Abstract
Inflammatory or degenerative pathology involving the vertebral bodies and/or ventral intervertebral joints has been described in numerous species, both captive and free ranging, including mammals, birds, and snakes, although never in amphibians. We described 15 cases of a newly recognized spinal arthropathy in adult cane toads (Chaunus [Bufo] marinus), an invasive species in Australia. Grossly appreciable lesions consisted primarily of ventral proliferation of bone and cartilage that resulted in ankylosis. Histologic examination of the entire vertebral columns of the 15 affected toads revealed 13 toads to have lesions at multiple intervertebral sites, totalling 41 intervertebral joints with lesions. Most lesions involved bone and cartilage proliferation that resulted in fusion of the joint, with (n = 15) or without (n = 9) associated pyogranulomatous inflammation. In the remaining 17 affected joints, histologic lesions were characterized by degeneration of articular cartilage without joint fusion. In addition, in one of these joints, there was also hemorrhage and pyogranulomatous inflammation. Bacterial culture of affected joints in 9 toads and grossly normal joints in 4 control toads resulted in isolation of Ochrobactrum anthropi in 7 affected toads but in none of the controls (P < .01). We proposed an interaction between degenerative and bacterial etiologies in the pathogenesis of this condition. Invasive toads may be predisposed to this condition because of their large size; increased rates of movement; and, possibly, immunosuppression resulting from inhabiting a novel environment.
Pathology involving the vertebral bodies and the ventral aspect of intervertebral joints include degenerative conditions, many of which are associated with advanced age or repeated trauma, and inflammatory conditions, which may be immune mediated or bacterial. 17,25,29,35,46 Bacterial infection involving the spine is typically hematogenous, and, although the pathogenesis is poorly understood, is thought to be predisposed in many cases by degenerative changes. 24,42 Inflammatory conditions of the vertebral bodies and intervertebral joints were reported in various captive species, including canids, swine, mink, poultry, and snakes, as well as in numerous species of free-ranging large mammals, including whales. 8,9,12,15,19,25,26,33,34,39,43 Despite intensive interest in skeletal abnormalities of amphibians, descriptions of degenerative diseases or spinal pathology are rare and limited to scoliosis. 1,28,36
Cane toads (Chaunus [Bufo] marinus) were introduced into coastal Queensland, Australia, from Hawaii in 1936 in a strategy to control the sugar cane beetle. 22 Since then, their range has expanded westward and the toads have evolved enhanced dispersal ability. 31 In 2005, they arrived in the Darwin region of the Northern Territory, 1900 km from their point of introduction. 31 Cane toads are large (up to 2 kg),heavy-bodied, ground-dwelling amphibians that feed on a variety of insects and small vertebrates. All life history stages of cane toads are toxic to naïve predators, therefore, the impact of invasive toads on native fauna is a major conservation issue. 22,30 Since their introduction, there has been considerable interest in studying diseases of free-ranging cane toads, with the hope of identifying pathogens that could be used to control the invasive population. 36 These studies identified numerous protozoal, bacterial, and fungal organisms associated with cane toads; however, published reports of disease in Australian cane toads are limited to occasional cases of fungal infection. 3,11,27,36,37
In this paper, we described the pathology of a newly recognized spinal arthropathy of free-ranging adult cane toads and propose the etiology to involve interaction between bacterial infection and preexisting degeneration of intervertebral joints.
Materials and Methods
The study animals consist of 15 toads with spinal arthropathy (affected toad Nos. 1–15) and 4 toads with grossly normal vertebral columns (control toad Nos. 1–4). All toads were captured in the Darwin region during 2006 and 2007. Affected toad Nos. 1–11 were in captivity for 1 to 2 weeks before euthanasia. Affected toad Nos. 12–15 and the control toads were captured 18 hours before euthanasia. The affected toads and control toads were all large adults of comparable sizes (Table 1). All toads were humanely euthanatized by using an overdose of pentobarbitone sodium into the heart or dorsal lymph sacs. A complete postmortem was performed on each toad.
Summary of toads with spinal arthropathy and controls.
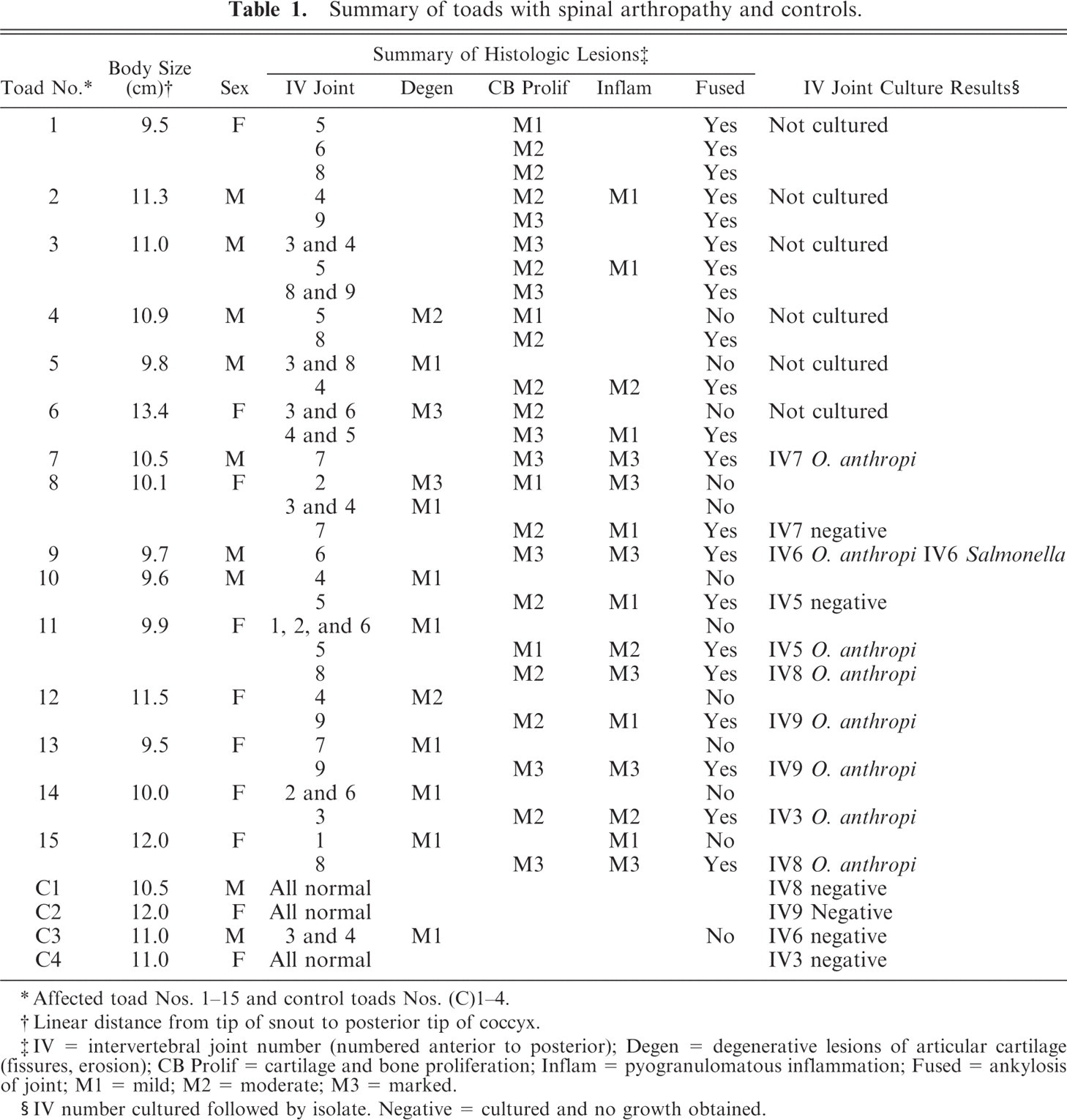
∗Affected toad Nos. 1–15 and control toads Nos. (C)1–4.
†Linear distance from tip of snout to posterior tip of coccyx.
‡IV = intervertebral joint number (numbered anterior to posterior); Degen = degenerative lesions of articular cartilage (fissures, erosion); CB Prolif = cartilage and bone proliferation; Inflam = pyogranulomatous inflammation; Fused = ankylosis of joint; M1 = mild; M2 = moderate; M3 = marked.
§IV number cultured followed by isolate. Negative = cultured and no growth obtained.
