Abstract
Three Swainson's Blue Mountain Rainbow Lorikeets (Trichoglossus haematodus moluccanus), ranging from 6 to 8 months of age, presented with lethargy, emaciation, and progressive neurologic signs. The first one died 24 hours after the onset of clinical signs, and the other two were euthanized 10 to 14 days after the onset of progressive neurologic disease. Clinical signs in these lorikeets included head pressing, hemiparesis, seizures, obtunded mentation, weakness, and lethargy. Two of the lorikeets had hepatomegaly, and one had splenomegaly on gross examination. Histopathology revealed disseminated microgranulomas in the liver, spleen, and brain, and lymphohistocytic perivascular encephalitis and cephalic vasculitis. Electron microscopic examination of macrophages in brain lesions revealed spherical to rod-shaped prokaryotic organisms with a trilaminar cell wall. Molecular analysis revealed a novel species of Coxiella. This is believed to be the first report of a Coxiella sp. causing disease in a lorikeet.
Lorikeets and lories are brilliantly colored nectivorous psittacine birds native to Australia, New Guinea, Indonesia, and surrounding islands. Most species have stable populations in the wild. The Swainson's Blue Mountain Rainbow Lorikeet (Trichoglossus haematodus moluccanus) is native to southeastern Australia and adjacent islands. 8 Because of their bright plumage, these birds are common exhibit species in zoologic facilities.
Coxiella burnetii, the causative agent of Q fever in humans and abortion in domestic ruminants, is an obligate, intracellular, Gram-negative bacterium that is able to replicate in large numbers within the phagolysosome of eukaryotic phagocytes. 18 Due to its unique survival strategies, it is resistant to drying and is able to survive in the environment for a long period of time. 1, 7 Based on recent 16S rRNA sequence analysis, the Coxiella genus has been reclassified to the gamma subdivision of Proteobacteria. The genera Legionella, Francisella, and Rickettsiella are now its closest relatives. 1, 3, 9, 11, 14, 22 This organism is highly infectious and has zoonotic potential. 1, 7, 12 In humans, C. burnetii is primarily acquired via aerosolized particles from infected placenta, bodily fluids, and contaminated dust. However, it also can be transmitted via arthropod vectors, such as ticks and lice. 1, 7, 19, 29 This modality allows C. burnetii to persist in the wild and provide a year-round reservoir for infection. Ticks are considered to be the most important vector and reservoir agent for this mode of transmission. Coxiella sp. can be carried by more than 40 species of ticks. Ticks can transmit the organism vertically within species, but only horizontally to other species, such as mammals, birds, and reptiles. 3, 7, 28 Genera known to be carriers include Ixodes, Rhipicephalus, Amblyomma, Dermacentor, and Haemaphysalis. 1, 7, 15, 17, 18 Due to their survival within the macrophage, Coxiella sp. have the ability to induce persistent infections in a wide variety of hosts, including humans, ruminants, other mammals, birds, and reptiles, but aside from C. burnetii, disease associated with these agents has not been documented. 1, 3, 7, 14, 17– 19
In the last decade, new species of Coxiella and closely related proteobacteria have been discovered in the wild. 15, 17, 19 All documented new species have only been discovered within arthropod vectors. Some, such as Rickettsiella grylli, have been found to be intracellular pathogens within grasshoppers. 3 Others have been found to be carried by ticks collected from different species of birds. 21, 22 This report describes fatal disease associated with Coxiella sp. infection in Swainson's Blue Mountain lorikeets (Trichoglossus haematodus moluccanus), and is believed to be the first report of fatal coxiellosis in birds.
Materials and Methods
Cases
All 3 lorikeets in this report were captive-raised at the Oklahoma City Zoo. Two of these birds were related (Nos. 1 and 3), and all three were hand-raised from 4 weeks of age to weaning as a group. All lorikeets were housed with other species of lorikeets in an indoor/outdoor zoo aviary.
Bird No. 1 was a 6-month-old male that presented for acute onset of lethargy late June 2006. Other lorikeets within the exhibit appeared clinically healthy at this time. The only abnormality found in physical examination was thin body condition. Despite supportive care with subcutaneous fluids, the bird was found dead the following morning.
Bird No. 2 was an 8-month-old that presented for acute onset of lethargy and weakness in August 2006. On physical examination, the bird was in thin body condition and had generalized weakness. It was treated supportively with subcutaneous fluids, broad-spectrum antibiotics (amoxicillin trihydrate/clavulanate potassium; 125 mg/kg orally twice a day), and antifungal medication (Terbinafine suspension; 15 mg/kg orally once a day). Blood was collected for a complete blood count (CBC) and biochemistry panel. CBC showed a normocytic, normochromic anemia (packed cell volume, 35%; reference range, 45–55%). Biochemistry panel was unremarkable. Whole-body radiographs showed no abnormalities. Despite treatment, progressive neurologic signs and weight loss were seen. On the seventh day following presentation, multiple seizurelike activities and head pressing behaviors were observed. Due to lack of improvement and suspected encephalitis, a dose of 2 mg/kg dexamethosone was given intramuscularly on the ninth day. Euthanasia was elected on the 10th day.
Bird No. 3 was an 8-month-old that presented for acute onset of lethargy and inappetance 3 days after No. 2. This bird was a clutch mate of No. 1. On physical examination, the bird was in thin body condition, and the feathers were ruffled. It was treated supportively with subcutaneous fluids and showed clinical improvement over the next 24 hours. Eight days after initial presentation, this lorikeet presented with progressive ataxia, hyperesthesia, intention tremors, hemiparesis, and weight loss. Supportive care, including subcutaneous fluids and gavage feedings, were administered. Toltrazuril (10 mg/kg orally once a day) was started for possible protozoal encephalitis. Central nervous system signs continued to worsen, and the bird was euthanized 14 days after initial presentation.
