Abstract
Melanosis of the urinary bladder is a very rare condition characterized by an abnormal black or brownish-black pigmentation of the organ. The pigmentary disorder can involve both the urothelial cell layers and/or the submucosa. The biologic potential of the melanosis of urinary bladder remains unknown because only a few cases have been reported in medical literature. So far melanosis of the urinary bladder is not known to occur in cattle. Here we describe the first case of melanosis of the urinary bladder in an inbred red-spotted, 7-year-old cow. Light, ultrastructural, and immunohistochemic investigations demonstrated melanin pigment in the submucosa and lamina propria but not the urothelium of the bladder. In addition, biochemical characterization of the pigment-laden cells demonstrated that the pigment of this disorder consisted mainly of eumelanin, thus corroborating the morphologic studies. Finally, virologic examination revealed the presence of bovine papillomavirus type 2.
Keywords
Melanosis of the urinary bladder is a very rare pigmentary disorder characterized by an abnormal black or brownish-black pigmentation of the organ. Since Alroy et al. 1 reported the first case of melanosis of the urinary bladder in man, only a few additional cases have been described. 4,6,10 In all of these cases, abnormal pigmentary deposits primarily involve urothelial cell layers. A very rare association between melanosis and malignant melanoma in the genitourinary tract has also been reported in human medicine. 8
The aim of the present paper is to describe light, immunohistochemic, ultrastructural, virologic, and biochemical findings of not previously reported melanosis of the urinary bladder observed in an inbred red-spotted, 7-year-old cow. The animal, after clinical evaluation, was slaughtered at the public slaughterhouse and found fit for human consumption. At the postmortem examination, the only pathologic findings were limited to the urinary bladder, a wide mucosal surface of which showed an abnormal black pigmentation. No similar lesions were seen in other organs, including meninges, known to be frequently anatomic sites of pigmentary disorders. 7 For histologic studies, several bladder samples were fixed in 10% buffered formalin and embedded in paraffin following routine procedures. Five-micron-thick sections were stained with hematoxylin and eosin (HE). For immunohistochemistry, the streptavidin-biotin-peroxidase technique with the following antibodies was used: S-100 (Biomeda, USA), Melan-A (Dako, Denmark). Briefly, deparaffinized sections were bleached with 0.25% potassium permanganate for 4 hours followed by 1% oxalic acid to avoid interferences of the diaminobenzidine immunoreactions with the melanin pigment; 5 the sections were blocked for endogenous peroxidase in 0.3% H2O2 in methanol for 20 minutes. Antigen enhancement was performed by pretreating with microwave heating (twice for 5 minutes each at 750 W). Slides were washed 3 times, then incubated with secondary antibody (biotinylated goat antirabbit IgG, Dako) for 30 minutes. Color development was obtained with 5–20 minutes of diaminobenzidine treatment. Sections were counterstained with hematoxylin. The primary antibody was omitted and replaced by phosphate-buffered saline in the corresponding negative control sections.
For ultrastructural investigations, bladder samples were immediately fixed in 4% paraformaldehyde in buffer phosphate. For transmission ultrastructural studies, they were postfixed in 1% osmium tetroxide in the same buffer, dehydrated in graded alcohol, and embedded in Spurr resin. Semithin sections were stained by 1% toluidine blue in water solution. The ultrathin sections obtained from chosen areas were counterstained by lead citrate and uranyl acetate and observed with a 902 Zeiss transmission electron microscope.
For scanning ultrastructural studies, samples were dehydrated in graded alcohol without any postfixation; they were critical point dried and, after mounting on aluminium stubs, they were sputtered by a gold–palladium mixture. The samples were observed with a Cambridge stereoscan 240 scanning electron microscope.
For the detection of bovine papillomavirus type 2 (BPV-2) DNA, specific primers for the E5 region were designed by Beacon Designer 2.0 software (Premier Biosoft International, USA). The forward primer BPV-2NS (5′-TACTGTTTCTGCTGCTATTT-3′) and the reverse primer BPV-2NAS (5′-ACAAATCAAATCCACATAATAGTA-3′) are known to amplify a fragment of 125 bp from 3943 to 4067 of complete genome of BPV-2. In each experiment, a blank sample consisting of reaction mixture without DNA and a positive sample consisting of BPV-2–cloned DNA (kindly provided by Dr. M. S. Campo) was included.
