Abstract
To contribute to the existing body of knowledge in the literature on the apparently rare extramedullary plasmacytoma in cats, lymphoid tumors with plasmacytic cellular morphology taken from nine cats were examined. The paraffin-embedded material was investigated by standard hematoxylin and eosin, and special staining techniques (Giemsa, Congo-red, and periodic acid–Schiff reaction). The tumors also were examined immunohistochemically for the presence of immunoglobulin G, immunoglobulin A, immunoglobulin M, immunoglobulin light chains (λ, κ), various amyloid proteins, and FeLV-antigen (p27 protein). An immunoglobulin- producing tumor of plasmacellular origin (extramedullary plasmacytoma [EMP]) could be diagnosed in all cases on the basis of immunohistochemical light-chain expression. All but one of the neoplasms occurred in the skin of older, predominantly male cats. As in humans and dogs, the following types could be identified according to their morphologic features: mature type (two), cleaved type (two), asynchronous type (four), and polymorphous type (one). The tumor tissue of three cats revealed amyloid deposits, which were immunohistochemically diagnosed as ALλ-amyloid in all three cases.
Plasmacytomas are neoplasms consisting mainly of plasma cells at varying stages of differentiation. 11 The solitary myeloma and multiple myeloma are additional types of plasma-cell–derived tumor. The latter are primarily located in the bone marrow. Extramedullary plasmacytomas (EMPs) are characterized by their extraskeletal location. 8 , 29 As defined by the World Health Organization, EMPs consist of atypical neoplastic plasma cells with monoclonal expression of immunoglobulin (Ig) light chains or heavy chains. 15 They differ from other non-Hodgkin lymphomas by their predominantly postmitotic and terminally differentiated plasma cells. The main cellular function, production and secretion of Igs, is largely unaffected. 22
Whereas the EMP is a frequent neoplasm in dogs, 4 , 13 , 16 , 23 , 25–27 it is a sporadic tumor in man. 3 , 7 , 29 , 30 Judging from the existing case reports involving only one or two animals, the incidence of EMP in cats seems to be low. Feline EMPs have been described as occurring in the skin, 4 , 5 , 13 , 19 gastrointestinal tract, 24 , 31 retroperitoneal space, 14 upper lip, gingiva, 9 and orbita. 28
Affected cats were generally more than 10 years old; the average age of the animals was 10.6 years. 13 , 19 A male predisposition could be clearly concluded from the literature (male to female ratio was 2:1). The European Short-Haired cat was the most commonly affected. One case each was reported in the Abyssinian, 28 the Long-Haired, the Persian, 9 and the Burmese 13 breeds. Tumor-associated monoclonal gammopathy was reported in three cases. 5 , 14 , 28
The literature is uniform in describing the morphology as a monomorphic population of plasma cells with isolated bi- or multinucleated giant cells. 4 , 28 , 31
In several cases, immunohistochemistry revealed monoclonal expression of λ-Ig light chains, 4 , 6 , 9 , 19 whereas κ-Ig light-chain expression was found in only one case. 9 Ig heavy chains of the immunoglobulin G (IgG) 9 and immunoglobulin (IgA) 31 types also have been confirmed in the tumor cells. Amyloid deposits in feline EMPs were demonstrated by several authors, 4 , 5 , 13 , 14 , 19 , 24 although the amyloid was identified as ALλ-amyloid in only three cats. 19 , 24 To exclude a multiple myeloma, some authors examined the bone marrow for tumor cell infiltrates in the cats. 5 , 10 , 14 , 31
The aim of this retrospective study was to contribute to the existing body of knowledge in the literature on feline EMP by reporting on the morphologic and immunohistochemical findings in nine cats.