Necropsy
All lorikeets were necropsied by a team of veterinary externs, staff technicians, and veterinarians using a routine avian necropsy protocol developed by the Animal Health Department at the Oklahoma City Zoo. Tissues collected at necropsy from each bird and fixed in 10% neutral-buffered formalin included skin, skeletal muscle, lung, heart, liver, spleen, kidney, pancreas, small and large intestine, ventriculus, proventriculus, crop, and esophagus. Brain was collected from Nos. 2 and 3.
Histopathology
Select tissues from each bird were fixed in 10% neutral-buffered formalin for 3–5 days, processed routinely, sectioned at 5 μm, mounted on frosted glass slides, and stained with hematoxylin and eosin (HE). Some sections also were stained with periodic acid–Schiff base (PAS), Fite acid fast, Gomori methylamine silver (GMS), Giemsa, Warthin Starry, and Gimenez techniques.
Immunohistochemistry
Blocks containing brain lesions from Nos. 2 and 3 and blocks containing spleen and liver from No. 1 were screened for Chlamydophila psittacii and C. burnetii antigen. The immunohistochemical procedure for C. burnetii was modified from an original report by Dilbeck et al. 4 using a Dako Autostainer Plus (Dako Corp., Carpinteria, CA). Deparaffinized 4-μm sections were quenched with 3% hydrogen peroxide in methanol, brought to water through graded alcohols, rinsed in water for 3–5 minutes, and then rinsed in a buffered rinse solution consisting of 1% BRIJ in Tris-buffered saline for 3–5 minutes. This buffered rinse for 3–5 minutes was used for all remaining rinse steps up to application of counterstain. The sections were blocked using 1% casein in rinse buffer, the primary human antiserum (1 : 500 dilution; Integrated Diagnostics Inc., Baltimore, MD) was applied for 60 minutes, rinsed, and a 1 : 200 dilution of biotinylated goat anti-human IgG (heavy and light chain; Vector Laboratories, Burlingame, CA) was applied for 30 minutes, followed by a buffer rinse and application of the avidin-biotin complex reagents (Vectastain Elite ABC kit; Vector Laboratories), a buffer rinse, and the addition of AEC (Dako AEC Substrate-Chromagen system; Dako). The sections were then rinsed in water for 3–5 minutes, counterstained with Mayer hematoxylin for 15–90 seconds, rinsed several times for 1–2 minutes in water, aqueous mounting media was applied, and the slides then coverslipped. The negative control consisted of a paired serial section using known negative human antisera to replace the primary human antisera (Integrated Diagnostics). The positive control was a section of goat placenta infected with C. burnetii confirmed previously via compatible gross and histopathology, the presence of Gimenez-positive pleomorphic intratrophoblastic organisms on impression smears coupled with a negative fluorescent antibody reaction for Chlamydophila on impression smears, and insignificant bacterial growth as previously reported. 18
For C. psittacii, formalin-fixed, paraffin-embedded tissue was sectioned at 4 μm and mounted on charged slides, quenched with 3% hydrogen peroxide in 95% reagent alcohol, and hydrated to water. Sections were treated with 0.4% pepsin at 37°C for 15 minutes, rinsed, and blocked with 0.5% casein in TBS-Tween for 10 minutes at room temperature. All further incubations were at room temperature, and all rinses were with TBS-Tween. An anti-chlamydia antibody (1681; ViroStat, Portland, ME), diluted 1:50 in 1% casein in TBS-Tween, was applied for 30 minutes and detected with a horseradish-conjugated anti-mouse polymer (K4001; DakoCytomation, Carpinteria, CA), followed by AEC (3464; Dako) for 10 minutes. Sections were counterstained with Mayer hematoxylin and coverslipped with an aqueous mounting media. Nonspecific staining was evaluated with a duplicate slide receiving normal mouse whole IgG in place of the chlamydia-specific primary antibody.
Electron microscopy
Formalin-fixed brain from No. 3 and formalin-fixed liver from No. 1 were transferred to half-strength modified Karnovsky before two washes in 0.2 M sodium cacodylate and postfixed in 2% osmium tetroxide reduced with 2.5% potassium ferrocyanide. 11, 29 Following fixation, tissue was washed in 0.2 M sodium cacodylate, dehydrated through a graded ethanol series before infiltration and embedment in Eponate 12s epoxy resin (Ted Pella Inc., Redding, CA). Thick sections were cut, mounted on glass slides, and stained with Toluidine blue O and examined by light microscopy. Thin sections were cut, mounted on 150-mesh copper grids, stained with 6% uranyl acetate in 75% ethanol, poststained in Reynold lead citrate, and examined in a Zeiss 906E transmission electron microscope (Zeiss Electron Microscopy, Thornwood, NY) at 60 kV accelerating voltage. 24
DNA extraction for PCR
DNA isolation from frozen brain tissue from bird No. 3 was performed, followed by polymerase chain reaction (PCR) amplification. DNA was extracted from the sample using a commercial kit (Qiamp DNA Minikit; Qiagen, Valencia, CA) according to the manufacturer's instructions. For fixed tissue blocks from bird Nos. 1 and 2, five 10-μm sections were cut from paraffin blocks containing brain and tissue pools that had been fixed in formalin for 24 hours. To avoid cross-contamination, the microtome was cleaned after each block was cut with Clear-Rite 3 (Richard-Allan Scientific, Kalamazoo, MI), followed by Eliminase (Decon Labs Inc., King of Prussia, PA) and 70% alcohol. A new blade was used for each block, gloves were changed after each block, and cut sections were placed into sterile microcentrifuge tubes with the aid of sterile, disposable, plastic forceps. DNA was extracted from these cut sections using a commercial kit (Qiamp DNA Minikit) according to the manufacturer's instructions, with the following variations: a sterile, disposable, plastic needle was used to scrape melted paraffin from the sides of the tube and mix it with the ATL lysis buffer after a few minutes at 60°C in the first step. After overnight incubation at 60°C with lysis buffer and 40 ml of 20 mg/ml proteinase K, the upper melted paraffin layer was removed as much as possible using a pipette tip. After the Buffer AL and ethanol steps, the tube was placed in a freezer at −20°C for 30 minutes. This caused the remaining wax to adhere to the sides of the tube so that the mixture could be pipetted into the Qiagen spin column without wax.