In order to identify the pyrrole-2,3,5-tricarboxylic acid (PTCA), the chemical marker of eumelanins, formalin-fixed tissues were treated with a 2:1 v/v chloroform methanol mixture for 2 hours, then dried at room temperature for 3 hours. The pigmented areas were carefully selected, weighted, and subjected to degradation according to the procedure previously reported. 8 In brief the tissue was suspended in 1M NaOH (0.1 ml/mg tissue) containing 1.5% H2O2. The reaction mixture was allowed to stand at room temperature with stirring for 24 hours, and then treated with 5% hydrogen sulphite (200 μl/ml), acidified to pH 4.0 with phosphoric acid and filtered through a 0.45-μm nylon membrane. The supernatant was analyzed by HPLC using a Shimadzu instrument equipped with a multiwavelength ultraviolet detector set at 280 nm. The mobile phase was as reported, an octadecylsilane column (Sphereclone, 25 cm × 0.46 cm, 5-μm particle size) was used at a flow rate of 0.7 ml/min. The yields of PTCA were estimated by measurement of peak areas and comparison, with external calibration curves obtained with an authentic sample prepared as reported. Analyses were run in triplicate. Data are expressed as the mean of 3 separate experiments ± SD.
Macroscopically, a diffuse black pigmentation making a wide mucosal surface abnormally thickened and rough was clearly evident (Fig. 1).
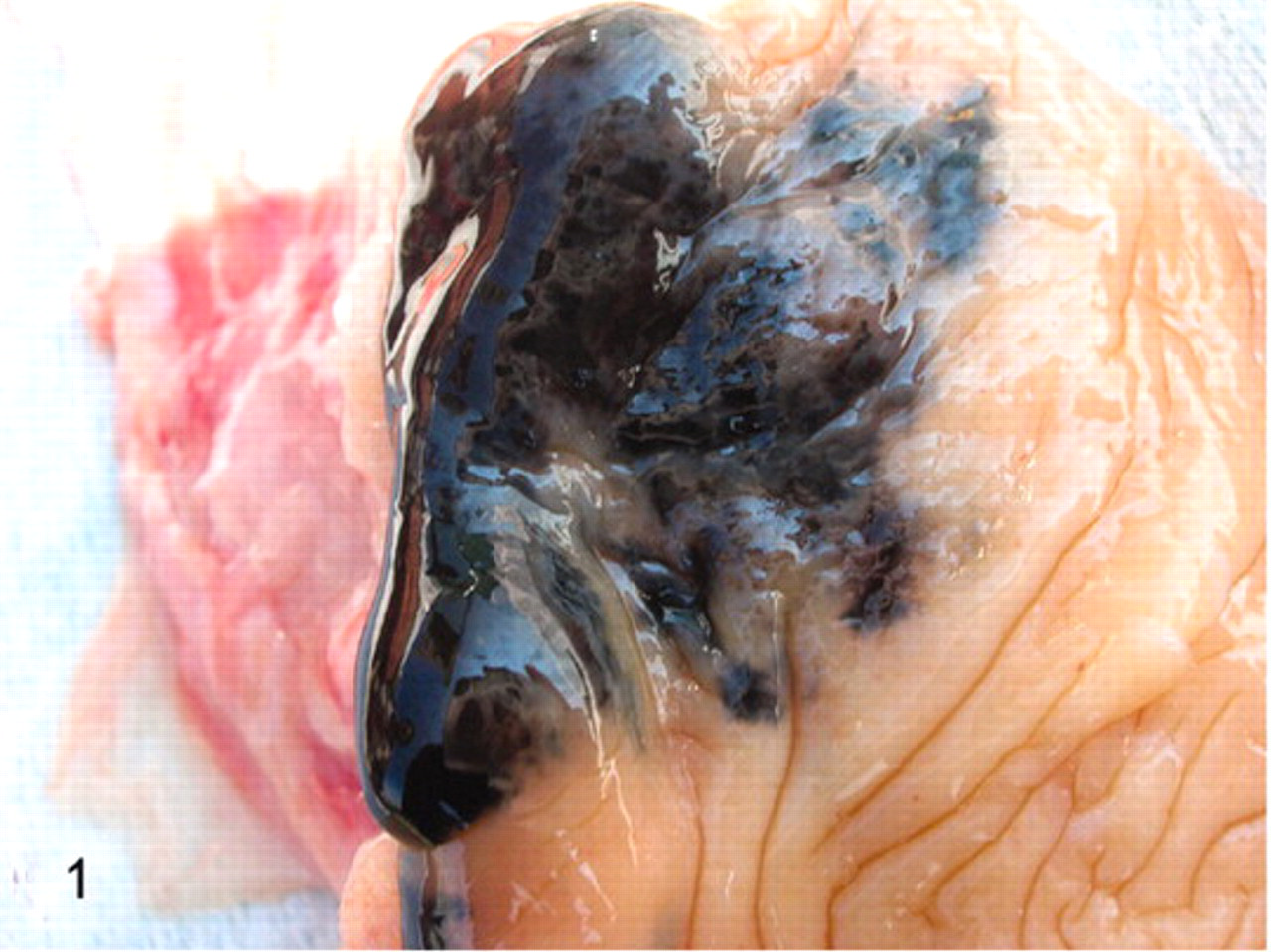
Urinary bladder; cow No. 13090. A diffuse black pigmentation appears to be macroscopically evident.
