Abstract
Toxicity following ingestion of the vagrant, foliose lichen Xanthoparmelia chlorochroa was identified as the putative etiology in the death of an estimated 400–500 elk on the Red Rim-Daley Wildlife Habitat Management Area in Wyoming during the winter of 2004. A single, unsubstantiated report in 1939 attributed toxicity of X. chlorochroa in cattle and sheep to usnic acid, a common lichen secondary metabolite. To test the hypothesis that usnic acid is the proximate cause of death in animals poisoned by lichen, domestic sheep were dosed PO with (+)-usnic acid. Clinical signs in symptomatic ewes included lethargy, anorexia, and signs indicative of abdominal discomfort. Serum creatine kinase, aspartate aminotransferase, and lactate dehydrogenase activities were considerably elevated in symptomatic sheep. Similarly, only symptomatic ewes exhibited appreciable postmortem lesions consisting of severe degenerative appendicular skeletal myopathy. The median toxic dose (ED50) of (+)-usnic acid in domestic sheep was estimated to be between 485 and 647 mg/kg/day for 7 days.
On February 6, 2004, the Wyoming Game and Fish Department (WGFD) received a report of 2 recumbent, presumably paralyzed, cow elk (Cervus canadensis) in the Red Rim-Daley Wildlife Habitat Management Area (RRWHMA) 10 miles southwest of Rawlins, Wyoming. This report marked the beginning of a 7-week episode in which an estimated 400–500 elk were similarly stricken and subsequently died. Clinical signs included red urine, ataxia, and muscular weakness that rapidly progressed to recumbency. Affected elk were usually discovered in sternal or lateral recumbency. Although the elk were unable to rise, they displayed normal cognition when approached. Once recumbent, they did not recover. Death resulted from dehydration, predation, or euthanasia. Myodegeneration was observed in elk recumbent for 2 or more days. Whether muscle damage was the primary cause of recumbency or secondary to exertion or recumbency could not be determined. Poisoning from ingestion of the lichen Xanthoparmelia chlorochroa (University of Nebraska at Omaha Lichen Herbarium #15631) was identified as the putative cause (Fig. 1). 6

Xanthoparmelia chlorochroa, the lichen putatively incriminated as the cause of elk mortalities during February 2000 in Wyoming.
Xanthoparmelia chlorochroa has been cited as an indicator of good pronghorn antelope range, as well as a source of forage during particularly harsh or long winters. 3,24 Cattle, sheep, pronghorn antelope, and elk have all been reported to consume X. chlorochroa. 2,6,24 Accounts of any lichen species being toxic in ruminants are extremely rare. Beath (1939) reported poisoning of Wyoming cattle and sheep by a lichen species identified as Parmelia molliuscula, now classified as X. chlorochroa. 11 The report by Beath describes a syndrome clinically similar to that seen in the RRWHMA elk. 2,16
Beath (1939) reported that the lichen contained 1.6% usnic acid (UA), a dibenzofuran secondary metabolite, which was speculated to be the toxic component. 2,14 In a subsequent feeding experiment, a lichen extract, purportedly containing only UA, as well as the portion of lichen remaining after carbon bisulfide extraction, was fed to 2 separate groups of animals. Animals receiving the UA extract showed signs similar to those observed in animals naturally affected by lichen. No clinical signs were noted in animals fed the lichen residue. 16 Many key points in this feeding trial are vague. The species of experimental animals is not mentioned, and it is unclear how UA was extracted, identified, or quantified. It is probable that along with UA, other compounds were extracted and subsequently fed.
The pathophysiologic effects of UA have not been well described either in vitro or in vivo, and only monogastric species have been used for testing. The limited data available suggest that (+)-UA is hepatotoxic and works by inhibiting hepatocellular mitochondrial respiration and oxidative phosphorylation, resulting in an increase of free radicals. 1,7,12,14 Reported intravenous lethal dose (LD50) include 25 mg/kg, 30 mg/kg, and 40 mg/kg in mice, rats and rabbits, and dogs, respectively. 14
To test the hypothesis that UA is the proximate toxin in X. chlorochroa responsible for the syndrome seen in lichen-poisoned animals, the UA concentration of toxic lichen was quantified and domestic sheep were used as a surrogate ruminant model. Ewes were dosed with purified (+)-UA following an up-and-down procedure for acute toxicity testing as previously described. 4
Materials and Methods
Extraction and quantitation of (+)-usnic acid
X. chlorochroa was collected by hand from the RRWHMA in March 2004, shortly after the elk mortality. Lichen was allowed to air dry on an outdoor concrete pad. Once dry, lichen was placed in sealed 50 gal containers and stored at ambient temperature.
UA was extracted from the toxic lichen for analysis as follows. Coarsely torn X. chlorochroa (30 g) was soaked in 500 ml acetone for 30 minutes. The extract was filtered (Whatman 4, Whatman Inc., Florham Park, NJ) and taken to dryness using a Rotavapor (Bűchi, Postfach, Switzerland). The extract was then dissolved using 100 ml dichloromethane and partitioned against 150 ml of deionized water. The dichloromethane fraction was evaporated to dryness under nitrogen, reconstituted in 200 ml acetone, and then transferred to a beaker covered by a watch glass. The acetone was allowed to evaporate slowly over approximately 1 week, after which time yellow crystals appeared. The crystals were removed from the beaker, placed on filter paper, and washed with ice-cold acetone.
The enantiomeric form of UA found in X. chlorochroa was determined via polarimetry (PerkinElmer model 341, Wellesley, MA). For the polarimetry reading, conducted at room temperature, 10 mg each of 98% pure (+)-UA (Sigma-Aldrich Co., St. Louis, MO) and UA crystals extracted from X. chlorochroa, as described above, were dissolved in 5 ml chloroform.
