Abstract
Nasal polyps in dogs are space-occupying soft-tissue masses that have been encountered concurrently with intranasal neoplasia in surgical biopsy specimens. The proportion of nasal polyp co-occurrence with primary nasal tumors was examined, and follow-up biopsies on dogs initially diagnosed with nasal polyp were reviewed. Histologic sections from 321 cases of intranasal neoplasia and 50 cases of nasal polyp from 2004 to 2017 were reviewed. Of the 321 cases of intranasal neoplasia, 51 (16%) had concurrent nasal polyps, and most of these (47/51) had intranasal carcinoma. Twenty-five of the 50 dogs with a primary diagnosis of nasal polyp were rebiopsied, and the diagnoses in these subsequent biopsies were nasal polyp in 15, malignant neoplasm in 9, and intranasal nematode in 1. Nasal polyps occurred frequently in conjunction with nasal carcinoma. In dogs with a diagnosis of nasal polyp, repeat biopsy to reveal possible neoplasia is warranted.
Nasal polyps in dogs occur as space-occupying soft-tissue masses that can be associated with turbinate destruction and extension through the nasal septum on diagnostic imaging. 4 Clinical signs in dogs with nasal polyps include sneezing, nasal discharge, epistaxis, and stertor. 4 These clinical signs and diagnostic imaging findings overlap with other intranasal diseases including neoplasia, inflammation, and fungal infection. Histopathology is required for a definitive diagnosis. 8,11 The cause of nasal polyps in dogs is not known, but this pattern may represent a nonspecific reaction pattern to injury in the nasal cavity. A lack of eosinophils and mast cells suggests these polyps are not related to allergy, as nasal polyps can be in humans. 4 As local edema is the predominant feature, a disruption in lymphatic drainage may play an important role.
The diagnostic biopsy service at the University of Pennsylvania School of Veterinary Medicine has seen several dogs initially diagnosed with nasal polyps but then diagnosed with nasal malignancy after a subsequent biopsy. Although rhinoscopy and rhinoscopy-assisted mucosal biopsy for inflammatory and neoplastic lesions in dogs is reportedly successful in 83% to 91% of cases, 6,10 repeated biopsies are frequently required for diagnosis of nasal cavity neoplasms. 3 In dogs in which there was a strong clinical suspicion of nasal neoplasia, a diagnosis of malignancy was made in 69% of cases that were rebiopsied after an initial benign diagnosis. 3
Clinical interpretation of benign nasal biopsy results can be challenging because various nasal diseases present with similar clinical signs. The presence of a mass lesion on magnetic resonance imaging or computed tomography is not specific for nasal neoplasia. 8 Sampling techniques including nasal hydropulsion and blind or rhinoscopy-assisted mucosal biopsy may either sample unaffected tissue or fail to sample pathologic lesions. The histologic pattern of canine nasal polyps may resemble the typical peritumoral nasal mucosa. In addition, other benign proliferations with very similar histologic features to canine nasal polyp, including respiratory epithelial adenomatoid hamartoma (REAH), chondroosseous REAH, and angiofibroma, have been described in the canine nasal cavity. 1,5,7 The aims of this study were (1) to identify the proportion of primary nasal neoplasms with concurrent nasal polyps and (2) in patients with a primary diagnosis of nasal polyp, to describe diagnoses made on subsequent biopsies.
Electronic medical records of nasal cavity biopsy samples from dogs submitted to the Penn Vet Diagnostic Laboratory between 2004 and 2017 were reviewed. Most samples were obtained via the nostril (eg, blind or rhinoscopy assisted), and approximately 5% of samples were obtained via rhinotomy. All nasal neoplasms and nasal polyps were identified using the following search terms: carcinoma, chondrosarcoma, osteosarcoma, hemangiosarcoma, lymphoma, histiocytic sarcoma, sarcoma, malignant neoplasia, malignant neoplasm, melanoma, malignant melanoma, multilobular tumor of bone, and polyp. Cases were included in the study if either archived slides or formalin-fixed paraffin-embedded tissues were available for review. Based on medical record and slide review, cases with tumors obviously arising from the nasal planum, paranasal sinus, maxillary bone, and oral cavity were excluded. For all cases with an initial diagnosis of nasal polyp, the biopsy database was searched for any follow-up biopsies.
All slides were reviewed for the hallmark histologic features of canine intranasal polyp, namely, a columnar to pseudostratified ciliated epithelium overlying a markedly edematous submucosal stroma with mucin accumulation, lymphangiectasia, and mild mixed inflammation. 4 A subset of the intranasal neoplasms included the polyp findings in the diagnosis or description of the original pathology report. All slides were reviewed for consistency and to identify cases in which only the neoplasm was reported.
The study population consisted of 220 cases of nasal carcinoma, 101 other nasal neoplasms, and 50 cases with a primary diagnosis of nasal polyp. Carcinomas outnumbered other tumors at a ratio of 69% to 31%, similar to the reported occurrence of tumors in the canine nasal cavity. 9 Typical specimens were several fragments of tissue ranging in size from 4 to 10 mm in diameter.