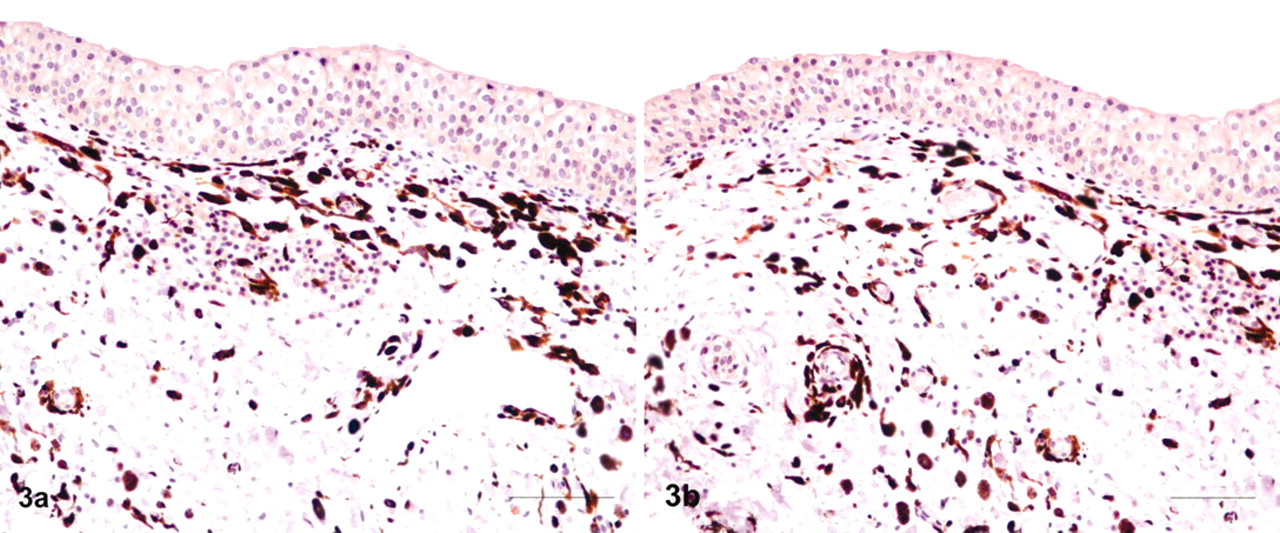
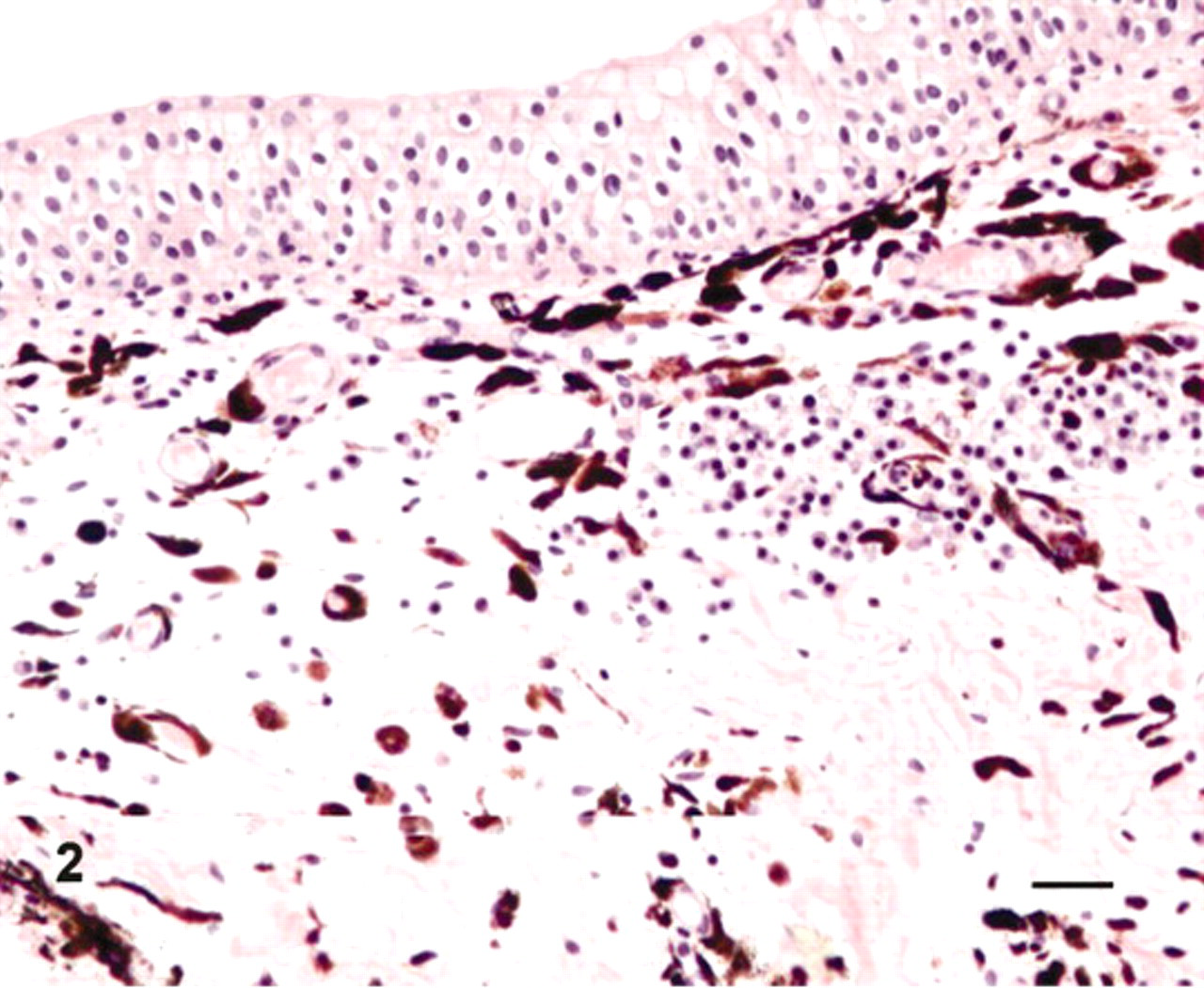
Microscopically, pigment-laden cells were identified as melanocytes and melanophages immediately beneath the basement membrane and around von Brunn nests, which are invaginations of the surface urothelium into the underlying lamina propria. An inflammatory process characterized by a few aggregates of lymphoid and mast cells as well as a mild edema and scattered small hemorrhages were also evident in the lamina propria of the mucosa (Fig. 2). Furthermore, pigment-containing cells were heavily scattered all over the submucosa. Pigment granules were also detected in the cytoplasm of the endothelial cells and the pericytes as well. No pigmented cells were seen among urothelial cell layers. Immunohistochemically, most cells were positive for S-100 and melan A, well-known melanocytic markers (Fig. 3A, B).
Urinary bladder; cow No. 13090. Anti-melan A (
Urinary bladder; cow No. 13090. Numerous pigment-containing cells are scattered in bladder wall without any involvement of the mucosa. Notice lymphoid and mast cell aggregates under the lamina propria. HE. Bar = 25 μm.
Ultrastructurally, the cytoplasm of all the pigment-containing cells was filled with stage IV melanosomes and, only occasionally, stage III melanosomes. These organelles were characterized by a strong pleomorphism and showed a remarkable size variability. Stage I and II melanosomes were never detected. Even if solitary melanosomes were observed, compound melanosomes, that is, intensely electron-dense rounded or elliptical melanosomes enveloped by a common limiting membrane, were the prevailing submicroscopic aspect of these organelles in the melanophages (Fig. 4). These complexes were also seen in the melanocytes, the endothelial cells, the pericytes, and some mast cells. Some giant melanosomes were also seen. Neither premelanosomes nor melanosomes were observed in the urothelial cells. No aberrant ultrastructural melanosomes as well as nuclear atypia and/or mitoses were seen in the pigmented cells. Pigment-containing cells were also investigated by scanning electron microscope interfaced with a dispersive energy spectrometer. Cytoplasmic overloading with melanosomes was responsible for clusters of 3-dimensional spheric structures projecting from some pigmented cells mostly in and around some vessels (Fig. 5). Light and ultrastructural findings showed an untransformed phenotype.