The UA concentration of the toxic lichen was determined using a gas chromatography-mass spectrometer (GC/MS) (CP-3800 Saturn 2200 Varian, Walnut Creek, CA) containing a 30-m Varian Factor Four column with an ID of 0.25 mm (CP 8944, Varian). Helium was the carrier gas at a flow rate 1 ml/min. The injector temperature was 200°C, and 1-μL injections were made for both the standards and the sample. The column oven initial temperature was 170°C/min (held 1 minute) and was ramped to 320° at 20°C/min (held 3.5 minutes). The trap temperature, manifold temperature, and transfer line temperature were 170°C, 40°C, and 250°C, respectively. X. chlorochroa was ground using a Thomas-Wiley Mill (model ED-5, Arthur H. Thomas Co., Philadelphia, PA), and 1 g was exhaustively extracted with 250 ml dichloromethane using a Soxhlet (model 6718, Ace Glass Inc., Vineland, NJ) apparatus. Subsequently, the extract was reduced to a final volume of 10 ml using a Rotavapor. The 10-ml lichen extract was spiked with 100 μl of 10,000 μg/ml chlorpyrifos (prepared from neat chlorpyrifos, AccuStandard, New Haven, CT) as an internal standard. Standards (125, 250, 500, 1,000, 2,000, 5,000 μg/ml) were similarly prepared from 98% pure (+)-UA in dichloromethane. Software used for quantitation was Varian MS Workstation (Version 6.5, Varian).
(+)-Usnic acid dosing
Nine adult Rambouillet-Suffolk cross ewes were used in this study, and animal identification was based on the order in which the ewes were dosed (i.e., the third ewe dosed is ewe No. 3). Ewes were offered municipal water and ground alfalfa hay ad libitum. Wood chips were used as bedding, and ewes were given 30 days to acclimate to the test environment. All ewes were weighed 1 day prior to dosing and, while on trial, were placed in a separate portion of the pen.
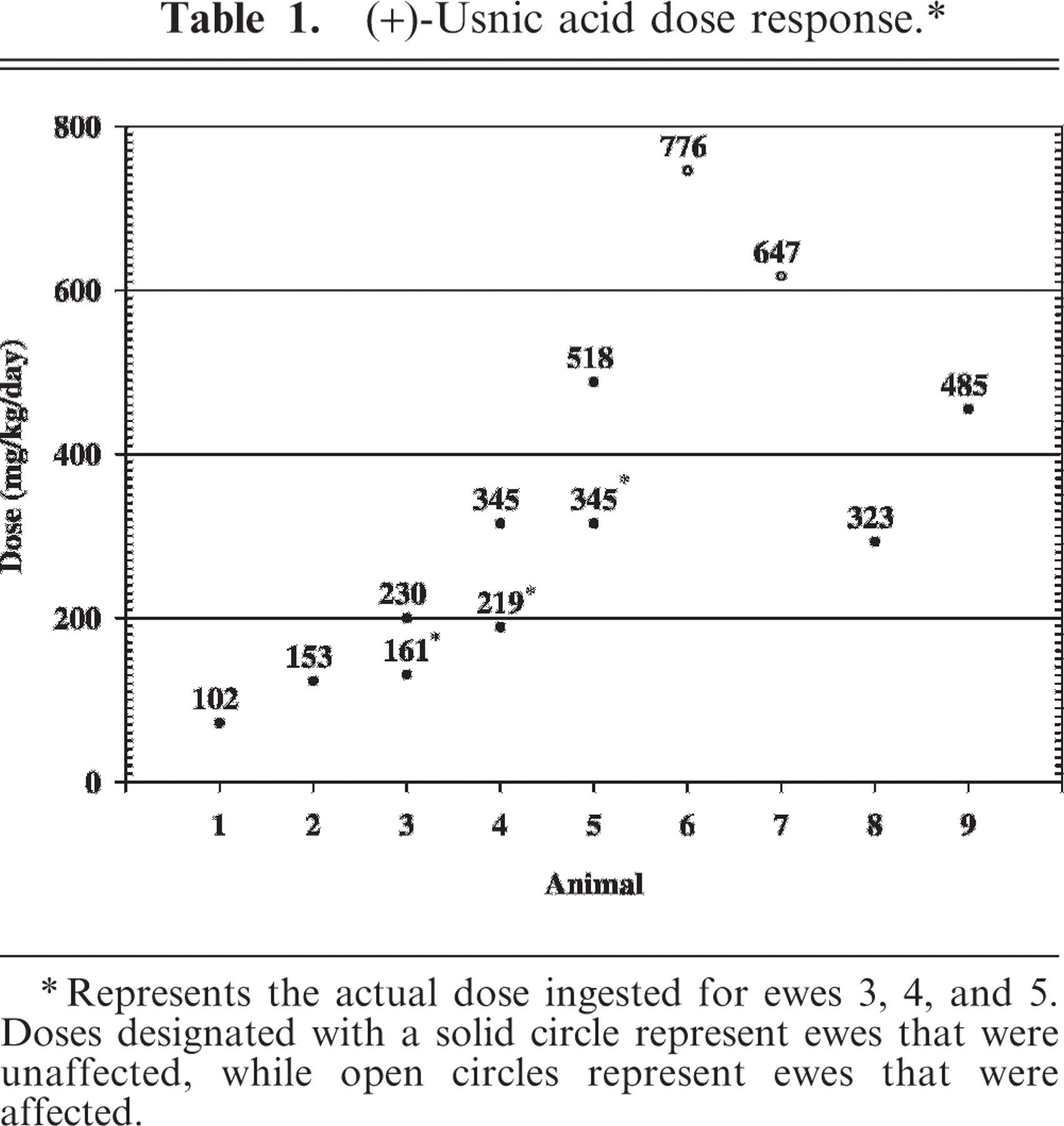
The dose administered was based on the up-and-down method for acute toxicity testing and was adjusted to body weight for each sheep. 4 Using this method, ewes were dosed in a sequential fashion, as the dose for each ewe was dependent upon the response of the previous ewe to (+)-UA. If ewe No. “n” failed to exhibit signs of ataxia or incoordination after a given dose of (+)-UA, the dosage for the subsequent ewe (n+1) was increased by 50%. If ewe No. n did exhibit signs, the subsequent dose was decreased by 50% of the difference between the doses given ewes n and n − 1. As a result, doses converge on a median toxic dose (ED50). The starting dose (102 mg/kg/day) was chosen as it was slightly less than what a ewe, consuming a diet of 100% lichen, would receive from lichen containing 1% (+)-UA. Dosing ceased when the ED50 had been bracketed twice (i.e., 2 positive and 2 negative responses).
