Abstract
Tumors of the nasal cavity or paranasal sinuses of 18 dogs were examined histopathologically, immunohistochemically, and histochemically. The tumors were classified histologically as 13 adenocarcinomas, 3 transitional carcinomas, 1 squamous cell carcinoma, and 1 adenosquamous carcinoma. Tumor cells were strongly immunoreactive for broad-spectrum cytokeratins in all cases, for cytokeratin 8/18 in 16 cases, and for cytokeratin 19 in 17 cases. None of the 18 carcinomas had cytologic or histologic features indicative of neuroendocrine differentiation, yet tumor cells in 5 of the 13 adenocarcinomas were argyrophilic and immunohistochemically positive for synaptophysin and chromogranin A. Results of this study indicate that neuroendocrine markers may be detected immunohistochemically and histochemically in canine nasal or paranasal adenocarcinomas despite the lack of typical histologic features of neuroendocrine differentiation.
Keywords
Tumors in the nasal cavity or paranasal sinuses are uncommon in dogs. 24 Malignant epithelial tumors in these regions are histologically classified as squamous cell carcinoma, transitional carcinoma, adenocarcinoma, adenosquamous carcinoma, adenoid cystic carcinoma, acinic cell carcinoma, undifferentiated carcinoma, olfactory neuroblastoma, and neuroendocrine carcinoma. 4 Among these tumors, adenocarcinoma is most common, followed by transitional carcinoma and then squamous cell carcinoma. 23 Neuroendocrine carcinomas are rarely reported. 16 The main growth patterns of adenocarcinomas are glandular, acinar, papillary, tubulopapillary, and solid. Neuroendocrine carcinomas in the nasal cavity and paranasal sinuses, like those in other regions, are characterized by growth patterns such as sheets, nests, or cords of cells separated by fibrovascular stroma. Tumor cells are small- to medium-sized, round to polyhedral, and have granular eosinophilic cytoplasm. 4 Definitive diagnosis of neuroendocrine carcinoma is based on immunohistochemical detection of neuroendocrine markers such as synaptophysin (Syn) and chromogranin A (CgA). Neuroendocrine differentiation has been reported in nasal or paranasal adenocarcinomas in humans, 1, 8 but not in dogs. Therefore, we examined nasal or paranasal neoplasms of 18 dogs to determine whether malignant epithelial tumors without typical histologic features of neuroendocrine carcinoma expressed neuroendocrine markers by immunohistochemistry and histochemistry.
Materials and Methods
Tumors
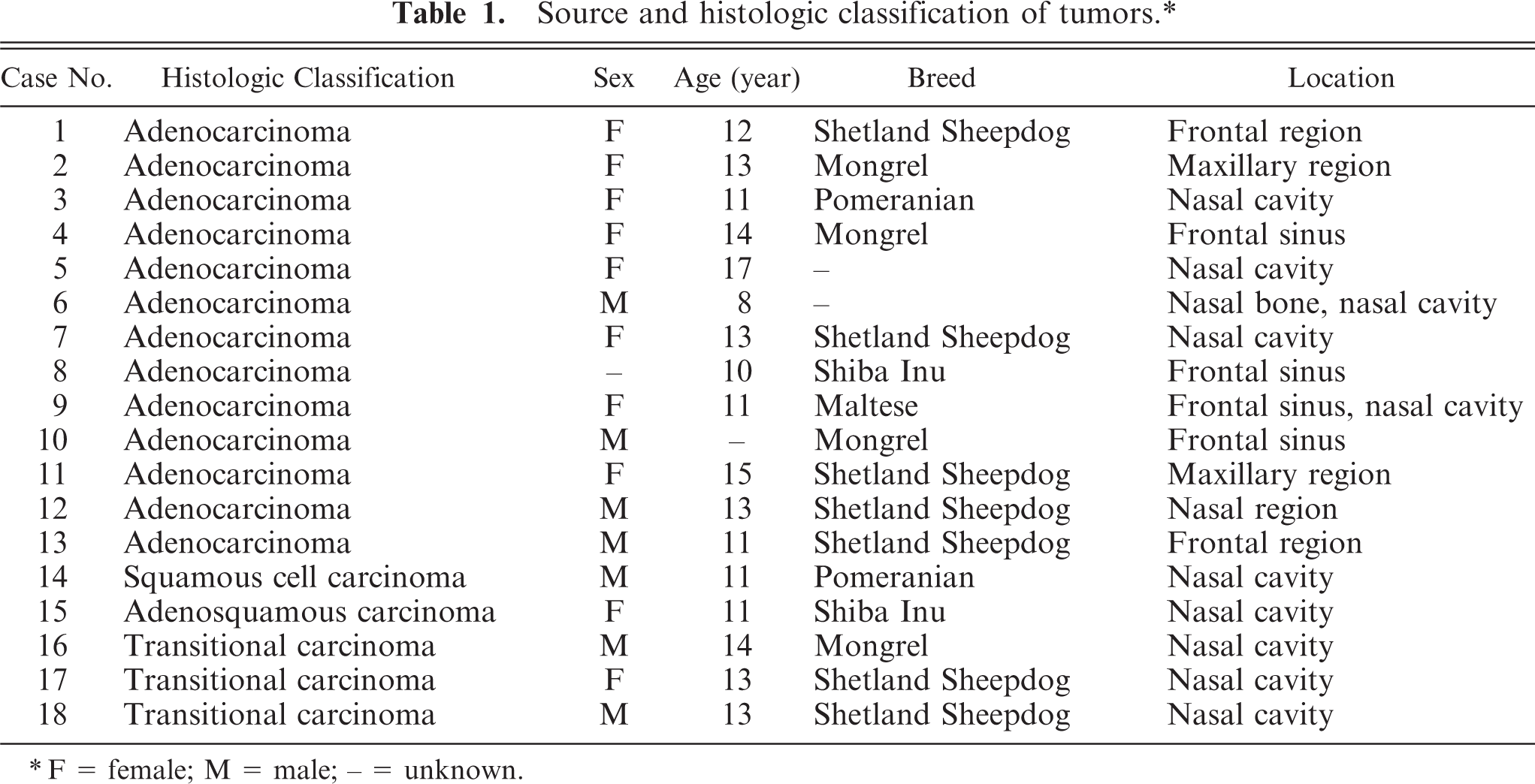
All tumors were surgically resected from the nasal cavity or paranasal sinuses of 18 dogs by veterinary practitioners, and formalin-fixed specimens were sent to our laboratory. The dogs consisted of 7 males, 10 females, and 1 of unknown sex; included 7 Shetland Sheepdogs and various other breeds; and were from 8 to 17 years of age. The tumors were located mainly in the nasal cavity or frontal sinus but extended widely from the nasal region to the frontal region (Table 1).
