Abstract
A captive-born 8-day-old male rainbow lorikeet (Trichoglossus haematodus) was found dead. Histologically, there were necrotizing hepatitis, myocarditis, and ventriculitis. Silver stain revealed argyrophilic filamentous bacilli within hepatocytes, smooth myofibers of the gizzard, and cardiac myofibers surrounding foci of necrosis. Immunohistochemistry using anti-Clostridium piliforme RT and MSK strain antisera reacted positively against bacilli within hepatocytes, cardiac myofibers, smooth myofibers of the gizzard, and splenic and intestinal macrophages. Polymerase chain reaction (PCR) assay of paraffin-embedded liver, heart, gizzard, spleen, and small intestine amplified the 196-bp DNA fragment specific to 16S ribosomal RNA of C. piliforme. The results of histopathology, immunohistochemistry, and PCR are consistent with C. piliforme infection in this lorikeet.
Tyzzer's disease has been reported in various laboratory, domestic, and nondomestic animals, 1 but there has been only one previous report of Tyzzer's disease in a bird. 5 In that case report, the identification of Clostridium piliforme was based upon histochemical and electron microscopic features of the bacilli. Here, we describe the second case of Tyzzer's disease in a bird and the first avian case diagnosed using immunohistochemistry and polymerase chain reaction (PCR) assay.
A captive-born 8-day-old male rainbow lorikeet (Trichoglossus haematodus) from a zoo in the southeastern USA was found dead in its nest. The bird had very poor weight gain over the previous 7 days. At necropsy, the liver was pale, friable, and enlarged, with rounded edges. The gastrointestinal tract was largely devoid of contents.
Samples of liver, heart, gizzard, lung, intestine, spleen, brain, trachea, eye, esophagus, skin, proventriculus, testis, kidney, and pancreas were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5.0 µm, and stained with hematoxylin and eosin (HE). Modified Gomori's methenamine silver (GMS) stain was used for visualization of bacteria in tissue sections.
For immunohistochemistry, 3.0-µm sections of paraffin-embedded liver, heart, gizzard, intestine, and spleen were stained using mouse antiserum to C. piliforme RT and MSK strains (Dr. S. Kawamura, Laboratory of Biomedical Science, Department of Veterinary Medical Science, University of Tokyo). Peroxidase-conjugated streptavidin (Histofine SAB-PO Kit, Nichirei Corp., Tokyo, Japan) and diaminobenzidine (substrate) were used to visualize the reaction.
Sections of paraffin-embedded liver, heart, gizzard, small intestine, and spleen were sectioned at 15 µm, placed in a 1.5-ml tube, and deparaffinized. PCR was performed using a previously published protocol. 3
Histologically, there were many scattered, coalescing foci of coagulative and lytic necrosis of hepatocytes admixed with fibrin and hemorrhage (Fig. 1). Many necrotic foci were surrounded by few macrophages and heterophils. Several hepatocytes encompassing areas of hepatocellular necrosis contained many intracytoplasmic 7–10-µm filamentous bacilli arranged in a criss-cross pattern. The intracytoplasmic bacilli were argyrophilic when stained with modified GMS stain (Fig. 2). There was multifocal degeneration and necrosis of cardiac myofibers, and the myocardial interstitium was infiltrated by low numbers of macrophages, lymphocytes, plasma cells, and extravasated erythrocytes. Multiple aggregates of plasma cells, lymphocytes, and macrophages associated with degenerate and necrotic smooth myofibers were in the muscularis externa of the gizzard. Similar argyrophilic bacilli were within cardiac myofibers and smooth muscle fibers neighboring the foci of necrosis.
