Abstract
Listeria monocytogenes has been recognized as an important food-borne pathogen in animals. Records of the disease caused by this bacterium in large felids are, however, rare. The nervous form of listeriosis was diagnosed in a 12-year-old male cougar (Felis concolor) with a several-day history of neurologic disease characterized by excess salivation, head pressing, and circling that progressed to recumbency and death. Microscopically, the main alteration in the brain and spinal cord was a variably severe meningoencephalomyelitis composed mainly of mononuclear cell aggregates with fewer neutrophils. L. monocytogenes was isolated from the brain by microbiological culture, and L. monocytogenes antigen was detected in formalin-fixed, paraffin-embedded sections of brain and spinal cord by immunohistochemical analysis. On the basis of the nucleotide sequence of the 16S rRNA gene, the isolated strain was determined to be serotype 1/2a. Food-borne transmission of the bacterium was suspected, but food was not available for testing.
Listeric infections, caused principally by Listeria monocytogenes, are widespread, having been recorded in countries in over six continents and in more than 40 species of wild and domesticated animals. 7 Listeriosis is of major veterinary importance in ruminants, and has frequently been associated with encephalitis and uterine infections, leading to abortion in these species. In monogastric animals, the disease is uncommon, but septicemia and meningoencephalitis have been reported. 7, 11 Clinical cases of listeriosis in large felids are restricted to reports of the septicemic form of the disease in a serval (Felis [Leptailurus] serval) 6 and a leopard (Panthera pardus). 4 We describe the findings in a cougar (Felis concolor) with listeric meningoencephalomyelitis.
A 12-year-old male cougar from an exotic feline rescue center developed anorexia, fever, and acute neurologic disease characterized by excess salivation, head pressing, and circling that progressed to recumbency. Despite antimicrobial and fluid therapy, the animal died 5 days after the onset of clinical signs of disease. The body was submitted to the Purdue Animal Disease Diagnostic Laboratory where complete necropsy was performed. The main gross findings consisted of gastric trichobezoar, moderate gastrointestinal nematodiasis (Toxascaris leonina), and bilateral thyroid cysts. Gross abnormalities were not detected in the central nervous system (CNS). Specimens of multiple tissues, including brain and spinal cord, were fixed by immersion in neutral-buffered 10% formalin, were processed in routine manner, and were embedded in paraffin.
Frozen specimens of brain were analyzed by the direct fluorescent antibody testing for feline coronavirus, feline herpesvirus, and canine distemper virus. Virus isolation was attempted. Bacterial culture was done using special media. Brain tissue was subjected to a multiplex reverse transcriptase-polymerase chain reaction analysis for the detection of West Nile (WN) virus, Eastern equine encephalomyelitis (EEE) virus, and Western equine encephalomyelitis (WEE) virus at the National Veterinary Laboratory Services, Ames, IA.
Microscopically, the main alteration in the brain and spinal cord was a variably severe, multifocal to coalescing meningoencephalomyelitis (Figs. 1, 2). Numerous lymphocytes, plasma cells, and macrophages, and fewer neutrophils were present in the gray and white matter of brain and spinal cord. Macrophages and fewer lymphocytes also expanded the Virchow-Robin space of many of the intralesional blood vessels. The parenchyma was multifocally rarefied; gitter cells and spheroids were seen in the most severely affected areas. Distended myelin sheaths were scattered throughout the white matter. Small areas of acute hemorrhage were present, often in association with hyperemic vessels. Leptomeninges and choroid plexus of the lateral ventricles were variably infiltrated with mixed inflammatory cell infiltrates similar to those described in the parenchyma. Histologic alterations were most severe in the sections of mid- and hindbrain, cerebellar white matter, and spinal cord. In the cranial lumbar region (L1 and L2), the most severely affected region of the spinal cord, the inflammatory infiltrate was also present in the perineurium and epineurium of spinal nerves. Rare gram-positive rods were detected within the inflammatory cell aggregates. Immunohistochemical staining 8 revealed the presence of large numbers of L. monocytogenes antigen in the cytoplasm of intralesional phagocytic cells, mainly in macrophages (Fig. 3). Other histopathologic findings in this cougar included focal, bilateral, thyroid adenomatous hyperplasia with cystic degeneration; vascular amyloidosis in sections of heart, kidney, and adrenal gland; and colonization of the gastric mucosa by Helicobacter-like organisms.