The (+)-UA dose was suspended in molasses diluted with tap water (7 : 3) (vol/vol). The suspension was then applied to 0.454 kg ground alfalfa hay using a Sureshot sprayer (Milwaukee Sprayer Manufacturing Co., Menomonee Falls, WI). The sprayer was then rinsed with 200 ml tap water, which was subsequently applied to the hay to ensure no (+)-UA remained in the sprayer. Ewes were to receive one 0.454-kg ration per day, for 7 days. All 7 rations were made at the same time and refrigerated until use.
Each morning, the ewe on trial received 0.454 kg of treated hay. After the treated hay was consumed, clean ground hay was offered to fulfill a 2% basal body weight diet requirement. If the entire treated ration was not consumed, the remainder was weighed and added to the next day's ration. As the dosages increased, the ewes began to refuse feed, which may have been due to the inability of molasses to sufficiently mask (+)-UA odor and taste. After ewe No. 5, it was decided that remaining ewes would be gavaged to ensure that ewes received the full dose. The daily dose was split in half, and ewes were gavaged twice daily at approximately 12-hour intervals. The (+)-UA dose was placed in a 140-ml syringe, which was then filled with tap water. The syringe was shaken well just prior to dosing to keep the (+)-UA suspended. Each (+)-UA dose was followed by a slurry of finely ground alfalfa and water (280 ml). If feed consumption decreased, the amount of alfalfa slurry administered was increased to maintain daily dietary intake. Ewes were observed several times throughout each day and evening. After receiving 7 consecutive doses, all ewes were observed for an additional 3 days. Each ewe underwent a neurologic exam during the final 3-day observation period, or whenever they exhibited any clinical signs. The exams were performed by a board-certified neurologist and included a visual exam for gait abnormalities as well as conscious proprioception tests.
Sample collection
Blood was collected prior to dosing to establish a baseline. Additional blood samples were collected at any sign of illness, or beginning on day 4 if no signs were present. Serum chemistries and a complete blood count were run on all blood samples. Complete blood counts were conducted using a Hemavet 850FS (Drew Scientific, Oxford, CT). A VETEx (Alfa Wassermann, Inc., West Caldwell, NJ) was used to measure serum chemistries, which included alkaline phosphatase, aspartate aminotransferase, creatine kinase (CK), gamma-glutamyl transferase, lactate dehydrogenase, albumin, calcium, glucose, magnesium, phosphorus, total serum protein, blood urea nitrogen, total globulins, sodium, and potassium. Analysis was done using manufacturer reagents and following manufacturer recommendations.
A complete postmortem examination was performed on all ewes immediately following death or after humanely euthanizing with an IV injection of sodium pentobarbital on day 10. A complete set of representative tissues from lymphatic, respiratory, cardiovascular, digestive, urinary, endocrine, skeletal, and nervous systems and eye were collected and immersed in 10% neutral buffered formalin or Davidson's fixative (eyes only). Skeletal muscles included semitendinosus, semimembranosus, vastus lateralis, rectus femoris, longissimus dorsi, psoas major, diaphragm, and tongue. Additional sections of skeletal muscle were collected if gross lesions were apparent. For the nervous system, samples included the entire brain, spinal cord, and sections of sciatic, peroneal, median, and radial peripheral nerves. To minimize artifact, skeletal muscle and peripheral nerve samples were extended and attached to an appropriately labeled wooden tongue depressor with staples. Skeletal muscles were then allowed to rest at room temperature for approximately 1 hour prior to being immersed in fixative. Tissues were allowed to fix for a minimum of 7 days while on an orbital shaker. Following fixation, the tissues were trimmed, processed for paraffin embedding, sectioned, and stained according to accepted histologic technique. Cross, as well as longitudinal, sections were trimmed for skeletal muscles and nerves. All sections were stained with hematoxylin and eosin for light microscopic examination.
Results
Subsamples of X. chlorochroa collected from the RRWHMA (Fig. 1) contained between 1 and 2% (+)-UA (1.55% ± 0.49%). Optical rotation of the (+)-UA standard was [∝]24 D = +518 (CHCl3, c 0.2). Rotation of UA crystals extracted from X. chlorochroa was [∝]24 D = +524 (CHCl3, c 0.2), thus validating the use of (+)-UA in the study.
Ewe Nos. 3, 4, and 5 refused to eat their entire ration and, as a result, did not receive their entire dose of (+)-UA each day. Treated rations were worked back into the diet over 2 to 3 additional days. Taking into account the additional time needed to consume all 7 rations, actual doses (mg/kg/day) consumed were calculated using weigh back data (Table 1). Ewe Nos. 6 and 7 received the highest doses, 776 and 647 mg (+)-UA/kg/day, respectively, and were the only ewes to exhibit clinical signs. During the week of dosing, both ewes displayed progressively worsening signs indicative of abdominal discomfort and lethargy. Ewe No. 6 became anorexic and displayed stiffness in her hind legs after the sixth dose of (+)-UA. Neither ewe became recumbent for any length of time. Ewe No. 6 was found dead the morning of day 7. Ewe No. 7 received all 7 doses and was found dead the morning of day 8. Red urine was never observed in any of the (+)-UA-dosed animals, and ewe Nos. 1–5, 8, and 9 remained asymptomatic during the entire course of the trial. The ED50 of (+)-UA was estimated to be between 485 and 647 mg/kg/day for domestic sheep.
