Abstract
Feline myeloma-related disorders (MRD) are rare neoplasms of plasma cells. The multistep transformation model of myeloma in humans is based on the premise that plasma cells undergo neoplastic transformation primarily within the intramedullary compartment and that over time they become poorly differentiated and metastasize to extramedullary locations. Historically, diagnostic criteria used for human multiple myeloma have been applied to the cat, with the assumption that feline MRD commonly arises in the intramedullary compartment. Our objectives were to describe the features of feline MRD confirmed by cytology, histopathology, histochemistry, and immunohistochemistry and to categorize these tumors. A priori hypotheses were 1) tumor category predicts survival and 2) cats with well-differentiated tumors commonly have extramedullary involvement in contrast to human myeloma patients. This multicenter, retrospective study identified 26 MRD cases. There was good agreement between histopathologic and cytologic tumor categorization. Histochemistry and immunohistochemistry were shown to be valuable adjunct tests in the diagnosis of MRD. Cats with well-differentiated tumors had increased median survival relative to those with poorly differentiated tumors (254 versus 14 days). We have reported that marked extramedullary involvement at initial clinical presentation is significantly more common in the cat than in human MRD patients. In this study, we demonstrate that cats with well-differentiated tumors more commonly have extramedullary involvement than human myeloma patients with well-differentiated tumors (90% versus 20%, P < 0.0002). These results contrast strongly with the human myeloma model of primary intramedullary neoplastic transformation and suggest that primary extramedullary neoplastic transformation may be more common in feline MRD.
Keywords
Introduction
Myeloma-related disorders (MRD) derive from the neoplastic transformation of a plasma cell or an immunoglobulin (Ig)-secreting B-lymphocyte precursor. MRD are rare in cats, accounting for 0.0012–0.0025% of all feline malignancies reported in 2 case series (
The first description of feline MRD is attributed to Holzworth and Meier (1957), who reported a reticulum cell myeloma in a cat. 33 Since then, a diverse array of sometimes confusing terminology and diagnostic criteria have been used for feline MRD, including multiple myeloma, lymphoma, plasmacytoid lymphosarcoma, plasma cell dyscrasia, plasmacytoma, plasma cell sarcoma, and plasma cell tumor. 18, 20, 30, 51, 52, 62, 77 In a previous report, 49 we classified reported feline MRD in the following diagnostic categories: 1) myeloma (intramedullary or combined intra- and extramedullary involvement), 49 2) cutaneous extramedullary plasmacytoma (CEMP), 49 3) noncutaneous extramedullary plasmacytoma (NCEMP), 49 4) solitary plasmacytoma of bone (SPB), 50 5) IgM (Waldenström's) macroglobulinemia, 45, 77 6) immunoglobulin secreting lymphoma (IgSL), 17 and 7) myeloma cell leukemia. 19, 54, 59
Few publications describe the cytologic or histologic features of feline MRD. Brief cytologic descriptions have been reported in single cases. 32, 39, 59 Many histopathologic reports of feline MRD simply describe myeloma cells as plasma cells. However, other terminology has been used: “plasmacytoid cells” have been reported in single cases of plasmacytoma, 9, 24, 43 and “lympho-plasmacytoid” appearance was described in 2 cases reported as lymphoma (IgSL) 62, 77 and 1 case reported as multiple myeloma. 20 Small to medium-sized lymphocytes were described in 1 case of IgSL. 17 More detailed cytologic and histopathologic descriptions were provided in a series of 15 cats, but that report did not present a tumor morphologic classification system and the diagnoses were not corroborated by immunohistochemistry. 54 The largest histopathologically and immunohistochemically confirmed case series reported thus far comprised 9 cats with cutaneous or mucocutaneous masses. 46 That study did not include details of clinical presentation, internal organ involvement, or outcome. The production of a single Ig type in a plasma cell infiltrate, as demonstrated by immunohistochemistry, is a surrogate indicator of monoclonality and a useful adjunct test in the distinction of neoplastic (myeloma) versus inflammatory (or reactive) plasma cell lesions. 40 Such immunohistochemistry has not been previously reported in cats with bone marrow and/or visceral MRD. 9, 40, 46 A rare form of T-cell/natural killer cell lymphoma with plasmacytoid features (but non-Ig secreting) has been reported in humans 23, 41 and in the dog 56 but is not currently included in the classification of MRD. Examination of CD3 and CD79a immunolabeling (T and B lymphocyte markers, respectively) has not been reported previously in feline MRD.
There is no internationally accepted standard for the morphologic categorization of human MRD, but a key feature of all reported systems is the division into well-differentiated, intermediate-grade or poorly differentiated tumors, depending on the proportions of myeloma cell types from mature, terminally differentiated myeloma cells to poorly differentiated plasmablastic cells. There is generally good agreement between cytologic and histologic categorization of tumors. 73, 79 As early as 1948, it was hypothesized that myeloma cell morphologic features could be correlated with prognosis, 7 and this has been borne out in numerous studies. 6, 12, 25, 79 In a study of 101 human myeloma cases, only 20% of patients with well-differentiated tumors had extramedullary metastases at necropsy, whereas a significantly higher proportion (69%) of those with poorly differentiated tumors had extramedullary involvement and shorter survival from time of diagnosis. 79
We have previously reported that marked extramedullary involvement at initial clinical presentation is significantly more common in the cat than in human patients (67% versus <5%,
Further aims of this study were 1) to describe the cytologic, histopathologic, histochemical, and immunohistochemical features of feline MRD and to morphologically categorize these tumors, 2) to examine the agreement between histopathologic and cytologic findings, and 3) to examine, by multivariate analysis, whether other selected variables (tumor location, CD79a+, presence of mitoses, treatment category, and gender) had an effect on survival.
Materials and Methods
Case Selection
Cases were retrieved by searching the databases and/or making direct contact with veterinarians of all major public and commercial UK referral centers and laboratories (see acknowledgements). A spectrum of disciplines (oncology, pathology, clinical pathology, internal medicine, ophthalmology, soft-tissue surgery, orthopedics, dermatology, and neurology) was included in an attempt to avoid selection bias. Search terms were as follows: plasma cell neoplasia, myeloma, plasmacytoma, macroglobulinemia, hyperglobulinemia, gammopathy, paraprotein. Cases were derived from 1990 to 2004. Permission for the use of the clinical records and patient material was obtained from the contributing veterinarians and their clients.
Cytology, Histopathology, Histochemistry, and Immunohistochemistry
Samples comprised antemortem cytologic and biopsy specimens, as well as postmortem material. Confirmation of the original report of a MRD was achieved by review of:
Cytology: Preparations were optimally stained with May-Grünwald-Giemsa, although some were received prestained with other Romanovsky stains (e.g., Rapi-Diff II, Bios Europe Ltd., West Pimbo, UK).
Histopathology: 4-μm–thick sections of formalin-fixed, paraffin-embedded tissues were stained with hematoxylin and eosin (HE).