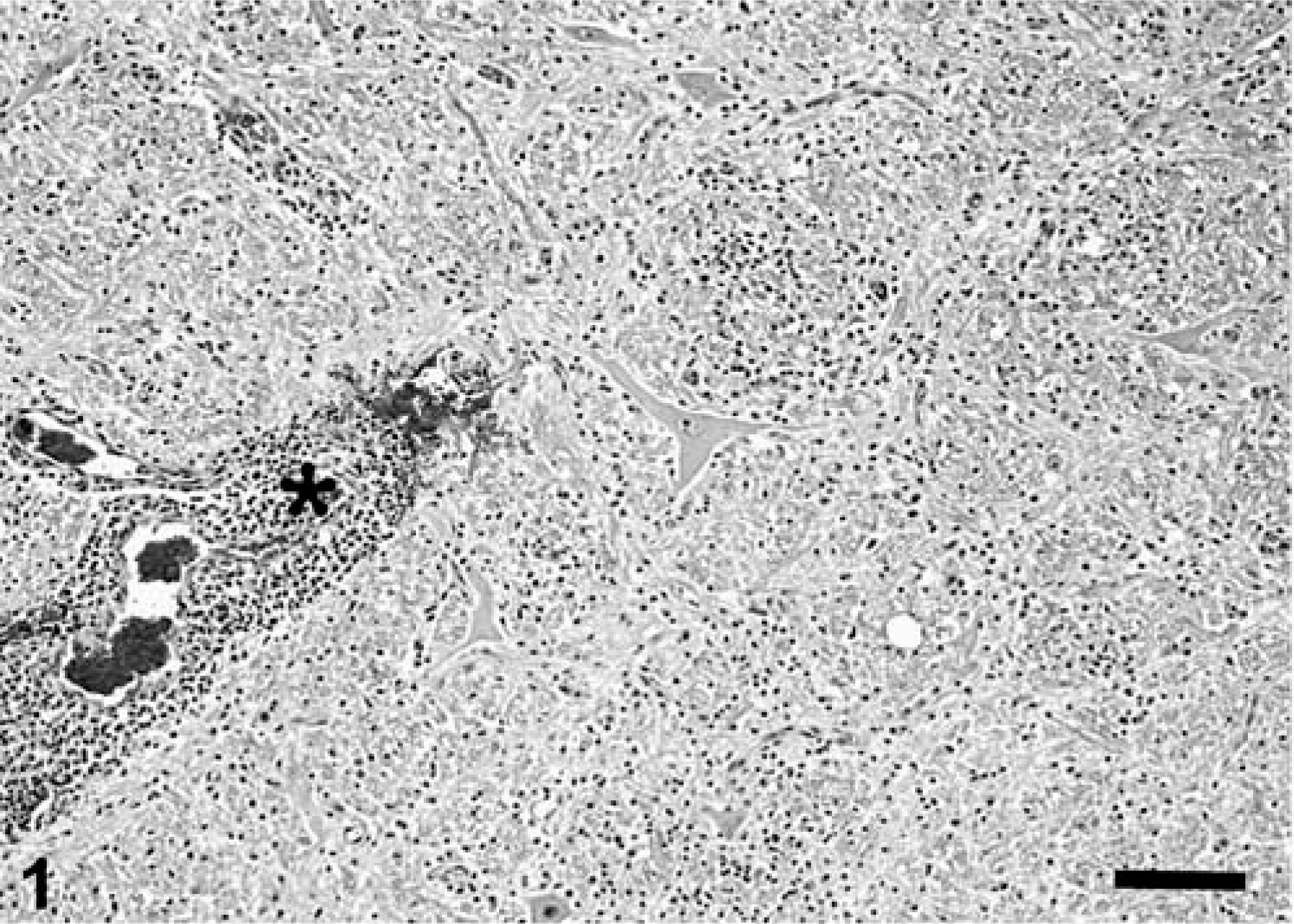
Brainstem; cougar (Felis concolor). Many inflammatory cells form thick perivascular cuffs (asterisk) and multifocal to coalescing aggregates in the neuropil. Small areas of acute hemorrhage are frequently seen in association with hyperemic vessels. HE. Bar = 135 µm.