∗∗ Represents the actual dose ingested for ewes 3, 4, and 5. Doses designated with a solid circle represent ewes that were unaffected, while open circles represent ewes that were affected.
Serum chemistry abnormalities were observed in one asymptomatic ewe (No. 8), as well as in both the symptomatic ewes (Nos. 6 and 7). Ewe Nos. 6 and 7 exhibited a continuous elevation of serum CK activity (9,950–101,500 μ/liter) beginning on day 4 (Table 2). Ewe No. 8, received 323 mg (+)-UA/kg/day, remained asymptomatic throughout the experiment, but exhibited a transient increase in serum CK (1,762 μ/liter) on day 6. All subsequent serum samples were within the normal range. Other abnormal serum chemistry results included markedly elevated lactate dehydrogenase (LDH) and aspartate aminotransferase (AST) in ewe Nos. 6 and 7 (Table 2). No definitive changes were observed in the other parameters measured nor were any abnormalities observed in the remaining, lower dosed ewes.
Serum analyte concentrations(μ/liter) in 3 (+)-usnic acid dosed ewes.
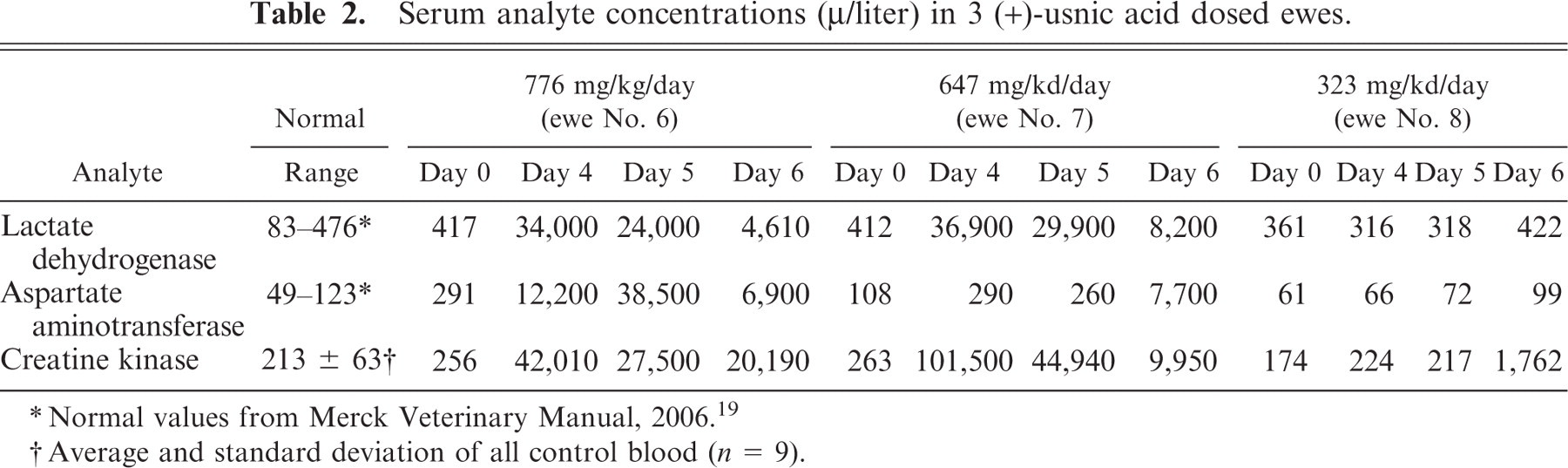
∗ Normal values from Merck Veterinary Manual, 2006. 19
†Average and standard deviation of all control blood (n = 9).
At postmortem examination, gross lesions were observed only in clinically affected ewes (Nos. 6 and 7). Both extensor and flexor muscle groups of limbs were dramatically affected. Lesions consisted of variable pallor and sometimes white chalky areas of mineralization (Fig. 2). Affected skeletal muscles were edematous. Affected skeletal muscles in rear limbs included quadriceps, semimembranosus, semitendinosus, biceps femoris, peroneous, and gluteus. Lesions in forelimbs involved the extensor carpi radialis and medial head of the triceps. Gross lesions were not evident in the axial skeletal muscles, tongue, or diaphragm.