Sections from each formalin-fixed, paraffin-embedded specimen were stained with hematoxylin and eosin. Tumors were classified according to the histologic criteria established by the World Health Organization (WHO) (Table 1). 4 Grimelius' staining was also performed on each specimen.
Immunohistochemistry
Each specimen was stained by the universal immunoenzyme polymer method. The universal staining system (Autostainer, Dako Japan Co. Ltd., Kyoto, Japan) was used for all primary antibodies except cytokeratin (CK) 19. The primary antibodies and antigen retrieval methods are detailed in Table 2.
Details of primary antibodies and pretreatment procedures for antigen retrieval.∗
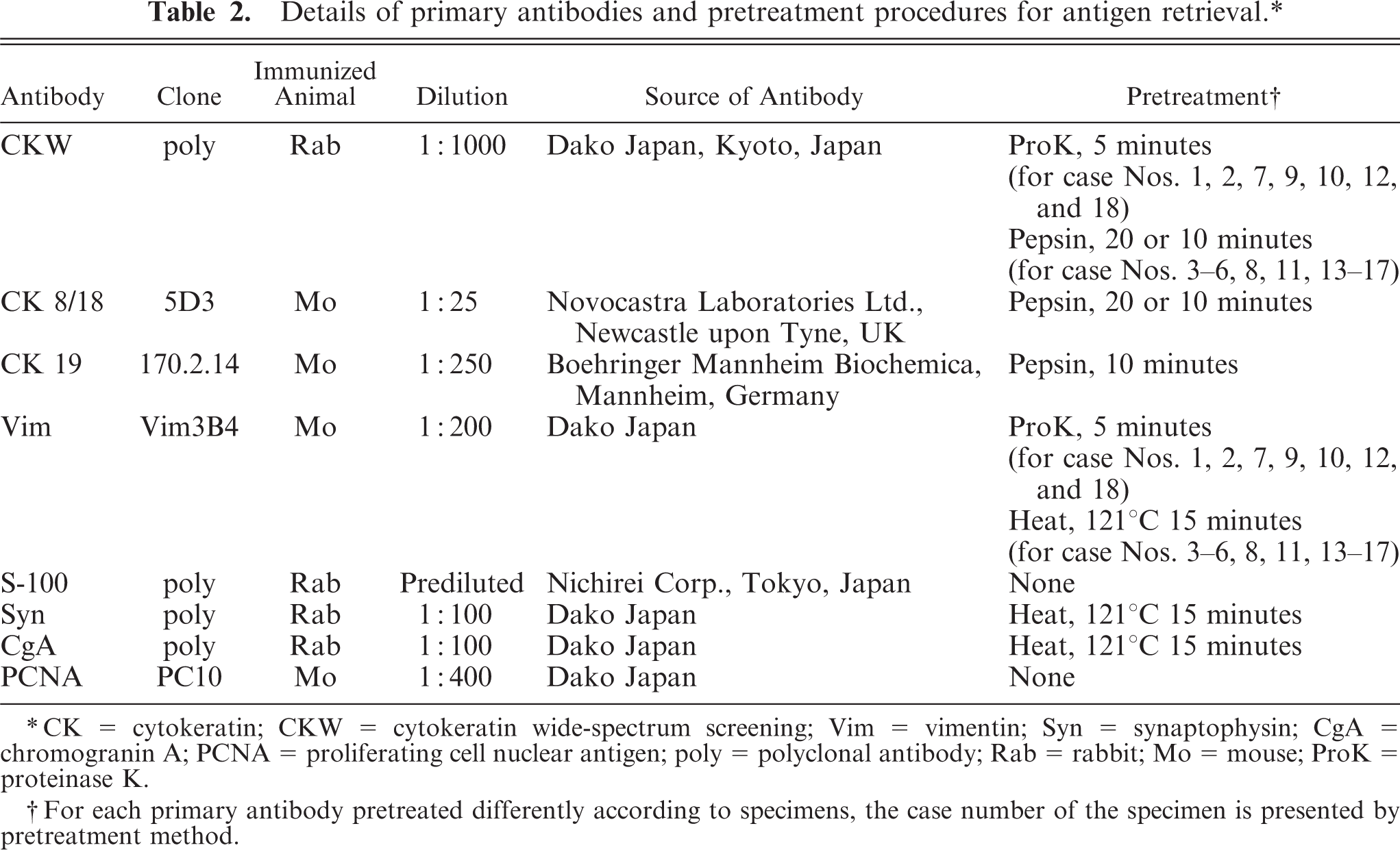
CK = cytokeratin; CKW = cytokeratin wide-spectrum screening; Vim = vimentin; Syn = synaptophysin; CgA = chromogranin A; PCNA = proliferating cell nuclear antigen; poly = polyclonal antibody; Rab = rabbit; Mo = mouse; ProK = proteinase K.
For each primary antibody pretreated differently according to specimens, the case number of the specimen is presented by pretreatment method.
Each section was deparaffinized and treated with heat pretreatment as needed. Sections were washed with purified water and buffer (Tris-buffered saline T, Dako Japan; phosphate-buffered saline for CK19), then treated with a peroxidase-blocking reagent (Dako Japan) for 5 minutes or (for CK19) 3% H2O2 for 10 minutes at room temperature. The sections were washed with the buffer, and enzymatic pretreatment was applied as needed. All sections were washed with buffer and incubated with the primary antibody for 30 minutes at room temperature. After washing with the buffer, the sections were incubated for another 30 minutes with Histofine Simple Stain Rat MAX PO (M) or (R) (Nichirei Corp., Tokyo, Japan) at room temperature. Finally, after washing with the buffer, all sections were treated with chromogen (3,3′-diaminobenzidine tetrahydrochloride liquid system [substrate], Dako Japan) and counterstained with hematoxylin.