Swabs for bacterial culture of intervertebral joints were obtained at postmortem from grossly enlarged intervertebral joints of 9 affected toads and a grossly normal intervertebral joint from each of the 4 control toads. Samples were obtained by making a transverse cut through the intervertebral joint region by using aseptic techniques and by applying a sterile swab to the cut surface. Blood samples were taken by cardiocentesis for blood culture from affected toad Nos. 12–15 and the 4 control toads immediately before euthanasia. Intervertebral swabs were inoculated onto tryptone soya agar with sheep blood and MacConkey agar (Oxoid, Thebarton, Australia). Blood was inoculated onto thioglycolate broth (Amyl Media Pty Ltd., Dandenong, Australia) immediately after sampling. The media plates and broths were incubated at 30 ± 2°C and checked for growth at 24 and 48 hours. Bacteria were identified by using the commercial phenotypic systems API 20 NE (bioMerieux, Marcy l'Etoile, France) and Microbact 24E (Oxoid, Hants, U.K.). The Biolog GN2 MicroPlate system (Biolog, Hayward, CA), which uses 95 discrete tests to produce a “metabolic fingerprint” based on usage of various carbon sources, was used to corroborate identification on 1 isolate.
For histologic examination, the vertebral columns of all affected toads and control toads were decalcified in a solution of 10% formalin with 9% formic acid for 72 hours. Either transverse sections at intervertebral joints or mid sagittal sections of the vertebral column were taken, embedded in paraffin, sectioned at 5 μm and stained with hematoxylin and eosin (HE). In affected toad Nos. 7, 8, 11, and 13, vertebral columns were also stained with Gram-twort, Ziehl-Neelsen acid-fast, and Gomori methenamine silver stains to enhance visualization of any etiologic agents. 10,38 These 4 toads were chosen for special stains, because the lesions were associated with substantial pyogranulomatous inflammation. Formalin-fixed tissues, including brain, eye, heart, lung, liver, spleen, stomach, intestine, pancreas, kidney, bladder, gonad, skeletal muscle, skin, and appendicular joint, from affected toad Nos. 1, 2, 7, 11, and 13 were processed as above, stained with HE, and examined microscopically to look for evidence of concurrent diseases.
Results
Unlike mammals, many anurans, including cane toads, have synovial joints between vertebral bodies, with a condyle on the posterior aspect of the vertebral body and a concave articular surface on the anterior aspect (procoelous joint morphology). 13 Cane toads normally have 9 vertebrae, with the ninth vertebra, or sacrum, having a bicondylar articulation with the coccyx, or urostyle, which represents the fused postsacral vertebrae. 13 On gross examination, when viewed from the ventral aspect, the joints between vertebral bodies of toads appeared as lucent, white, well-defined transverse straight or slightly undulating lines between adjacent vertebral bodies, with the epiphyses of some vertebral bodies flaring slightly at the joint (Fig. 1). In normal toads, manipulation of the vertebral column produced obvious movement at intervertebral joints.
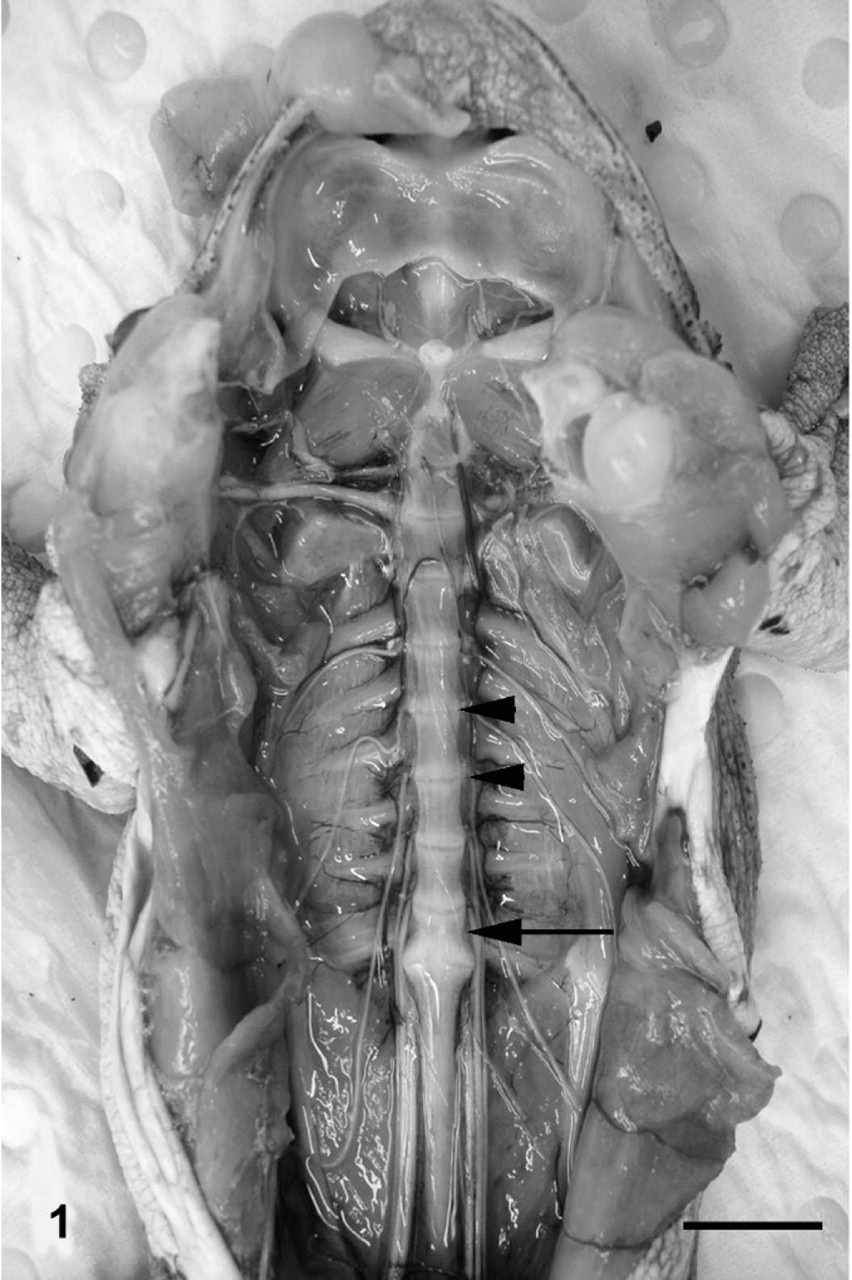
Vertebral column; cane toad. Normal toad (control No. 1) with viscera removed exposing ventral aspects of vertebral bodies. Nine vertebrae are present; arrowheads depict 2 adjacent normal intervertebral joints. The arrow indicates the sacral vertebra, which articulates posteriorly with the elongate coccyx. Bar = 1 cm.
Gross examination of affected toad Nos. 1–15 revealed 2 toads with lesions at single intervertebral joints, and 13 toads with lesions at up to 5 joints. Affected joints in toads with multiple lesions exhibited variable appearance. Joint spaces were indistinct or absent, with varying degrees of smooth, moderately firm to rugose bony swelling (Figs. 2– 4). In a few toads, the swelling was focally soft or red (Fig. 3). The bony swellings usually bridged adjacent vertebral bodies, resulting in ankylosis of the joint. The swelling was roughly symmetrical when it involved a joint anterior to the articulation of the sacrum and the coccyx. Swelling at the sacrococcygeal joint was frequently asymmetrical, primarily involving only 1 condyle. In a few toads, there were subtle gross changes in intervertebral joints that were perceptible on gross images only after histologic evaluation revealed the joints to be abnormal. These included joints with a slightly irregular joint space of increased width and joints with a subtly abnormal shape compared with adjacent joints (Figs. 2, 4). Toads with spinal arthropathy did not have gross lesions that involved the dorsal intervertebral joints or appendicular joints. There was no evidence of trauma or wounds associated with affected intervertebral joints. Other gross postmortem findings in affected toad Nos. 1–15 were limited to poor body condition in 1 toad and scattered, 1- to 2-mm, white, caseous foci in the gastric wall of 5 toads.
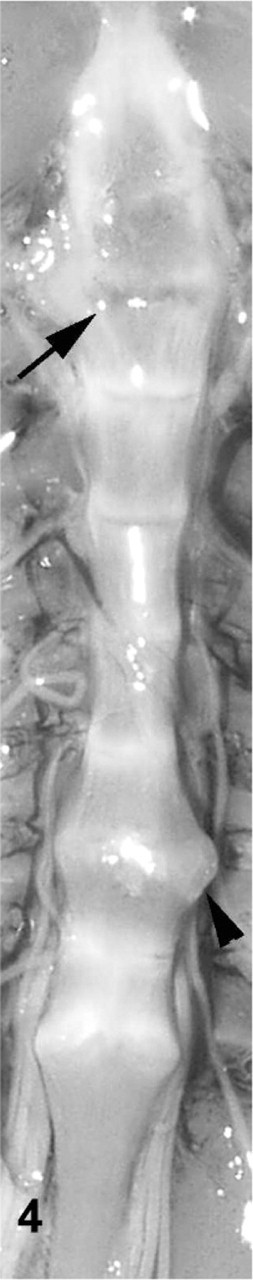
Vertebral column; cane toad No. 8. Mild irregularity and expansion of second intervertebral joint space (arrow) depicted histologically in Figs. 10, 11. Moderately expanded, fused seventh intervertebral joint (arrowhead).