Material and Methods
The tissue samples of nine feline EMPs were either from the files of the Institute of Veterinary Pathology of the University of Munich (
Eight tumors occurred in the skin (see Results for details), and one had been removed from the oral cavity. Tissue samples were fixed in 5% buffered formaldehyde and embedded in paraffin. Subsequently, staining of 3-μm-thick sections was carried out with hematoxylin and eosin (HE) and Giemsa stain, as well as the PAS-reaction and Congo-red staining method. 21
Immunohistochemistry
For immunohistochemistry, an indirect peroxidase method was chosen. 17 The following cross-reacting rabbit-derived polyclonal antibodies were used: anti-human IgG (DAKO diagnostika GmbH, Hamburg, Germany; diluted 1:500); anti-human IgA (DAKO; diluted 1:500); anti-human IgM (DAKO; diluted 1:500); anti-human λ light chains (DAKO; diluted 1:750); and anti-human κ light chains (DAKO; diluted 1:500). Incubation with the primary antibody was followed by incubation with peroxidase–conjugated swine anti-rabbit Ig (DAKO; diluted 1:50). Tumors also were examined for the presence of feline leukemia-virus antigen (p27 protein) by incubation with polyclonal goat anti-p27 antibody (Biodesign, Dunn Labortechnik GmbH, Asbach, Germany; diluted 1:2,000). The secondary antibody was peroxidase–conjugated rabbit anti-goat Ig (DAKO; diluted 1:400). The reaction cascade was observed by incubation with diaminobenzidine-tetrahydrochloride (Fluka, Feinchemikalien GmbH, Neu-Ulm, Germany). Finally, the slides were counterstained with hematoxylin.
To identify the biochemical nature of the amyloid, monoclonal antibodies cross-reacting with animal amyloid were used together with the unlabelled immunoperoxidase (peroxidase–anti-peroxidase [PAP]) method, as summarized previously 12 : anti-human ALλ (HAR; diluted 1:8,000); anti-human ALλ (ULI; diluted 1:2,000); anti-equine