Abstract
A 5-y-old spayed female Golden Retriever dog was referred because of severe normocytic normochromic nonregenerative anemia and thrombocytopenia. Serum analysis revealed hyperproteinemia and monoclonal or oligoclonal gammopathy. Fine-needle aspiration of the spleen revealed a highly erythrophagocytic population of neoplastic round cells, morphologically suggestive of plasma cells. After euthanasia, histologic assessment of the spleen and liver revealed an erythrophagocytic round cell tumor. Immunohistochemical analysis of the tumor population was positive for MUM1p and negative for CD3, CD20, and Iba-1, confirming the plasma cell origin of the tumor. Erythrophagocytic multiple myeloma is a very rare neoplastic condition in dogs.
Keywords
A 5-y-old spayed female Golden Retriever dog was referred by her veterinarian for evaluation of severe anemia. Physical examination was unremarkable except for pale oral mucosae. A CBC revealed severe normocytic, normochromic, nonregenerative anemia and thrombocytopenia (Table 1). A basic serum biochemistry profile revealed hyperproteinemia (plasma protein 95 g/L; RI: 57–80 g/L) with hyperglobulinemia (globulin 80 g/L; RI: 25–45 g/L) and hypoalbuminemia (15 g/L; RI: 28–40 g/L). Other parameters were within RIs (aspartate aminotransferase, alkaline phosphatase, total bilirubin, glucose, cholesterol, urea, creatinine, phosphorus). Serum protein electrophoresis revealed a monoclonal or oligoclonal gammopathy (Fig. 1). Urinalysis revealed borderline isosthenuria, proteinuria (urine dipstick 500 mg/dL; RI: <30 mg/dL) with urine specific gravity of 1.014, and mild hematuria.