Evaluation by immunohistochemistry and histochemistry
The specimens were examined with a light microscope (AX80, Olympus Corp., Tokyo, Japan) and photographed at 400-fold magnification (DP70 and DP control, Olympus Corp.). Positive cells in 5 photomicrographs were counted. The real size of each field was approximately 219 μm × 165 μm. For tumors with multiple growth patterns and pattern-dependent immunohistochemical or histochemical results, examination of 5 fields was performed for each pattern. Immunoreactivity was rated on the following 4-point scale: − (negative), + (< 30% positive cells), ++ (30 to 70% positive cells), +++ (> 70% positive cells).
Results
The tumors were classified as 13 adenocarcinomas, 3 transitional carcinomas, 1 squamous cell carcinoma, and 1 adenosquamous carcinoma (Table 1). The growth patterns in each tumor are presented in Table 3. The predominant growth pattern in adenocarcinomas was tubular (4/13, Fig. 1), tubulopapillary (1/13, Fig. 2), acinar (1/13, Fig. 3), or solid (6/13, Fig. 4). In the remaining adenocarcinoma, tubular and solid patterns were almost equally prominent. Multiple growth patterns were observed in 9 of the 13 adenocarcinomas. The squamous cell carcinoma consisted entirely of a squamous pattern. In the case of the adenosquamous carcinoma, a predominantly squamous pattern was combined with a tubular pattern. All 3 transitional carcinomas had a predominantly stratified pattern that was combined in 1 tumor with a tubular pattern and, in another tumor, with an acinar pattern. None of the 18 cases had a palisading or rosette pattern, suggestive of neuroendocrine differentiation. The cells of the adenocarcinomas had eosinophilic or pale eosinophilic homogeneous cytoplasm and varied in shape (Figs. 1–4). None of these cells had distinctly granular eosinophilic cytoplasm.
Growth patterns in tumors.∗
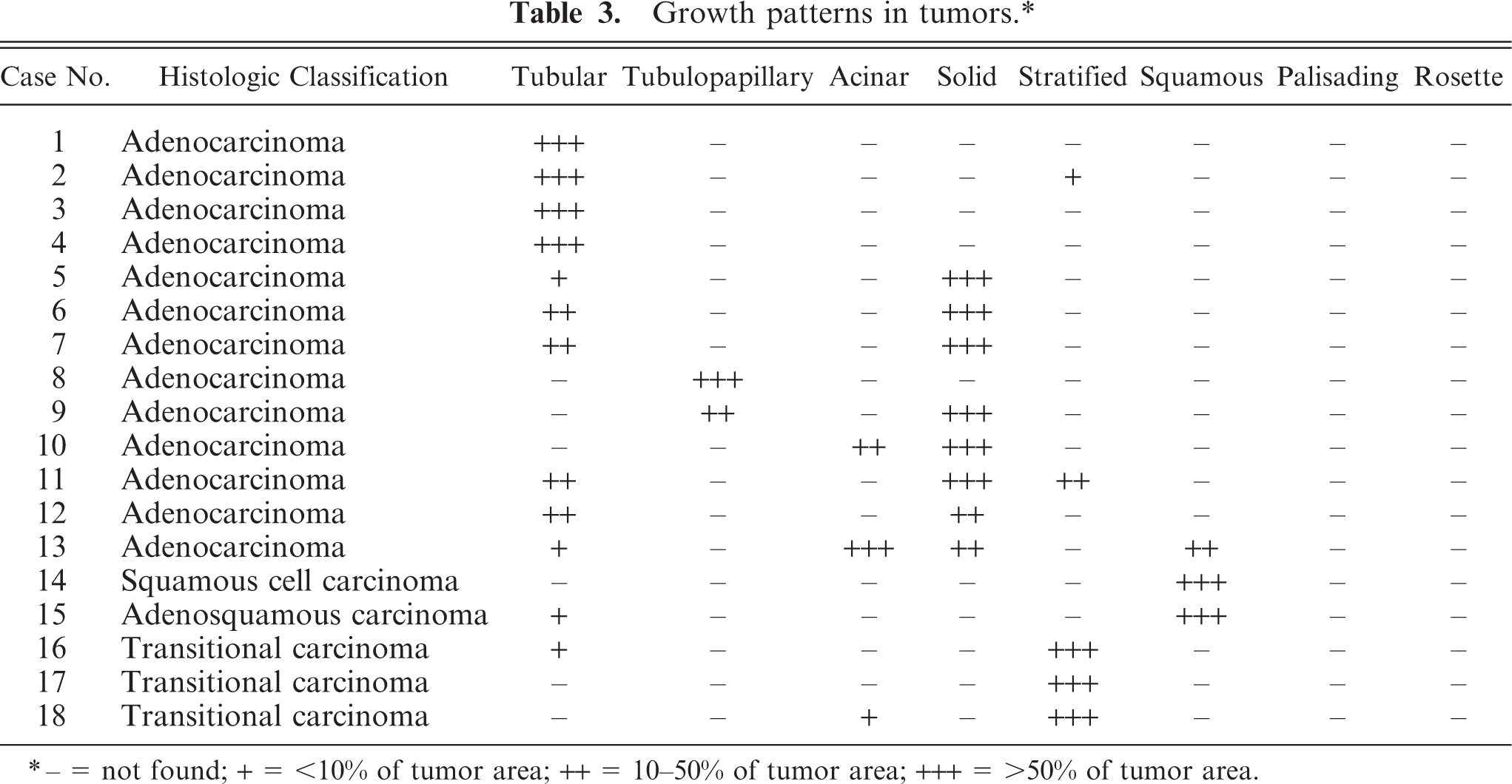
− = not found; + = <10% of tumor area; ++ = 10–50% of tumor area; +++ = >50% of tumor area.
The results of immunohistochemistry and histochemistry are presented in Table 4. All 18 tumors were positive for broad-spectrum CK and proliferating cell nuclear antigen, but negative for vimentin. CK8/18-positive cells were found in 16 cases, excluding the squamous cell carcinoma and 1 of the 3 transitional carcinomas; regions of squamous differentiation in the adenocarcinoma and adenosquamous carcinoma were negative. CK19-positive cells were found in 17 cases, excluding the squamous cell carcinoma. Syn-positive cells were found in 9 of the 13 adenocarcinomas, the adenosquamous carcinoma, and 1 of the 3 transitional carcinomas (Fig. 5). CgA-positive and Grimelius' staining–positive cells were confirmed in 6 and 3 of the 13 adenocarcinomas, respectively (Figs. 6–8). In the adenosquamous carcinoma, Syn-positive cells were observed only in the regions with a tubular pattern and not in the regions with a squamous pattern. Reactivity for multiple markers including Syn, CgA, and argyrophilia by Grimelius' staining was evident in 5 of the 13 adenocarcinomas (case Nos. 2, 7, 9, 11, and 13). S100-positive cells were found in 2 of the 3 transitional carcinomas.