Ancillary histochemistry and immunohistochemical analysis: If the diagnosis of an MRD was in question, we carried out further histochemistry and/or immunohistochemistry prior to inclusion or rejection: toluidine blue, astra blue, lysozyme (polyclonal rabbit anti-human lysozyme, clone EC3.2.1.17, DakoCytomation Ltd. [DC], Ely, UK); fascin (monoclonal mouse anti-human fascin, clone 55K-2, DC); MHC II (monoclonal mouse anti-human HLA-DR alpha-chain, clone: TAL.1B5, DC); Melan A (monoclonal mouse anti-human Melan A, clone A103, DC); cytokeratin (monoclonal mouse anti-human cytokeratin, clone MNF116, DC); and BLA.36 (monoclonal mouse anti-human B lymphocyte antigen 36kD, clone A27-42, DC) according to standard or manufacturers' techniques.
Immunoglobulin heavy- and light-chain immunohistochemical labeling: 2-μm–thick sections of formalin-fixed, paraffin-embedded tissues were stained for α, γ, and μ heavy chains and λ and κ light chains in the presence of controls (Table 1) according to standard or manufacturers' techniques. In a subjective assessment of representative sections, case specimens were considered positive if the majority of the neoplastic population exhibited cytoplasmic labeling.
Details of epitope retrieval, anti-immunoglobulin reagents, and controls for immunohistochemical labeling of myeloma-related disorders.
All cytologic specimens were reviewed by a panel of cytologists (JA, EJV, RMP, PJM). A cytologic diagnosis in bone marrow aspirates was achieved if representative slides contained >20% myeloma cells as a proportion of the total number of myeloid and lymphoid cells examined. Where there was discordance between cytologic and histopathologic findings in the same animal, the histopathologic and immunohistochemical categorization was accepted on the basis that evaluation of repeat sections from a larger tissue sample was more likely to be representative than aspirated cells.
All histopathologic and immunohistochemical specimens were reviewed by a panel of histopathologists (SH, KCS, TJS, PEM, DF) who were blinded to the cytology results. A histopathologic diagnosis from bone marrow core biopsies was achieved if representative bone core sections contained >20% myeloma cell infiltration and myeloma cell clustering. Sections displaying single Ig light-chain immunolabeling were regarded as consistent with a diagnosis of MRD, whereas sections displaying mixed κ and λ light chains were diagnosed as reactive plasma cell lesions and were excluded from further analysis. Histopathologic specimens that did not label with anti-Ig antibodies were still included in the case series on the basis of unequivocal cytologic findings and/or histopathologic, ancillary histochemical, and immunohistochemical findings.
All confirmed MRD histopathologic specimens were also examined for expression of CD79a (monoclonal mouse anti-human CD79αcy, clone HM57, DC) and CD3 (monoclonal mouse anti-human CD3, clone F7.2.38, DC) in the presence of controls according to standard techniques. Where HE sections revealed material consistent with amyloid deposition, the presence of Ig light-chain associated amyloid (AL) was confirmed by examination of Congo red-stained, permanganate oxidized sections under polarized light. Mitotic rate (average of 10 fields at ×400 total magnification) was assessed in histopathologic sections only.
Morphologic Categorization of Feline MRD
Cytologic and histopathologic sections were categorized according to an adaptation of the method described by Wutke and others.
79
Myeloma cells were classified as mature myeloma cells, proplasmacytes, plasmablasts, giant myeloma cells, or lymphoma cells (Table 2, Figs. 1–8). Other illustrations of these cells and further information can be found at
Cellular classification of myeloma cells.

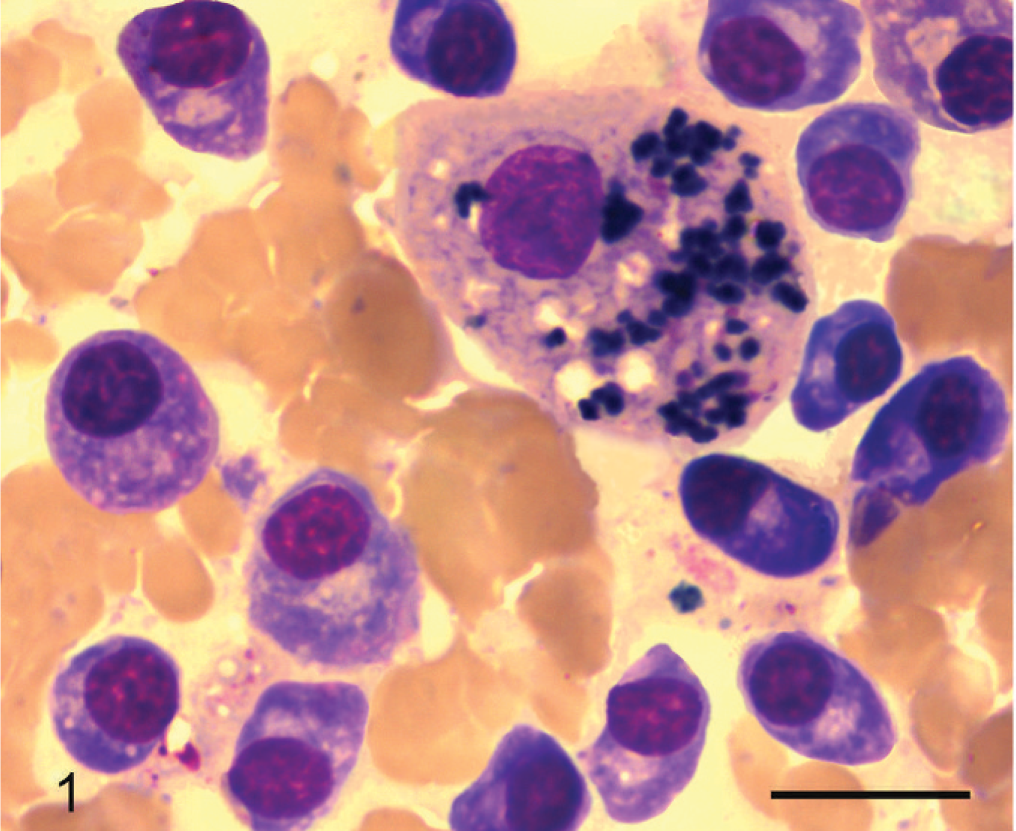
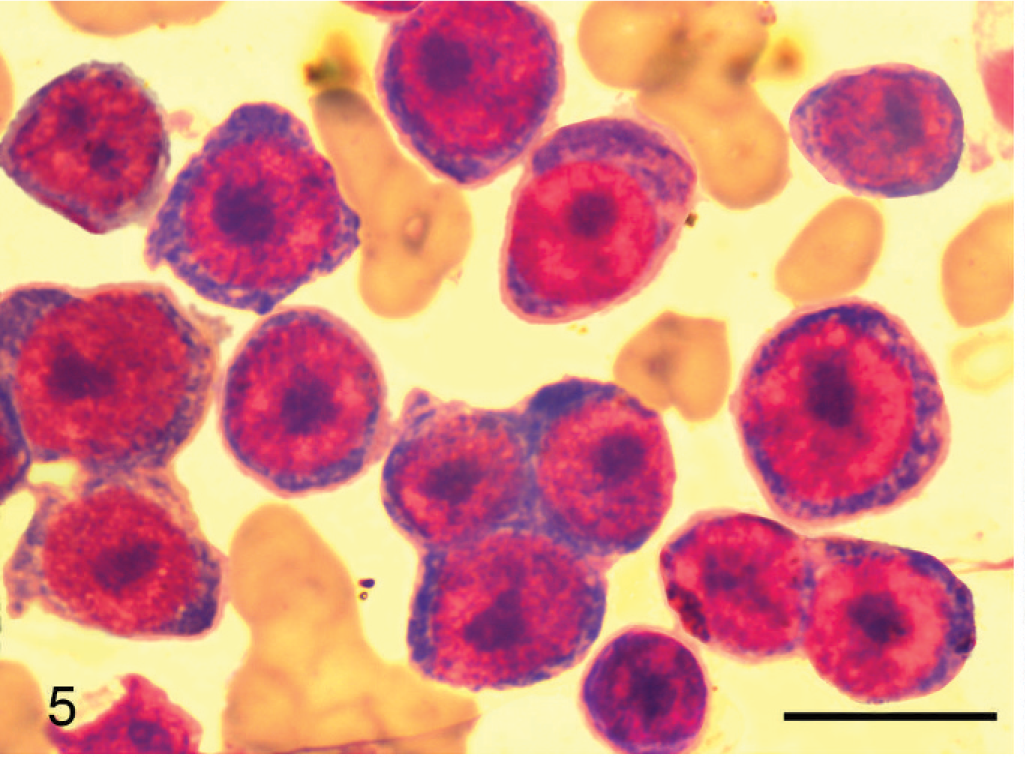
Liver; cat No. 1. Mature plasma cells; cytologic appearance. Mature myeloma cells (classical, terminally differentiated neoplastic plasma cells) are clustered around a hepatocyte. They have an eccentrically located round-to-oval nucleus that is typically 1–1.5 times the diameter of a red blood cell. There is minimal anisokaryosis. The nucleus has a dense chromatin pattern and no nucleoli. May-Grünwald-Giemsa stain. Bar = 10 μm.
Lymphoma (IgSL) could be included as a MRD if accompanied by hyperglobulinemia (total serum globulin >52 g/L) attributable to a myeloma (M) proteinemia, as evidenced by serum protein electrophoresis (SPE). All MRD were then categorized according to the proportion of plasmablasts as follows: 1) well-differentiated MRD, 2) intermediate-grade MRD, and 3) poorly differentiated MRD (Table 3).
