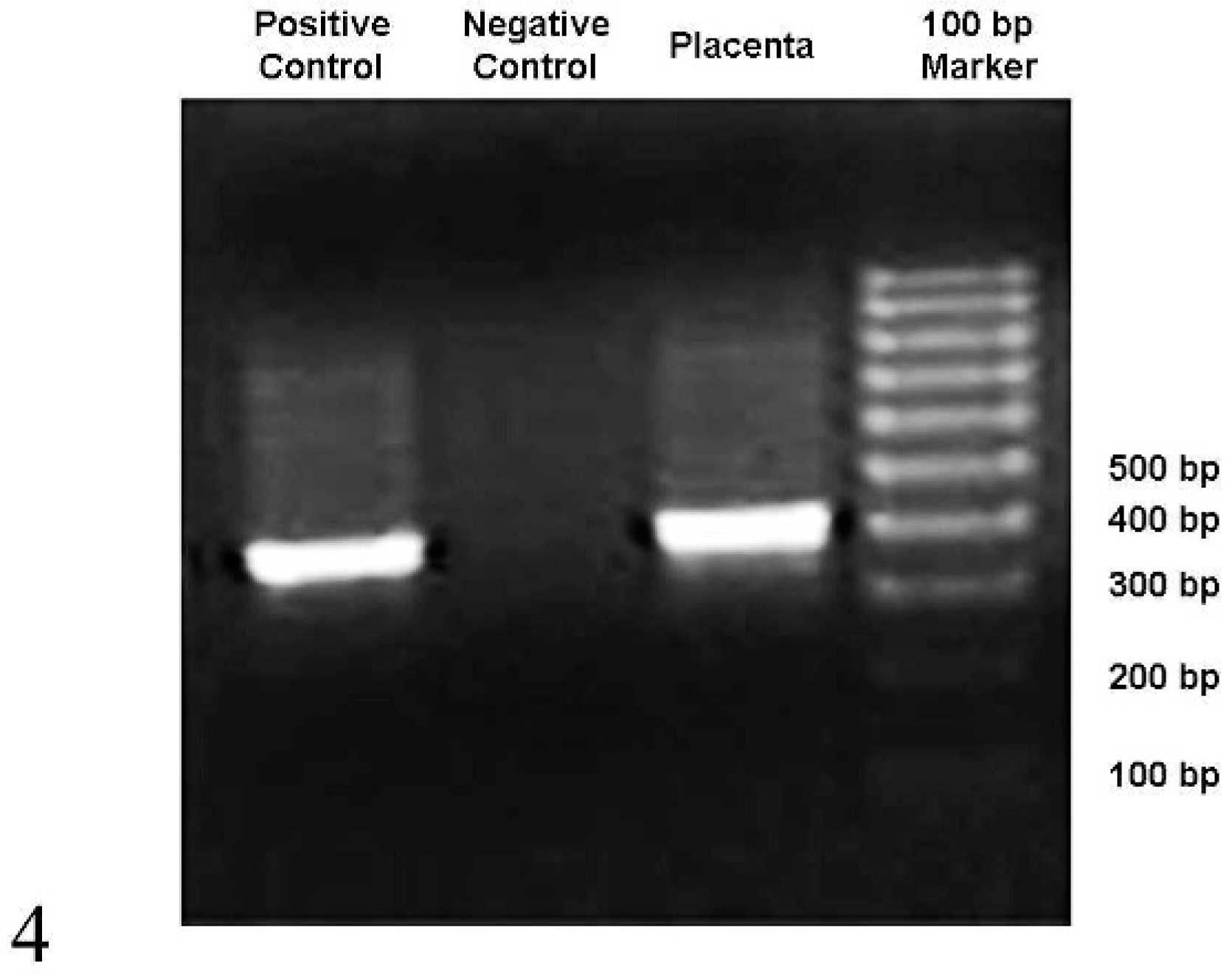
Detection of E. cuniculi DNA in the alpaca on a 1% agarose gel stained with ethidium bromide after PCR amplification. Numbers at the right indicate the 100-bp DNA ladder fragment sized in base pairs.
Footnotes
Acknowledgements
We thank Phyllis Lockard for assistance with electron microscopy, and the Histopathology and Molecular Diagnostic Laboratories at Purdue University Animal Disease Diagnostic Laboratory for histochemical stains, and PCR and sequencing, respectively.