ALλ (HIP; diluted 1:2,000); anti-human AA (mc1; cell culture supernatant diluted 1:10); and anti-human AA (mc129; cell culture supernatant diluted 1:10). The bridging antibody was a sheep anti-mouse IgG prepared in-house (diluted 1:20), and the third antibody was a PAP complex (DAKO, diluted 1:100). The reaction was developed with aminoethylcarbazole and the slides were counterstained with acid hemalum.
The primary antibody was replaced by nonimmune sera of the same species for control purposes.
Results
An extramedullary plasmacytoma was diagnosed in the case of all nine tumors on the basis of the cellular morphology and the immunohistochemical reaction pattern with light-chain restriction, i.e., the monoclonal expression of an Ig light-chain type was considered to be a decisive diagnostic criterion (Figs 1–7). Six of the affected animals were European Short-Haired cats and two were Siamese cats. No information was available regarding the breed of the ninth cat. The cats were 6–20 years old, with an average of 8.2 years. Four of them were females and five were males. Eight tumors were localized in the skin of various body regions (paws, thorax, neck, shoulder, tail, metatarsus, and nose); one was removed from the oral cavity. The EMPs varied in diameter, the largest being 3.5 cm wide. The cut surface of the tumors had a firm consistency, and the epidermis of the cutaneous EMPs was ulcerated in six cats.
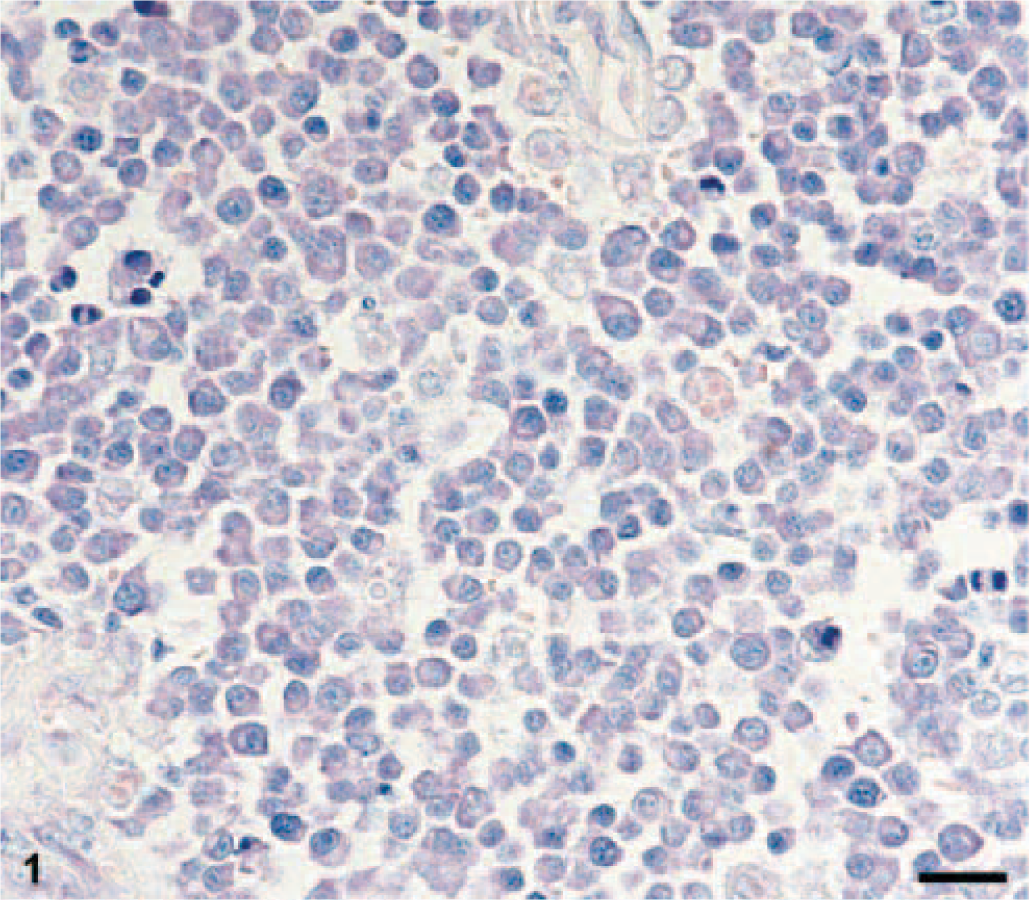
Skin, paraffin section; European Short-Haired cat, 7-year-old spayed female. Feline extramedullary plasmacytoma (EMP, asynchronous type of tumor cells) with basophilic cytoplasm, a clear perinuclear halo, and a blastic nucleus with a central nucleolus. Giemsa. Bar = 20 μm.
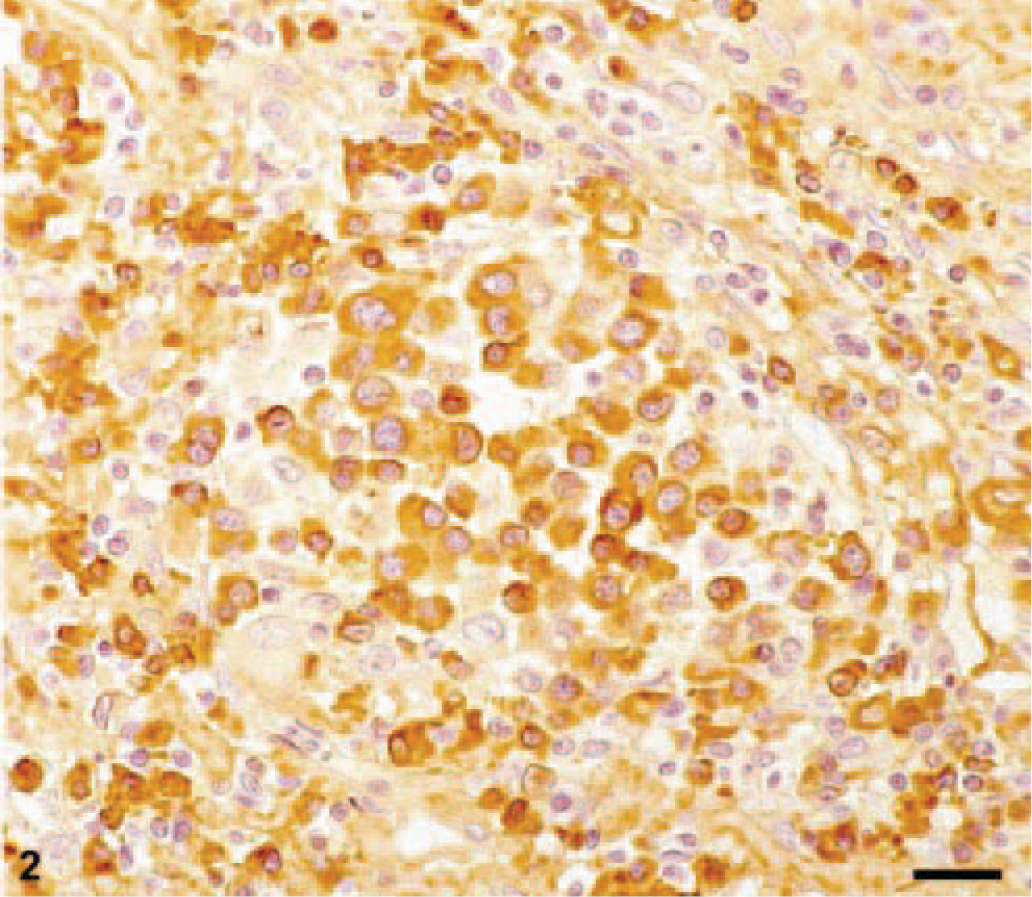
Immunohistochemical identification of ALλ amyloid with the anti-ALλ antibody ULI. Same cat as in Fig. 1. Bar = 20 μm.