Categorization of myeloma-related disorders (MRD) by proportion of plasmablasts.

Statistical Analysis
To test the first hypothesis, the association between tumor morphologic category and survival was examined by both 1) log rank analysis of Kaplan-Meier survival curves, followed by Holm-Bonferroni corrections for multiple comparisons
44
(significance was set at
In the same model, Cox proportional hazards analysis was used to assess the association between other independent variables and survival (censored and all-cause mortality). Because of the small sample size, these were selected, dichotomized variables as follows: 1) tumor location (visceral and/or bone marrow versus cutaneous or subcutaneous mass), 2) CD79a expression (versus absence), 3) presence of mitoses (1–10 per field at ×400 total magnification versus absence), 4) treatment category (chemotherapy or surgical excision versus no treatment or glucocorticoids alone), and 5) gender (male and male neutered versus female and female spayed). Forward variable selection with
To test the second hypothesis, the 2-sided
Results
Reports for 56 cases of feline MRD were obtained. The original reports from the referring cytopathologists/histopathologists described myeloma cell, plasmacytoid cell, or lymphoid neoplasms but provided no specific MRD morphologic classification or correlation of morphologic features with prognosis. In 41 cases, we obtained the original cytologic/histopathologic preparations for review and conducted further histochemical and immunohistochemical analysis as previously described. Four cases with both κ and λ light-chain immunolabeling were excluded from further analysis because these were interpreted as reactive (rather than neoplastic) plasma cell accumulations. An additional 10 cases were found to have been misdiagnosed as MRD when ancillary histochemical and immunohistochemical analyses supported diagnosis of other round cell neoplasms (4 mast cell tumors, 3 histiocytic tumors, 2 poorly differentiated round cell tumors, and 1 lymphoma [non-Ig secreting]). Twenty-seven cases were provisionally diagnosed as MRD. Of these, 7 had both cytologic and histopathologic specimens, 7 had cytologic slides only, and 13 had histopathologic specimens only. Subsequently, 1 case with discordant cytologic versus histologic/immunohistochemical results was discarded (see below). Finally, 26 cats had confirmed MRD. Signalment, historical, clinical, clinicopathologic, and imaging features, as well as response to treatment for 24 of these confirmed cases, have been reported elsewhere. 49
Site Distribution of Tumors
Distribution of the tumors by organ system is given in Table 4. In total, 21 cats had extramedullary involvement and 10 cats had intramedullary involvement when assessed at any time point from presentation to death. Cats with abdominal organ or bone marrow involvement did not have cutaneous masses.
System involvement by histopathologic/cytologic demonstration of a myeloma-related disorder (MRD).
Total number of cases in which the tissue of interest was histopathologically/cytologically examined, except for skin and oral masses, in which it was assumed that all 26 cats underwent cutaneous and oral clinical examinations for gross abnormalities. Full postmortem examinations were carried out on 2 cats by a university pathologist and on 5 cats by a general practitioner.
Cytology Versus Histopathology
In 7 cats, concurrently obtained cytologic and histopathologic preparations were available for review. One case cytologically diagnosed as intermediate-grade MRD was histopathologically and immunohistochemically identified as a histiocytic neoplasm and was excluded from further analysis. An examination of 22 specimens (11 paired cytologic and histopathologic specimens) from a variety of tissues from the remaining 6 cats revealed excellent concordance in 11 of 11 pairs with respect to morphologic categorization of the tumor and 10 of 11 pairs with respect to diagnosis of an MRD. Discordance in diagnosis occurred in 1 case in which a marrow core biopsy specimen was diagnosed as MRD, but a marrow aspirate was negative for MRD. In this animal, concurrent paired sampling of other organs resulted in cytologic and histopathologic agreement in the diagnosis of a MRD.
Overall, 7 cases were diagnosed as MRD on the basis of cytology alone. Adjunctive clinicopathologic evidence for a diagnosis of MRD was present in the form of a M-proteinemia in 6 of 6 cases examined by SPE. Only one case lacked supportive evidence for a diagnosis of MRD, but that cat had >80% bone marrow infiltration with predominance of proplasmacytes and plasmablasts.