Specific diagnoses for the carcinomas included 89 adenocarcinomas, 54 transitional carcinomas, 19 undifferentiated carcinomas, 19 squamous cell carcinomas, 1 acinic cell carcinoma, 1 adenosquamous carcinoma, 1 adenoid cystic carcinoma, and 36 carcinomas not otherwise specified. Nonepithelial tumors were as follows: 40 chondrosarcomas, 21 osteosarcomas, 24 spindle cell sarcomas, 5 lymphomas, 5 hemangiosarcomas, 2 malignant melanomas, 1 leiomyosarcomas, 1 multilobular tumor of bone, 1 esthesioneuroblastoma, and 1 transmissible venereal tumor.
The median age at diagnosis for epithelial neoplasms was 11 years (range: 1–17 years), for nonepithelial neoplasms 9 years (range: 0–14 years), and for nasal polyps 10 years (range: 4–17 years; Table 1). The most commonly submitted breed in each category was mixed breed, followed by Golden Retriever and Labrador Retriever. The male-to-female ratios for the cases of neoplasia, both epithelial and nonepithelial, approached 1.0 (1.16 for nasal carcinoma and 0.95 for all other neoplasms). Nasal polyp diagnoses were made more frequently in male dogs, with a male-to-female ratio of 1.42.
Demographic Data for the Study Population and Polyp Co-occurrence.
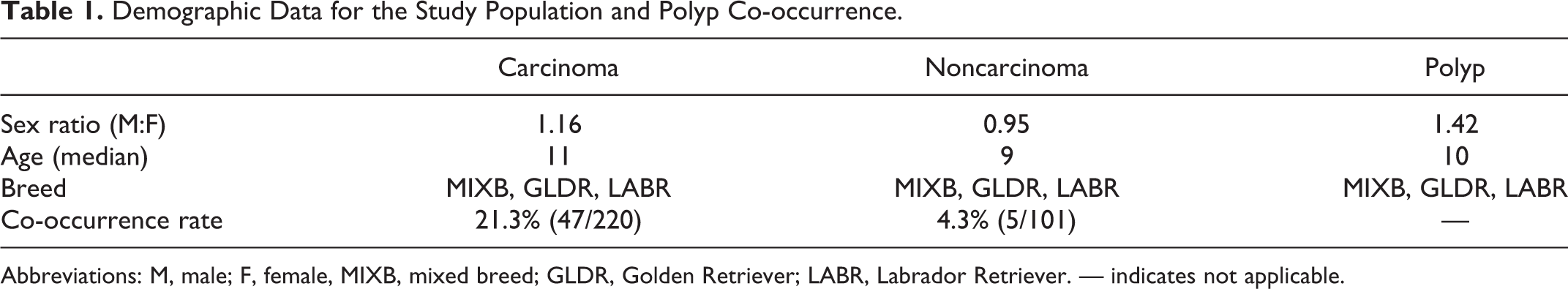
Abbreviations: M, male; F, female, MIXB, mixed breed; GLDR, Golden Retriever; LABR, Labrador Retriever. — indicates not applicable.
Concurrent nasal polyp was found in 47 of 220 (21.4%) cases of nasal carcinoma. The median age of dogs with carcinoma and polyp was 11 years, with a male-to-female ratio of 1.14. Concurrent nasal polyp was found in 5 of 101 (5.0%) of nonepithelial nasal neoplasms. The median age of dogs with noncarcinoma neoplasms and polyp was 10, with a male-to-female ratio of 3. The noncarcinoma neoplasms with concurrent polyp included chondrosarcoma (2), leiomyosarcoma (1), and spindle cell sarcoma (1).
A primary diagnosis of nasal polyp was made in 50 cases. A second biopsy was submitted in 25 of these cases, and the diagnoses on the second biopsy were nasal polyp (15/25, 60%), carcinoma (6/25, 24%), chondrosarcoma (2/25, 8%), osteosarcoma (1/25, 4%), and nematode parasite (presumptive Eucoleus boehmi; 1/25, 4%). In this cohort of dogs with nasal disease, we found that nasal polyps frequently occurred concurrently with nasal carcinomas and rarely with other neoplasms. Thus, repeated biopsies of nasal polyps most frequently resulted in a concordant diagnosis of nasal polyp again; however, in 9 of 26 cases (35%) with an initial diagnosis of nasal polyp, the second biopsy revealed a malignancy (Figs. 1–4).