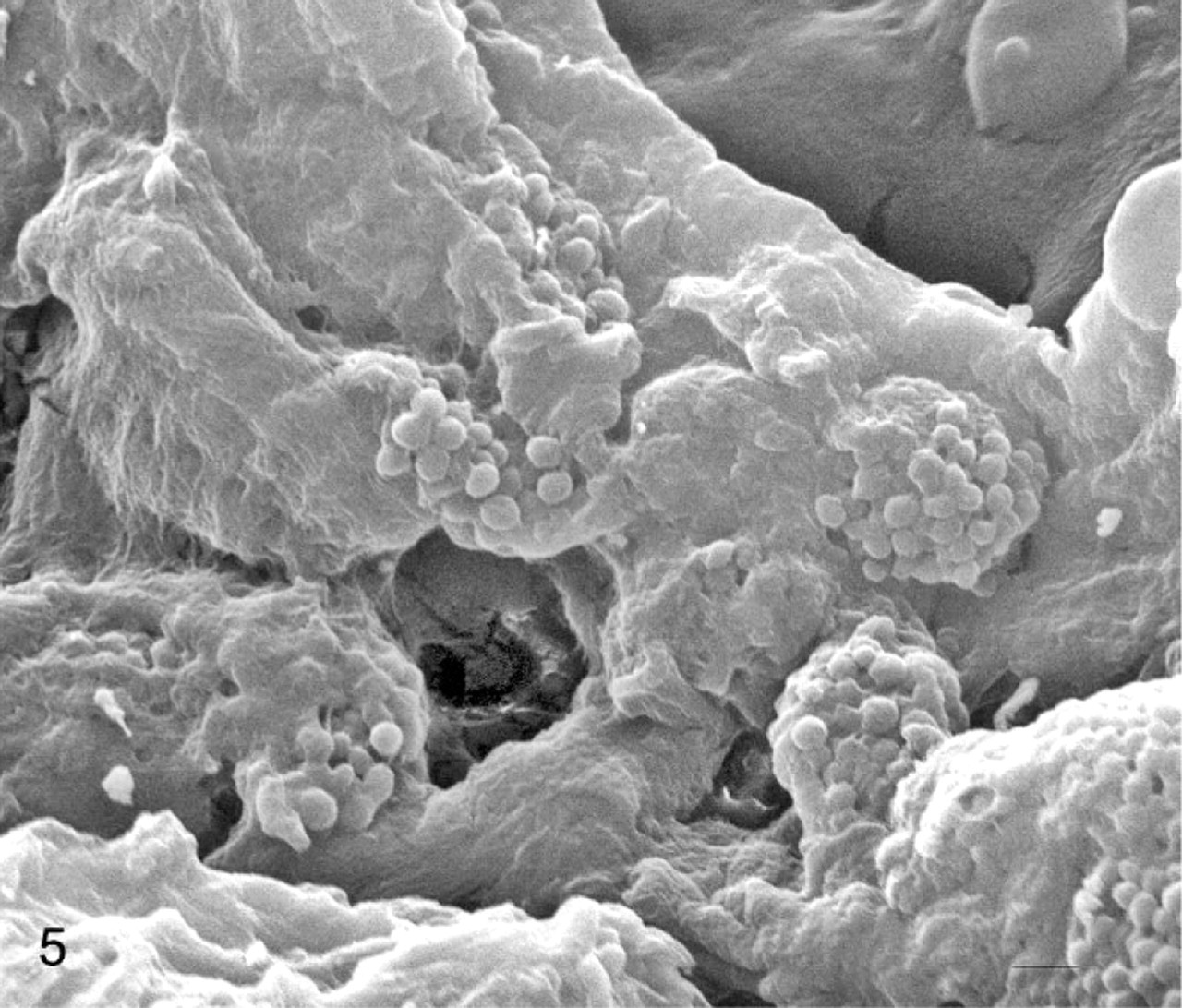
Urinary bladder; cow No. 13090. Scanning electron micrograph demonstrating numerous pigment-containing cells mostly located around some vessels with projecting spheric structures. Bars = 0.5 μm.