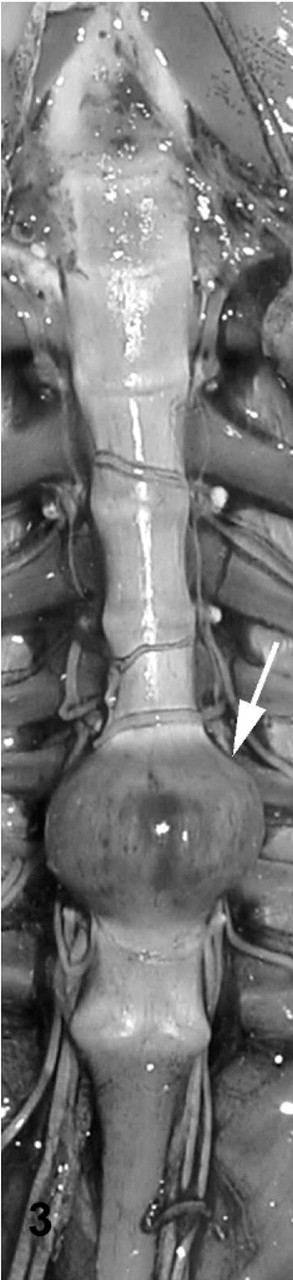
Vertebral column; cane toad No. 7. Markedly enlarged, fused seventh intervertebral joint (arrow).
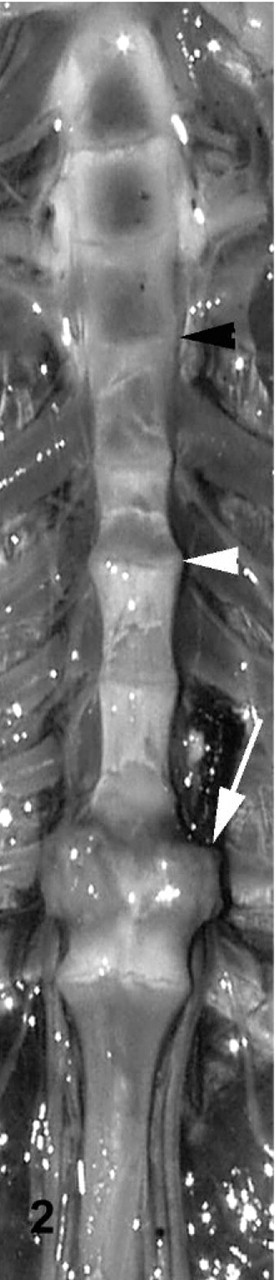
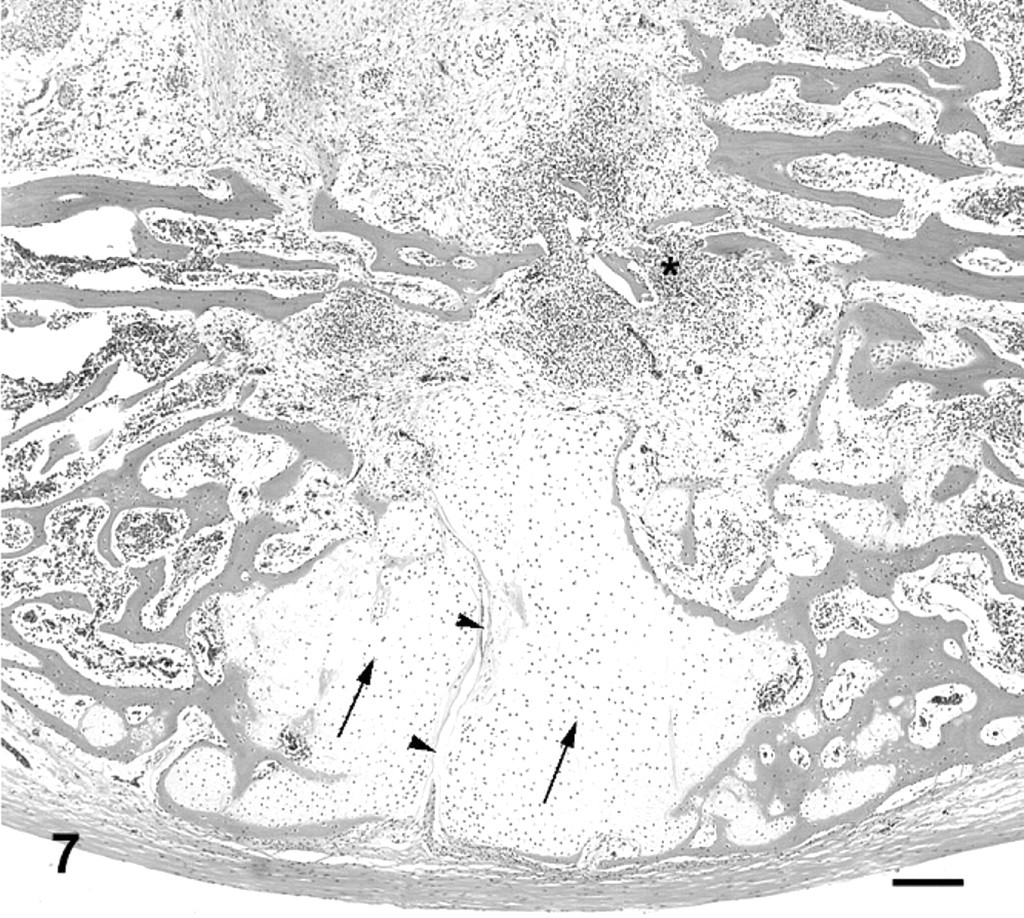
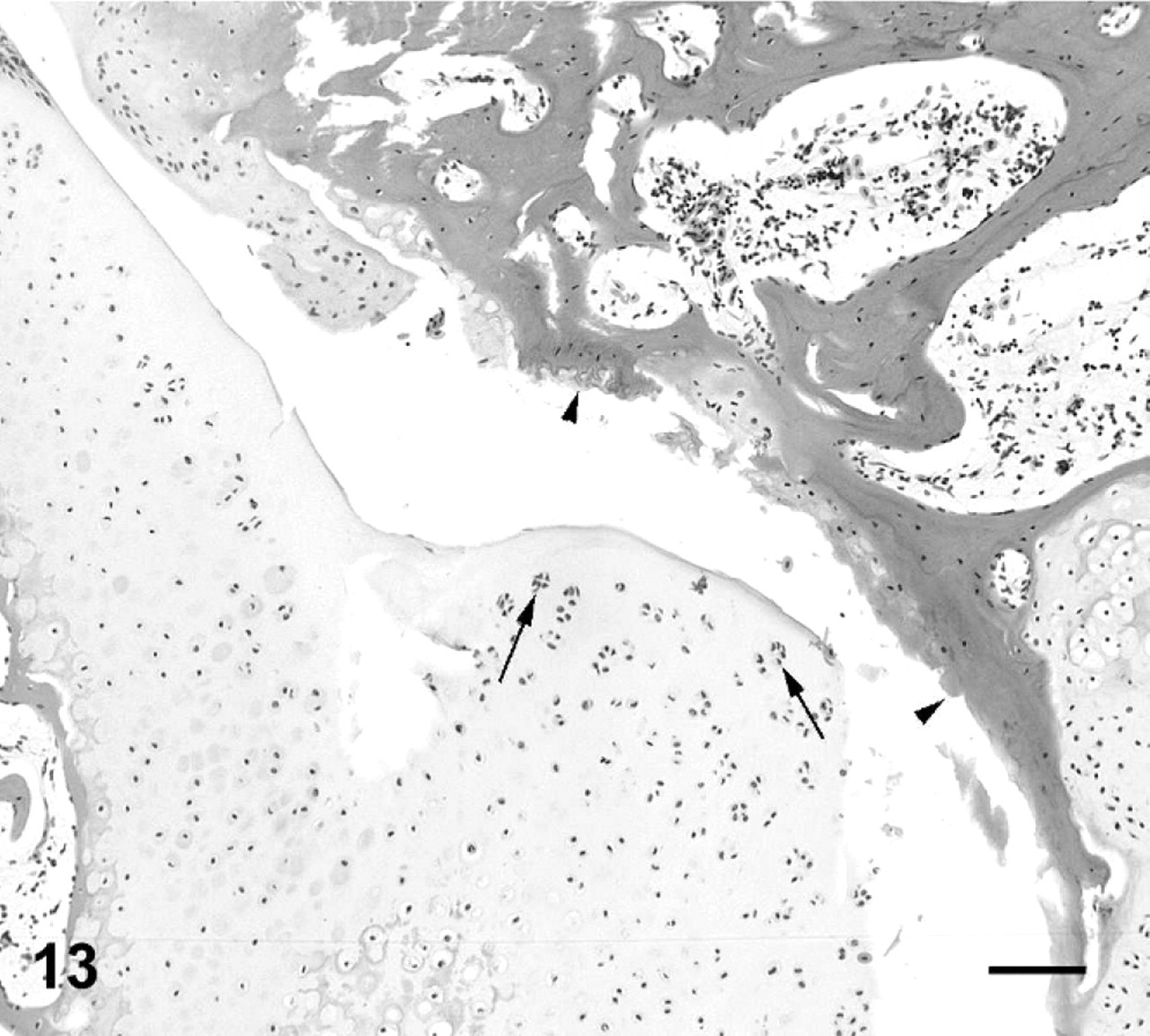
Vertebral column; cane toad No. 4. Normal third intervertebral joint depicted histologically in Figs. 5, 6 (black arrowhead). Fifth intervertebral joint with mild degenerative changes (white arrowhead) depicted histologically in Fig. 13. Eighth intervertebral joint fused with bone and cartilage (white arrow) depicted histologically in Fig. 9.