Histology
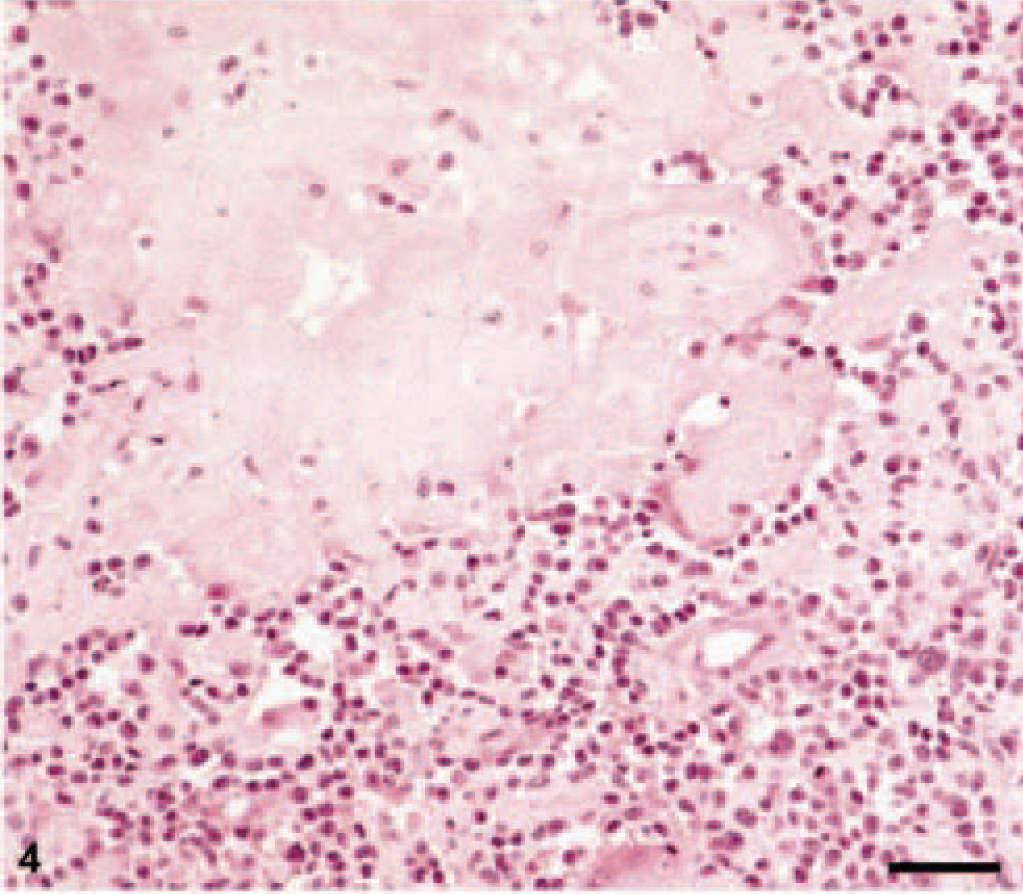
Histologically, the tumors were located in the corium and the subcutis of the skin or in the lamina submucosa of the oral cavity. They frequently expanded into the surrounding tissue, but a clearly infiltrative and destructive growth pattern was evident in only four cases. Invasion of blood vessels was not seen in any of the tumors. Inflammatory cellular infiltration at the tumor periphery consisted of macrophages, a few lymphocytes, and mast cells. In the areas of ulceration, tumor tissue was infiltrated by neutrophilic granulocytes.
According to the classification scheme of Bartl et al. 2 and Platz et al., 20 four different types of EMPs could be ascertained on the basis of histologic and cytomorphologic criteria. They are as follows.