Morphologic Classification
All tumors contained a mixture of myeloma cell types (Table 2). Tumors were classified according to the criteria outlined in Table 3. Most cats in this series had well-differentiated tumors (
Morphologic categorization of myeloma-related disorders (MRD) and number of cases in selected organ systems.
Other Morphologic Features
Occasional giant myeloma cells (Figs. 7, 8) were observed in 2 cutaneous cases (1 intermediate-grade, 1 poorly differentiated) that were confirmed by cytology, histopathology, and immunohistochemistry. Mitoses were seen in histopathologic specimens in 5 cases only and all at <10 per field (at ×400 total magnification). Cases with mitoses were recorded in all tumor morphologic categories for both cutaneous and abdominal masses. Intratumoral light-chain amyloid was confirmed in 3 cats; in 1 of these cases, there was extratumoral light-chain amyloid deposition in the liver and kidneys without concurrent myeloma cell infiltration in these organs.
Skin mass; cat No. 2. Mature plasma cells; histopathologic appearance. Mature myeloma cells are classical, terminally differentiated neoplastic plasma cells. They have an eccentrically located small, round-to-oval nucleus. There is minimal anisokaryosis. The nucleus has a dense chromatin pattern. Nucleoli are not evident. HE. Bar = 50 μm.
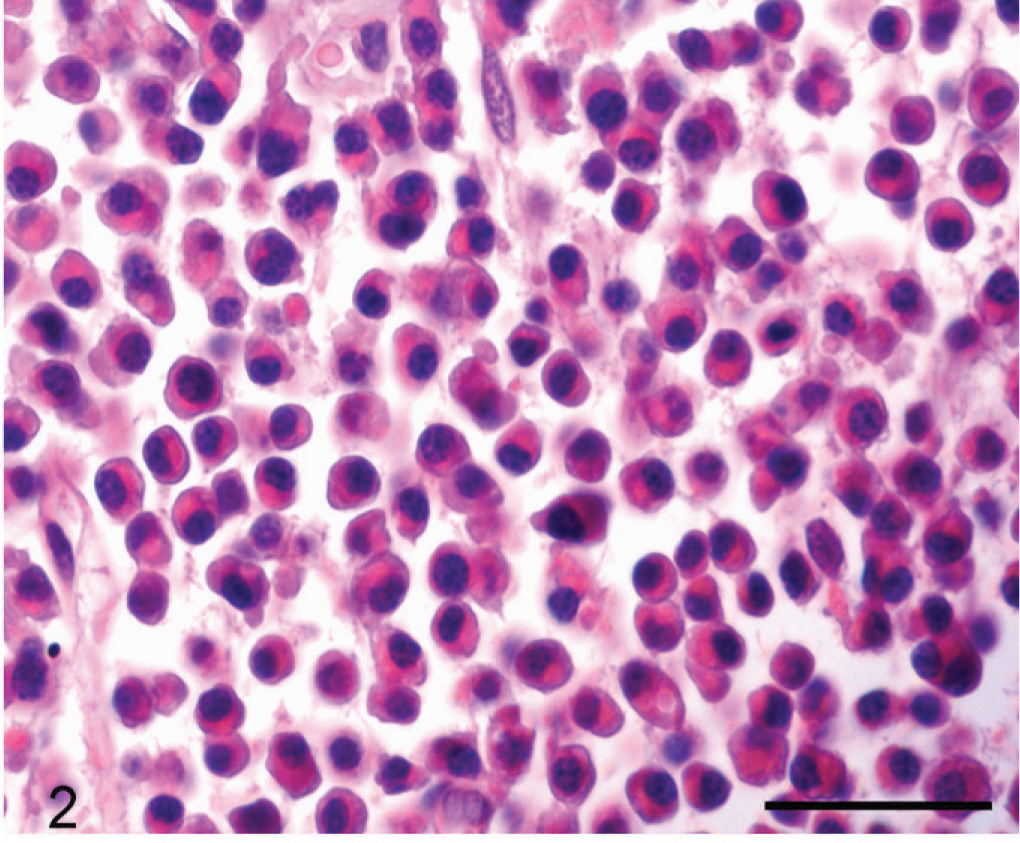
Liver; cat No. 3. Proplasmacytes; cytologic appearance. Proplasmacytes are larger than mature plasma cells, show greater anisocytosis, and display asynchronous nuclear and cytoplasmic maturation. They have an eccentric large nucleus with indistinct or small nucleoli. May-Grünwald-Giemsa stain. Bar = 10 μm.
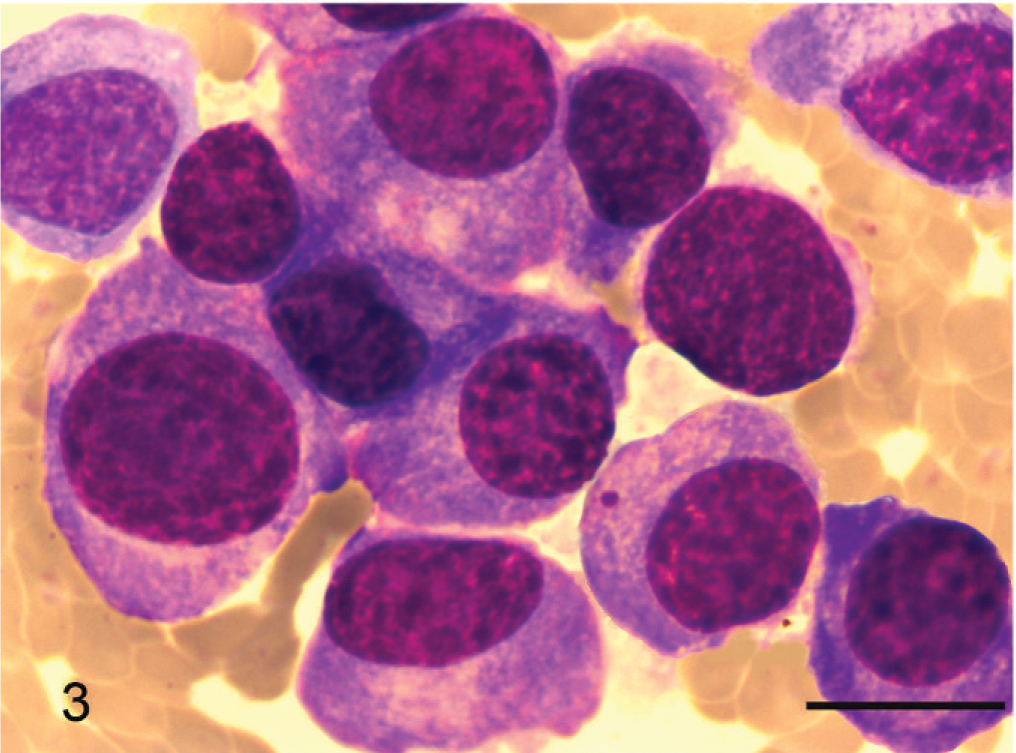
Skin mass; cat No. 4. Proplasmacytes; histopathologic appearance. Proplasmacytes are larger than mature plasma cells with greater anisocytosis and asynchronous nuclear and cytoplasmic maturation. The eccentric nucleus is large with indistinct or small nucleoli. HE. Bar = 50 μm.