On histologic examination, the intervertebral joints of normal cane toads exhibited similar features to synovial joints of mammals, with a joint cavity, synovium lining a fibrous joint capsule, and articular cartilage characterized by flattened elongate chondrocytes oriented parallel to the articular surface. There was a subarticular layer of cartilage undergoing interstitial growth, continuos with an epiphyseal ossification center (Figs. 5, 6).
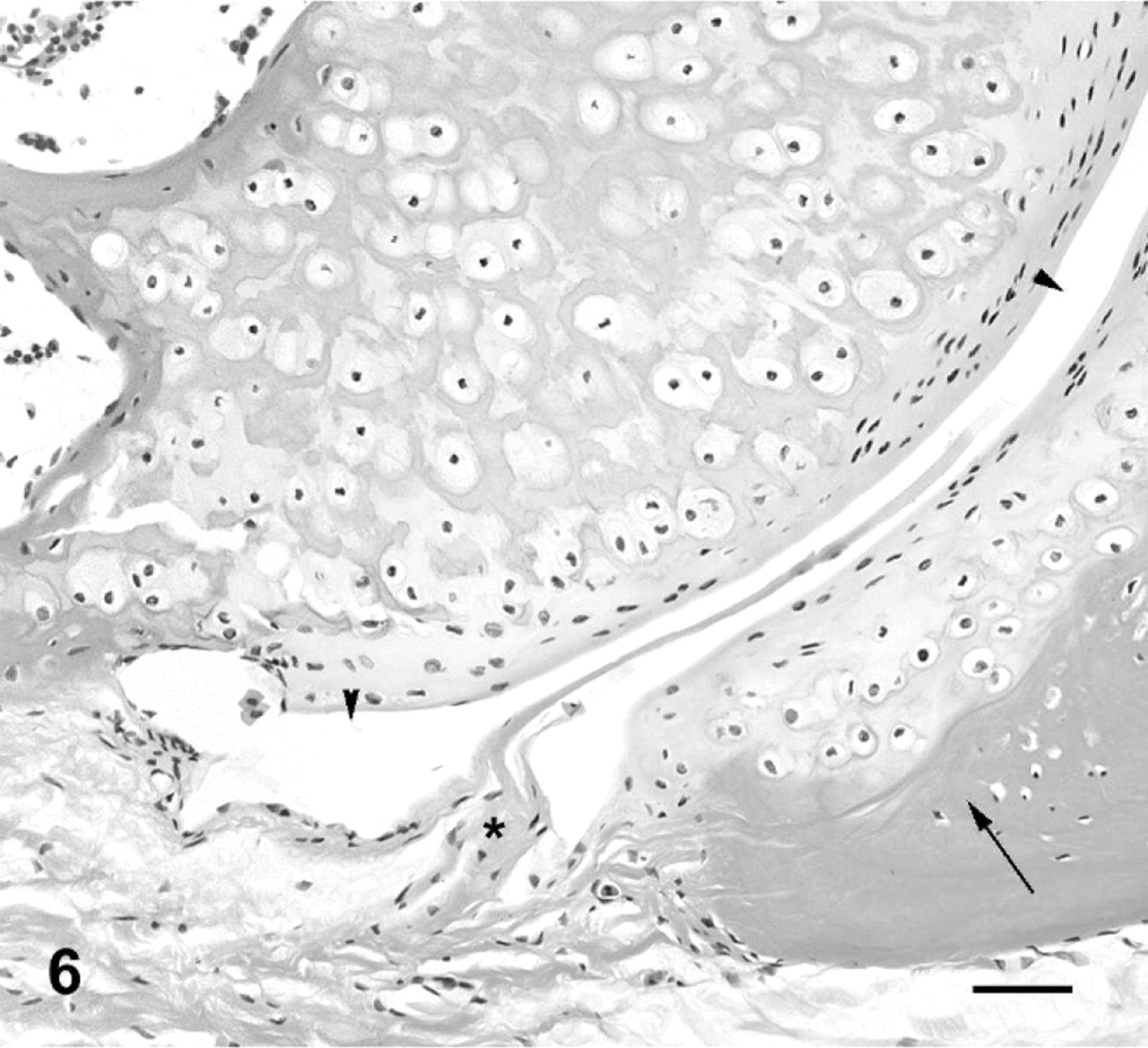
Third intervertebral joint; cane toad No. 4. Higher power view of ventral aspect of joint depicted in Fig. 5. Joint space (arrowheads), subchondral bone (arrow), synovium (asterisk). HE. Bar = 50 μm.
Vertebral bodies and third intervertebral joint; cane toad No. 4. Normal intervertebral joint with joint space (arrowheads) lined by articular cartilage, synovium (asterisks) and subchondral bone trabeculae (arrows). HE. Bar = 200 μm.
Histologic lesions in affected toad Nos. 1–15 are summarized in Table 1. The 15 toads exhibited histologic lesions at 41 intervertebral spaces. The majority of lesions that were grossly visible as enlarged fused joints were similar in appearance. There was partial or complete loss of the joint space, with woven bone and irregular islands of hyaline cartilage breaching the space and extending ventrally as wide bridges of bone and cartilage between adjacent vertebrae. In 15 lesions, these changes were accompanied by inflammation (Figs. 7, 8), whereas, in 9 lesions, the joint was fused with no associated inflammation (Fig. 9). In cases with inflammation, the inflammatory infiltrate consisted primarily of dense aggregates of macrophages that were focally degenerated and necrotic. Heterophils and neutrophils were present less consistently. Necrotic bone fragments and remains of cartilage were frequently intermingled with the inflammatory cells (Figs. 7, 8). Inflammatory cells replaced hematopoietic cells in bone marrow adjacent to the joint, were variably present in the remains of joint spaces, and expanded joint capsules. Bone and cartilage proliferation did not extend into the spinal canal, although, in 1 toad, the periosteum projected into the spinal canal because of expansion with inflammatory cells but with no histologic evidence of spinal-cord compression.