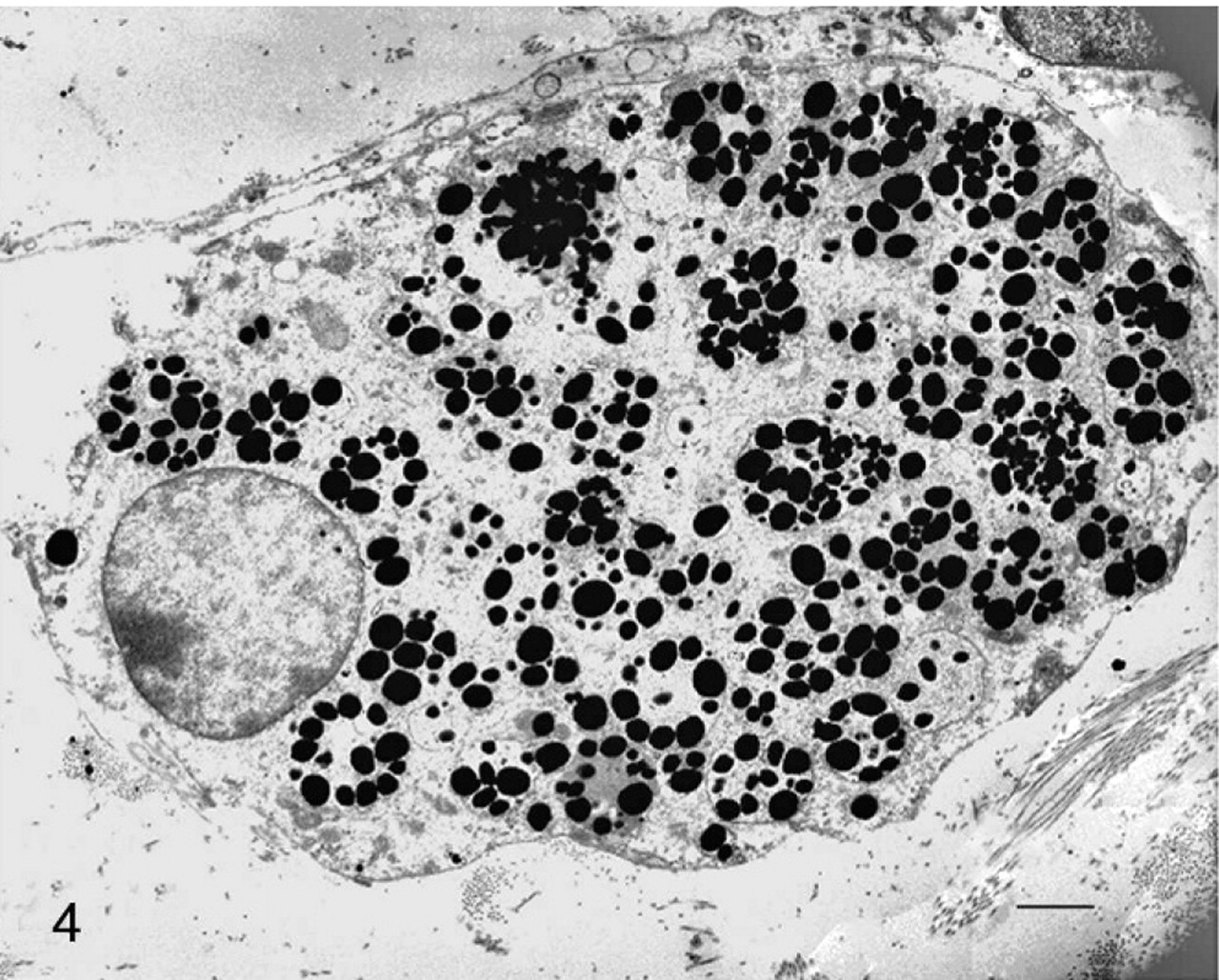
Urinary bladder; cow No. 13090. Transmission electron micrograph demonstrating pigment-containing cells showing single and compound stage IV melanosomes. Bars = 0.5 μm.
Virologic analysis showed a band corresponding to the amplified sequences of BPV-2 in the pigmented samples (Fig. 6). To validate the PCR result, the amplified products were also sequenced.
