Abstract
A 6-year-old, sterile, Blanca Celtibérica breed adult doe was referred to our faculty. The doe had external female genitalia, a short anogenital distance, and normally shaped udders. Masculinization signs in the head shape and male behavior were also noted at the time of referral. Genetic analysis demonstrated normal 2n = 60 XX karyotype and an absence of the sex-determining region Y (SRY). The animal was homozygous for a DNA deletion responsible for the Polled Intersex Syndrome (PIS). A uterus and 2 uterine horns were present at the postmortem examination. Gartner's ducts and degenerated Wolffian derivatives persisted. There were 2 intra-abdominal testicle-like structures, one of which consisted of epididymal and deferent ducts. An advanced Leydig cell tumor, resulting in almost total destruction of the intratesticular structures, was also observed. Leydig cell tumors usually produce testosterone. Thus, these histologic findings are compatible with the evident virilization.
Polled Intersex Syndrome (PIS) is a cause of intersexuality in goats. Affected does show abnormal horn development and intersexuality, resulting in sterility. The genetic bases of PIS have been previously described. 8 Briefly, the PIS gene regulates the transcription of 2 genes involved in gonadal development: PIS-regulated transcript 1 (PISRT1) and forkhead transcription factor gene (FOXL2). This regulatory function is lost when a 11.7-kb deletion occurs in the PIS gene in homozygosis. As a result, XX embryos affected by PIS are masculinized.
Detailed descriptions of prepubertal and pubertal PIS-affected animals are reported in the literature. 10 In all cases, testicular-like structures (seminiferous tubules, Sertoli cells, and intertubular tissue) exist, whereas lack of germ cells and ovarian structures is reported.
Because they are sterile, PIS does are usually slaughtered at a young age, and data on their vital evolution are not available. We report a detailed, genetic, anatomic, and histopathologic description of a sterile 6-year-old Blanca Celtibérica doe maintained in an ex situ conservation program. Its external genitalia were found to have a female phenotype, with a normally shaped udder. The anogenital distance was 6 cm. It showed a 25-cm beard, even though Blanca Celtibérica females are usually beardless or show very short beards (up to 5 cm). The dorsal hair also resembled that of a buck. There were no horns (Fig. 1), and the animal exhibited masculinized behavior, fighting with the bucks and trying to mate with other does.

External aspect of the PIS affected doe. Polledness, long beard, and male aspect of hair and head are visible.
Chromosome analysis of this doe was based on the study of 50 metaphase plaques obtained by following standard procedures. All of them were 2n = 60XX. A polymerase chain reaction (PCR) analysis was also performed to discard the presence of the sex determining region (SRY) in a translocated position. SRY was partially amplified by means of the primers GCTTAGAGCCTGGGCTTTCT and TTTCACAGTGGGAACAAAAGG (both in 5′→3′ direction) to obtain a 660 bp fragment. MgCl2 concentration was 1.5 mM. PCR cycles were: 94°C, 4 minutes + 36 x (94°C, 30 seconds; 63°C, 30 seconds; 72°C, 1 minute). Normal and fertile does and bucks were used as positive and negative controls in the amplification protocols. SRY was not detected in the affected doe (Fig. 2B).
A PCR reaction was also designed to amplify DelE, a central 147-bp DNA fragment of the region deleted in the PIS animals. Primers were TTCCACTGCTTTTGGTGTGT and AACAAGAGAGGTGCCCTGAA (both in 5′→3′ direction). MgCl2 concentration was 1.5 mM. Amplification cycles were: 94°C, 4 minutes + 36 x (94°C, 30 seconds; 63°C, 45 seconds; 72°C, 10 seconds). As can be observed in Fig. 2A, no amplification was obtained for DelE in the affected doe while the expected 147-bp DNA fragment was obtained in the control animals.
Because neither DelE nor SRY are expected to be amplified in PIS affected does, the OarFCB0020 microsatellite was used as an additional control. 3 This marker was correctly amplified in the DNA sample obtained from the affected animal; therefore, correct laboratory functioning was verified (Fig. 2C).