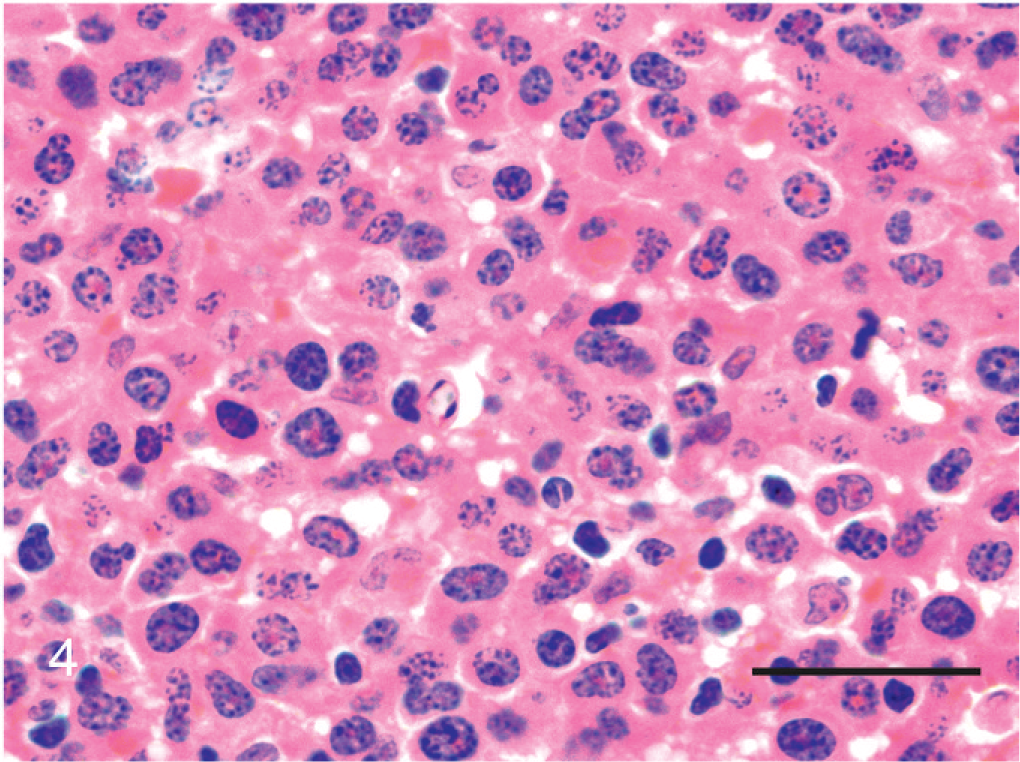
Liver; cat No. 5. Plasmablasts; cytologic appearance. Plasmablasts are larger than mature plasma cells with greater anisocytosis. They have a small rim of basophilic cytoplasm and less distinct or inapparent perinuclear clear zone. They have a high N : C ratio, a large and immature round to ovoid nucleus with anisokaryosis, and ≥1 prominent nucleolus. May-Grünwald-Giemsa stain. Bar = 10 μm.
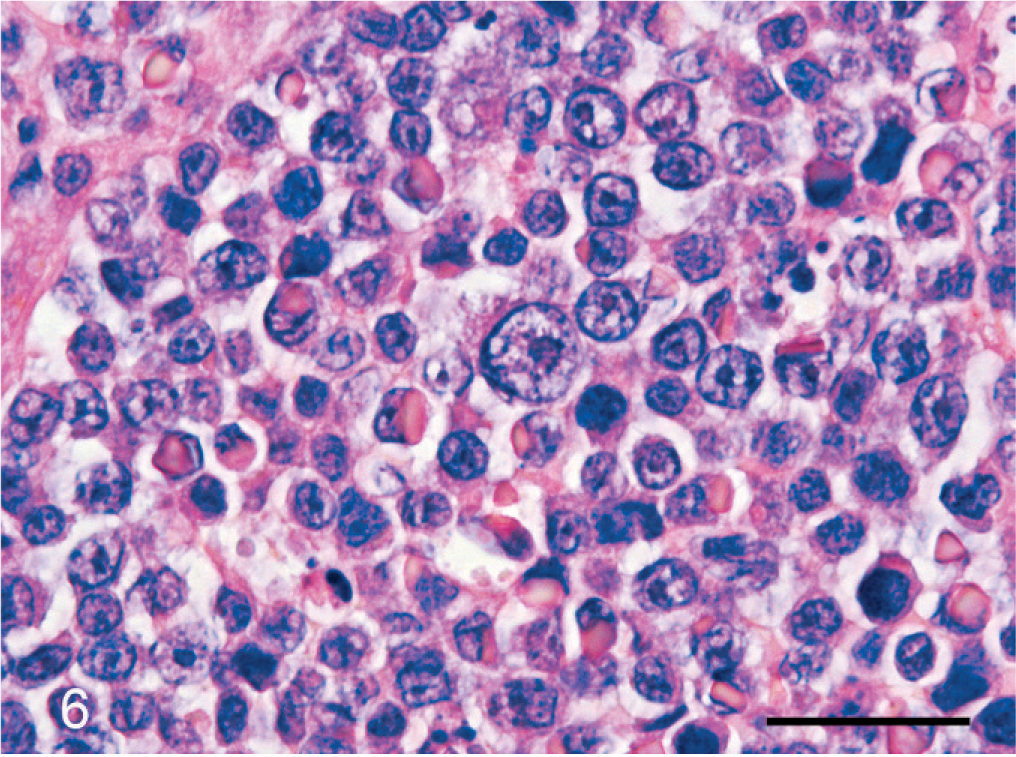
Liver; cat No. 5. Plasmablasts; histopathologic appearance. Plasmablasts are larger than mature plasma cells with greater anisocytosis. They have a narrow rim of cytoplasm and a less distinct or inapparent perinuclear clear zone. Some cells contain large, eosinophilic cytoplasmic inclusions of immunoglobulins (Russell bodies). They have a high N : C ratio, a large and immature round to ovoid eccentric nucleus with anisokaryosis, and ≥1 prominent nucleolus. HE. Bar = 50 μm.