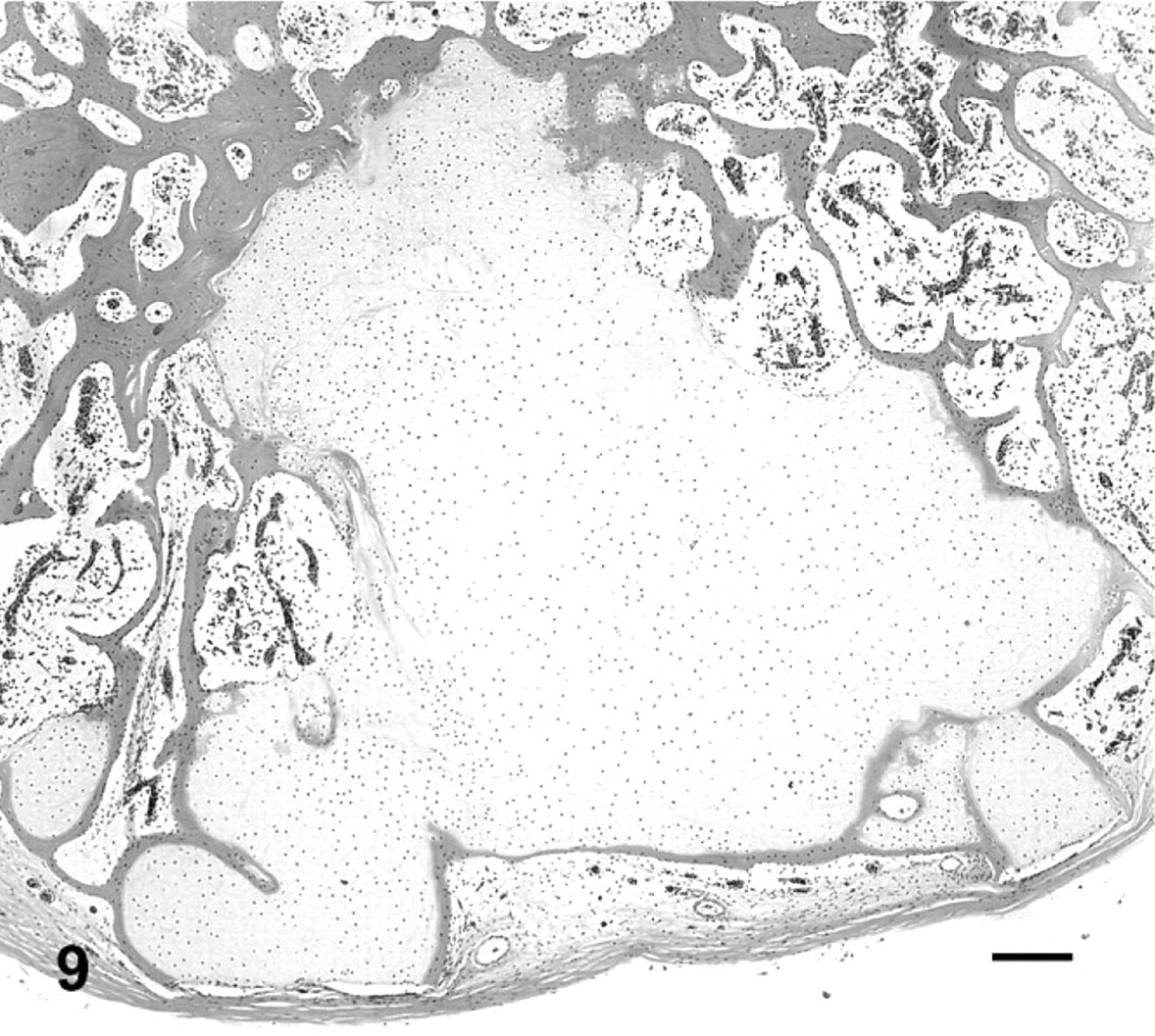
Fused vertebral bodies, eighth intervertebral joint; cane toad No. 4. An irregular mass of cartilage surrounded by bone occupies previous site of intervertebral joint. HE. Bar = 200 μm.
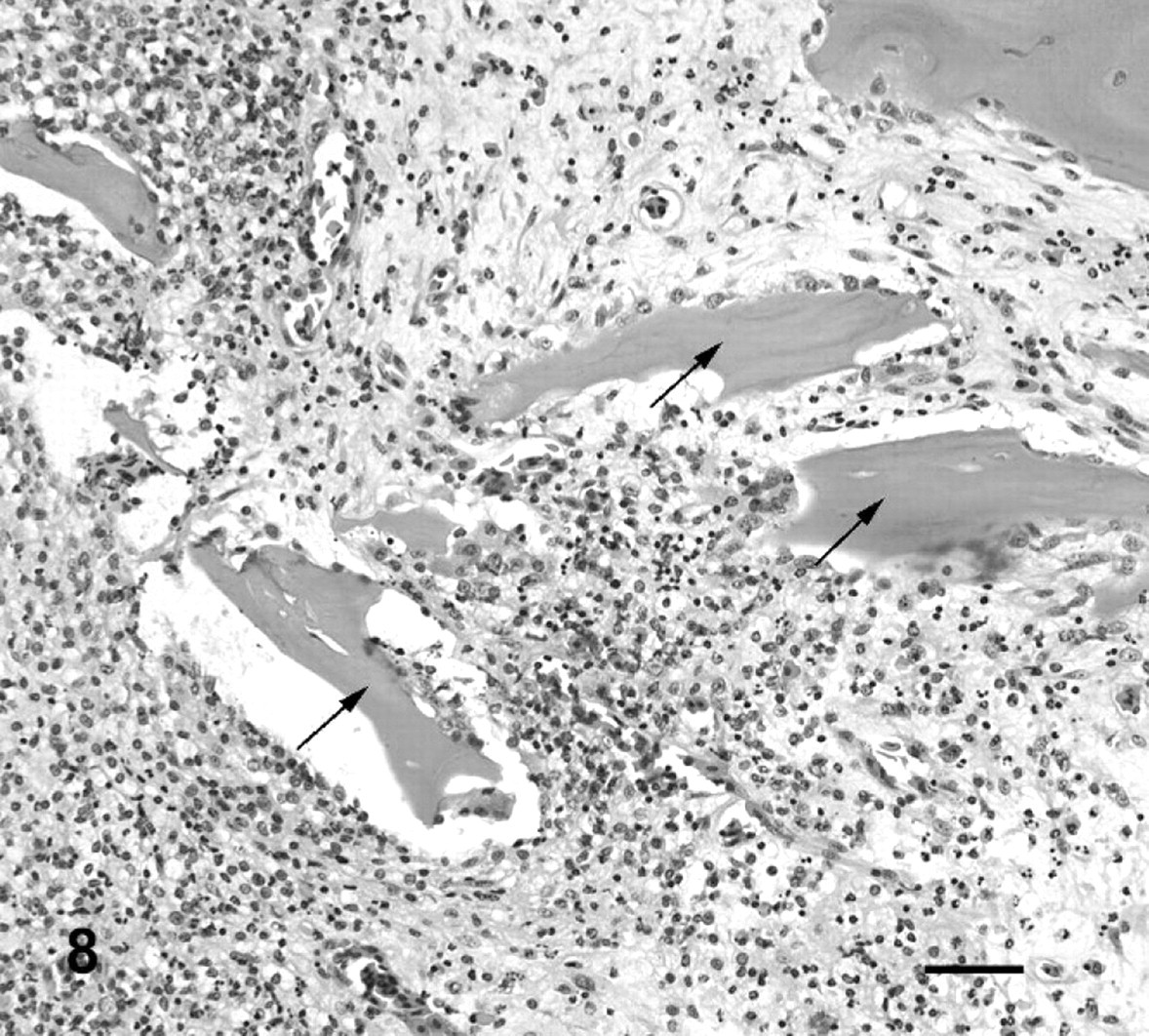
Fused vertebral bodies, fourth intervertebral joint; cane toad No. 5. Higher power view of area with asterisk from Fig. 7. Necrotic fragments of bone (arrows) surrounded by pyogranulomatous inflammation. HE. Bar = 50 μm.
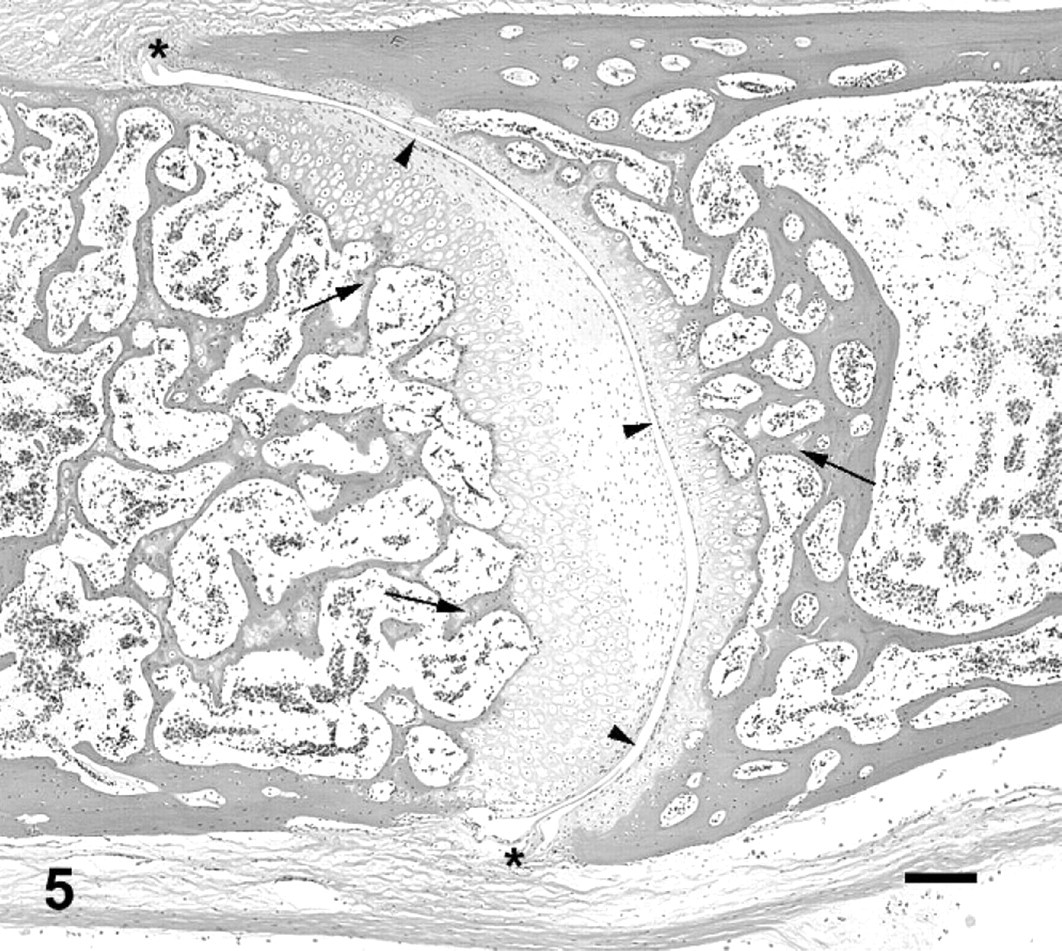
Vertebral bodies and fourth intervertebral joint; cane toad No. 5. The joint is almost entirely fused, with a small section of remaining joint space ventrally (arrowheads). Proliferative cartilage surrounding ventral aspect of joint (black arrows). Increased cellularity in midsection of fused vertebral bodies (asterisk). HE. Bar = 200 μm.