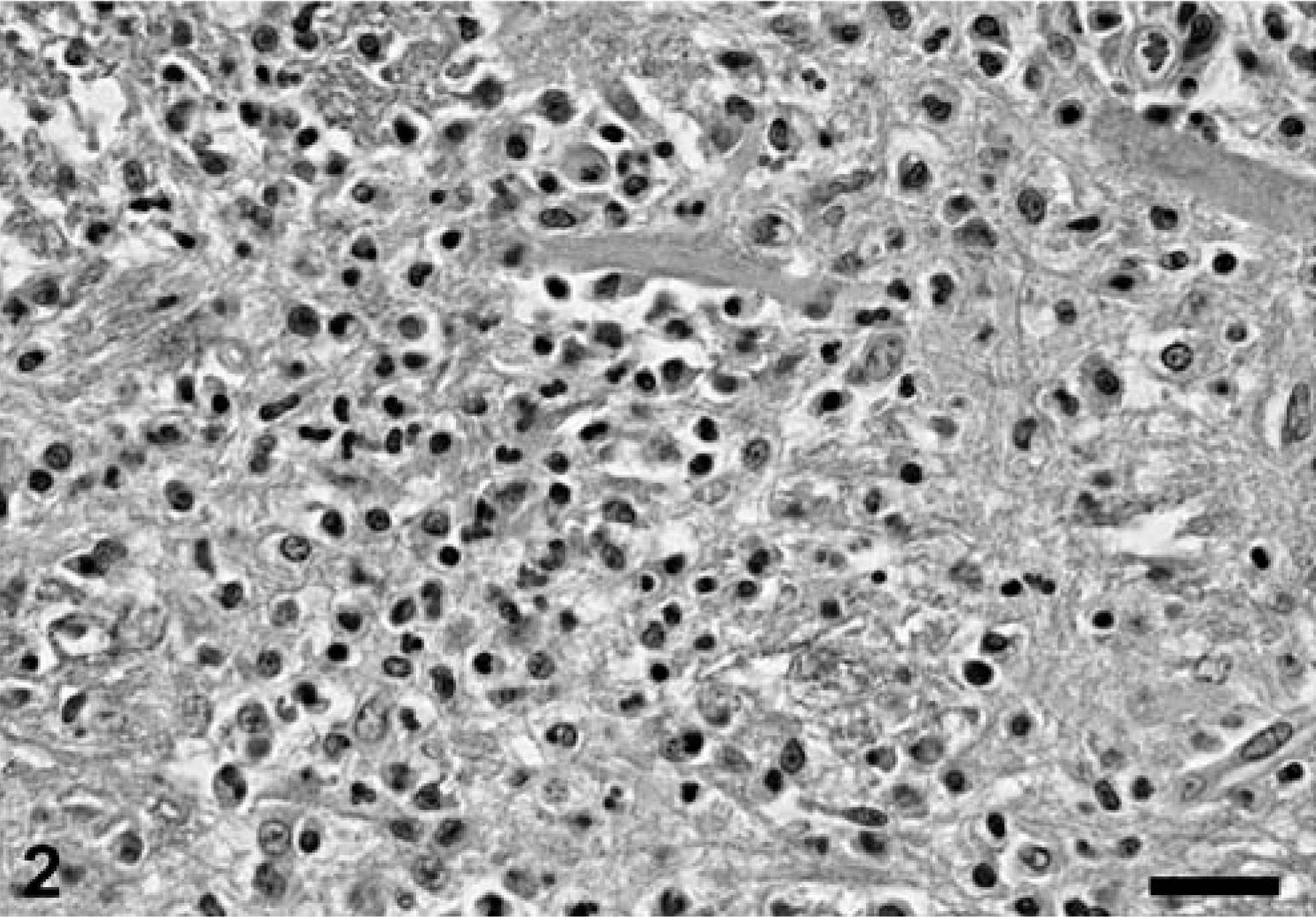
Brainstem; cougar (Felis concolor). The inflammatory cell infiltrate consists mainly of mononuclear cells. HE. Bar = 33 µm.