Standard rickettsia PCR assays, which failed to amplify DNA from the sample, included those targeting “gene D,” a 3065-nucleotide open reading frame from Rickettsia species, citrate synthase, and dnaJ. 13, 27, 31 A PCR amplicon was obtained from the fresh brain tissue (bird No. 3) when PCR was performed using universal rickettsia primers 5′-CGAAAGCCTGATCCAGCAAT-3′ and 5′-TAAGGGCCATGATGACTTGA-3′ to amplify an 812–base pair portion of the 16S ribosomal RNA gene, as previously described. 26 These primers did not yield an amplicon from the fixed tissues, so a specific PCR for the lorikeet agent was designed, based on the sequence from sample No. 3, using the Primer 3 program online. 28 Primers Loricox 835F (5′-GTAGTCCACGCTGTCAACGA-3′) and Coxspp1029R (5′-CTGTCACTCGGTTCCCAAAG-3′) were used to amplify a 194–base pair portion of the 16S ribosomal RNA gene. The PCR reactions with a total volume of 50 μl consisted of 10 μl Invitrogen buffer A (5× buffer at 300 mM Tris-HCl, 7.5 mM MgCl2, 75 mM ammonium sulfate, pH 8.5), 2 μl of 10 mM dNTPs, 1.5 μl each of 40-μM stocks of primers, 30 μl sterile DEPC water, 0.5 μl Taq (Fermentas), and 4.5 μl sample DNA. PCR conditions entailed 4 minutes of denaturing at 95°C, followed by 36 amplification cycles consisting of 30 seconds of denaturing at 94°C, 45 seconds of annealing at 59°C, and 1 minute of extension at 72°C, with a final extension cycle of 7 minutes at 72°C. DNA from bird No. 3 was used as a positive control. Extracted sterile DEPC water was used as a negative control, and water was used as a no-template control for all PCR reactions.
PCR amplicons were visualized on 1.5% agarose gels containing ethidium bromide, excised using a sterile scalpel blade under ultraviolet illumination, and purified using a Freeze ‘n’ Squeeze kit (BioRad, Hercules, CA) according to the manufacturer's directions. The amplicon DNA was sequenced directly on both strands by Amplicon Express (Pullman, WA) using the Perkin-Elmer Applied Biosystems Prism Dye Terminator Kit, with analysis on an ABI 373 DNA Sequencer. Forward and reverse sequences were aligned using a Clustal W program online. 37 GenBank search for similarities was then accomplished using the NCBI BLAST program online. 20
Results
Necropsy findings
Bird No. 1 was emaciated and had mild hepatomegaly. Bird No. 2 had multiple serosal ecchymoses throughout the proximal proventriculus. Bird No. 3 had mild splenomegaly and hepatomegaly. No other gross abnormalities were noted in these birds.
Histopathologic findings
Histologic examination (Figs. 1, 2) revealed disseminated moderate to severe microgranulomatosis in liver and spleen (3), perivascular lymphohistiocytic encephalitis with microgranulomas (3), and mild lymphohistiocytic enteritis (2). A consistent finding in the birds was the presence in the brain of transmural and perivascular histiocytic inflammation and the formation of small microgranulomas adjacent to affected vessels. The histiocytic cells frequently had displaced nuclei and smudged eosinophilic cytoplasm (Fig. 2, inset). Iron sequestration was occasionally seen in the splenic and hepatic microgranulomas. The Gimenez stain identified single-to-multiple, approximately 1- to 4-μm intracytoplasmic magenta inclusions that sometimes fused to form larger granules within macrophages of the brain in Nos. 2 and 3 (Fig. 3) and liver and spleen of No. 1. Inclusions also had a positive PAS reaction, although with this stain the inclusions were considerably larger, from 3–10 μm in diameter (Fig. 4). Fite acid fast, Warthin Starry, Brown and Brenn, and Giemsa stains did not identify organisms in the lesions.
Electron microscopy and immunohistochemistry
Electron microscopic examination of brain lesions from No. 3 and hepatic microgranulomas from No. 1 revealed spherical to rod-shaped prokaryotic organisms within lysozomes. These organisms were approximately 400 nm in diameter, and rod-shaped forms were up to 900 nm in length. The organisms had a trilaminar cell wall similar to that of a Gram-negative bacterium (Fig. 5). Immunohistochemical stains specific for C. burnetii and C. psittacii antigen were negative in brain lesions of Nos. 2 and 3, and in liver and spleen of No. 1.
PCR detection and sequencing
The sequence from bird No. 3 most closely matched that of an unnamed species of Coxiella that was a symbiont of Rhipicephalus sanguineus ticks (GenBank accession no. D84559.1, 97.3% sequence identity). The next closest matches were to C. burnetii strain VR145 and nine-mile strain (96.4% sequence identity with accession numbers AY342037.1 and Y11502.1). Fixed brain and tissue pool samples from bird Nos. 1 and 2 did not yield amplicons of sufficient quality for direct sequencing, so the agent-specific PCR was used for these samples. This PCR yielded amplicons of the expected 194–base pair size. These amplicons were then directly sequenced in both directions, and their sequences exactly matched that of the Coxiella sp. from No. 3 (Fig. 6).