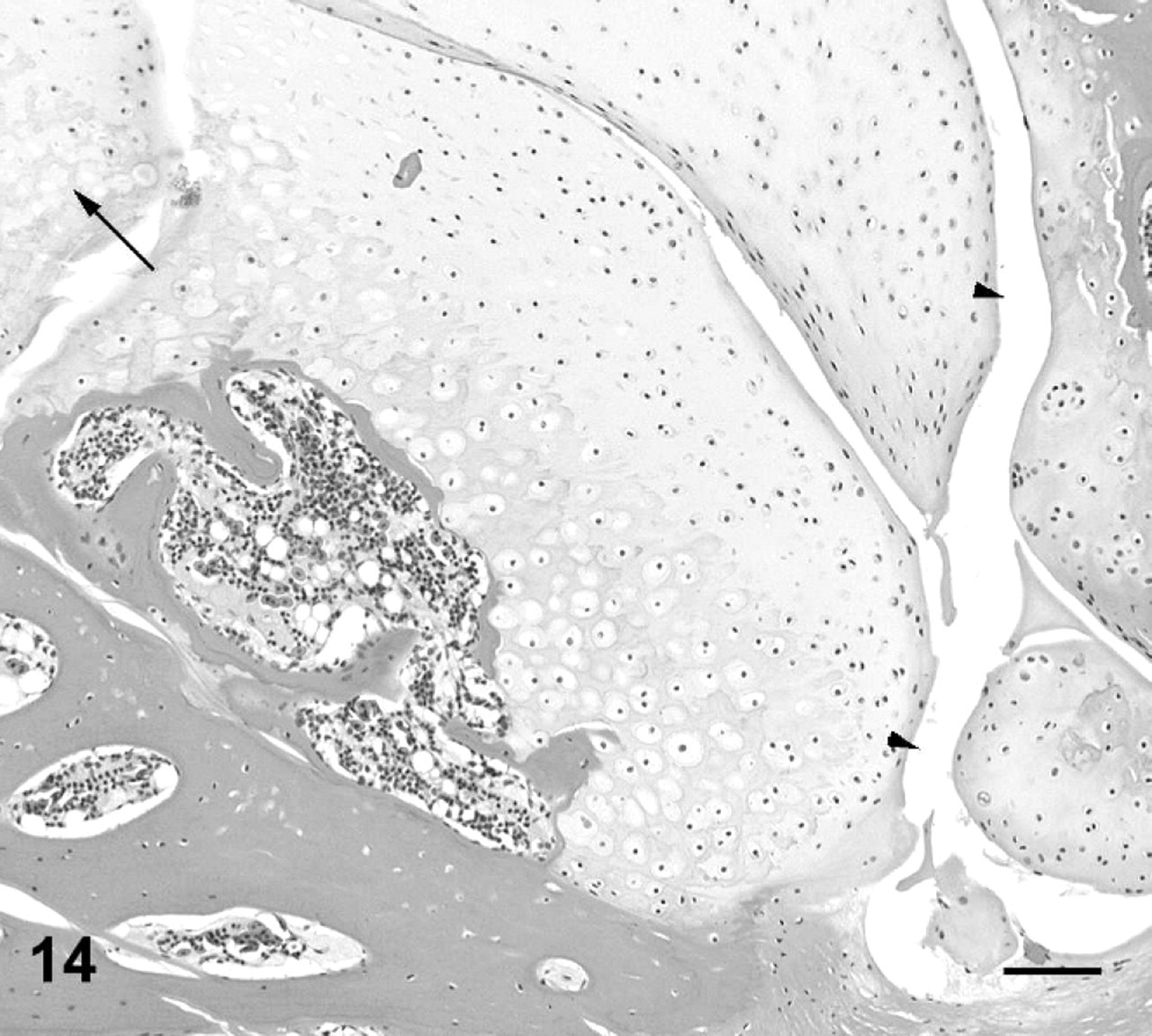
In addition to lesions that resulted in gross enlargement of the intervertebral joint, there were numerous lesions appreciable only by histologic examination. In affected toad No. 8 (Fig. 4), there was severe erosion and ulceration of articular cartilage, with hemorrhage and necrotic-cell debris in the joint space at intervertebral joint 2, along with a typical proliferative ankylosing lesion at intervertebral joint 7. At intervertebral joint 2, cartilage proliferation was limited to the ventral margins of the joint, and there was marked infiltration of the marrow spaces, synovium, and joint capsule with neutrophils, heterophils, and macrophages (Figs. 10, 11). Among the 15 affected toads, there were also 16 joints with relatively mild lesions in which an inflammatory infiltrate was minimal or absent but articular cartilage was fissured, focally eroded, or necrotic, with or without concurrent mild-to-moderate cartilage proliferation that had not progressed to joint fusion (Figs. 12–14). Special stains (Gram-twort, Ziehl-Neelsen acid-fast, and Gomori methenamine silver) did not reveal organisms in affected joints in affected toad Nos. 7, 8, 11, or 13.
Sixth intervertebral joint; cane toad No. 6. Irregular proliferation of articular cartilage surrounding joint space (arrowheads) with deep fissures and necrotic foci in cartilage (arrow). HE. Bar = 100 μm.
Fifth intervertebral joint; cane toad No. 4. Ulceration of articular cartilage of posterior aspect of joint, with exposure of subchondral bone (arrowheads). Multinuclear chondrones in irregular proliferative articular cartilage in apposing anterior aspect of joint (arrows). HE. Bar = 100 μm.
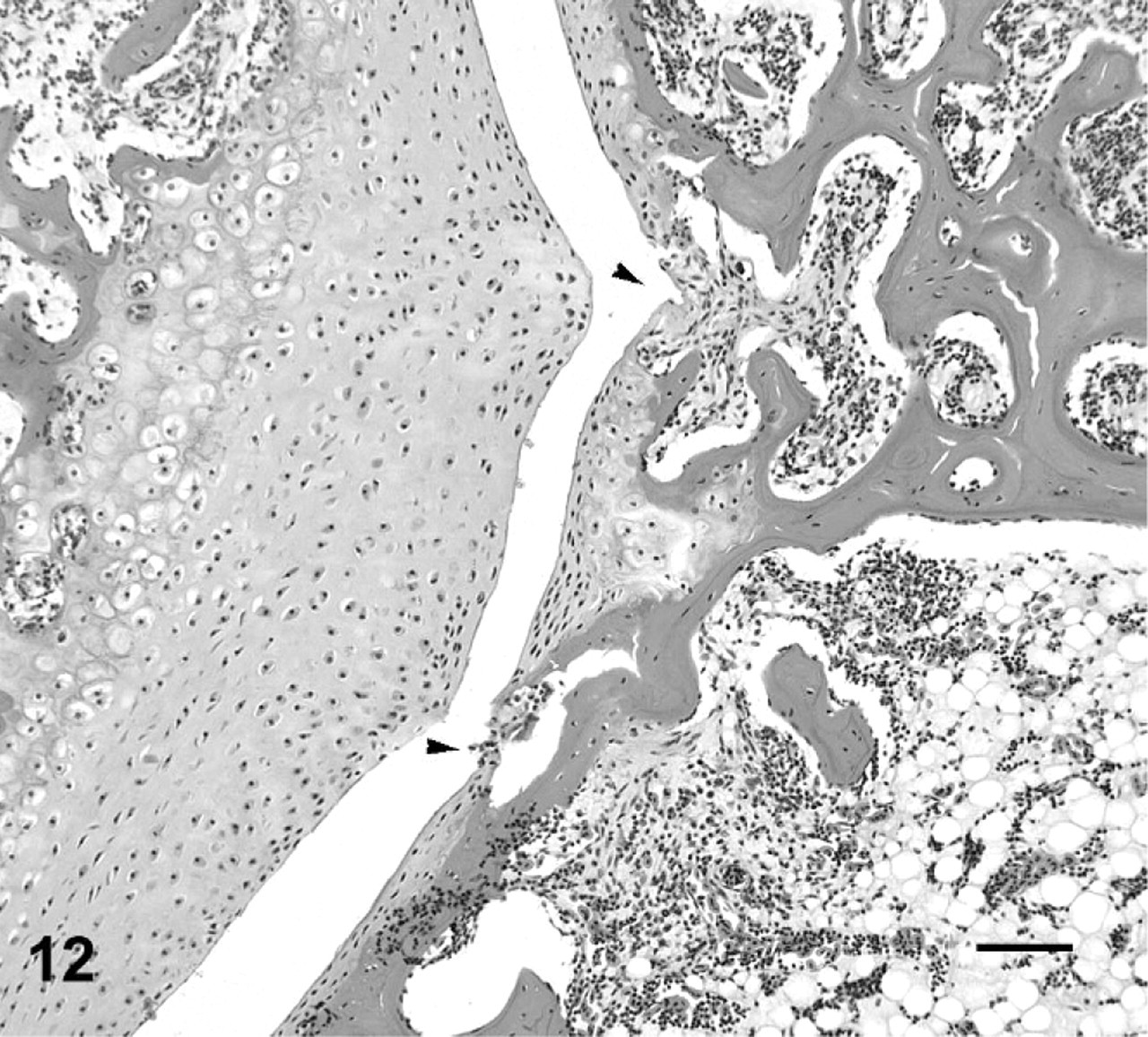
Second intervertebral joint; cane toad No. 11. Ulceration of articular cartilage on posterior aspect of joint (arrowheads) with irregular apposing articular cartilage of anterior aspect of joint. HE. Bar = 100 μm.