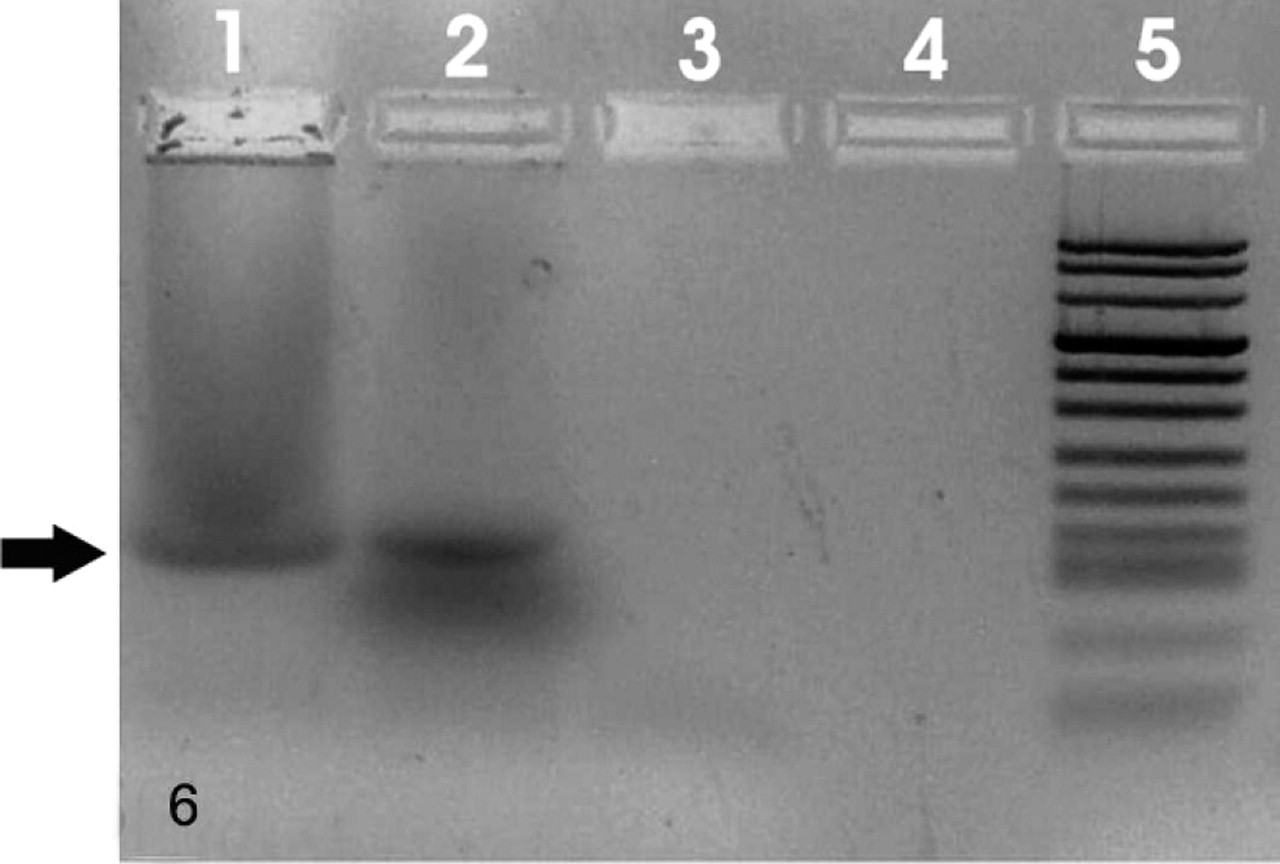
Urinary bladder; cow No. 13090. Detection of BPV-2 DNA sequences. An amplicon of 125 bp is evident in pathologic sample. Lane 1: positive control; lane 2: sample from melanotic bladder; lanes 3–4: samples from normal bladder (negative controls); lane 5: molecular size marker (DNA Molecular Weight Marker VIII, DIG-labeled, Roche Applied Science, USA).
Oxidative degradation of the formalin-fixed tissue showed the formation of PTCA, the typical marker of eumelanins. This was identified and quantified in yields of approximately 5.67 ± 0.6 μg/mg of pigmented tissue by HPLC analysis (Fig. 7). On the other hand, no detectable amounts (<0.01 μg/mg of tissue) of 1,3-thiazole-2,4,5-tricarboxylic acid or 6-(2-amino-2-carboxyethyl)-2-carboxy-4-hydroxybenzothiazole, typical markers of the reddish sulphur-containing pheomelanins, were obtained. Control experiments were run on formalin-treated and untreated skin tissues to assess possible interference of products deriving from degradation of the fixed tissue on the analysis of the melanin markers.