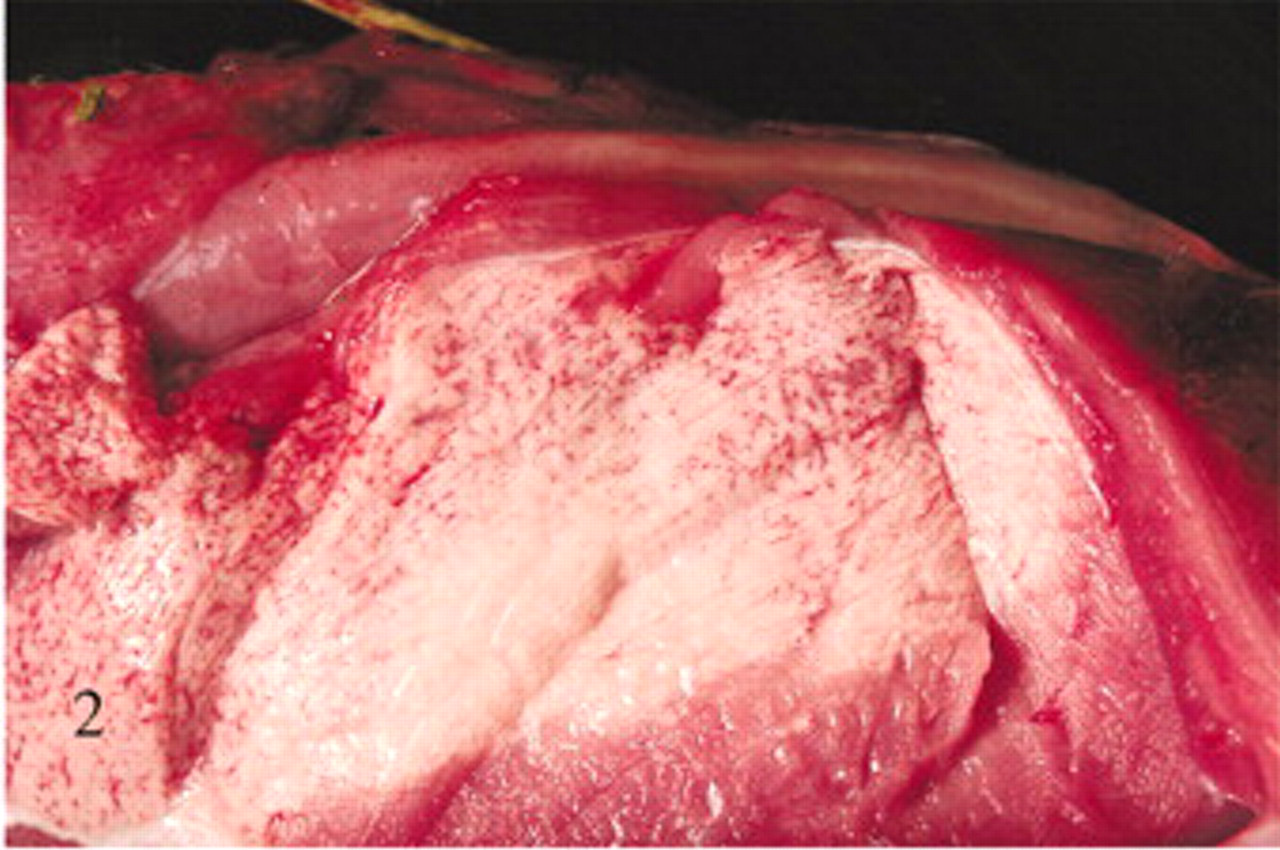
Caudal thigh muscles; sheep No. 6 receiving an usnic acid dose of 776 mg/kg/day. Marked pallor indicates areas of myodegeneration and mineralization.
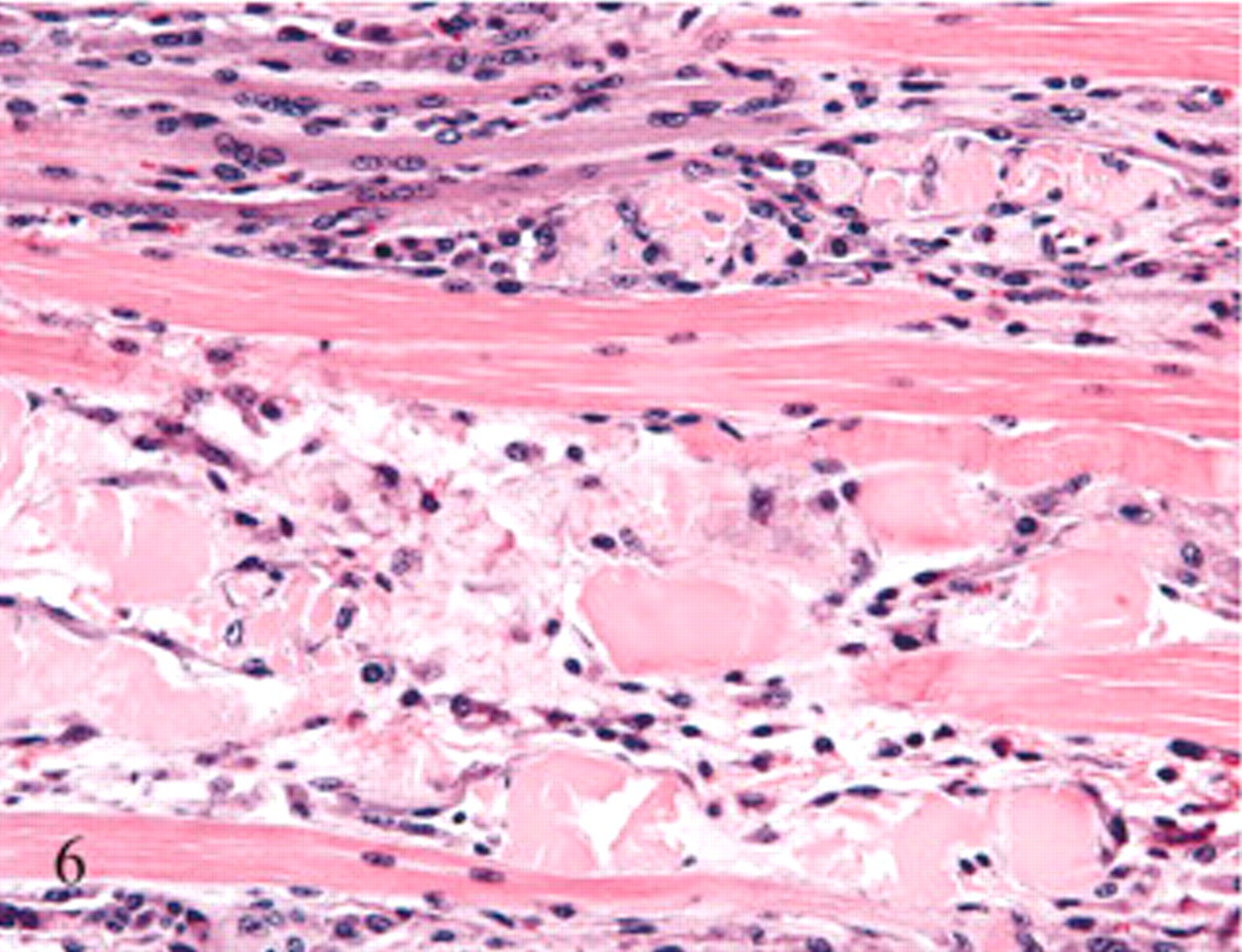
Histologic changes were limited to skeletal muscle. In asymptomatic ewes, changes were minimal and included rounding or swelling of individual myocytes accompanied by increased cytoplasmic eosinophilia, some evidence of hypercontraction, and variable loss or blurring of cross-striations in cross and/or longitudinal sections. In symptomatic ewe Nos. 6 and 7, microscopic lesions were severe and confined to muscles with gross lesions. Lesions in individual muscles represented a spectrum of acute as well as more protracted skeletal muscle damage (Figs. 3– 6). Acute lesions included swollen, hypereosinophilic and/or hypercontracted myofibers with central vacuolation. Fragmentation and flocculation of myofibers were pronounced. Macrophages progressively infiltrated in and around the degenerate myofibers, and cytoplasmic mineralization was prominent in some areas of myodegeneration. Early myocellular regeneration, evidenced by centralization of nuclei, multinucleation and/or cytoplasmic basophilia occurred in muscles with more acute degeneration suggesting an ongoing process (Fig. 6).