Discussion
Based on histologic and molecular findings in all birds and electron microscopic findings in the brain of bird No. 3 and the liver of bird No. 1, a Coxiella-like organism was determined to be the etiologic agent of the systemic inflammatory disease in these lorikeets. Lesions in all tissues were moderate to severe and account for the clinical presentations. Histologic examination and application of Gimenez and PAS stains were useful for achieving a preliminary diagnosis of Coxiella infection, especially after the investigators were familiar with the agent and associated lesions in the brain. The pattern and cellular constituents of the brain lesion are especially supportive, particularly the scattered histiocytic cells and formation of microgranulomas adjacent to foci of perivascular cuffing or vasculitis. Smudged intracytoplasmic inclusions with associated nuclear displacement were commonly visualized in the histiocytic cells, especially in the brain. Interestingly, these inclusions resemble those in placental trophoblasts of ruminants infected with C. burnetii. 18 Cytoplasmic inclusions were detectable using Gimenez and PAS stains in areas corresponding to smudged cytoplasm of histiocytes. The Gimenez stain was mostly likely staining the cell membrane of the organism. It is believed that the PAS technique stained the organism's cell wall in addition to the membrane of the phagolysozome or adjacent rough endoplasmic reticulum, which may account for the slightly larger size of the inclusions detected by this method. The inclusions of C. psittacii also stain magenta with Gimenez, but in our experience are much smaller, have sharper outlines, and are not especially prevalent in brain tissue. The morphologic and tinctorial features of this agent in histologic section, in conjunction with electron microscopic findings and negative staining for Chlamydophila antigen by immunohistochemistry, indicate that this agent is not C. psittacii.
Immunohistochemical staining for C. burnetii antigen was based on a published protocol, 4 and C. burneiti–infected goat placenta was used as a positive control. 18 The antibody for this procedure is known to cross-react with gut bacteria, plant material, some fungi, and lipofuscin. 4 It appears from this study that the agent in these lorikeets does not cross-react with C. burnetii using this technique. This is a conservative interpretation, because of prolonged tissue fixation in formalin (up to 5 days) and it is unknown whether the C. burnetti antibody will cross-react with antigen in avian tissue. Because Coxiella spp. have not previously been diagnosed in birds by immunohistochemistry, a positive avian control was not available.
The newly discovered species of Coxiella appears to adopt survival strategies similar to those used by C. burnetii, the causative agent of Q fever. 1, 3, 10, 12, 16 For example, C. burnetii can be acquired via inhalation of aerosolized particles from infected placenta, bodily fluids, or contaminated dust, or through an arthropod vector, such as ticks and lice. 1, 3, 9, 13, 17, 21– 23, 35, 36 These different strategies have allowed C. burnetii to persist in the wild and provide a year-round reservoir for infection. Ticks are considered to be the most important vector and reservoir agent for this mode of transmission of C. burnetii. This appears to be the case as well with the newly discovered Coxiella species. It has been found that Coxiella species are carried by more than 40 species of ticks. Genera known to be carriers include Ixodes, Rhipicephalus, Amblyomma, Dermacentor, and Haemaphysalis. 1, 9, 17, 21, 22 Ticks have the added benefit of transmitting the organism vertically as well as horizontally to other species, such as mammals, birds and reptiles. 3, 9, 35 No ticks were found in the exhibits or on the three lorikeets of this report, and the mode of transmission has not yet been discovered.
Birds have been recognized as important wildlife reservoirs for Coxiella, and they have been implicated in the transmission of this organism to humans. 6, 7, 17, 19, 25, 32, 35, 36, 38 They also have been incriminated as a point source of Legionella pneumophila, the causative agent of Legionnaire's disease in humans, and will seroconvert following infection. 17 Nonetheless, clinical disease attributed to Coxiella species or other similar organisms is rare in birds and has been described briefly in one report. 5
Clinical signs in humans with the acute form of Coxiella infection can range from flulike symptoms to those associated with hepatitis, pneumonia, endocarditis and, on rare occasions, central nervous system dysfunction due to meningoencephalitis. 1, 30 Treatment with doxycycline, fluoroquinolones, rifapin, or newer macrolides appear to resolve the acute phase of the infection. 1, 9, 12 The lorikeets in this report had clinical and pathologic signs similar to infected humans. The etiologic diagnosis of Coxiella infection was not made until after all birds were dead, so specific antibiotics for this disease were not administered. Although antibiotics effective for treatment of coxiellosis in humans were not used in these lorikeets, supportive care that included anitibiotic therapy was not successful for 2 of the birds. Further studies would be required to evaluate the effectiveness of antimicrobial therapy for Coxiella in birds.
Molecular analysis revealed that the organism seen in these 3 birds is closely related to C. burnetii. All of the recently discovered Coxiella spp. have not been reported to have caused disease in their respective hosts. To the authors' knowledge, this appears to be the first report of disease caused by Coxiella sp. in lorikeets. Additional sequencing of longer segments of the 16S rRNA gene from multiple infected lorikeets is ongoing, to more closely define the phylogeny of this organism.