Immunohistochemistry
Immunohistochemically, monoclonal λ light-chain reactivity of the cytoplasm was ascertained in all nine tumors (Fig. 2). In addition, three cats also expressed the IgG heavy chain.
Immunohistochemical demonstration of Ig-λ-light-chains in the cytoplasm of tumor cells. Same cat as in Fig. 1. Bar = 20 μm.
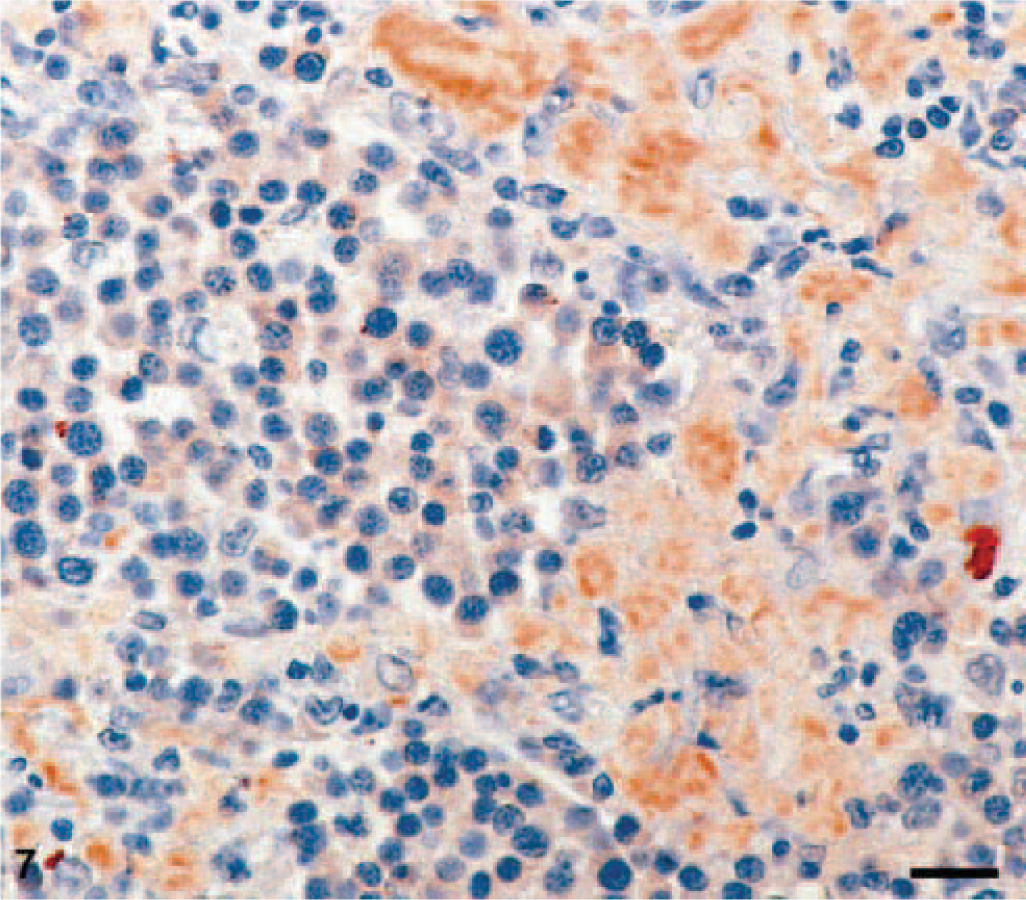
Amyloid was found in three EMPs in a diffuse pattern of distribution within the tumor tissue (Figs. 3, 4). Stained with Congo red, the amyloid deposits revealed the typical light-green birefringence when observed under polarized light (Figs. 5, 6). Two of the amyloid-containing EMPs were assigned to the asynchronous type, the third was of mature type. Immunohistochemistry showed that the amyloid deposits were ALλ-amyloid (Fig. 7) in all the three cats.