Extensor carpi radialis muscle, forelimb; sheep No. 6. A longitudinal section of the same muscle as depicted in Fig. 5. Basophilic myofibers with rowing of nuclei notable at the top of the figure indicate early myoregeneration. HE.
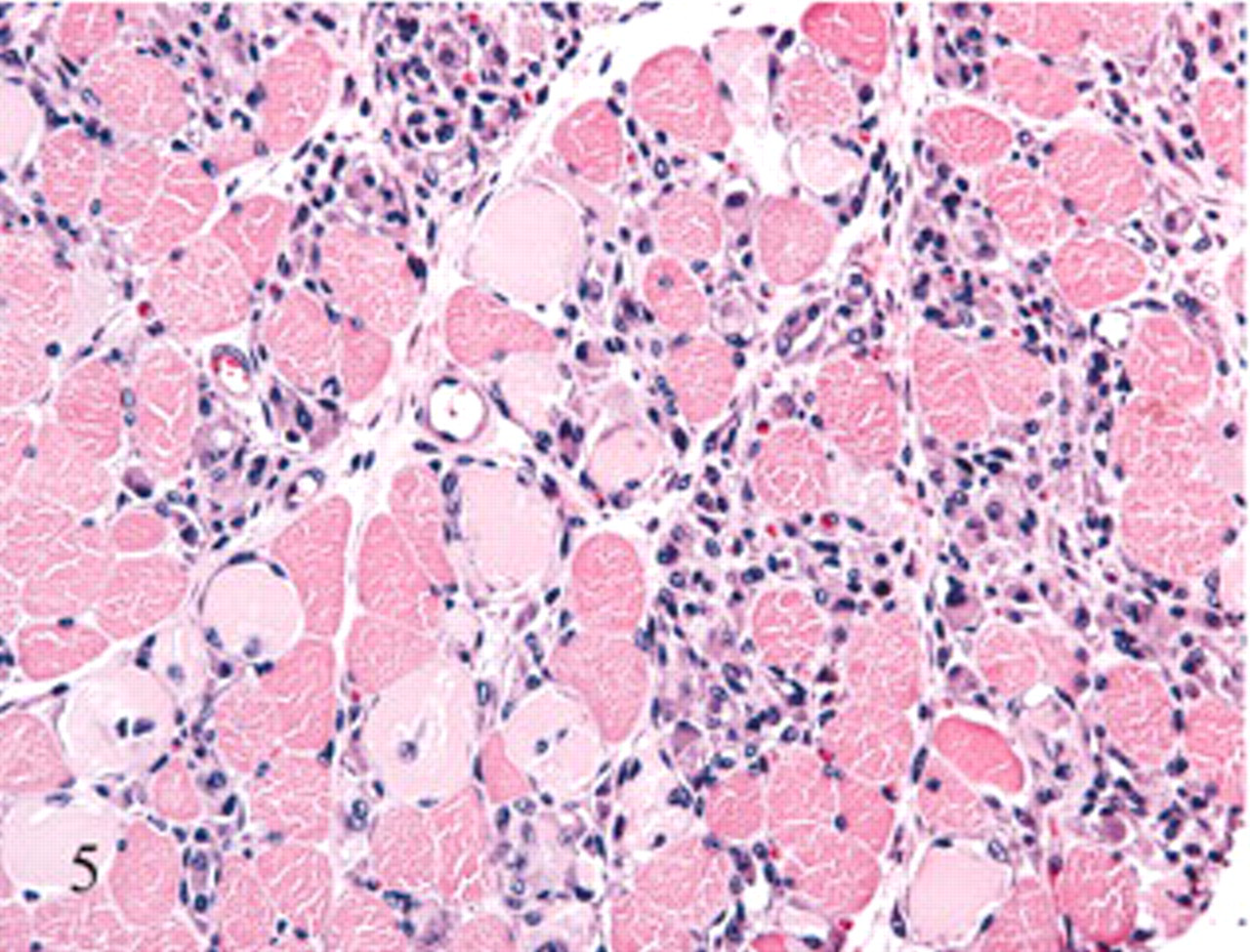
Extensor carpi radialis muscle, forelimb; sheep No. 6. Lesions are the same in this cross-section as noted for Fig. 3 but with a more intense macrophage response. HE.