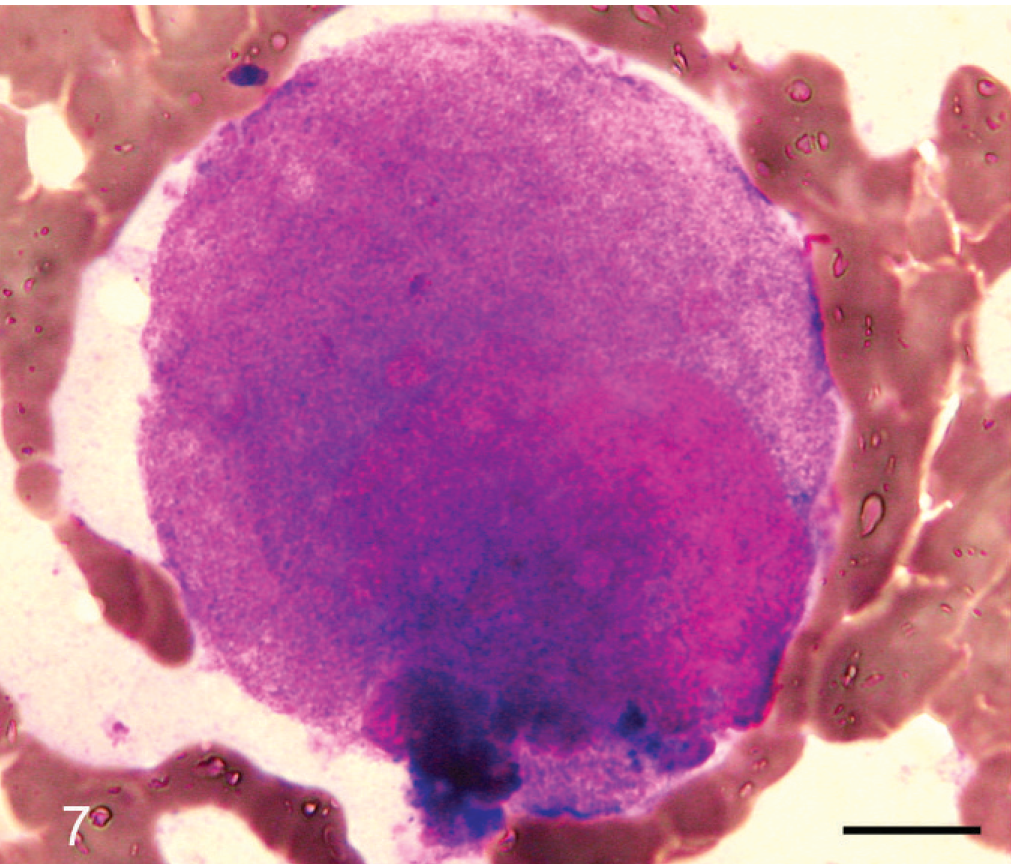
Skin mass; cat No. 6. Giant myeloma cell; cytologic appearance. May-Grünwald-Giemsa stain. Bar = 10 μm.
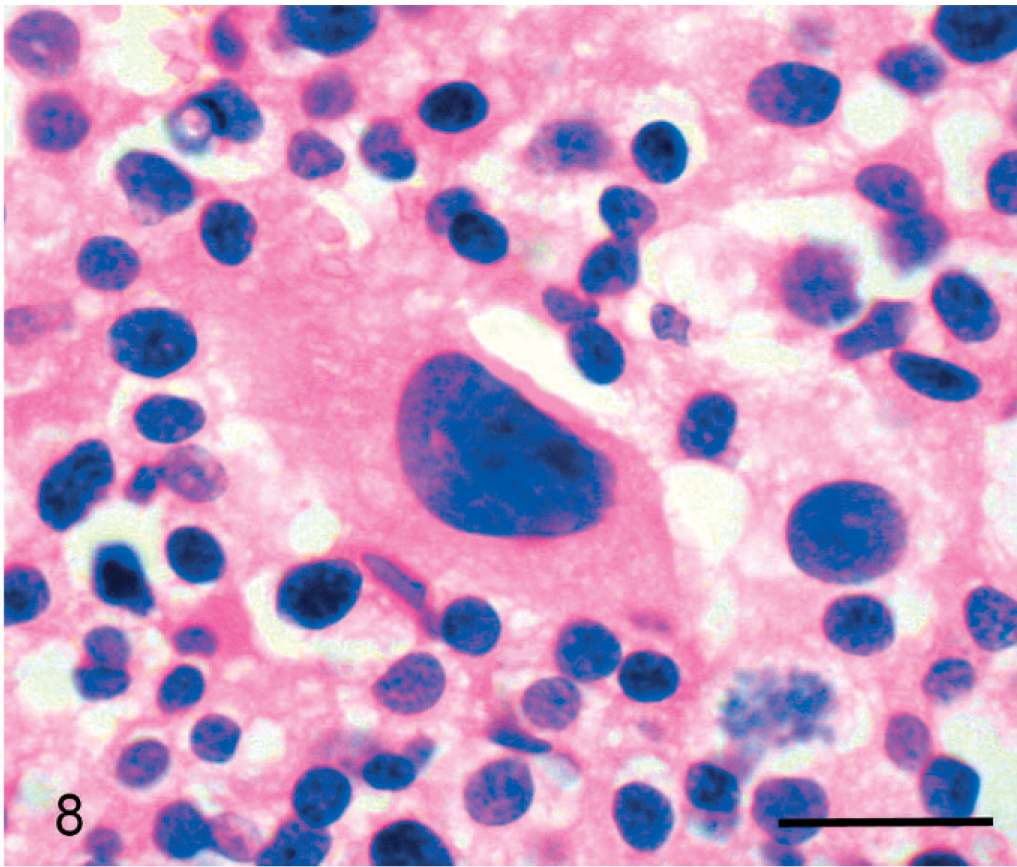
Skin mass; cat No. 7. Giant myeloma cell; histologic appearance. A giant myeloma cell is surrounded by a mixed population of mature myeloma cells and proplasmacytes. HE. Bar = 20 μm.
Immunohistochemistry
Eighteen of the 19 MRD cases for which histopathologic specimens were available had confirmatory Ig immunolabeling. Eighteen cases demonstrated light-chain labeling:
Twelve of 19 MRD cases did not label with either CD79a or CD3. Strong CD79a expression was seen in 6 cases, across all tumor morphologic categories, in both cutaneous and abdominal masses. The single IgSL case showed mixed CD79a and CD3 expression (see discussion). No other specimens showed CD3 labeling.