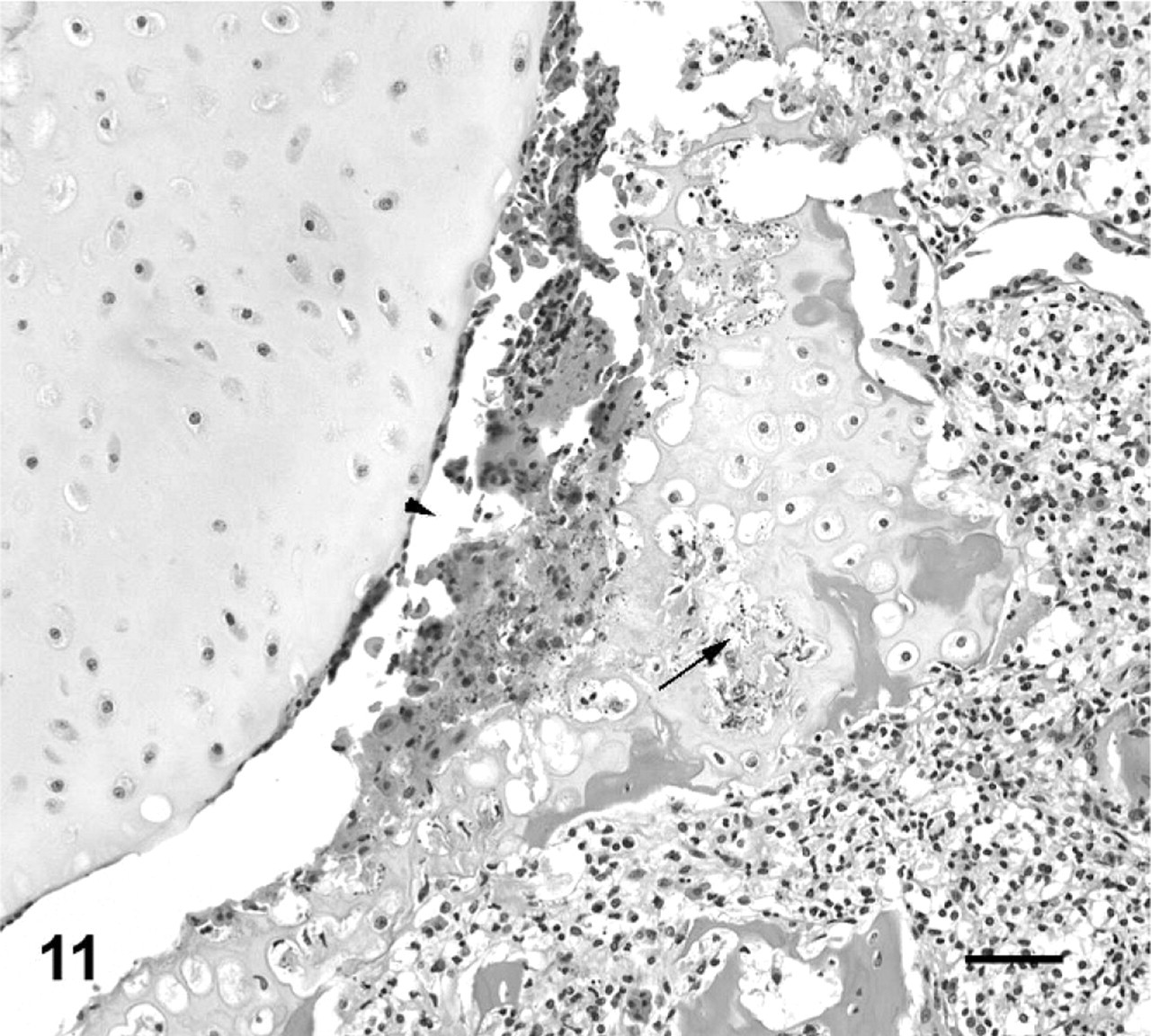
Second intervertebral joint; cane toad No. 8. Higher power view of joint space from Fig. 10. Hemorrhage and necrotic cellular debris in joint space (arrowhead) adjacent to eroded and necrotic cartilage (arrow). HE. Bar = 50 μm.
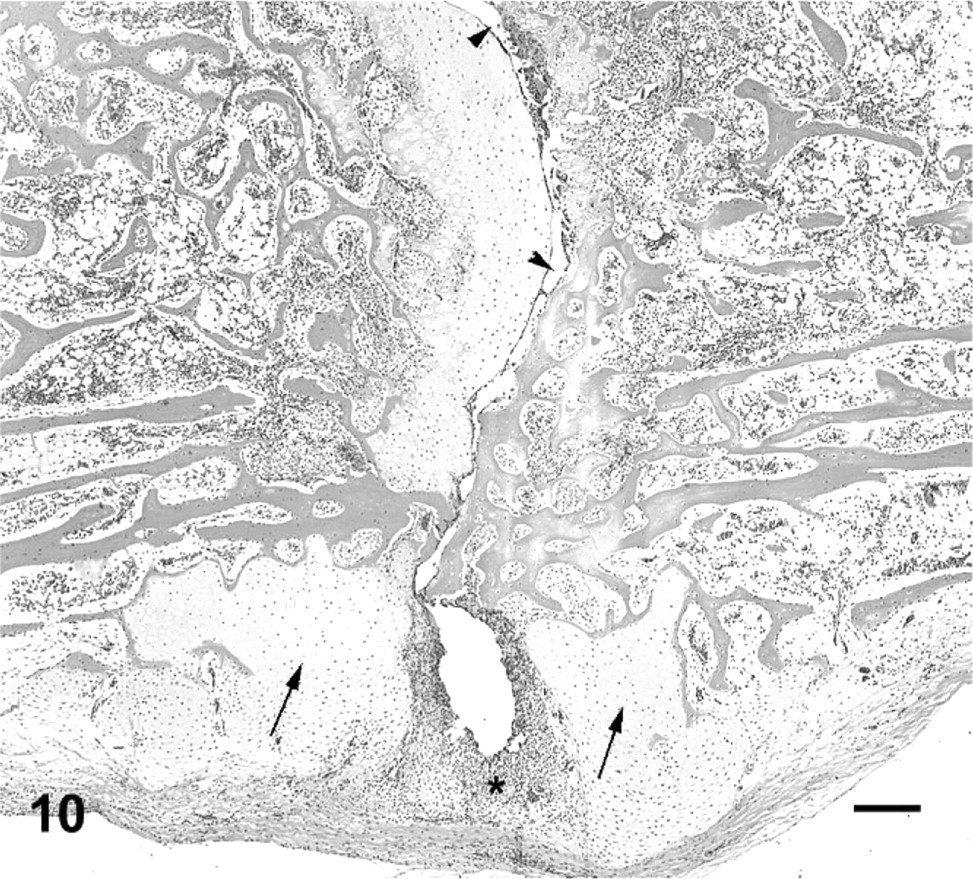
Vertebral bodies and second intervertebral joint; cane toad No. 8. Irregular joint space (arrowheads) with scant articular cartilage remaining on posterior (right) articular surface. Irregular proliferative cartilage ventral aspect vertebral bodies at the joint (arrows). Pyogranulomatous cellular infiltrate ventral synovium and joint capsule (asterisk). HE. Bar = 200 μm.