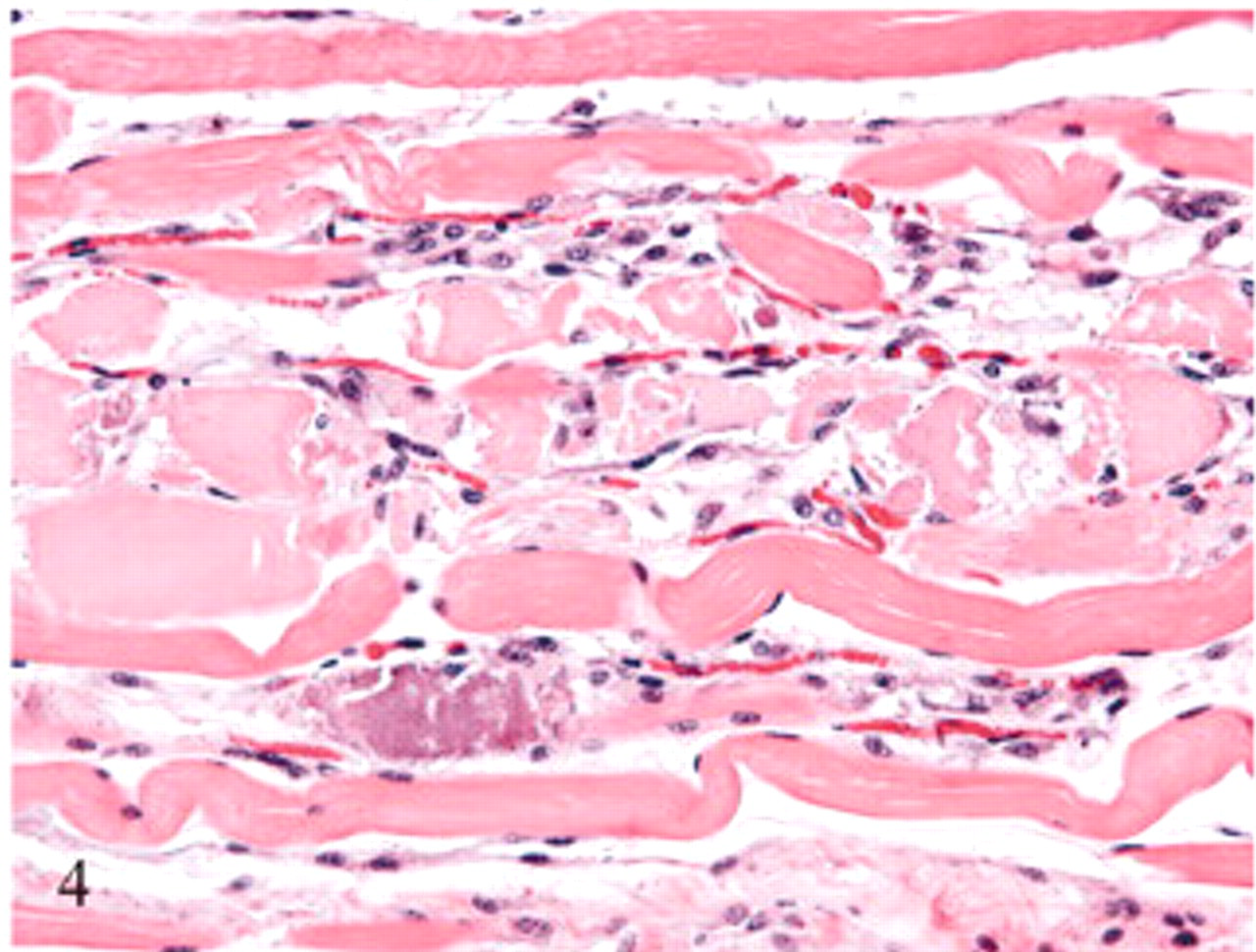
Semitendinosus muscle, hindlimb; sheep No. 7. A longitudinal section of the same muscle as depicted in Fig. 3. In this section, the loose organization of the muscle fibers indicates edema as noted grossly. HE.
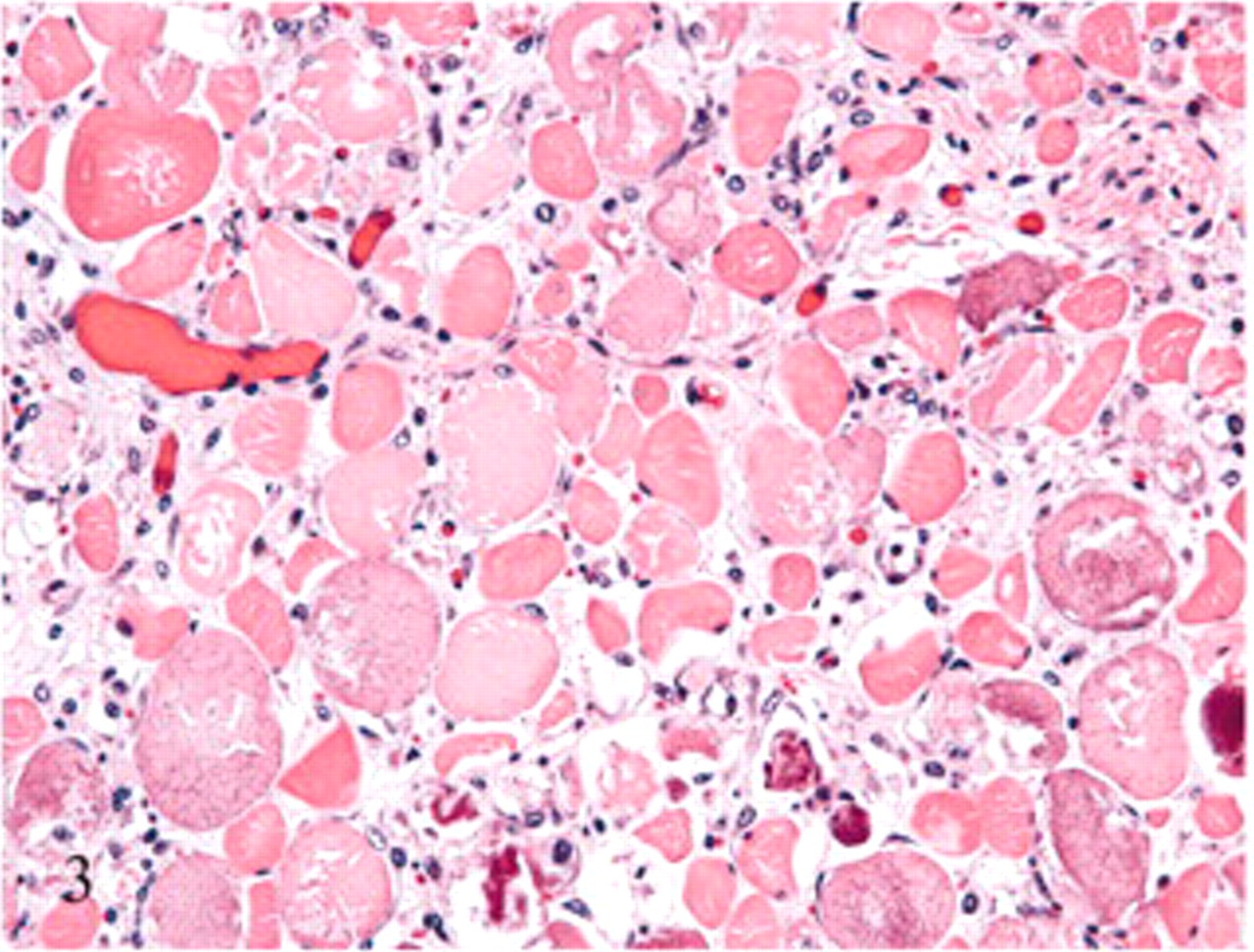
Semitendinosus muscle, hindlimb; sheep No. 7. Pale swollen myocytes with flocculation and fragmentation can be seen in this cross-section of the muscle. Also present are myocytes with granular basophilic cytoplasmic mineralization and an infiltrate of macrophages. Unaffected myocytes are scattered among the degenerating cells. HE.
Discussion
Previous research and several case studies indicate that (+)-UA is potentially hepatotoxic in monogastric animals and in humans. 1,7,8,12,22,23 In contrast, the current (+)-UA feeding study in sheep suggests a primary myotoxic effect in ruminants. This discrepancy is not surprising considering the differences in monogastric versus ruminant physiology. Toxicity of a compound may be reduced or amplified as a result of rumen metabolism. 5 Ruminants, for example, are more resistant to the effects of certain mycotoxins (i.e., ochratoxin A) as a result of the ruminal microbes' ability to degrade the toxins. 13 While it is unknown at this time whether (+)-UA itself or a metabolite of microbial fermentation is the proximate toxin, this study shows that high doses of (+)-UA can cause skeletal muscle damage.
Gross myodegeneration observed in the 2 symptomatic, high-dose (+)-UA-fed ewes was striking and resembled lesions observed in several myopathic syndromes in animals including nutritional and exertional myopathies. 15,18 Microscopically, only minimal changes of undetermined significance were observed in the clinically unaffected ewes. Rounded, “swollen,” hypereosinophilic and hypercontracted myofibers are considered artifactual and occurred in some ewes despite efforts to minimize these changes. 18 These artifactual changes may occur with some frequency in individuals where increased “irritability” of the muscle is suspected, including pigs and humans with malignant hyperthermia and in humans with Duchenne's muscular dystrophy. 9,18,26 Ultimately, the significance of this change is undetermined in the current ewes. The microscopic lesions in the 2 ewes with grossly evident myodegeneration are, however, irrefutable and suggest an active, ongoing process occurring over several days. This interpretation is supported by the continuously elevated serum CK, LDH, and AST activities. In these ewes, various stages of degeneration occurred in the same sections, ranging from acute segmental lesions of swollen hypereosinophilic and hypercontracted fibers, flocculation, and fragmentation that progressed through stages of macrophage infiltration and mineralization and eventuating in some apparent regeneration.