Results of immunohistochemistry and histochemistry.∗
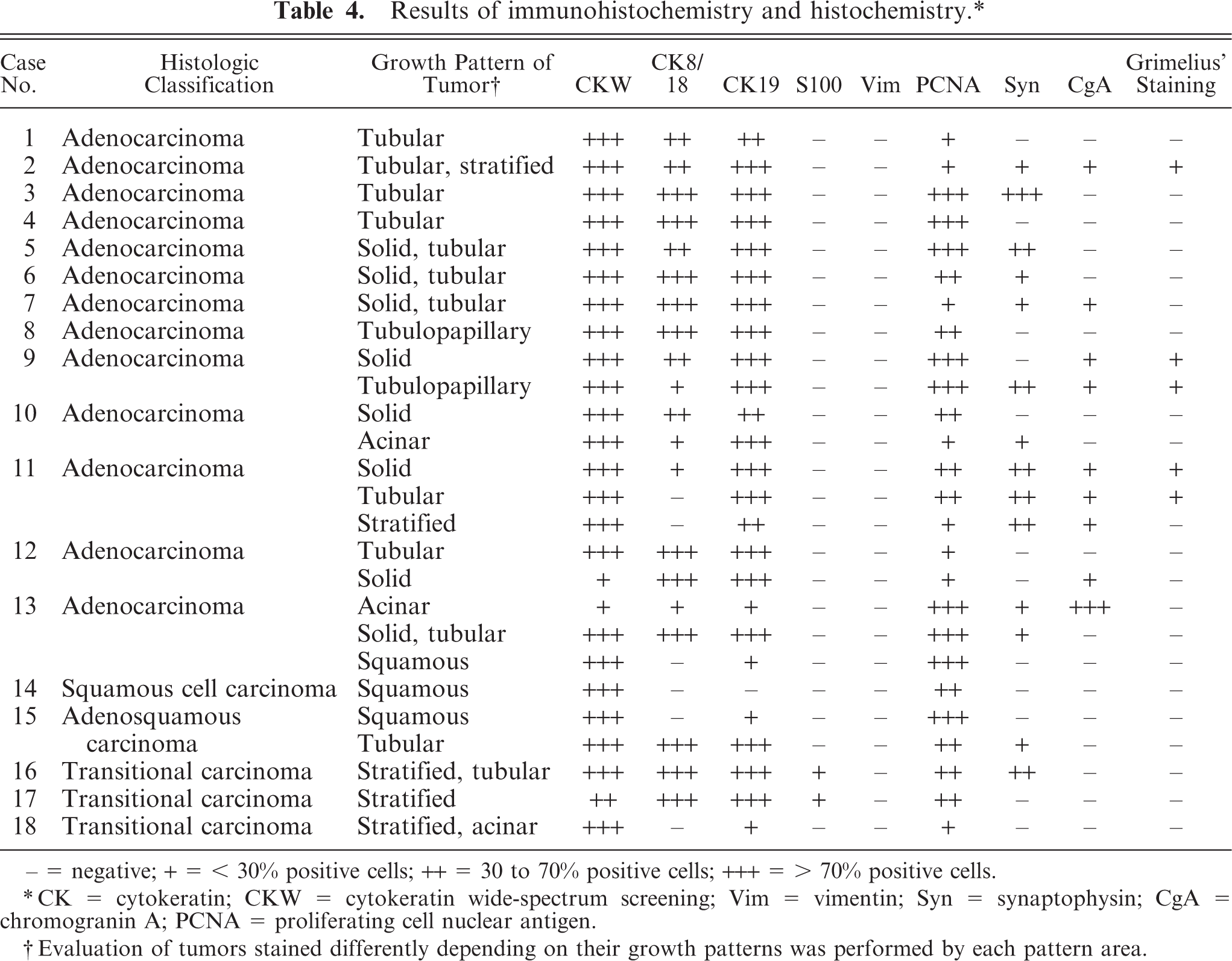
− = negative; + = < 30% positive cells; ++ = 30 to 70% positive cells; +++ = > 70% positive cells.
CK = cytokeratin; CKW = cytokeratin wide-spectrum screening; Vim = vimentin; Syn = synaptophysin; CgA = chromogranin A; PCNA = proliferating cell nuclear antigen.
Evaluation of tumors stained differently depending on their growth patterns was performed by each pattern area.
Discussion
Neuroendocrine carcinomas have been classified separately from epithelial or mesenchymal tumors of nasal or paranasal origin. 23 However, cytologic and histologic features of neuroendocrine carcinomas are similar to those in nasal epithelial tumors, and neuroendocrine carcinomas are classified as malignant epithelial tumors in the WHO histologic classification. 4 Furthermore, cells that express neuroendocrine markers are occasionally encountered in canine nasal epithelial tumors.
In the present study, although all 18 carcinomas lacked morphologic evidence of neuroendocrine differentiation, 5 of the 13 adenocarcinomas had diffuse or focal immunoreactivity for multiple neuroendocrine markers including Syn, CgA, and argyrophilia by Grimelius' staining. These findings suggested that the 5 adenocarcinomas had neuroendocrine differentiation. Although Syn and CgA are generally used as markers of neuroendocrine tumors, they are not specific for neuroendocrine cells and are also positive in a wide range of tissues, including those of the nervous system. In the present study, we required immunoreactivity for multiple neuroendocrine markers to document neuroendocrine differentiation.