Relevant hematological findings in a dog with an erythrophagocytic myeloma.
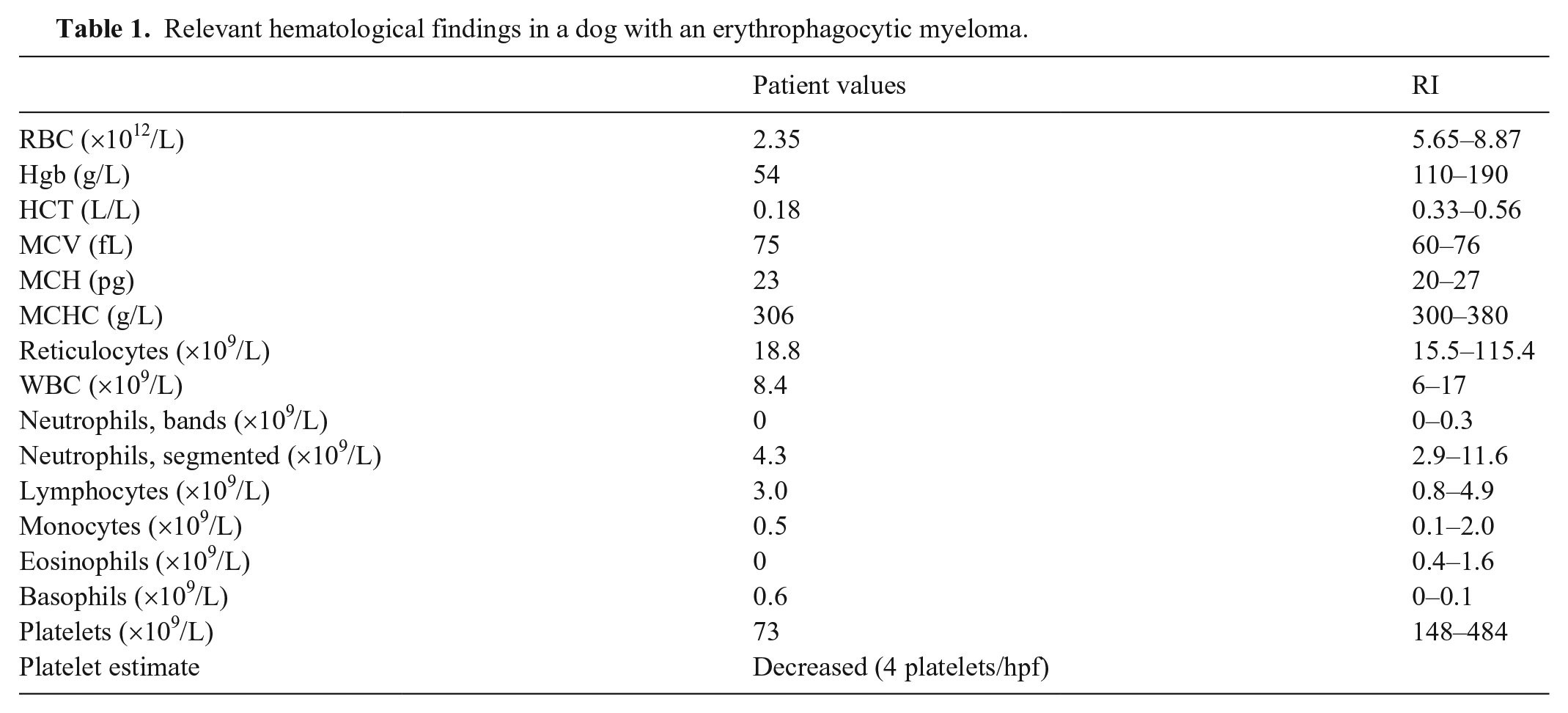
Electrophoretic profile of serum from a dog with erythrophagocytic multiple myeloma. The capillary zone electrophoresis trace revealed monoclonal gammopathy.
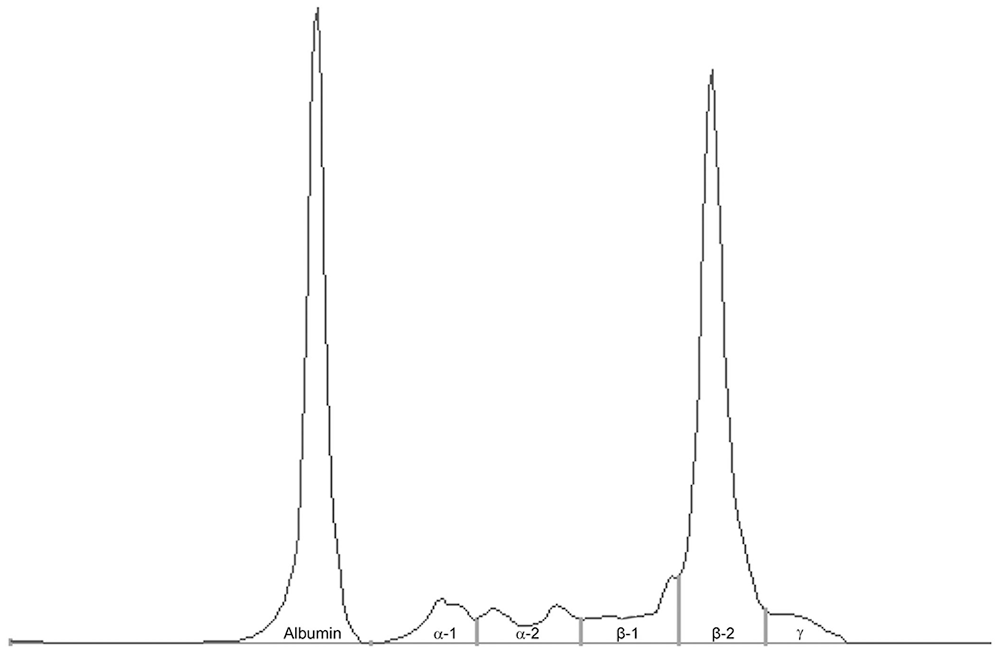
Abdominal ultrasound revealed severe splenomegaly; the spleen had rounded margins and finely nonhomogeneous parenchyma without focal lesions. Mild abdominal effusion was present but could not be sampled. The liver had no significant ultrasound findings. Echocardiography revealed normal kinetics and morphology. Pericardial effusion was not present. Fine-needle aspiration of the splenic parenchyma was performed; the sample was hemodiluted but highly cellular, with a predominance of atypical, 18–30-µm, round cells, characterized by a moderate amount of blue cytoplasm with a pale perinuclear halo. Cells contained single, eccentrically located, round nuclei, sometimes with irregular nuclear membranes and some indentation, with a moderate nuclear:cytoplasmic (N:C) ratio. Binucleation was rare (~4%). Nuclei contained coarse-to-clumped chromatin with prominent nucleoli. Some of these cells had a pink pericellular halo (“flame cells”) and erythrophagocytosis (Fig. 2). Similar neoplastic infiltrates were present in the liver.