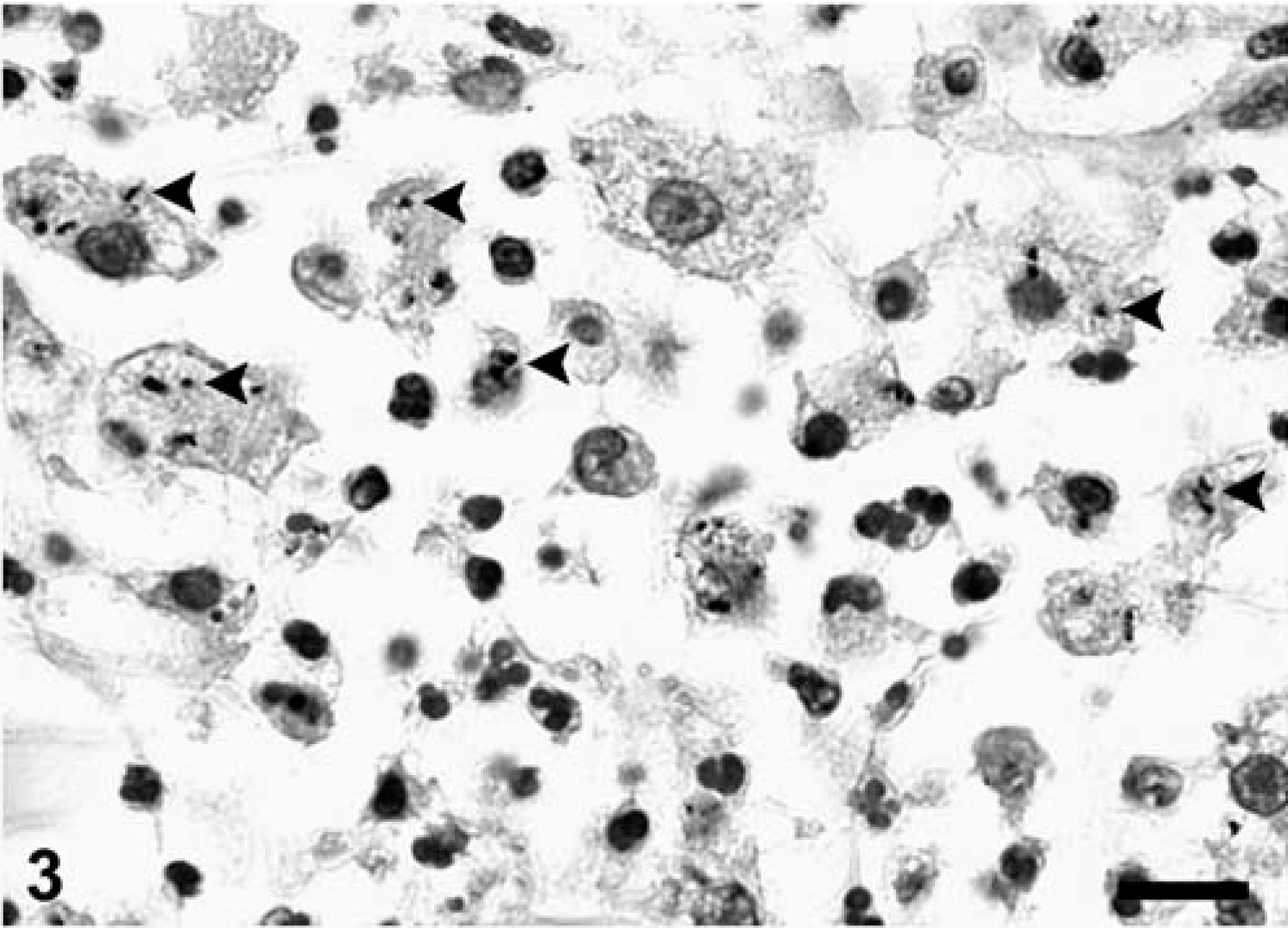
Brainstem; cougar (Felis concolor). Immunohistochemical demonstration of Listeria monocytogenes antigen (arrowheads) within macrophages. Immunoperoxidase-DAB, Mayer's hematoxylin counterstain. Bar = 10 µm.
L. monocytogenes was isolated from the brain. Results of culture for Haemophilus spp. were negative. On the basis of analysis of the nucleotide sequence of 16S rRNA gene, the isolated strain had the highest degree of similarity with the strain SLCC53 from L. monocytogenes, and was determined to be serotype 1/2a. 3 Results of the fluorescent antibody tests for feline coronavirus, feline herpesvirus, and canine distemper virus, as well as that of immunohistochemical staining for feline coronavirus, were negative. The WN, EEE, and WEE viral RNA were not detected in the brain, and evidence of viral infection was not observed in any of the cell cultures.