This study was not designed to identify the mechanism of action of (+)-UA or pathogenesis of the myopathy. Few studies have examined the effect of UA on muscle. UA has been shown to shorten the action potential duration at 50 and 90% of repolarization in rat heart ventricular tissue, presumably by blocking or activating ion-selective channels. 25 UA reduced contraction of rat aorta induced by KCl and phenylephrine, possibly as a result of inhibiting Ca2+ influx into the cell. 21 Although these studies were conducted with smooth muscle, the effect of UA on ion channels may contribute to the skeletal muscle damage observed in (+)-UA-poisoned ewes. Several toxins including scorpion toxins, gliotoxin, and scabrosin are known to affect Ca2+ homeostasis resulting in skeletal muscle damage. 10,17,20
Given the (+)-UA concentration measured in toxic lichen and the estimated ED50 (485–647 mg/kg/day for 7 days), it would be impossible for a ewe to eat enough lichen to approach a toxic dose of (+)-UA. It would seem that other compounds in the lichen, possibly in addition to (+)-UA, interact to produce the specific syndrome observed in lichen-poisoned animals. This could be one or more additional lichen substances or rumen metabolites. While (+)-UA is reportedly hepatotoxic in monogastrics, presumably as a result of mitochondrial dysfunction, 1,7,12,14 effects in ruminants seem selective for skeletal muscle.
Footnotes
Acknowledgements
We would like to thank Dr. Kurt Dolence of the University of Wyoming, School of Pharmacy for polarimetry assistance. We would also like to thank Dr. Robert Egan of the University of Nebraska at Omaha for his assistance with lichen identification, and Brent Larson of the University of Wyoming for providing and transporting the study animals. Finally, we thank Katie Bardsley of the Wyoming State Veterinary Laboratory for all the clinical pathology work, and Dr. Todd Cornish for his scientific input. Funding was provided by the Wyoming Agricultural Experiment Station, the Pharmaceutical Manufacturers Association, and the Department of Veterinary Sciences, University of Wyoming.