Hypothesis 1: Tumor Category Correlates with Survival
The MST for poorly differentiated tumors was 14 days versus 254 days for well-differentiated MRD (Fig. 9). Log rank analysis of Kaplan-Meier survival curves revealed a significant difference between poorly differentiated and well-differentiated MRD (
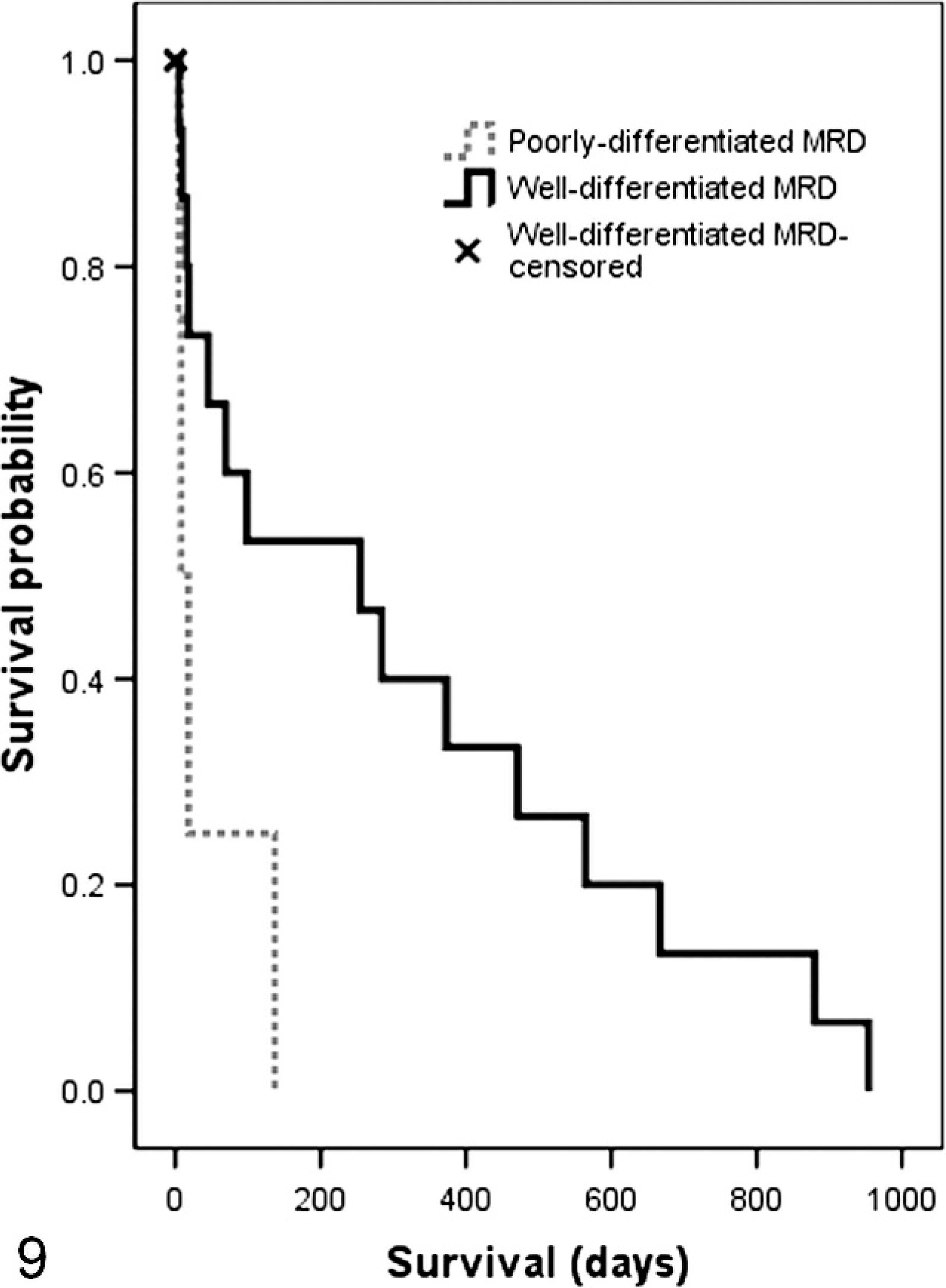
Kaplan-Meier survival curves for poorly differentiated MRD versus well-differentiated MRD.
However, in a multivariate analysis, Cox proportional hazards demonstrated an increased relative risk for dichotomized tumor morphologic categorization with respect to censored survival (RR 8.2, 95% CI 1.8–37.7) (Table 6). Note that although the RR was high, the confidence intervals were broad in this small sample set. When all-cause mortality was examined (i.e., no cases were censored), the RR was lowered and the CI included 1 (Table 6).
Cox proportional hazards analysis: the effect of dichotomized events on survival.

Correlation Between Other Independent Variables and Survival
Cox proportional hazards analysis also revealed that dichotomized treatment category had a significant effect on censored survival (RR 12.9, 95% CI 2.7–60.9) (Table 6). The MSTs for the treatment categories were as follows: none/glucocorticoids alone = 42 days, combination chemotherapy alone = 281 days, surgical excision alone = 562 days. No animals received both surgical excision and chemotherapeutic/glucocorticoid drugs. Log rank analysis of Kaplan-Meier curves confirmed significant differences between those receiving no treatment/glucocorticoids alone and 1) chemotherapy-treated animals (
Hypothesis 2: Cats with Well-Differentiated Tumors Commonly Have Extramedullary Involvement in Contrast to Human Patients
Of the 10 cats that underwent cytologic/histopathologic assessment of both intra- and extramedullary compartments, 9 (90%) had well-differentiated tumors with extramedullary involvement (Table 5). A
Discussion
The histopathologic, histochemical, and immunohistochemical panel review of original specimens in multiple tissues; presence of Ig expression; and absence of CD3 expression provided a high degree of confidence in the final diagnosis of a MRD in this series. Similarly, when only cytologic slides were available (
In total, 15 of 41 cases with an original reported diagnosis of an MRD proved to be either reactive plasma cell lesions or non-MRD neoplasms after review. Only 1 of these cases was incorrectly diagnosed at review as a MRD by cytology, when histopathologic and immunohistochemical studies indicated a diagnosis of histiocytic neoplasia. Immunocytochemistry can be used to confirm the cytologic diagnosis of a MRD, but the technique requires fresh cytologic specimens 71 and was not performed in this retrospective study. Histochemistry and immunolabeling are valuable adjunct diagnostic methods and should be used in conjunction with clinical and morphologic findings in the diagnosis of feline MRD.