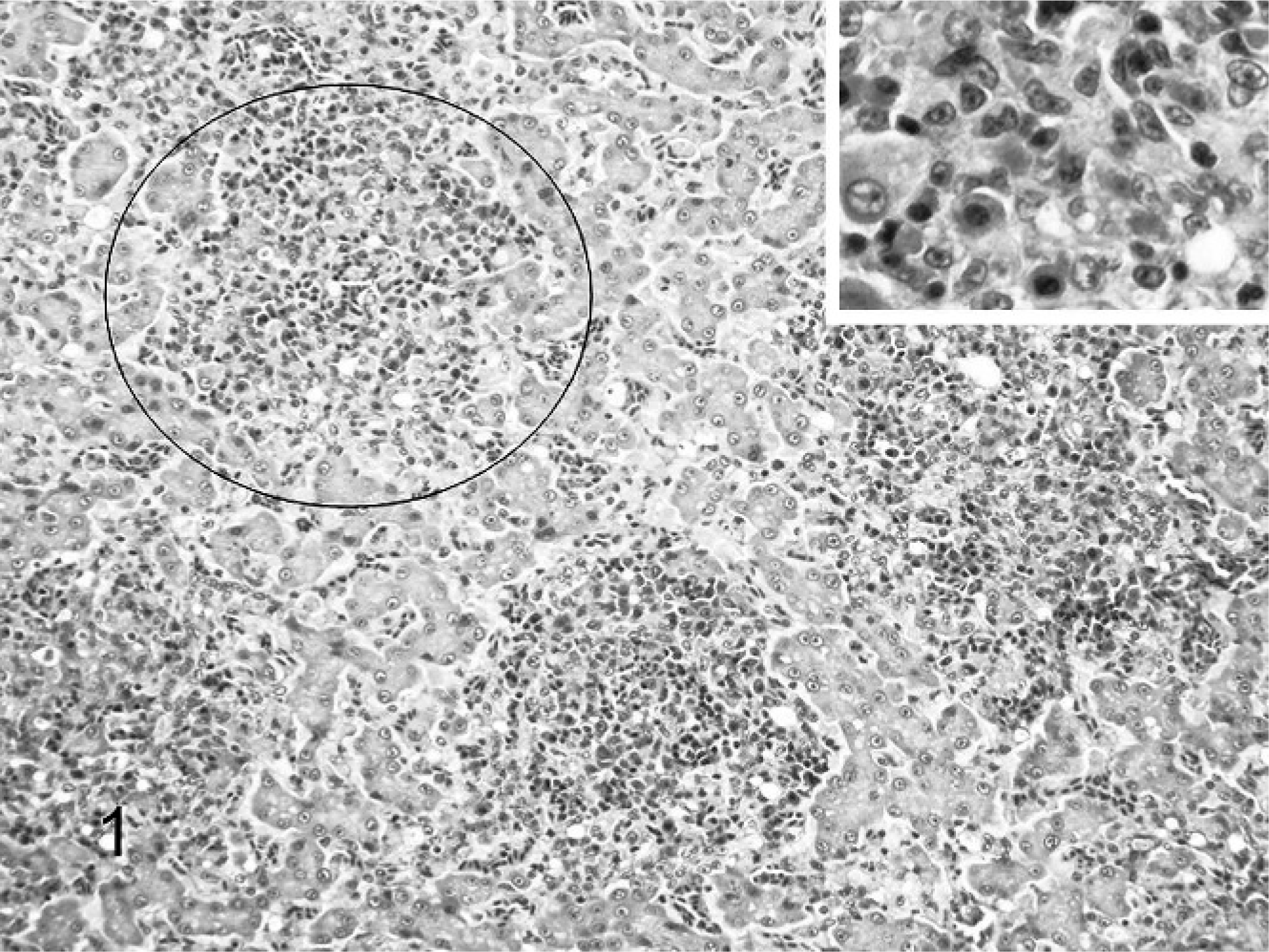
Liver, lorikeet. Note random circumscribed foci (circled) of inflammation distributed throughout the hepatic parenchyma. HE. Inset: Higher magnification of hepatic inflammatory cell foci. Note that inflammation is predominantly histiocytes with fewer lymphocytes and plasma cells. HE.
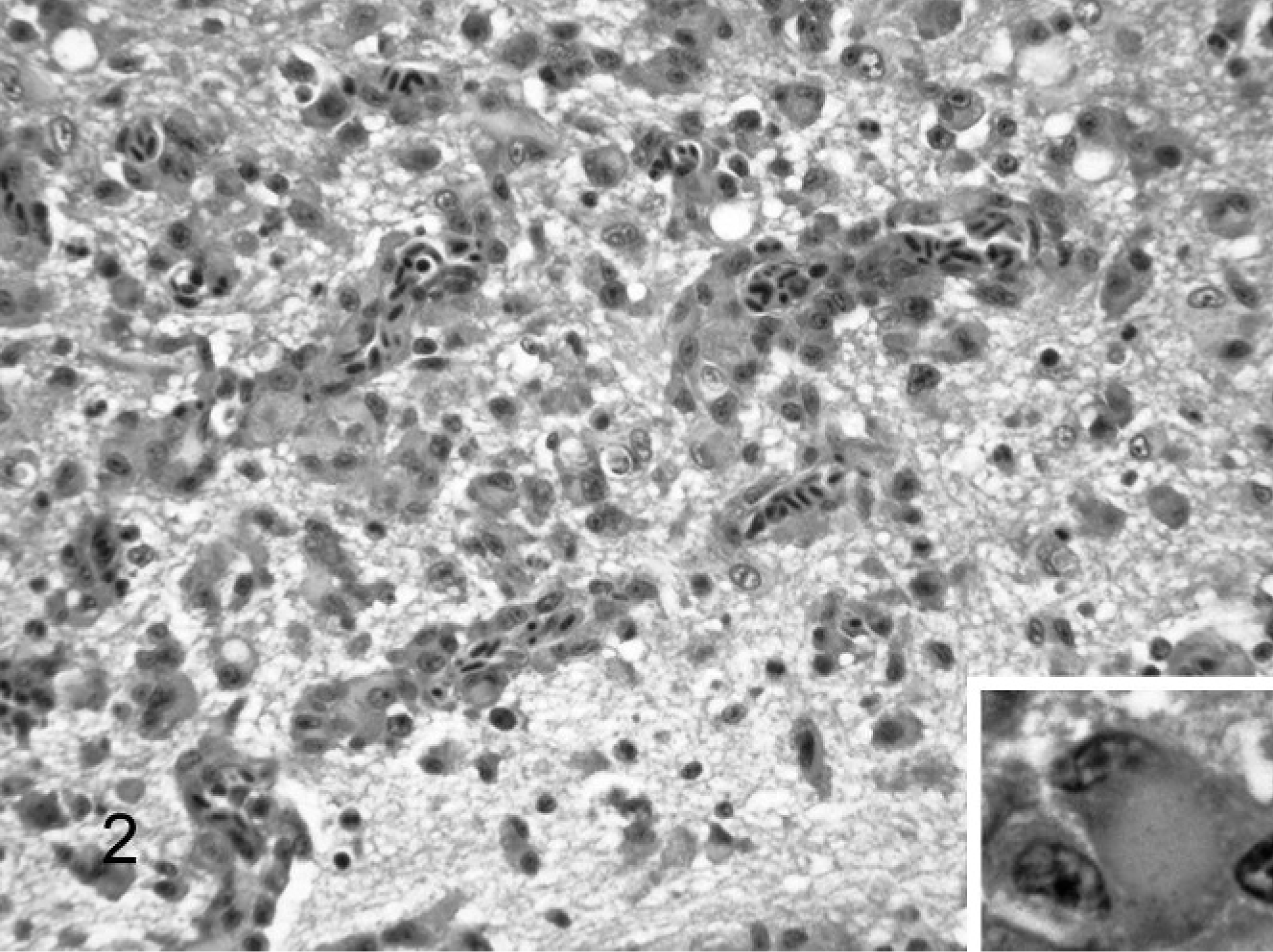
Brain, lorikeet. Note infiltrates of histiocytic cells within and around blood vessels and extending into the adjacent neuropil. HE. Inset: Higher magnification of histiocytic cell showing smudged cytoplasm and displaced nucleus. HE.