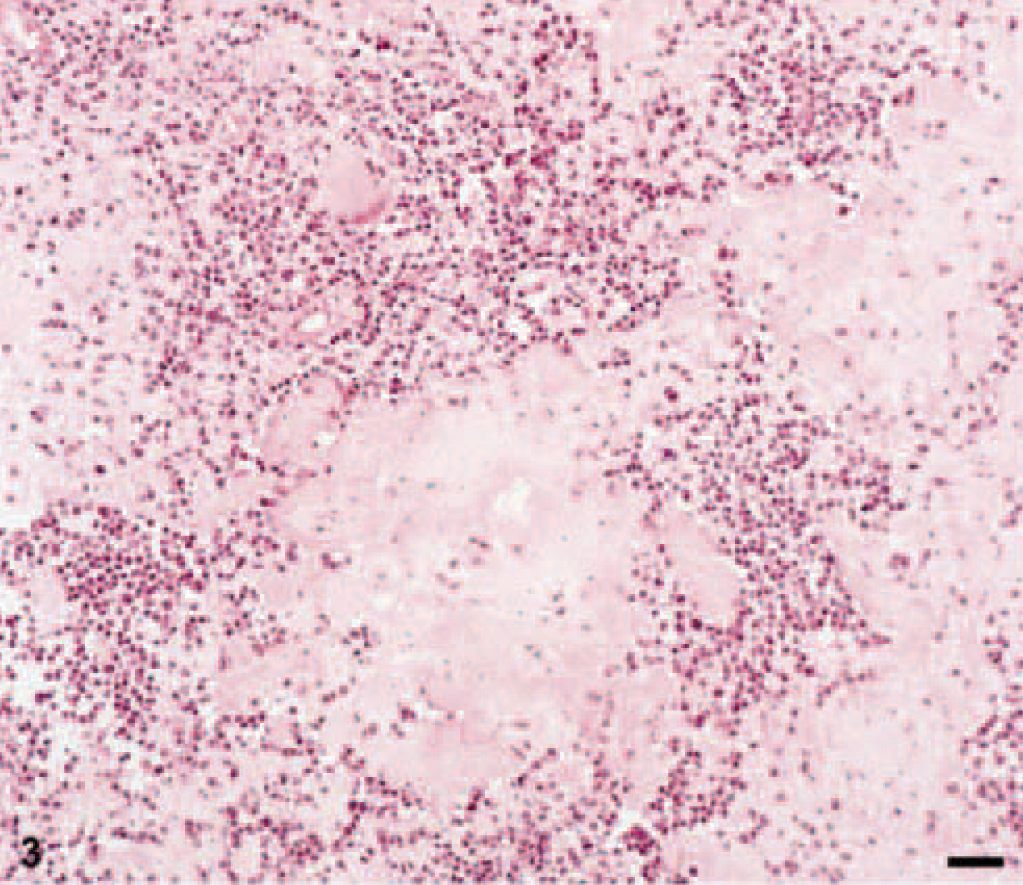
Feline EMP with homogenous eosinophilic amyloid deposits. HE. Bar = 50 μm.
Homogenous deposition of amyloid surrounded by plasmacytic tumor cells. HE. Bar = 50 μm.
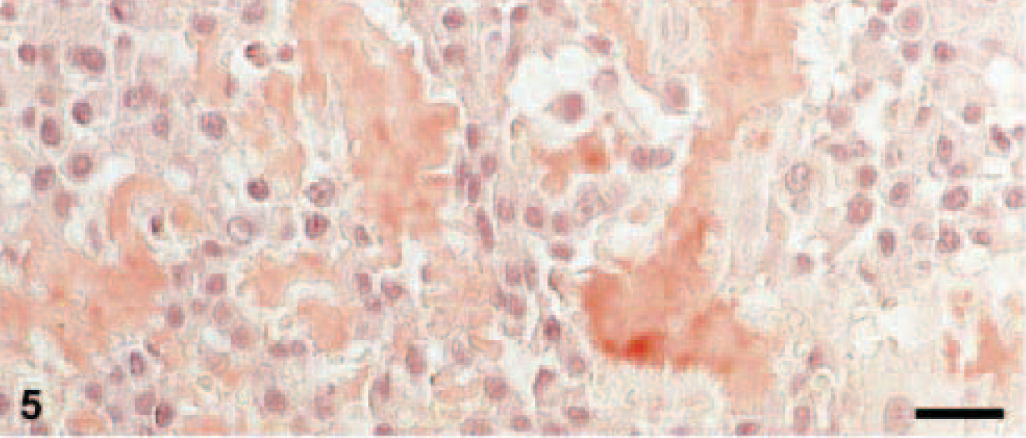
Skin; European Short-Haired cat, 13-year-old castrated male. Congo red–staining of EMP, mature type of tumor cells. Bars = 20 μm.