Neuroendocrine carcinomas are malignant epithelial tumors with neuroendocrine differentiation that have been reported in a wide range of tissues, including the liver, bile duct, intestines, paranasal sinuses, lung, and skin of domestic animals. 3, 5, 11, 13– 15, 17, 20, 21 Sako et al. reported neuroendocrine carcinomas of the nasal cavity in dogs that had typical growth patterns, i.e., sheets, nests, ribbons, or rosette formations, and had cells with eosinophilic cytoplasmic granules. 16 In their report, all neuroendocrine carcinomas were CK AE1/AE3–positive, but were negative for CK8 and CK19; some tumors were strongly positive for both Syn and CgA; others were only weakly positive for both neuroendocrine markers or were positive only for CgA. 16 A nasopharyngeal neuroendocrine carcinoma in a dog 12 had typical neuroendocrine patterns, i.e., groups, sheets, or palisading; tumor cells were negative for CK AE1/AE3 and positive for Syn; immunohistochemistry for CgA was not performed. The neuroendocrine carcinomas in those 2 reports differed in histologic pattern, cytologic features, and CK expression from the adenocarcinomas with neuroendocrine differentiation in the present study.
In humans, adenocarcinomas with neuroendocrine differentiation in the nasal cavity or paranasal sinuses have been reported. 1, 8 In those reports, CgA and/or Syn were used as markers of neuroendocrine differentiation. The results of the present study are consistent with the immunohistochemical findings for human tumors and indicate that there are adenocarcinomas with neuroendocrine differentiation in the nasal cavity or paranasal sinuses in dogs as in humans. Neuroendocrine differentiation in adenocarcinomas of the nasal cavity or paranasal sinuses was associated with higher mortality in humans. 8 Further studies are required to determine whether neuroendocrine differentiation of canine nasal adenocarcinomas warrants a worse prognosis.
Most adenocarcinomas with neuroendocrine differentiation in humans were categorized as intestinal type adenocarcinomas with morphologic characteristics of normal or neoplastic intestinal epithelial cells. 1, 8 In the present study, however, no correlation between histologic pattern and neuroendocrine differentiation was recognized. The 5 adenocarcinomas with neuroendocrine differentiation had various histologic patterns, including tubular, solid, tubulopapillary, stratified, and acinar. The other 8 adenocarcinomas similarly had no common pattern. On the other hand, tumor cells of the squamous cell carcinoma and squamous regions of the adenocarcinoma or adenosquamous carcinoma were negative for any neuroendocrine markers. This could indicate squamous differentiation and neuroendocrine differentiation are mutually exclusive.
The anatomic structures of the nasal cavity and paranasal sinuses are complicated, especially in dogs. 19 The nasal mucosa consists of 4 types of epithelium, namely squamous, transitional, respiratory, and olfactory; the distribution of these epithelia is also complicated. 2, 9, 22 Transition from 1 type of epithelium to another is gradual in nasal mucosa. The nasal epithelium has considerable plasticity in its differentiation in reaction to stimuli. 7, 10 Neuroendocrine cells are widely scattered in the epithelium of the respiratory tract. 6, 12, 18 The histologic variety of malignant epithelial tumors in the nasal cavity and paranasal sinuses may reflect these anatomic complexities of the nasal mucosa.
Our study demonstrated that some adenocarcinomas in the nasal cavity or paranasal sinuses of dogs undergo neuroendocrine differentiation. However, no correlation between neuroendocrine differentiation and other characteristics of these tumors, such as cytologic features, histologic pattern, proliferation index, tumor location, or signalment of the dogs, was apparent. Thus, the possibility of neuroendocrine differentiation should be considered in nasal adenocarcinomas, even if histologic features of neuroendocrine tumors are not obvious. Grimelius' staining for argyrophilia and immunohistochemical detection of Syn and CgA expression can be used to detect neuroendocrine differentiation in adenocarcinomas without typical histologic features of neuroendocrine tumors.
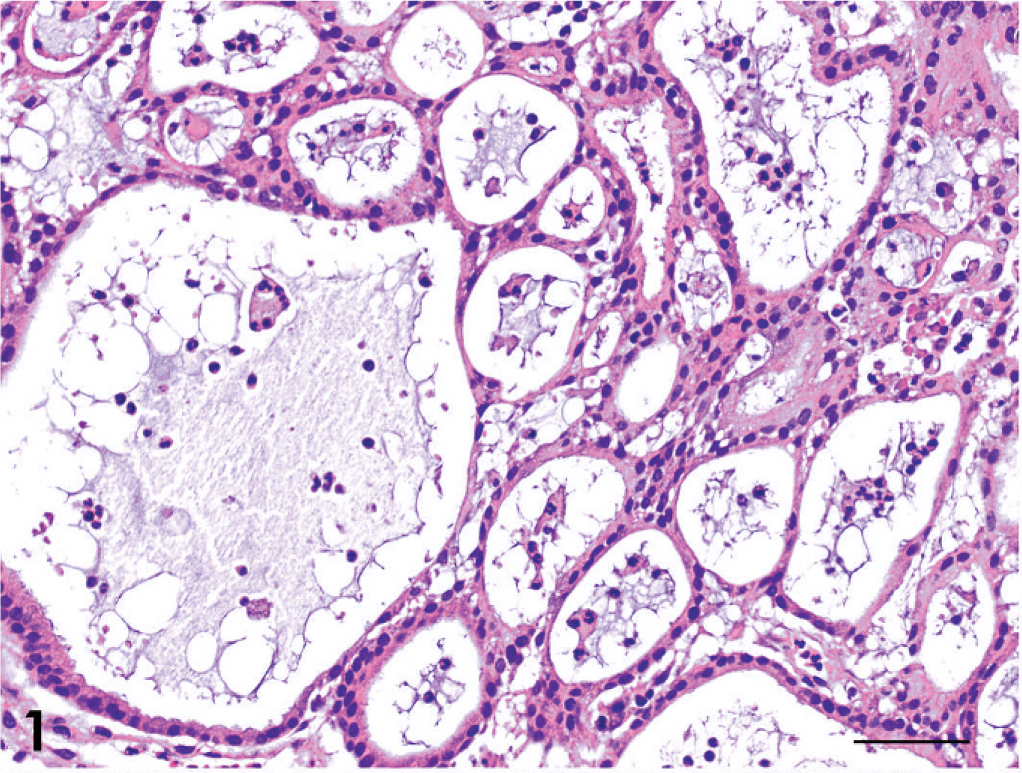
Adenocarcinoma; dog No. 4. Tubular pattern. HE. Bar = 50 μm.