On the basis of several ancillary tests, L. monocytogenes was determined to be the cause of the encephalomyelitis in this cougar. Clinical signs of listeric encephalitis are principally a consequence of the lesions in the brain stem. High rectal temperature, walking in circles, and drooling of saliva caused by partial pharyngeal paralysis are common reported signs of disease. 7 In small ruminants, recumbency develops and death occurs within 2 or 3 days, but in cattle, the duration of illness is often longer. 7 In this case, the clinical course lasted 5 days. Clinical signs of disease in this cat were thought to be mainly associated with the lesions in the brain stem, although the myelitis likely contributed to the tetraparalysis.
Gross lesions of the brain are rarely observed and were not present in this case; histologic alterations are, however, considered to be pathognomonic. 7 Lesions typically are asymmetric and extend from the thalamus to the cervical part of the spinal cord, being usually centered in the pons and medulla oblongata. 7, 9 In the cougar, the inflammatory process was present in all sections of brain and spinal cord examined, indicating generalized involvement of the CNS. This extensive distribution of the lesions is an unusual finding in cases of listeriosis. The inflammation is commonly a mixture of nonsuppurative and suppurative patterns; the formation of microabscesses is considered a hallmark of the infection. Meningitis, which is often present in cases of Listeria encephalitis and was also seen in this animal, is thought to generally develop after the parenchymal lesions. Involvement of the choroid plexus and spinal cord, other than the cervical segment, as was seen in this case, is rarely associated with listeriosis. 7
L. monocytogenes serotype 1/2a was identified by 16S rRNA gene sequence analysis. The use of this sequence as a distinct signature for a bacterium has become a universal method, allowing discrimination of strains within the species. 3
Food-borne transmission is the predominant means of infection with L. monocytogenes. Results of recent studies have confirmed the presence of this bacterium in a wide variety of foodstuffs. 7 The serotype 1/2 (specifically 1/2a, 1/2b, and 1/2c) is most often isolated from meat products. 5 The animal involved in this case was fed poultry, and fresh and frozen raw meat from various species (mainly horses). One of the other two cats that shared the same enclosure died several days later, after manifesting clinical signs of disease similar to those reported for this case. Although that cat's body was not submitted for necropsy, it is possible that the cat also contracted listeriosis after ingestion of a contaminated food product; however, none of the food given to these animals in the weeks that preceded the illness was available for further testing. The exact source of the pathogen could, therefore, not be established. Since L. monocytogenes can also infect wild rodents, 11 which are commonly seen in the large felids' enclosures in the rescue center, these represent another possible source of the organism.
To our knowledge, the pathogenesis of listeric encephalitis has not been conclusively defined yet. 7, 9 On the basis of the constant involvement of the brain stem in animals, most investigators have favored the hypothesis that the organism reaches the brain especially via the trigeminal or hypoglossal nerves after invasion of the oral or nasal mucosae. 11 A recent study described evidence of intraaxonal spread of L. monocytogenes from the periphery to the CNS along the sciatic nerve. 2 Sections of sciatic nerves were microscopically unremarkable in the cougar, making the travel of the pathogen along these nerves an unlikely route of infection. In general, the entry of the bacterium into the CNS via hematogenous spread is considered to be less likely. 9 Nevertheless, in a murine model of the disease in people, repeated oral dosing with L. monocytogenes caused lesions restricted to the CNS, without gastrointestinal tract or generalized disease. 1 The authors speculated that repeated oral intake of sublethal doses of the bacterium caused a persistent subclinical bacteremia leading to the development of CNS listeriosis in the experimental mice. On the basis of the diffuse involvement of the CNS by the inflammatory process in the cougar, this pathogenetic mechanism was considered to be most likely.
Among the most common infectious diseases reported in large felids, 10 those that can result in neurologic disorders include rabies, canine distemper, feline infectious peritonitis (FIP), feline leukemia viral infection, feline immunodeficiency viral infection, feline herpesvirus type-1 infection, aberrant ascarid larval migration, toxoplasmosis, and miscellaneous bacterial infections. Of all of these, the only disease that is characterized by lesions similar to those seen in this cougar (namely, perivascular pyogranulomatous inflammation) is FIP. Virologic tests and immunohistochemical staining for feline coronavirus were used to rule out infection with this virus.
In conclusion, this case report describes the microscopic and ancillary laboratory findings in a 12-year-old cougar with the nervous form of listeriosis. To the best of our knowledge, this form has not yet been reported in large felids. Although apparently rare, listeriosis can cause signs and lesions that mimic other infectious diseases in these species. It should, therefore, be included in the differential diagnosis for CNS diseases in these species.