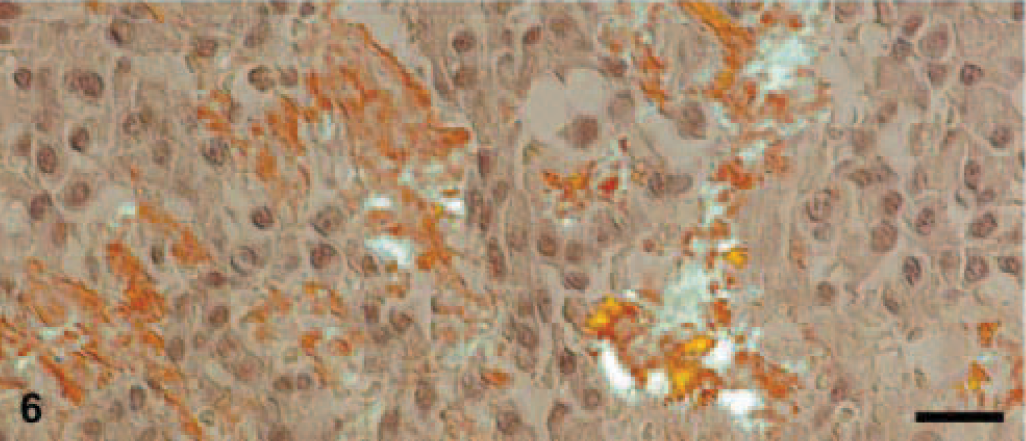
All Congo-red positive areas within the tumor are identified as amyloid by green-birefringence in polarized light. Same cat as in Fig. 5. Bars = 20 μm.
Immunohistochemical examination for FeLV (p27 protein) was negative for all the tumors.
Discussion
In humans, the extramedullary plasmacytoma is uncommon and accounts for less than 5% of all plasmacellular neoplasia. 7 , 29 , 30 Although EMPs are commonly found in dogs, mostly in the form of skin and mucosal neoplasms, 20 they seem to be rare in cats. The latter conclusion can be drawn both from the fact that so far only single-case reports exist in the literature and from the incidence of cutaneous EMPs, as estimated from the number of biopsies submitted to this department for histologic examination. The incidence of EMPs in other parts of the body besides the skin remains unknown, although, according to other authors, it can be assumed to be low. 4 , 5 , 13 From our own series, it also can be confirmed that EMPs in cats are mainly cutaneous neoplasms, as observed previously. 4 , 5 , 9 , 13 The occurrence of EMPs in other organs, e.g., digestive tract 24 , 31 and liver, 10 is rare. With an average age of 8.2 years, the EMP occurs predominantly in older cats. This, as well as the preference for the male sex, agrees with the result in the literature. 4 , 5 , 13
As in dogs, a typing of EMPs based on morphologic criteria also is possible in cats. With the exception of the hyaline type described in dogs, the same types of EMP found in dogs were found in cats. The absent hyaline type might be found in cats, if a larger number of tumors could be examined. Because of the small number of cases and a lack of follow-up studies, no claim can be made currently concerning the diagnostic significance of the typing of feline EMPs.
Immunohistochemistry of the feline EMPs mainly revealed λ light-chain expression. This is very similar to the physiologic distribution of light chains in normal feline plasma cells with primary expression of the λ light chain. 1 Monoclonal κ light-chain expression was reported in only one case. 9 With respect to the immunohistochemical results in our series, the possibility that the tumor plasma cells could express Ig heavy chains not detectable with the routinely used antibodies cannot be excluded.
According to the literature, amyloid is more frequently found in feline EMPs than in canine EMPs. Six out of 12 published cases of EMPs in cats report on amyloid deposits. 4 , 5 , 14 , 19 , 24 , 28 In the present study, three of the nine EMPs contained amyloid, identified as ALλ-amyloid. This is consistent with the result of the only study published to date of an immunohistochemically investigated feline EMP. 24
As reported by other authors, 5 , 10 , 14 immunohistochemical examination for FeLV-antigen (p27 protein) was negative for all tumors. Communication of the above results may contribute to the already existing knowledge regarding extramedullary plasmacytomas in cats.