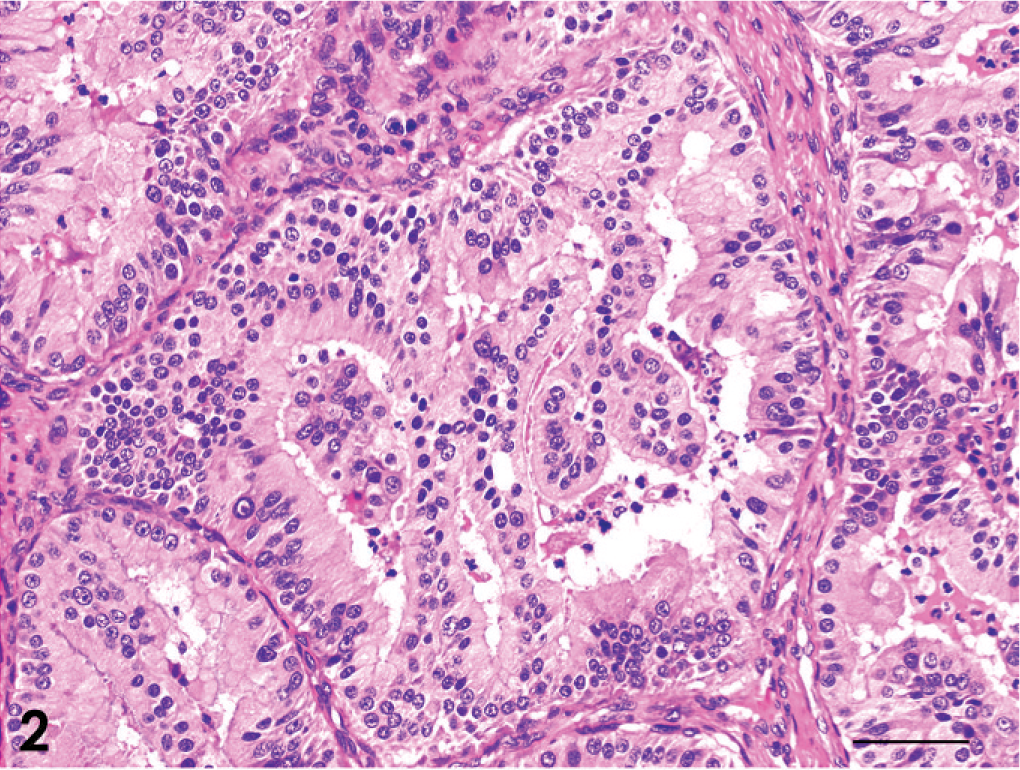
Adenocarcinoma; dog No. 8. Tubulopapillary pattern. HE. Bar = 50 μm.
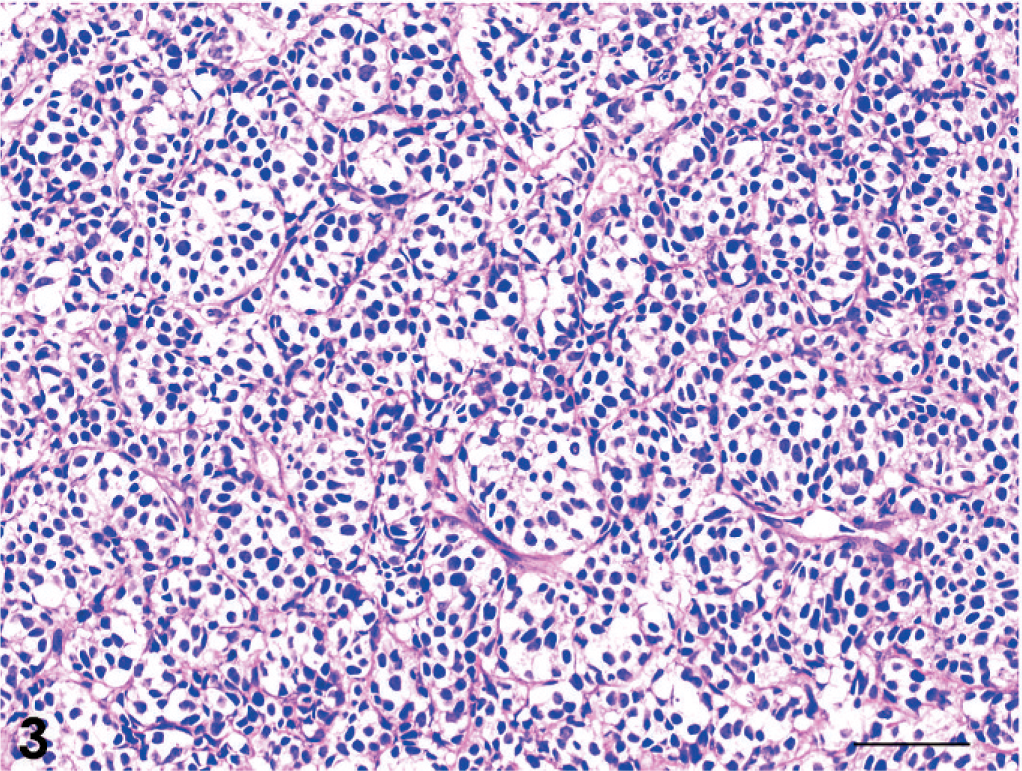
Adenocarcinoma; dog No. 13. Acinar pattern. HE. Bar = 50 μm.