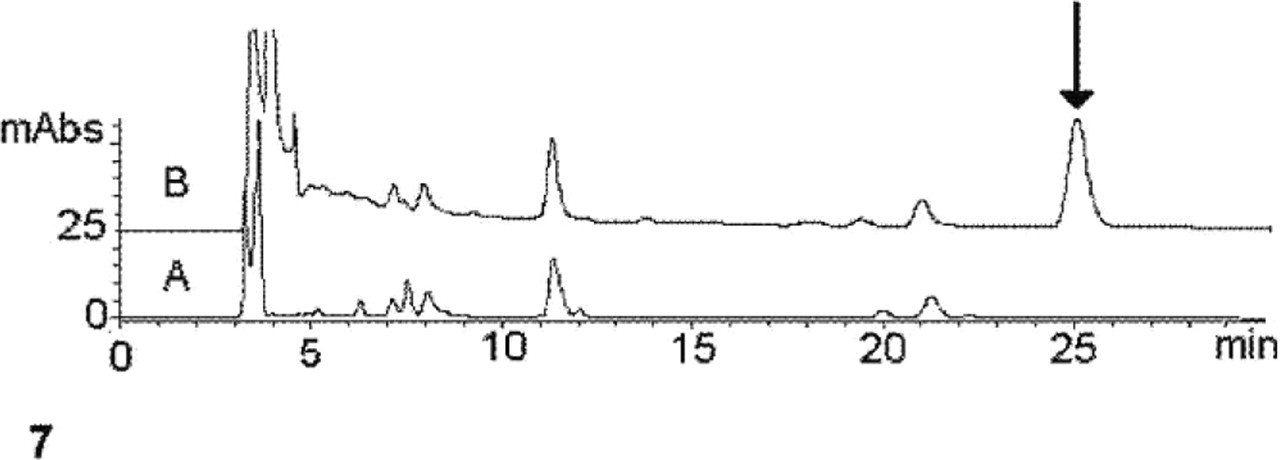
Urinary bladder; cow. HPLC chromatogram of the degradation mixture obtained from the (A) nonpigmented and (B) pigmented regions of the urinary bladder. Arrow denotes the elution peak of PTCA.
The term “melanosis” identifies a group of conditions characterized by an abnormal black or brownish-black pigmentation of an organ or part. It does not imply that the pigment is melanin. In melanosis coli the pigment granules contain lipofuschin, in melanosis ilei pigment granules may contain hemosiderin, and melanosis esophagi is characterized by melanin pigment.
Both non-neoplastic and neoplastic melanocyte lesions of the urinary bladder are very rare in comparative pathology, including man. We report a case of bladder melanosis in a cow that, to the best of our knowledge, has not been reported before. Furthermore, microscopic, immunohistochemic, submicroscopic, virologic, and chemical findings make this case unique because some remarks have been carried out for the first time in medical literature.
However, both morphologic and chemical investigations allow us to show that the abnormally accumulated pigment is clearly composed of melanin, particularly eumelanin. Microscopic and submicroscopic patterns of this case appear to be quite different from the ones reported in man. We describe a pigmentary disorder that appears to take place in the lamina propria and the submucosa without any involvement of urothelial mucosa, the latter being a site of pigmentary accumulation in all of the 7 cases of bladder melanosis described in man. 1,4,6,10 In addition, we did not identify any premelanosomes as was described in humans. 10 A peculiar ultrastructural feature of our case was the presence of compound melanosomes, both as heterolysosomes and autolysosomes. They can be interpreted as an ultrastructural aspect of the melanosome disintegration/degradation pathway. It is worthwhile noting that melanosome disintegration is known to occur both in autophagosomes of melanocytes and in heterophagosomes of macrophages as well as of endothelial cells and mast cells of several pigmentary disorders. 2 Compound melanosomes have never been reported in melanosis of the urinary bladder. Indeed, these complexes were well-documented by Yamazaki et al. 13 in melanosis esophagi in man, with which our case shares many ultrastructural findings. We detected the presence of BPV-2, a common agent responsible for severe bladder pathology in cattle, including cancer. 3 It is reasonable to believe that the mild interstitial cystitis we found may be related to BPV-2 but not to melanosis, although abnormal pigmentations of the urinary bladder associated with chronic interstitial cystitis are known to occur. 11
The biologic potential of bladder melanosis is unknown. It has been suggested that melanosis of the urinary bladder might represent a premalignant lesion. 4 Malignant melanoma arising in vesicovaginal melanosis has also been described. 8 Furthermore, the only recent follow-up study shows the potential severity of the lesion. It has been recommended that patients with bladder melanosis undergo periodic cystoscopies and repeated biopsies as therapeutic investigation in the follow-up study of the lesion. 4,6,10
We suggest that determining the type of melanin by biopsy examination and/or voided urine since melanuria can be seen in patients suffering from melanosis of genitourinary tract 12 is a further intriguing procedure useful for monitoring the clinical progression of melanosis of the urinary bladder, thus evaluating its potential oncogenic risk.
Footnotes
Acknowledgments
This work was supported in part by grants of the Italian Ministry of University and Scientific Research and the Regione Calabria, Dipartimento Agricoltura, Foreste, Forestazione, Caccia e Pesca. We wish to thank Dr. La Rizza Rosario for his helpful contribution in providing this case.