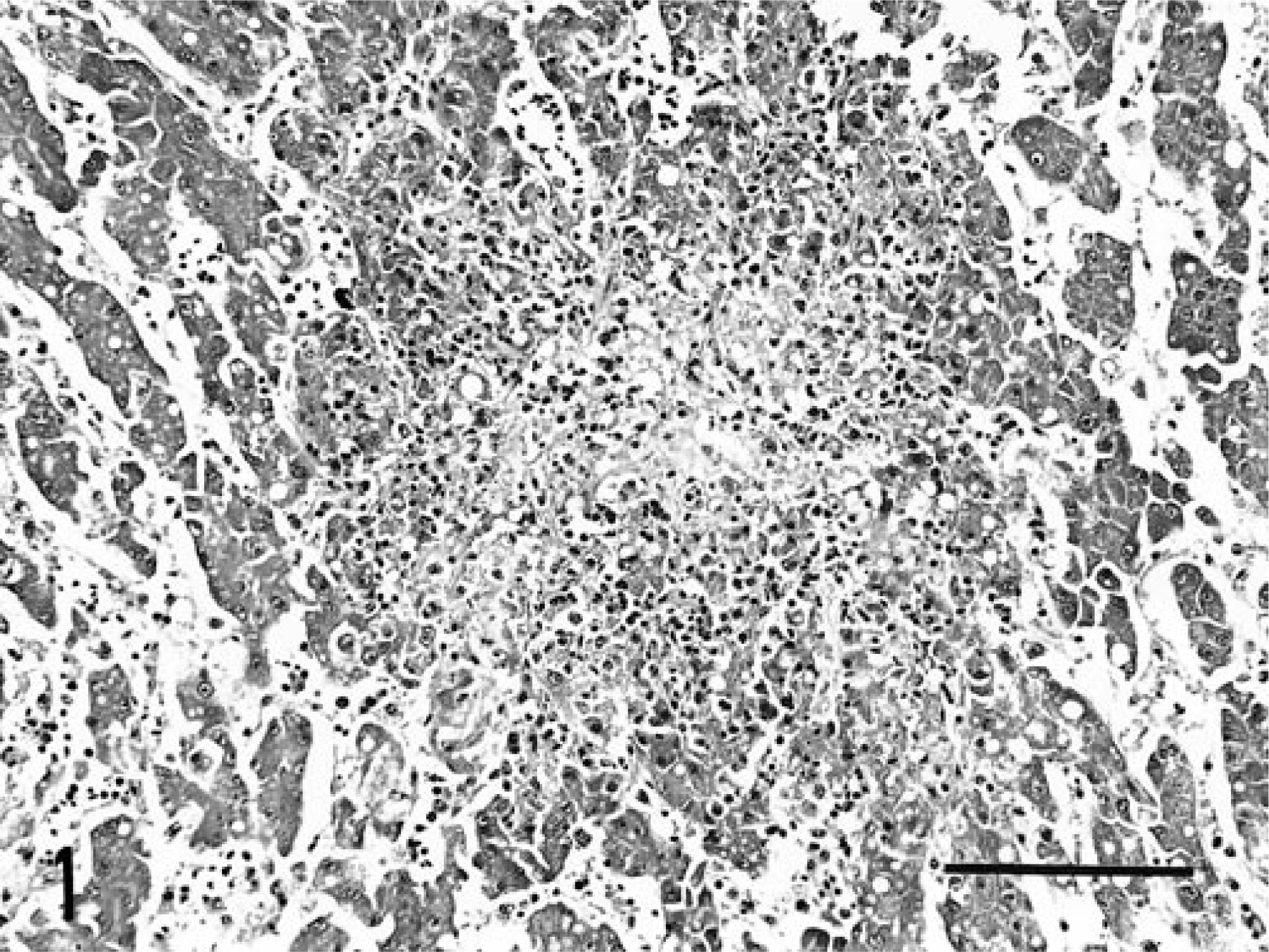
Liver; lorikeet. Focus of hepatocellular necrosis disrupts hepatic cords. HE. Bar = 200 µm.

Liver; lorikeet. Several hepatocytes contain numerous intracytoplasmic, argyrophilic bacilli (arrowheads). GMS. Bar = 50 µm.
Immunohistochemistry using anti-RT and anti-MSK antisera revealed positively immunostained intracytoplasmic bacilli within hepatocytes, smooth myofibers of the gizzard, and cardiac myofibers adjacent to necrotic foci. Immunopositive bacilli were also noted within macrophages in the splenic parenchyma and mucosa of the small intestine. Using PCR, the 196-bp DNA fragment specific to 16S ribosomal RNA of C. piliforme was amplified from liver, heart, spleen, gizzard, and small intestine (Fig. 3).
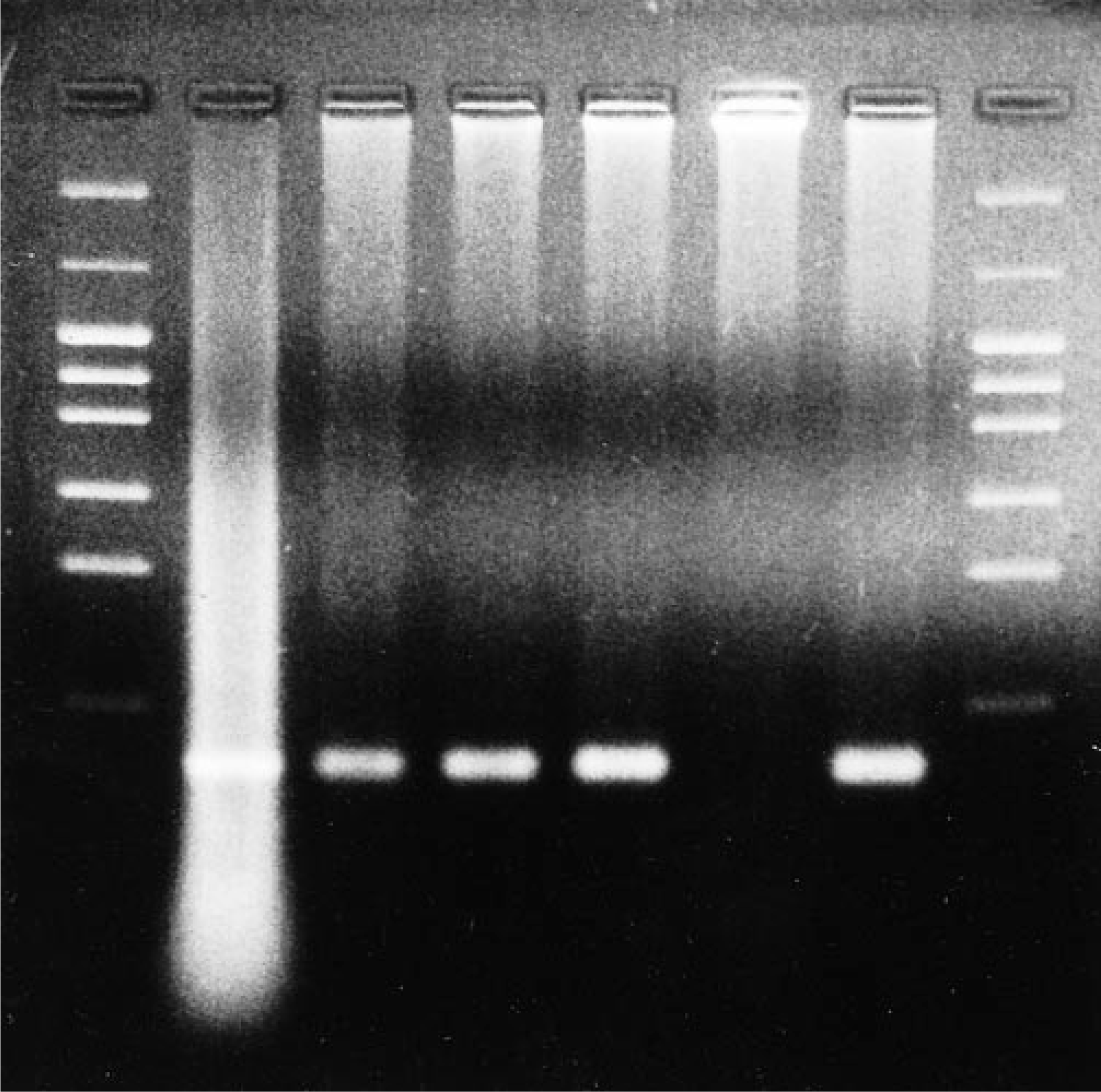
PCR for C. piliforme 16S ribosomal RNA. Lanes 1, 8 = 50-bp ladder DNA molecular weight marker; lane 2 = mouse liver infected with C. piliforme MSK strain (positive control); lane 3 = liver of the infected lorikeet; lane 4 = gizzard of the infected lorikeet; lane 5 = heart of the infected lorikeet; lane 6 = normal liver from parakeet (negative control); lane 7 = small intestine of the infected lorikeet (spleen not shown). Lanes 2–4, 5, and 7 show the specific bands at 196 bp.
The diagnosis of C. piliforme infection in this lorikeet was based upon results of histopathology, histochemistry, immunohistochemistry, and PCR study. This is only the second reported case of Tyzzer's disease in a bird. This lorikeet and the previous affected cockatiel 5 were both captive-born psittaciforms less than 9 days of age, indicating a possible susceptibility of select neonatal psittaciforms to infection with C. piliforme.