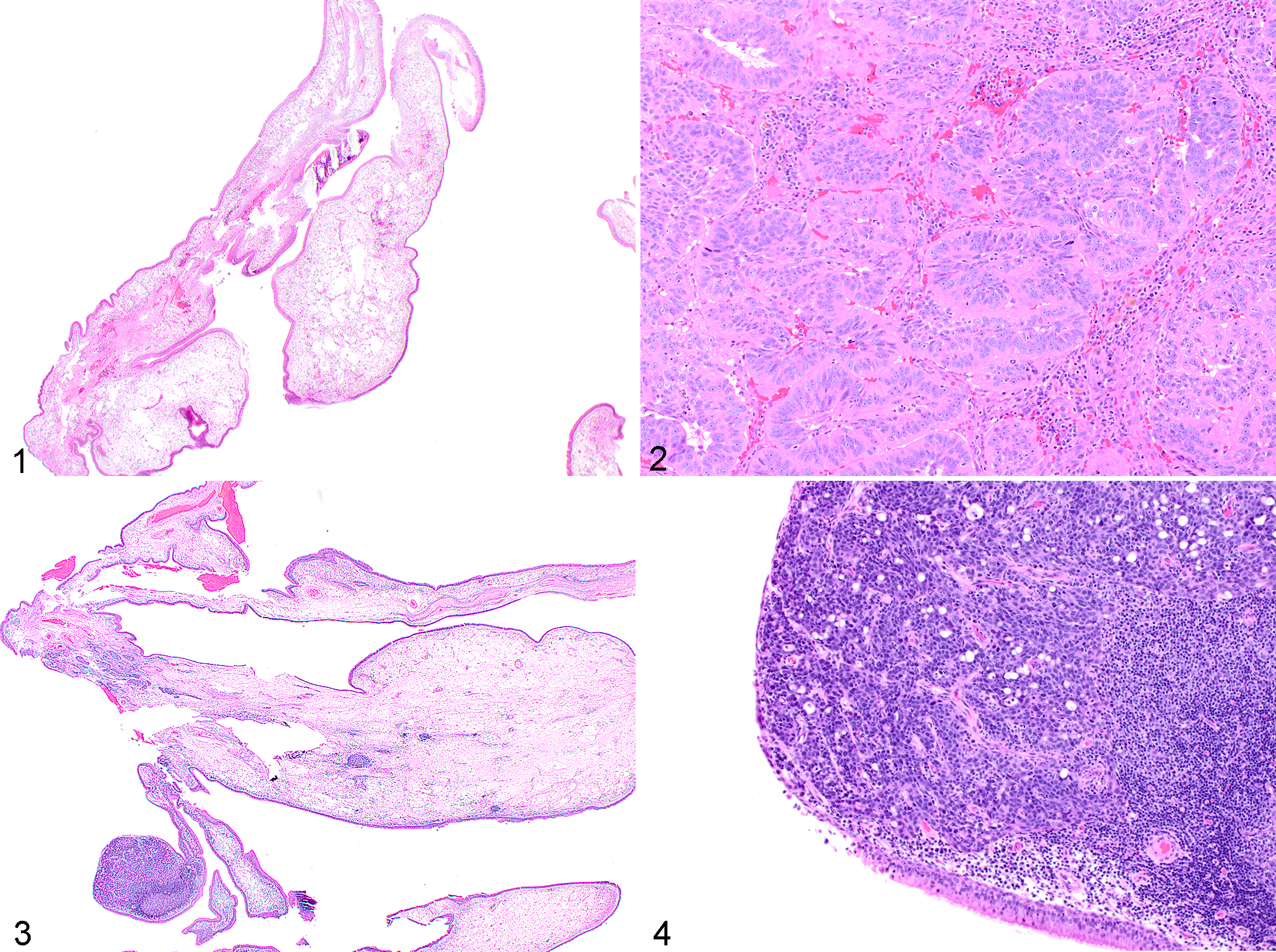
Nasal polyp with subsequent diagnosis of adenocarcinoma, nasal tissue, dog. Hematoxylin and eosin.
Accurately diagnosing nasal cavity masses is challenging. A study of 521 preoperative endoscopic biopsies of nasal masses in humans found a 43.7% sensitivity for malignant neoplasms. 2 Although the size of bioptic specimens was not recorded in that study, the low diagnostic yield of nasal biopsy suggests that larger or more specimens would be better. Given the relatively nonspecific histologic appearance of canine nasal polyps, we suggest that this diagnosis be made provisionally with the caveat that the hallmark features of marked mucosal edema with seromucinous gland hyperplasia are commonly encountered alongside malignant nasal neoplasms, most frequently carcinomas. The relationship between nasal malignant neoplasms and nasal polyps is unclear. The authors speculate that nasal carcinomas may obstruct lymphatic drainage, via extramural compression and/or plugging by neoplastic emboli, leading to regional edema and expansion of a nasal turbinate to form a nasal polyp.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985819854438 - Co-occurrence of Nasal Polyps and Neoplasms of the Canine Nasal Cavity
Supplemental Material, DS1_VET_10.1177_0300985819854438 for Co-occurrence of Nasal Polyps and Neoplasms of the Canine Nasal Cavity by James C. Tarrant, David E. Holt and Amy C. Durham in Veterinary Pathology
Footnotes
Acknowledgements
We thank Caroline Solomon, Alea Agrawal, and Jonathan Nagel for technical assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