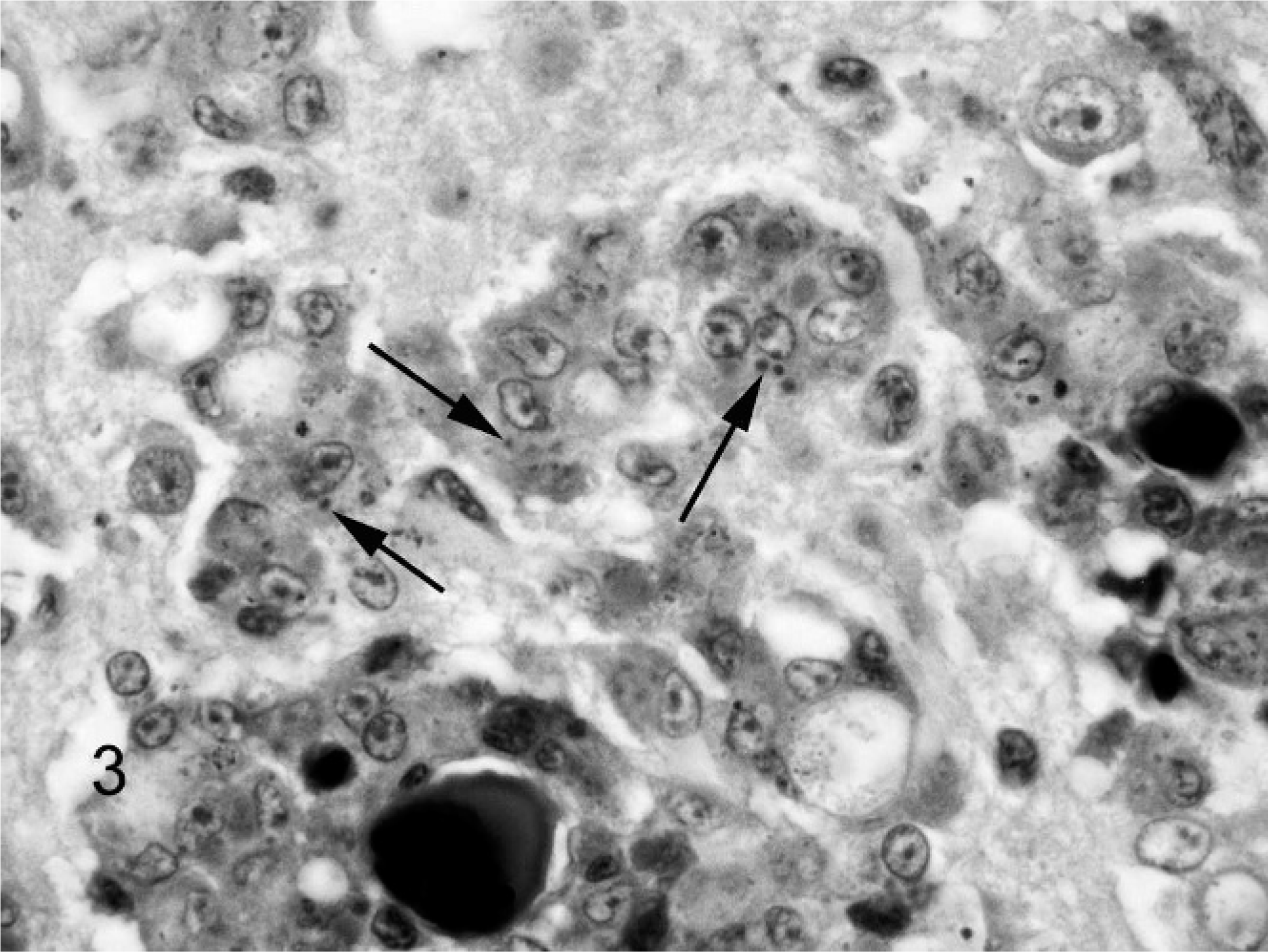
Brain, lorikeet. Note variably sized cytoplasmic inclusions (arrows) within histiocytic cells. Gimenez.