Erythrophagocytic multiple myeloma in a dog.
Cytology was performed on bone marrow aspirated from the costochondral junction. Bone marrow was highly cellular and had findings similar to the splenic sample, with ~10% mixed myeloid and erythroid hematopoietic precursors.
Based on the clinicopathologic findings (>20% plasma cells in bone marrow, and monoclonal gammopathy),1,20,21 multiple myeloma (MM) with unusual erythrophagocytosis was diagnosed. The owner declined additional investigations and chemotherapy. A few weeks later, the dog was euthanized given its poor clinical condition, and the owner authorized the sampling of liver and spleen for histopathology.
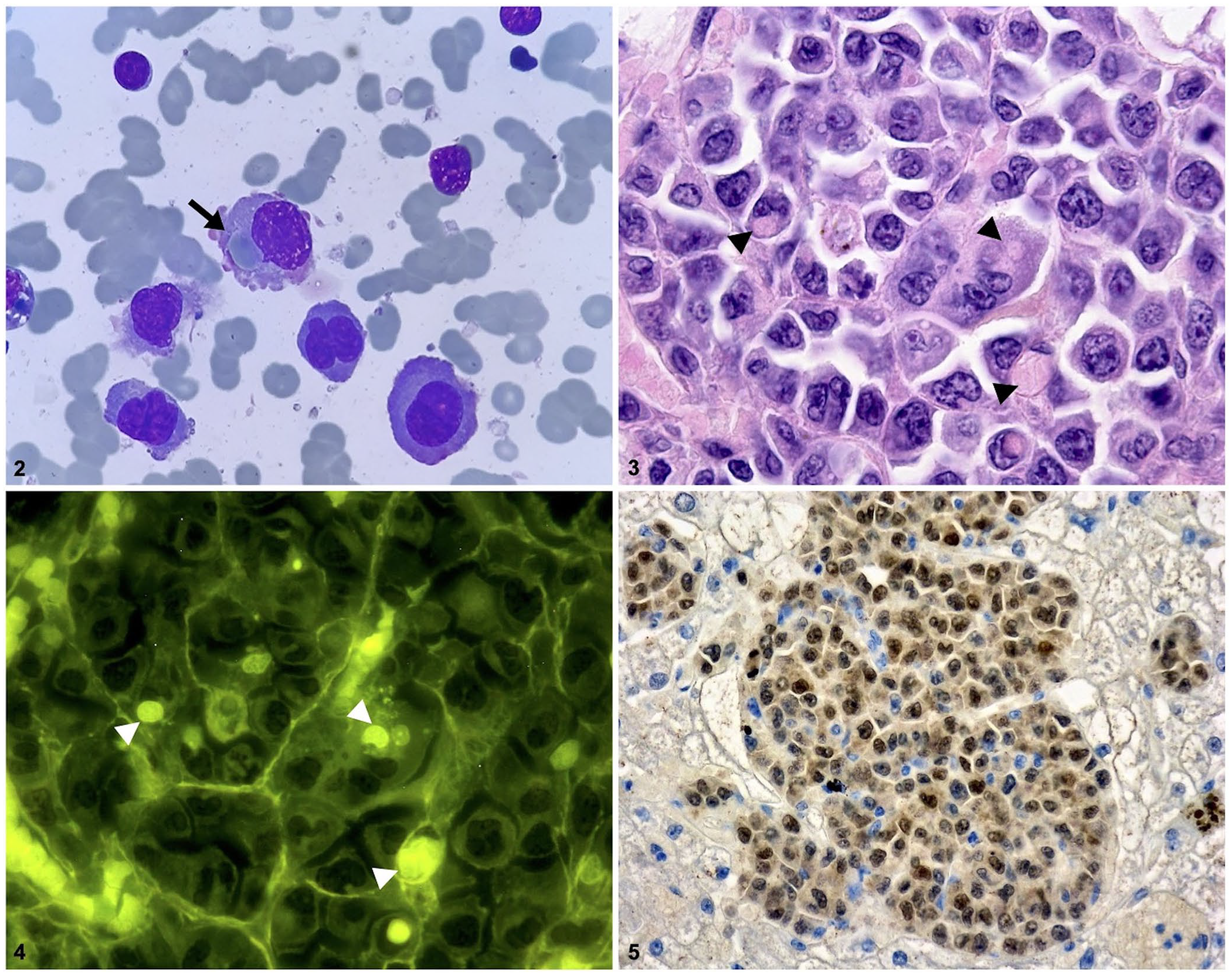
Histologically, the splenic red pulp and periportal and centrilobular areas of the liver were infiltrated by sheets of neoplastic non-cohesive round cells (Fig. 3). Neoplastic cells were 15–30 µm diameter, with distinct cell borders, high-to-intermediate N:C ratio, scant-to-moderate amounts of eosinophilic cytoplasm, occasionally with a perinuclear clear halo, eccentric round-to-indented nuclei, with granular chromatin and indistinct small nucleoli. Anisocytosis and anisokaryosis were moderate. The mitotic count was 4 in 2.37 mm2. 12 Occasionally, bi- and multinucleate cells and erythrophagocytosis were found. Intracytoplasmic erythrocytes were further confirmed through observation with a fluorescence microscope (Fig. 4). Hemosiderin-laden macrophages were present multifocally in the splenic red pulp.
Neoplastic cells were present as single cells within hepatic sinusoids. The liver was additionally affected by severe hepatocellular vacuolar degeneration, mainly involving periportal and midzonal regions. By immunohistochemistry (IHC), neoplastic cells had positive nuclear staining for MUM1p/IRF4 (rabbit monoclonal, clone BC5, Biocare; Fig. 5) and were negative for CD3 (rabbit polyclonal; Dako), CD20 (rabbit polyclonal; Thermo Scientific), and Iba-1 (rabbit polyclonal; Wako). Based on the appearance of the round cells, less-likely differential diagnoses included lymphoma and histiocytic neoplasia, but the presence of several flame cells was highly suggestive of plasma cell origin, which was confirmed by the MUM1p nuclear expression on IHC.