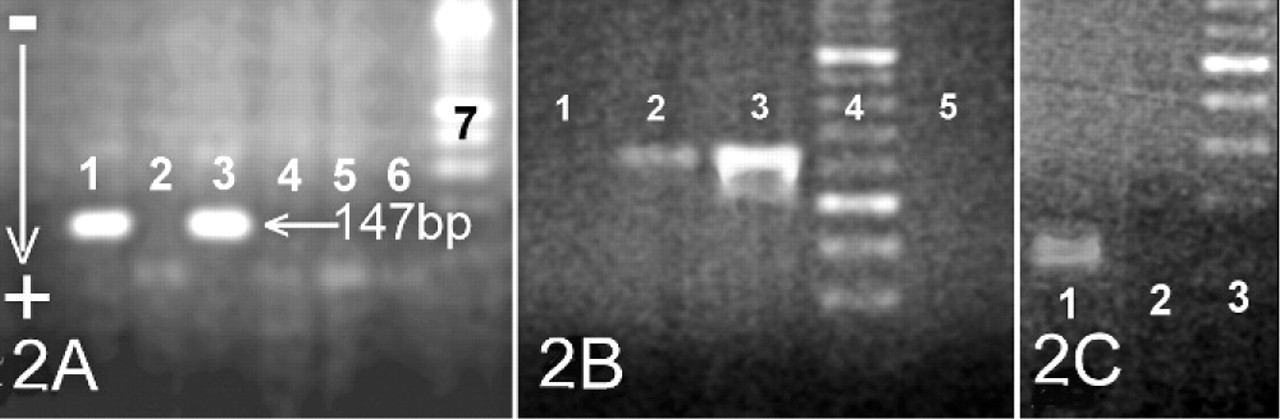
PCR products analysis.
It was concluded that the animal was a 2n = 60 XX, lacking the DelE region, the Y chromosome and the SRY region—in other words, a doe affected by PIS.
After euthanasia, opening of the abdominal cavity revealed a uterus, uterine horns, and 2 oval structures. However, Fallopian tubes and ovaries were not present.
The first oval structure (10 × 12 cm) was located on the right side and cranial to the right uterine horn. It was attached to the mesosalpinx by a prominent vascular plexus and covered by a thick, smooth, bright and highly vascularized capsule. The cut surface showed a trabecular structure consisting of irregular sheets that branched and linked each one with the others, forming intercommunicating spaces filled with thick, brown-colored blood. This structure collapsed as the content of the spaces flowed out. The histologic study confirmed that the capsule had the same structure as that of the testicular tunica albuginea, including the presence of the tunica vasculosa, the innermost layer of the testicular capsule (Fig. 3A). Furthermore, we observed fibrous projections from the capsule, similar to the septa that usually support normal testicular parenchyma (Fig. 3B). This oval structure was clearly related to a testicle. The inside of the right oval structure consisted of a large tumor composed of polyhedral cells. These cells had abundant eosinophilic cytoplasm with small or large spaces caused by lipid droplets (Fig. 3C). Nuclei were centrally located, round-shaped, regular in size, with single nucleoli. These cells were identified as interstitial cells (Leydig cells). Mitotic cells were rarely seen. Two different neoplastic growth patterns were observed: solid and cystic types. In the solid tumor areas, cells were diffusely arranged and formed masses surrounded by thin bands of collagenous connective tissue (Fig. 3D). Focal necrosis (Fig. 3B) and hemorrhage (Fig. 3A) were common. In the cystic areas, interconnecting strands of tumor cells surrounded spaces filled with homogeneous and amorphous eosinophilic material, often including erythrocytes (Fig. 3E).