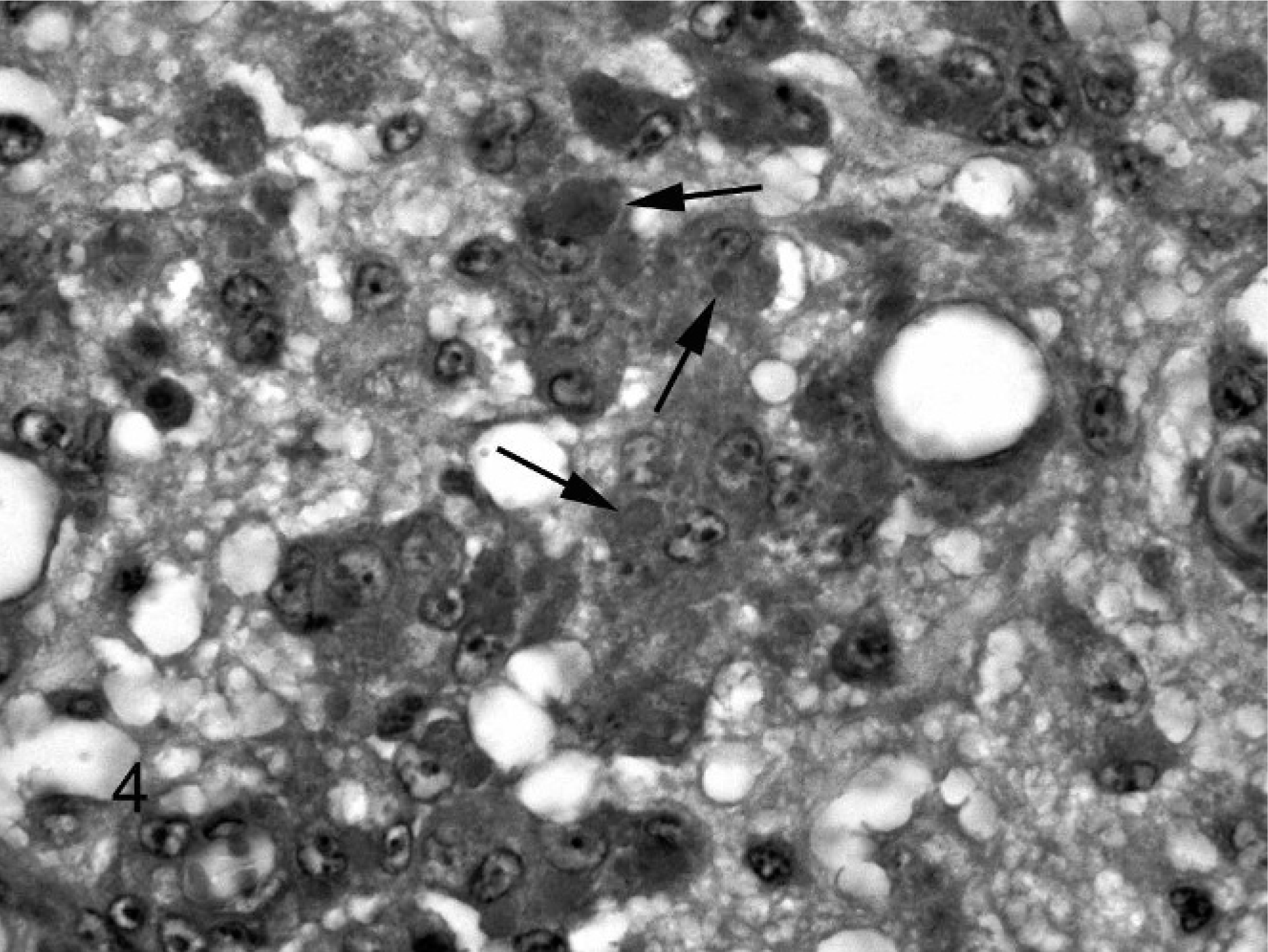
Brain, lorikeet. Note variably sized but considerably larger cytoplasmic inclusions (arrows) than those stained with the Gimenez technique. PAS.
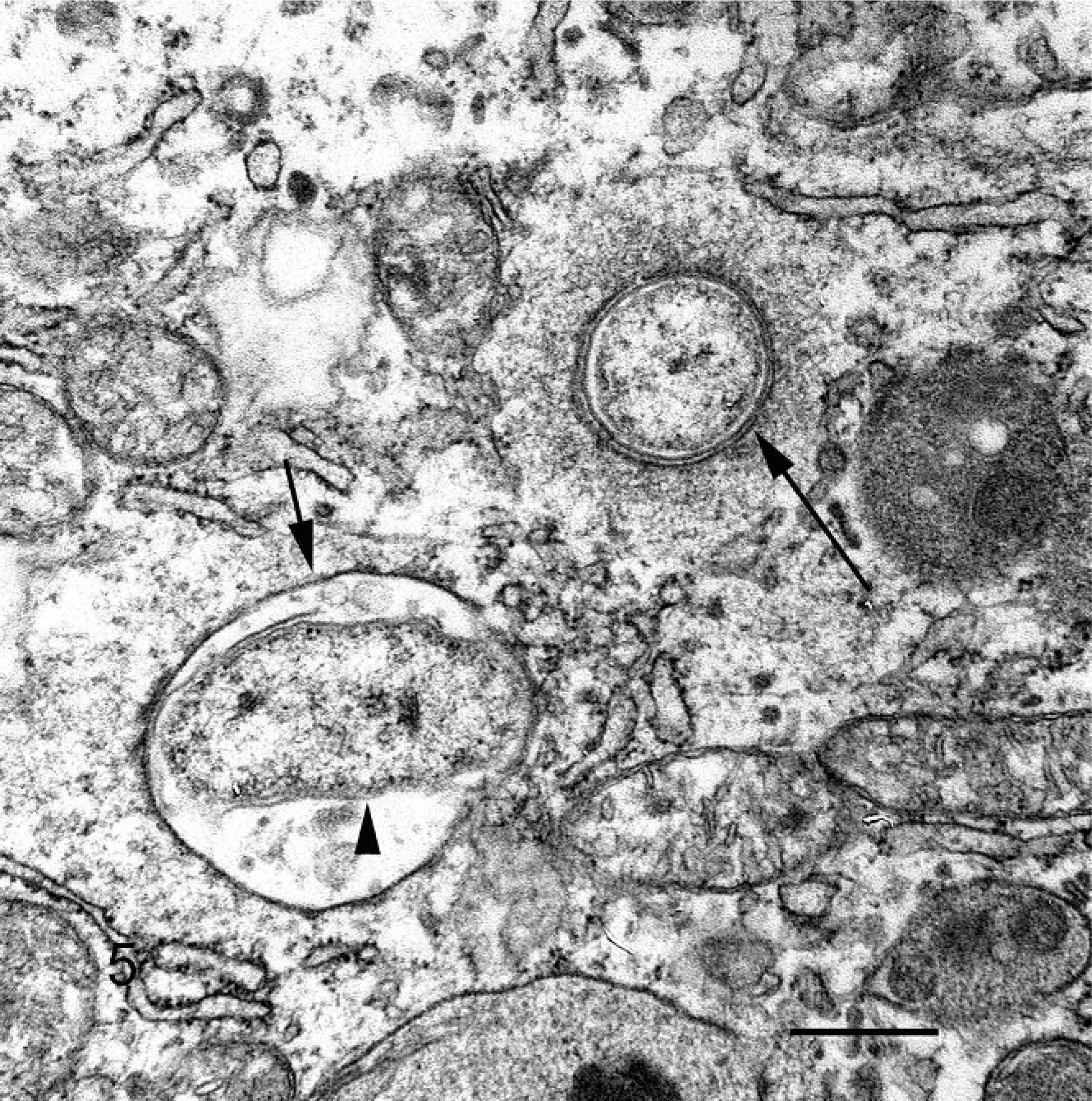
Histiocytic cell, brain, lorikeet. Note degenerative, rod-shaped bacterium (arrowhead) within a phagolysozome (small arrow), and a coccoid bacterium free in the cytoplasm (large arrow). The bacteria have a trilaminar cell wall. Uranyl acetate and lead citrate, bar = 300 μm.