Overall, there was good concordance between cytology and histopathology in the morphologic categorization of MRD in this series, as has been reported in human myeloma patients. 73, 79 Discordance between bone marrow aspiration and core biopsy, seen in 1 cat, was most likely attributable to variations in myeloma cell density at different sampling sites and demonstrates the value of performing both procedures concurrently when possible. 73
A number of other notable morphologic and immunohistochemical features were observed. (1) Well-differentiated MRD was the most common tumor morphologic category in this series. In a series of histopathologically (but not immunohistochemically) confirmed feline MRD by Patel and colleagues, 54 only 2 of 12 feline MRD cases were described as having mature plasma cells versus 10 of 12 cases that were described as having atypical plasma cells. That report did not present a tumor morphologic classification system, and the actual proportions of atypical cells required to classify these cases as “atypical plasma cell tumors” were not recorded. 54 (2) Large multinucleated plasma cells have been described in the cat. 54 We report for the first time the presence of immunohistochemically confirmed, giant myeloma cells (segmented nuclei or ≥4 nuclei) in cats (Fig. 8). Giant myeloma cells are a rare but well-described cell type in human MRD. 5, 13, 15 Giant myeloma cells can be mistaken for megakaryocytes but are seen in atypical sites, that is, they are noted outside of the bone marrow or typical sites of extramedullary hematopoesis. They have been associated with intermediate-grade and poorly differentiated tumors in human myeloma, 79 as was the case in the cats in the present study. (3) Amyloid was uncommonly observed; this observation was consistent with the literature. 10, 16, 46, 47, 55, 59, 63, 75 (4) The λ light chain was more commonly expressed by myeloma cells than the κ light chain in this series. This is consistent with the physiologic distribution of light chains in normal feline plasma cells, in which preferential expression of λ is typical. 3 (5) IgG MRD were more commonly encountered than IgA or IgM tumors, consistent with the literature. 16, 17, 21, 45, 46, 77 (6) Only 32% of feline MRD specimens were CD79a+ in this series. CD79a is expressed from early B-cell progenitors onwards and is variably expressed by human myeloma cells. 27
Two cases proved difficult to categorize. In 1 case, the tumor exhibited both IgG and IgA immunolabeling. Dual heavy-chain labeling has not previously been described in cats, although it has been reported in human myeloma cases. 4, 34 Unequivocal single light-chain immunolabeling was used to establish the diagnosis of an MRD in this cat. 60 The second case was an IgSL. Histopathology supported a diagnosis of small lymphocytic lymphoma 74 with hepatic, intestinal, mesenteric lymph node and marrow involvement. Biopsy specimens taken at initial presentation had mixed CD79a and CD3 expression with absence of Ig labeling (even after repeated analysis) despite hyperglobulinemia and M-proteinemia. Nine months later the cat was still M-proteinemic, and additional biopsy specimens again lacked Ig labeling (even after repeated analysis). However, increased numbers of CD79a+ lymphocytes were observed, and CD3 expression was no longer detected. The differential diagnosis included T-cell-rich MRD with progression to B-cell-only MRD and MRD with aberrant CD3+ expression. T-cell-rich B-cell lymphoma (TCRBCL) has been described in cats. 14 Progression of TCRBCL to B-cell lymphoma has been reported in dogs. 2 Aberrant T-cell antigen coexpression has been reported in rare cases of myeloma 61, 68 and B-cell lymphoma in humans. 1
Morphologic categorization has been shown to correlate strongly with prognosis in human myeloma.
12,
69,
79
In particular, poorly differentiated tumors are consistently associated with a lower MST.
25,
26
In the present series, log rank analysis of Kaplan-Meier curves demonstrated a significant difference between poorly differentiated and well-differentiated feline MRD (
In humans, the full morphologic and functional transition of a B-lymphocyte to a normal, terminally differentiated long-lived plasma cell occurs after exiting lymph node and splenic germinal centers and upon maturation in bone marrow. 28 Genetic and phenotypic studies have revealed that myeloma cells originate from these antigen-selected, postgerminal plasma cells and that neoplastic transformation occurs in the intramedullary compartment. 28, 70 A multistep transformation model is hypothesized for the development of human myeloma. 28, 67 Myeloma cells develop karyotypic instability, and it appears that translocations in IgH loci are an early and possibly universal event. 28 The initial clinical manifestation of this transformation is monoclonal gammopathy of unknown significance (MGUS). 28, 80 Over time, myeloma cells accumulate further translocations, gene deletions, and activating mutations; these have been correlated with the onset of clinical disease (typically MGUS to intramedullary myeloma then extramedullary metastases) and a worsening prognosis. 28, 65, 67 Furthermore, studies have clearly established correlations between accumulated cytogenetic aberrations and progressively poorly differentiated myeloma cells. 22, 76 These poorly differentiated cells may arise through dedifferentiation of mature myeloma cells, although there is increasing evidence that they are likely to be derived from a stem cell tumorigenic clone. 8, 48, 53
Historically, feline MRD has been assumed to resemble human myeloma with intramedullary involvement as a key diagnostic feature.
16,
29,
39,
45,
54
However, we and others have recently reported that extramedullary involvement is more common at initial presentation in feline than in human MRD (67% versus <5%,
In this study, we set out to challenge the presumption that feline MRD with extramedullary involvement is simply myeloma that has metastasized from the intramedullary compartment. If feline MRD is similar to human myeloma, then few well-differentiated tumors will have extramedullary involvement and conversely most poorly differentiated tumors will have extramedullary involvement.
65,
67,
79
Instead, 90% of the cats in this study with well-differentiated MRD that underwent cytologic/histopathologic assessment of both intra- and extramedullary compartments had extramedullary involvement versus human patients with well-differentiated tumors, of whom only 20% have extramedullary involvement. A 2-sided
In conclusion, this multicenter study represents the largest peer-reviewed case series of feline MRD, describing the cytologic, histopathologic, histochemical, and immunohistochemical features of these rare tumors. Cytology is a useful complementary tool in the diagnosis and staging of feline MRD. In general, there was good concordance between cytopathologic and histopathologic/immunohistochemical diagnosis. However, there should be an awareness of the potential for discordant results. Our study highlights the value of adjunct histochemistry and immunolabeling in discrimination of MRD from other round cell tumors. Furthermore, Ig light-chain immunolabeling proved useful in discriminating reactive plasma cell lesions from neoplastic myeloma cell lesions. A simple-to-use morphologic categorization system readily identified all categories of feline MRD seen in our series. Cats with well-differentiated tumors had longer survival than those with poorly differentiated tumors (MST 254 days versus 14 days). Cox proportional hazards analysis confirmed that survival was predicted by tumor cytologic or histopathologic categorization. We previously reported that marked extramedullary involvement at initial clinical presentation is significantly more common in cats than in human MRD patients (67% versus <5%,
Footnotes
Acknowledgements
The authors thank the cat owners and colleagues in primary practice for case referral; staff at UK veterinary schools (Cambridge, Edinburgh, Glasgow, Bristol, Liverpool, and London), the Animal Health Trust, Davies Veterinary Specialists, Wey Referrals, Willows Referrals, N. Kent Referrals, N. Downs Referrals, Veterinary Referrals Cancer and Critical Care Center, Godiva Referrals, Stone Lion Veterinary Center, Animal Medical Center, Oxford Cat Practice, Nine Lives Referrals, Finn Pathologists, Cytopath Ltd., Rest Pathology, IDEXX Laboratories Ltd., TDDS Laboratories, Axiom Laboratories, and Leeds Veterinary Laboratory Ltd; J. Miller, Animal Health Trust, for immunohistochemical assistance; Drs. F. Heath and T. J. McKinley (Cambridge Infectious Diseases Consortium, Department of Veterinary Medicine, University of Cambridge) and C. Lamb (Department of Veterinary Clinical Sciences The Royal Veterinary College) for statistical assistance; P. Pion, Veterinary Information Network, for help in establishing The Veterinary Myeloma Website. Financial support from the University of Cambridge Veterinary School Trust and Pet Plan Charitable Trust is gratefully acknowledged. Hills Pet Nutrition Ltd. kindly sponsored P. Mellor's clinical scholarship at the University of Cambridge. Portions of this study were presented at the European Society of Veterinary Clinical Pathology Annual Conference, September 2006 and the Veterinary Cancer Society Annual Conference, November 2007.