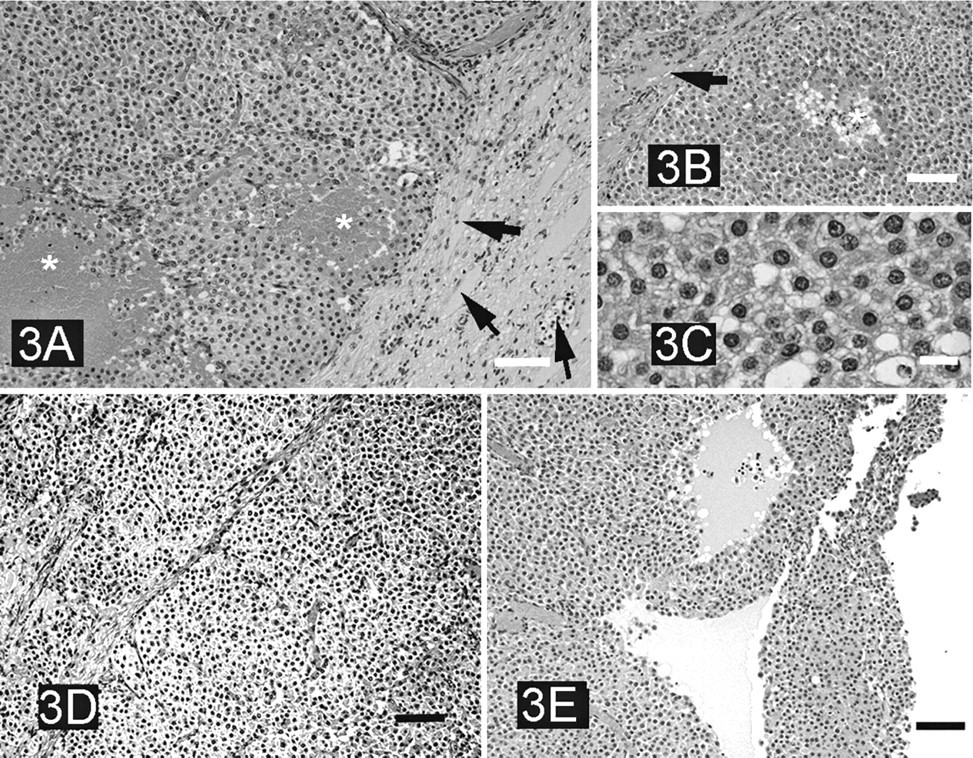
Interstitial (Leydig cell) tumor in the affected doe.
The second abdominal oval structure (4–4.5 cm diameter) was located on the left side and showed a “wrinkled” surface. It contained a single cyst encapsulated by a thick, firm, whitish wall, with areas of calcification. As in the right oval structure, solid and cystic interstitial Leydig cell tumor areas were observed around the large central cyst. Two peduncles had their origins in the left oval structure. One of them was composed of blood vessels resembling a pampiniform plexus. The other peduncle consisted of convoluted ductules similar to those forming the coni vasculosi in the head of the epididymis. Incompletely developed ductus epididymidis, lined by ciliated cylindrical cells, were embedded in abundant connective tissue. This indicated incomplete involution of the mesonephric duct (Wolff's duct).
Several cysts, ranging from 1–4 mm in diameter, were found along the cranial portion of the uterine horns. Two symmetric, solid, cordlike structures (1–1.5 mm in diameter) were arranged parallel to the length of uterine horns and to the lateral sides of the body of the uterus. They were identified as vestiges of the Gartner's duct. The thickness of the endometrium and myometrium was notably reduced.
PIS is not the only origin of goat intersexuality. Chimerism, usually caused by freemartin syndrome in opposite-sex twin pregnancies, also results in intersex animals, and even in true hermaphroditism in sheep and goats. 1,2 Polledness is not linked to chimerism.
Testicular neoplasia is usually caused by the proliferation of germ cells (more than 95%). Leydig cell tumors are uncommon; they only account for 1–3% of all testicle tumors. 7 To our knowledge, this is the first report on Leydig cell tumor in goats. PIS-affected goats do not carry spermatogonia; therefore, germ cells tumors should not be found in this kind of animal. 8,10 Extensive information is available on testicular tumors in humans and animals. Leydig cell tumors occur in domestic animals either in older age groups or in cryptorchid testicles. 5 Indeed, cryptorchidism is the most significant risk factor for testicular cancer. The ectopic testicle is up to 13 times more susceptible to neoplasia than a normally situated one. 6 Enlarged abdominal structures from male genital origin, compatible with neoplasia, have also been described in caprine chimeras. 9
Since Leydig cells secrete testosterone, they may be responsible for the hyperandrogenism symptoms observed in this case. Leydig cells appear to be active in 3-month-old XX PIS kids, ensuring testosterone secretion from an early age. 10 Different authors have reported reduced and increased levels of steroid synthesis in Leydig cells of intra-abdominal testes in bucks. 4 Some Leydig cell tumors have been shown to produce excess androgens. 5 The affected animal described in the present report is a XX female. Increase in testosterone levels caused by the tumor could have resulted in signs of masculinization.