Histologic examination of the spine of control toad Nos. 1–4 did not reveal inflammation of the intervertebral joints or ulceration of articular cartilage. However, in 1 toad (control toad No. 3), in 2 joints, there was erosion and/or fissuring of articular cartilage. In one of these joints, there was also hemorrhage and a few small clusters of degenerated cells, with small, round, dark nuclei (likely macrophages or synoviocytes) and rare granulocytes.
Histologic examination of a full range of tissues from affected toad Nos. 1, 2, 7, 11, and 13 did not reveal lesions that were consistent among toads, apart from all toads exhibiting mild-to-moderate granulomatous gastritis associated with at least 2 species of nematodes encysted in the submucosa and/or muscular tunics. In affected toad Nos. 11 and 13, there was mild granulomatous hepatitis. In addition, in affected toad No. 11, there was moderate granulomatous cystitis.
Culture of intervertebral swabs from 6 of 9 affected toads produced light or moderate pure growths of Ochrobactrum anthropi (identified by using API 20 NE, typical code 1243374), whereas the swab from 1 affected toad produced a light growth of O. anthropi and Salmonella sp. (identified by using Microbact 24E) (Table 1). The API 20 NE system identified O. anthropi, with an interpretation of very good identification (a 99.9% identification and T index of 0.72) after a 48-hour incubation (apiweb software, bioMerieux, Marcy l'Etoile, France). Two O. anthropi isolates were randomly selected for an extended API 20 NE identification strip incubation period of an additional 48 hours to differentiate O. anthropi from Brucella sp., but there were no changes in their biochemical signatures. The O. anthropi isolates were motile on microscopy, nonacid fast on modified Ziehl-Neelsen stain, and were able to grow on MacConkey agar, further differentiating them from Brucella sp. When a very good classification is reached with the API 20 NE system, for Gram-negative bacteria and O. anthropi in particular, the results correlate well with molecular identification of the isolate. 7,41 The Biolog MicroPlate system was used in an attempt to corroborate the identification of 1 O. anthropi isolate. This system failed to provide a definitive identification but listed O. anthropi as the first probable organism. There was no bacterial growth from intervertebral joints of the 4 control toads. The difference in presence or absence of bacterial growth from intervertebral swabs taken from control versus affected toadswas significant when using a chi-square test (χ2 = 6.74, df = 11, P < .01). The blood cultures of affected toad Nos. 12–15 and control toad Nos. 1–3 were negative. Salmonella spp. was isolated from the blood of control toad No. 4.
Discussion
The range of lesions of intervertebral joints suggested a progression from acute to chronic. An acute lesion could be represented by intervertebral joint 2 in affected toad No. 8, in which the joint space is still evident but with eroded or ulcerated articular cartilage, intense pyogranulomatous inflammation, and early cartilage and bone proliferation (Figs. 10, 11). As lesions appear more chronic, represented by the majority of lesions in this series, the inflammatory infiltrate becomes more focal and granulomatous, with partial or complete fusion of the joint with cartilage and bone (Figs. 7, 8). The joints that were completely fused with minimal associated inflammatory infiltrate, may represent a later stage of the lesion (Fig. 9).
The exact pattern of tissue reaction and inflammation in these lesions will undoubtedly differ somewhat from those of vertebral osteomyelitis and diskospondylitis in mammals, because of the synovial nature of ventral intervertebral joints in cane toads. However, the lesions still share many of the features associated with both spondylitis and bacterial arthritis, as described in various other species. 29,35 Perhaps the most notable difference in the toads was the exuberant cartilage and bone proliferation associated with the lesions, likely a general feature of amphibian tissue reaction. 16
There is a significant positive association between the inflammatory lesions and culture of O. anthropi from the affected tissue. This, and the abundant pyogranulomatous inflammation associated with the lesions, suggests that bacterial infection played a substantial role in the pathogenesis of the condition. Bacterial spondylitis and diskospondylitis are well described in other species, and the predisposition for bacterial localization in subchondral bone or cartilaginous endplates is thought to be because of the presence of capillary loops and vascular sludging in this region. 12,29,42 Diskospondylitis in dogs is associated with various opportunists, including Staphylococcus sp., Streptococcus sp., and Escherichia coli. 8,42 In pigs, diskospondylitis is associated with various bacteremic organisms, such as Erysipelothrix and Streptococcus sp. 12 In humans, pigs, and dogs, Brucella sp. are also commonly associated with diskospondylitis. 6,20,42 In humans, spondylodiskitis and vertebral osteomyelitis are diagnosed with increasing frequency, assisted by advancement of magnetic resonance imaging. Staphylococcus aureus and various Gram-negative bacteria are commonly isolated from these lesions. 21,24 Although the pathogenesis of spinal arthropathy in snakes is controversial, bacteria, most commonly Salmonella sp., are associated with the lesions. 15,19 The varied histologic appearances of the spinal lesions in snakes were proposed to be related to chronicity. 19
The route of infection in vertebral infections and diskospondylitis is usually assumed to be hematogenous from various sources, such as wounds, abscesses, skin infection, or the urinary tract. 8,21,42 Histologic examination of various tissues in 5 affected toads did not reveal consistent lesions among them to suggest a primary source of bacteria; however, the granulomatous cystitis in 1 toad was interesting, given the association of diskospondylitis with urinary tract infection in mammals. 42 The mild granulomatous hepatitis in 2 affected toads could be related to intervertebral joint infection, either as a source or the result of periodic bacteremia. Gastric mural parasitic granulomas similar to those seen in affected toads are also common in free-ranging cane toads that do not have spinal arthropathy (C. Shilton unpublished) and are of dubious significance to the pathogenesis of spinal arthropathy.
Spondylitis occurs subsequent to bacterial septicemia in poultry and was associated with positive blood cultures in various species, including snakes, mink, and dogs. 2,4,9,19,26 In affected toad Nos. 12–15 from which O. anthropi was recovered from intervertebral lesions, the blood cultures were negative, indicating that concurrent bacteremia was not present. However, this did not negate a possible hematogenous route of infection, given the small number of blood cultures performed and the chronic appearance of the majority of lesions at the time of culture.
O. anthropi (formerly known as “Achromobacter” CDC group Vd) is a Gram-negative soil organism, closely related to Brucella sp. but considered to be of low virulence. 14,18,45 Published reports of the organism causing this disease describe occasional opportunistic infections in humans, primarily associated with immunosuppression or other predisposing factors, such as prosthetic heart valves or indwelling catheters, and a few cases of osteomyelitis or arthritis in humans. 32,44 We are not aware of published reports of the bacterium causing disease in other animals. The only published reference to O. anthropi in association with amphibians is its isolation, along with other species of bacteria, from the skin of free-ranging newts involved in a mortality event, although the investigators dismissed it as a likely contaminant. 5 Bacterial infections in captive amphibians in general are often considered to be opportunistic by Gram-negative environmental bacteria and associated with stressors such as changes in husbandry or inappropriate temperature. 40 Given the isolation of O. anthropi in pure culture from intervertebral lesions in most toads cultured in this study, this bacterium may either have a predisposition for bone or joint tissues or may have a unique ability to evade toad immune defences and gain access to the blood stream.
In addition to the spinal lesions with pyogranulomatous inflammation described above, there were changes in grossly normal joints of toads with spinal arthropathy and in one of the normal control toads that were similar to osteoarthritic degenerative changes seen in mammals. 23,29,35,46 These changes included thinning; erosion; and ulceration of articular cartilage, with plugging of defects with fibrocartilage, fissuring, and necrosis of cartilage; and proliferation evidenced by multinuclear chondrones (Figs. 12–14). It is interesting to speculate that these early degenerative changes may predispose toads to infection. This pathogenesis has been proposed in various species, although it appears poorly understood. The proposed pathogenesis centers around vascular damage, with alterations in blood flow or hemorrhage that may occur as cartilage degenerates or becomes necrotic, or subchondral bone is damaged or undergoes microfracture. 4,8,24
Cane toads have evolved to disperse more rapidly since their introduction to Australia. 31 The increased levels of activity of toads at the invasion front raises the possibility that excessive physical activity is predisposing to degenerative joint changes, as it does in humans. 23 Several interactions could be occurring in toads. Immunosuppression is associated with increased likelihood of spinal infection in several species. 9,21,42 The physiologic stresses in an invasive species adapting to a new, potentially hostile, marginally suitable, environment or allocating energy resources to migration may be among the possible causes of immunosuppression that allow O. anthropi to elude toad immune defences and establish infection in early degenerate spinal tissue.
Footnotes
Acknowledgements
We thank John Humphrey for helpful comments on the manuscript; Haley Bowcock and Jerry Hemphill for toad collection; and the staff of Beatrice Hill Farm for property access. Litchfield Veterinary Clinic assisted in radiographing toads during preliminary investigations. Funding provided by the Australian Wildlife Health Network facilitated identification of the index case. Funding for further investigation of toad spinal arthropathy was provided by the Australian Research Council.