Necrotizing and inflammatory lesions in the gastrointestinal tract, liver, and heart are typical pathologic findings for Tyzzer's disease in mammals. 1 Both this lorikeet and the cockatiel with Tyzzer's disease had characteristic lesions of C. piliforme infection in the liver. This lorikeet also had lesions in the heart that are common in mammals with Tyzzer's disease.
Infection with C. piliforme is most commonly via an oral route through ingestion of bacterial spores. 2 It was not determined how this lorikeet became infected, and no other birds in the exhibit have died from Tyzzer's disease. The finding of C. piliforme and associated necrotic lesions in the wall of the gizzard indicates that the gizzard is a portal of entry for C. piliforme in birds. Intestinal leiomyositis has been described in mammals with Tyzzer's disease and is believed to be caused by lymphatic involvement of C. piliforme. 2 Enteritis was not seen in either avian case of Tyzzer's disease, but silver-positive bacilli were reported in the wall of the small intestine of the cockatiel, and this lorikeet had a few immunopositive bacilli within mucosal macrophages. Therefore, the small intestine probably also serves as an access point for C. piliforme infection in birds.
Immunosuppression usually plays a role in the development of infection with C. piliforme in mammals. 4 In addition, stressful events often precede outbreaks of Tyzzer's disease in mammals. 2 This lorikeet may have been immunocompromised by various physiologic and environmental stressors associated with captive rearing. The bursa of Fabricus was not examined microscopically in this lorikeet. The spleen had a marked absence of lymphoid tissue, which may have been due to the young age of the bird.
Footnotes
Acknowledgements
We thank R. Brown for histology and Dr. S. Kawamura for providing mouse antiserum to Clostridium piliforme strains RT and MSK.