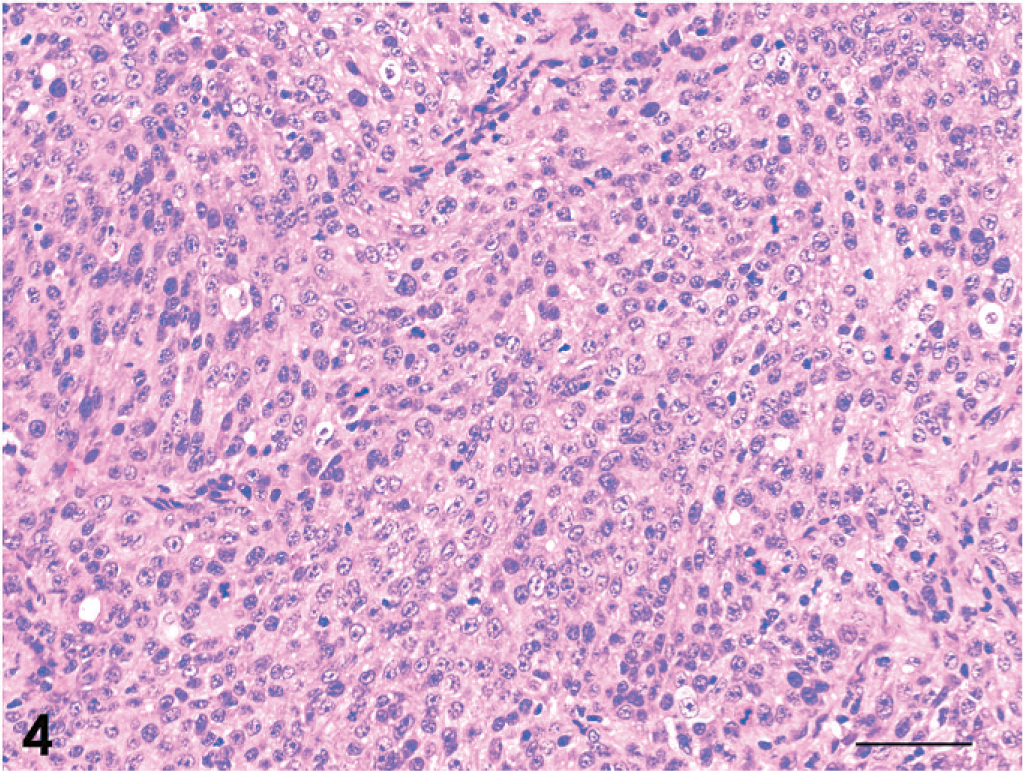
Adenocarcinoma; dog No. 9. Solid pattern. HE. Bar = 50 μm.
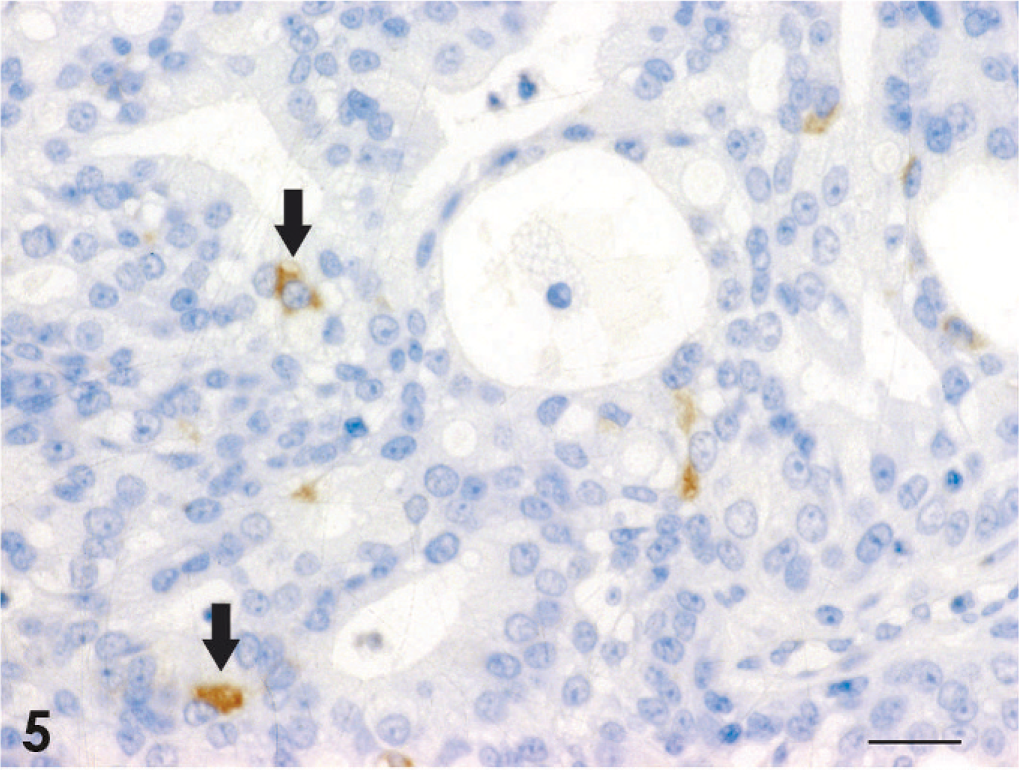
Adenocarcinoma; dog No. 2. Scattered Syn-positive tumor cells are observed (arrows). Universal immunoenzyme polymer method, DAB Chromogen, hematoxylin counterstain. Bar = 20 μm.
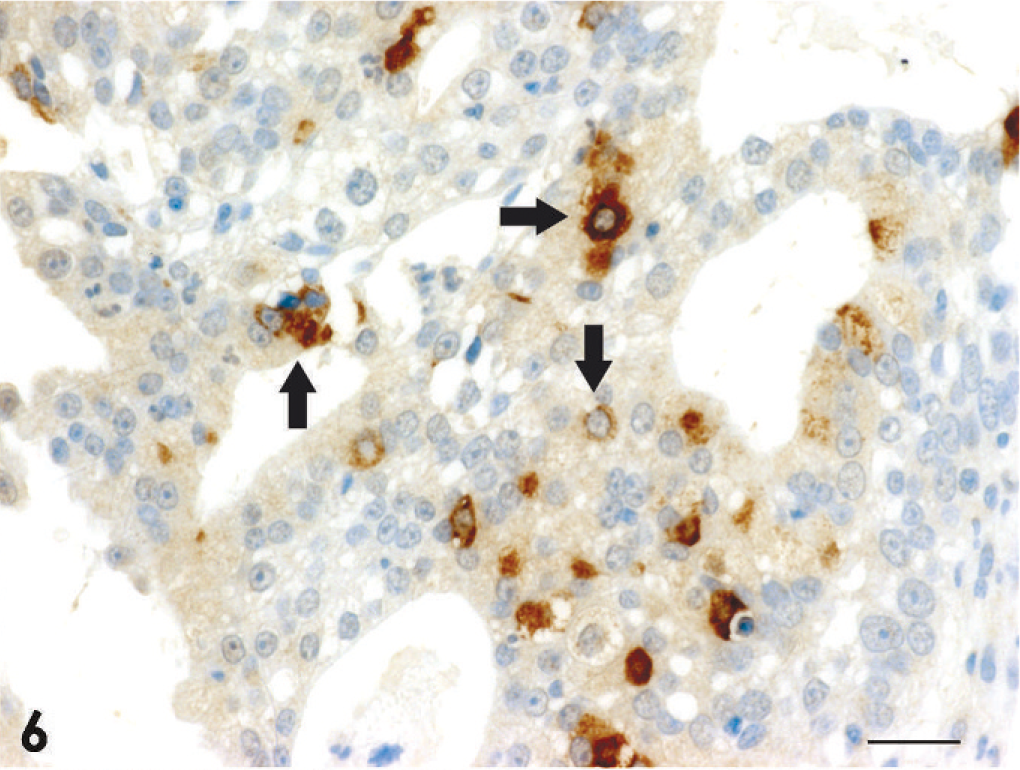
Adenocarcinoma; dog No. 2. Scattered CgA-positive tumor cells are observed (arrows). Universal immunoenzyme polymer method, DAB Chromogen, hematoxylin counterstain. Bar = 20 μm.