MM is a systemic proliferation of malignant plasma cells or their precursors, which arises as a clone from a single cell. MM usually involves multiple bone marrow sites, but may originate from extramedullary sites.13,21 Although MM is uncommon in veterinary medicine, MM has been reported in cats, dogs, and horses. 12 MM accounts for 0.3% of all malignancies, 8% of hematopoietic tissue tumors, and 3.6% of bone marrow tumors in dogs.11,12 A high prevalence of this tumor has been reported in the German Shepherd breed. 11 Age at diagnosis is 8–12 y, 12 with no apparent sex predisposition. Factors associated with the development of MM in companion animals have not been identified. Genetic predisposition, molecular aberrations, viral infections, chronic immune stimulation, and exposure to carcinogen stimulation have all been suggested as contributing factors. 21 The clinical signs of MM vary with the level of plasma cell proliferation, the localization and spread of the neoplastic plasma cells, and the nature and extent of proteinuria. 12 The clinical signs are usually nonspecific and include lethargy, weight loss, anorexia, diarrhea, vomiting, fever, and increased susceptibility to infections, as well as renal failure and hemostasis abnormalities. 12
Various degrees of cytopenia can be observed in association with MM. The dog in our report had normocytic, normochromic, nonregenerative anemia and thrombocytopenia. Normocytic, normochromic, nonregenerative anemia is present in ~67% of dogs with MM, 21 and can result from myelophthisis, blood loss from coagulation disorders, anemia of chronic inflammation, or increased erythrocyte destruction secondary to serum hyperviscosity. 21 In our case, erythrophagocytosis very likely contributed to the severe anemia.