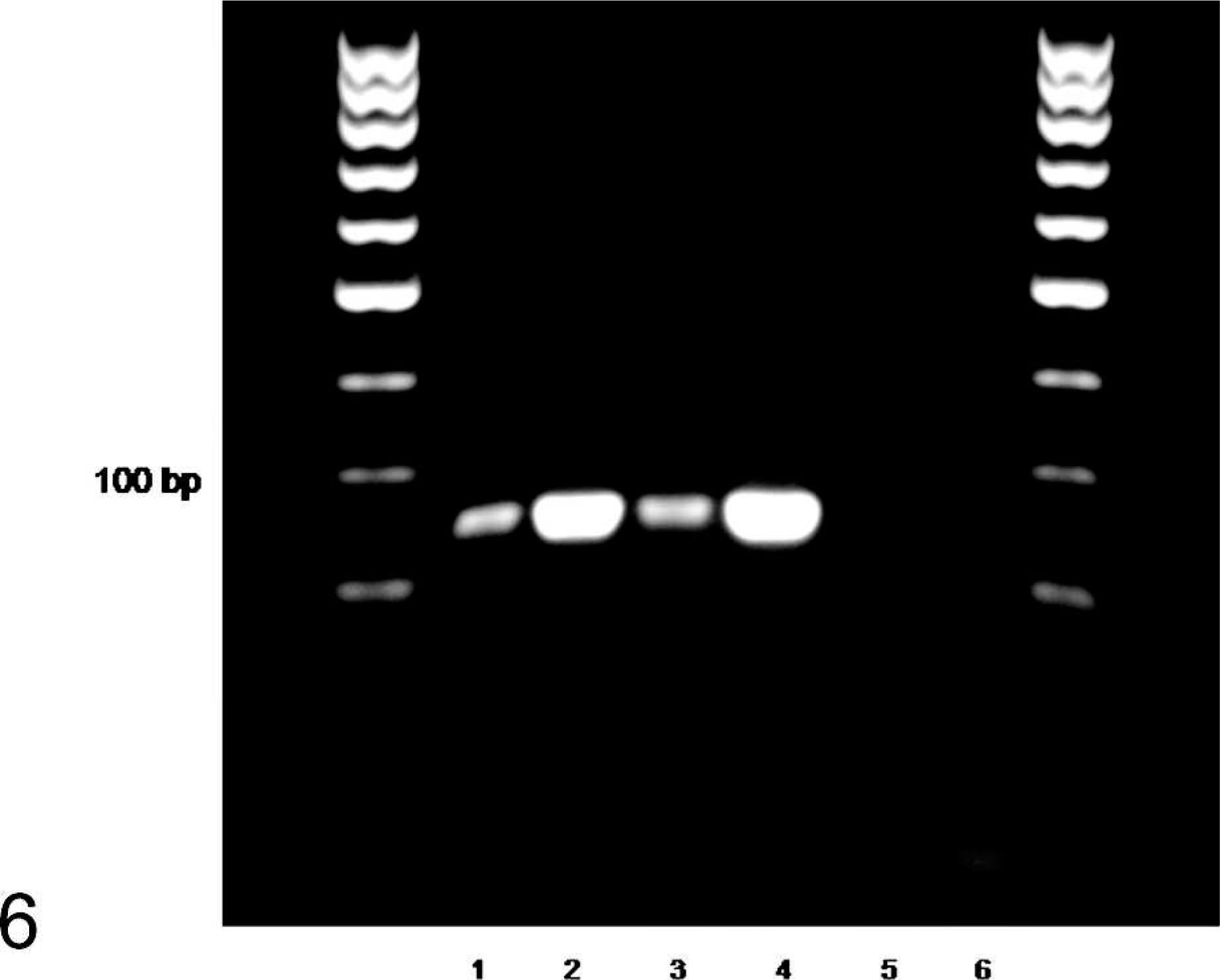
Coxiella-specific PCR on DNA extracted from lorikeet samples. Molecular size marker 100 bp; Lane 1, bird No. 1 fixed tissue; lane 2, bird No. 3 fresh brain; lane 3, bird No. 2 fixed tissue; lane 4, positive control (previously sequenced DNA from bird No. 3); lane 5, negative extraction control; lane 6, no-template control; M, 100-bp molecular size marker.
Footnotes
Acknowledgements
We are indebted to Histology Consulting Service for superb preparation of histology slides; the CAHFS, Davis, histology laboratory for technical support regarding the immunohistochemistry; Jamie Kinion and Susan Hinton for data retrieval; and Christie Buie for photo editing and electronic manuscript submission.