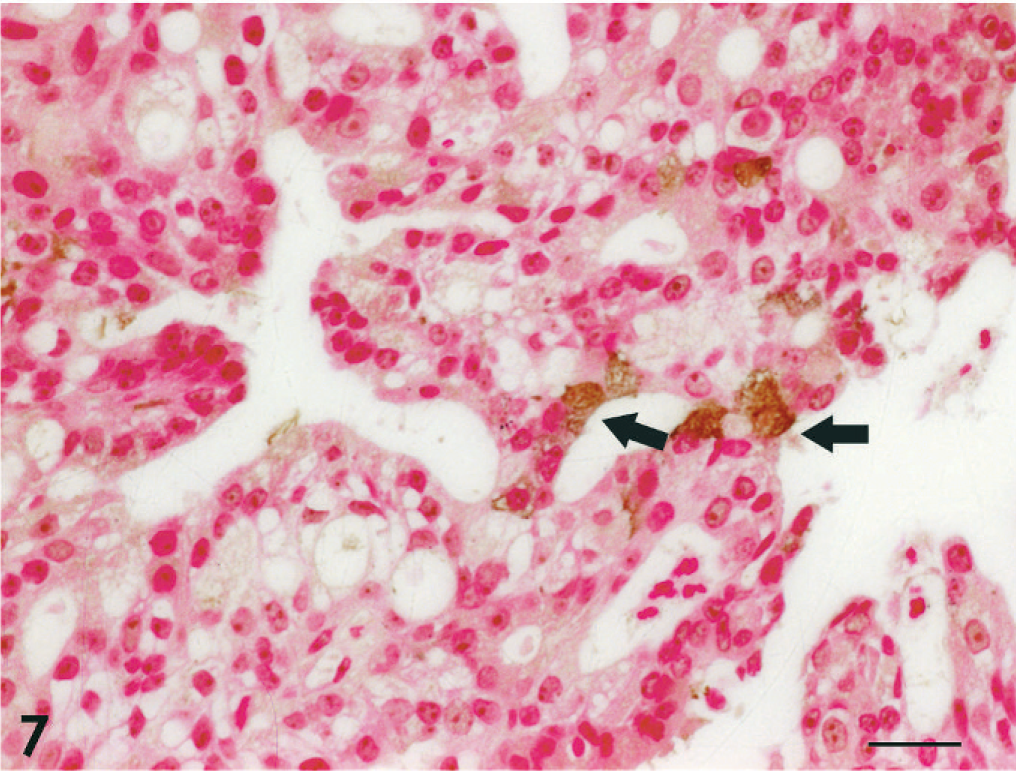
Adenocarcinoma; dog No. 2. Scattered argyophilic tumor cells are observed (arrows). Grimelius' stain with Kernechtrot counterstain. Bar = 20 μm.
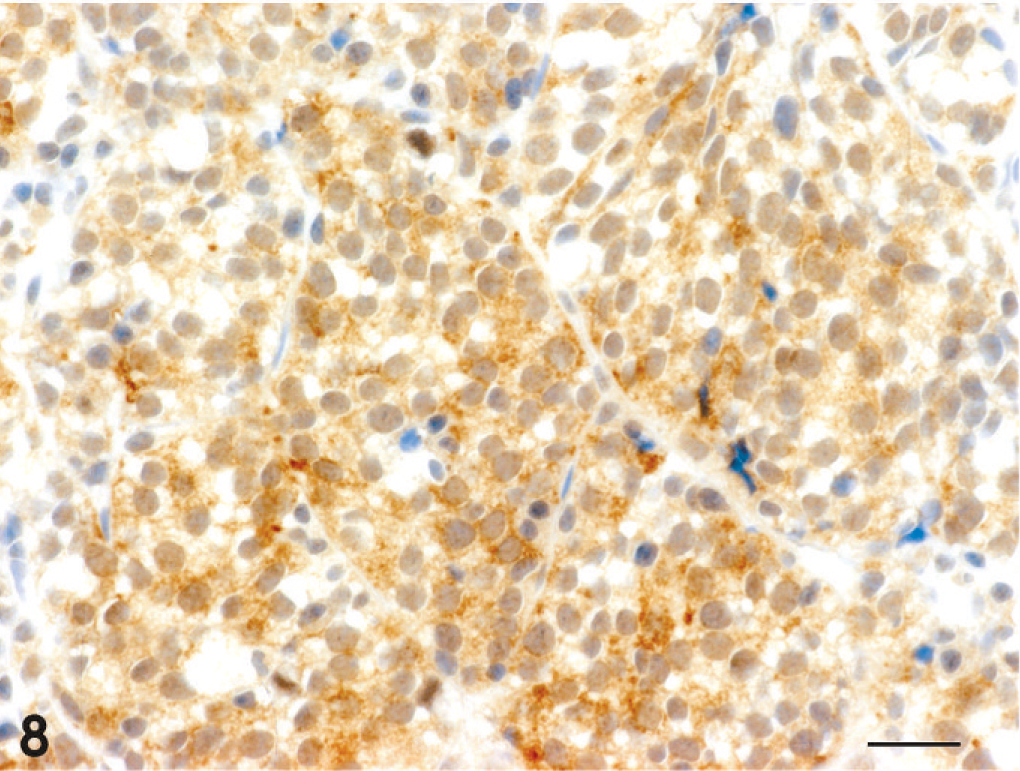
Adenocarcinoma; dog No. 13. Diffuse CgA-positive tumor cells are observed in a region with an acinar pattern. Universal immunoenzyme polymer method, DAB Chromogen, hematoxylin counterstain. Bar = 20 μm.
Footnotes
Acknowledgement
The authors thank Mrs. Hiromi Yanagawa for technical support.