Thrombocytopenia is present in ~33% of all canine patients with MM.4,10,19 However, there are many other reports of MM in veterinary medicine with normal numbers of platelets.1,21,23 Thrombocytopenia resulting in bleeding disorders can be related to infiltration of bone marrow by malignant plasma cells or consumption of platelets as part of a thrombohemorrhagic syndrome, such as disseminated intravascular coagulation. Other mechanisms, not yet proven in veterinary medicine, may include shortened platelet half-life or immune-mediated destruction.12,17 Moreover, platelet adhesiveness may be impaired by hyperviscosity and paraprotein interference.3,6 In these cases, thromboelastometry or platelet function analysis might have provided additional information about thrombocytopenia and platelet function.
Our patient had hyperproteinemia and protein dyscrasia, specifically hypoalbuminemia and hyperglobulinemia, which are common, but not invariable, features of MM.5,10,14 Indeed, there are some reports of serum albumin concentrations within the RI in animals.1,12 In our case, proteinuria contributed to hypoalbuminemia. In humans, hypoalbuminemia is related primarily to the extent of plasma cell proliferation, and it is therefore of diagnostic and prognostic importance. 12 Several studies have suggested that hypoalbuminemia is correlated with increased serum concentrations of interleukin-6, a potent myeloma cell growth factor, reflecting disease severity and cell proliferation. 12 Interleukin-6, a multifunctional, pro-inflammatory cytokine that stimulates B-cell maturation and proliferation and overproduction, has been demonstrated in a variety of B-cell malignancies. 12
Proteinuria detected by routine urinalysis is present in 90% of human patients with MM, 12 and it has been also detected in horses, 12 dogs, 17 and cats 24 with MM, using a dipstick test. In our case, proteinuria was investigated using a dipstick test, but not with the urine protein:creatinine ratio (UPC) and urine protein electrophoresis, as the literature suggests. 12 Indeed, the amount of protein loss in our case is unknown.
The appearance of malignant plasma cells can vary in cytologic preparations and histologic sections. The degree of differentiation ranges from cells that resemble normal plasma cells in late stages of differentiation to very large, anaplastic round cells with a high mitotic count, representing early stages of differentiation. 21
MUM1p is considered a specific immunohistochemical plasma cell marker for normal and neoplastic plasma cells in dogs15,18; labeling is nuclear with a weak cytoplasmic component. MUM1 is an interferon regulatory factor that is required for immunoglobulin light-chain rearrangement, and is expressed in B cells, plasma cells, activated T cells, and a subset of macrophages and dendritic cells in humans. 22 The lack of CD20 expression in our case was not unexpected given that only 20% of plasma cell tumors are reported to express CD20. 15
To our knowledge, only 2 cases of plasma cell tumor demonstrating erythrophagocytic activity have been reported in dogs9,24 and cats.4,23 On the contrary, there are several reports of phagocytic plasma cells in human patients with MM. 16 The mechanism underlying erythrophagocytosis in MM is unclear, given that plasma cells are not phagocytic under normal circumstances. Results of the direct antiglobulin test in humans are almost always negative, suggesting that erythrophagocytosis by neoplastic cells is not related to autoimmunity. 16 It has been speculated that phagocytic plasma cells may arise from an expansion of a rare B-cell clone with innate phagocytic potential. 16 A single case of phagocytic plasma cells aberrantly expressing CD15—normally found on neutrophils and monocytes and involved in phagocytosis—has been reported in humans. 8
In addition to the routine bright-field observation of the H&E-stained section, we took advantage of RBC autofluorescence to confirm erythrophagocytosis by neoplastic cells. The intensity of the autofluorescence of RBCs, which is attributed to products of peroxidation of phospholipids and glycation of proteins, increases with the age of the cells. 7
Unfortunately, the prognosis is poor for companion animals with MM. 12 The mean survival time in dogs with MM treated with melphalan, cyclophosphamide, and prednisone is 540 d after diagnosis. 20 In dogs, negative prognostic factors include extensive bone lysis, hypercalcemia, and light-chain proteinuria.13,21
Erythrophagocytosis is an uncommon feature of round cell tumors, in particular of plasma cell tumors because plasma cells do not naturally have phagocytic activity. Once erythrophagocytosis is identified in a round cell tumor it is then important to differentiate lymphoma, histiocytic neoplasia, and osteosarcoma 2 from plasma cell tumor via IHC and clinicopathologic findings (e.g., monoclonal or oligoclonal gammopathy).
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interests with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
