Abstract
Intoxication of sheep and cattle by Astylus atromaculatus recently occurred in Uruguay and Argentina in association with severe drought. Although the disease was experimentally reproduced in sheep in the 1970s, there is limited information on clinical and pathologic findings of sheep experimentally intoxicated by this beetle. Here, we described the clinical, gross, and microscopic findings in 3 sheep orally dosed with A. atromaculatus (treatment group, TG) and in 2 control sheep (control group, CG) dosed with distilled water. Anorexia, lethargy, ruminal stasis, reluctance to move, prolonged recumbency, and bruxism were observed in the TG but not the CG sheep. Gross postmortem lesions were only observed in TG sheep and included fibrinonecrotizing enteritis affecting the duodenum, jejunum, and ileum, and multifocal hemorrhages in rumen, omasum, and abomasum. Microscopically, all 3 TG animals had multifocal necrosis in the small intestine; the lesions were most severe in the jejunum. Multifocal necrosis was seen in the mucosa of the rumen, omasum, and abomasum. No significant gross or microscopic abnormalities were observed in the 2 CG sheep. The study supports the role of A. atromaculatus in acute, fatal gastrointestinal disease like that previously described in experimental and spontaneous cases in sheep.
The pollen beetle Astylus atromaculatus (Melyridae family) is an insect native to South America, commonly found in crops (Fig. 1).1,3,13 During the summer of 2023, several outbreaks of A. atromaculatus-associated gastroenteric disease were described in sheep and cattle grazing on pastures heavily invaded by A. atromaculatus in Uruguay and Argentina.4,10 No toxic principle for this insect is known, and previous reports failed to identify cantharidin, a well-known gastrointestinal irritant contained in beetles belonging to the family Meloidae, or any other toxic substances.4,5,9,12 A disease clinically and pathologically similar to that observed in sheep and cattle spontaneously intoxicated by this beetle was reproduced in cattle dosed orally with A. atromaculatus. 5 However, to our knowledge, there is only one record from more than 50 years ago on experimental intoxication with A. atromaculatus in guinea pigs and sheep, resulting in catarrhal to pseudomembranous and hemorrhagic enteritis and typhlitis. 6 The study referred to 6 provided a limited description of the clinical signs and lesions. The aim of this study was to experimentally reproduce the disease caused by A. atromaculatus in sheep to better characterize the clinical, gross, and histologic lesions of this condition.

Astylus atromaculatus beetles (adults) infesting a maize plant. The insects have yellow elytra and bilateral symmetric black spots.
Five female merino and corriedale sheep were used in this study. The signalments and experimental design are presented in Table 1. All experimental procedures using animals were performed following the guidelines of the Institutional Committee for Care and Use of Experimental Animals from Instituto Nacional de Tecnología Agropecuaria, Argentina (CICUAE; protocol 254/2023) and the Veterinary Faculty (Universidad de la República, Montevideo, Uruguay) (protocol 1753).
Signalment, inoculation dose, inoculation-death interval, and form of death in 3 sheep experimentally inoculated with adult Astylus atromaculatus and in 2 control sheep.
Three animals (treatment group, TG) were orally dosed with a total of 4 g (n = 2) or 5 g (n = 1) of A. atromaculatus per kg of body weight (BW). The beetles were collected from farms where outbreaks of intoxication in livestock grazing alfalfa heavily infested with A. atromaculatus had recently occurred (Fig. 1).4,8 The beetles were chopped and blended in an aqueous suspension of 0.33 g/ml of distilled water before giving them to the sheep by a stomach tube. Two additional animals (control group, CG) were similarly inoculated with 3 or 6 ml of distilled water. All sheep were clinically evaluated before and every 4 hours after inoculation. The 2 animals in the CG were euthanized 48 and 72 hours after inoculation, respectively.
Postmortem examination was performed in all animals immediately after death, and samples of rumen, reticulum, omasum, abomasum, small and large intestine, liver, kidney, urinary bladder, mesenteric lymph nodes, heart, lung, and brain were collected and fixed by immersion in 10% buffered formalin for 72 hours. All tissues were processed by standard techniques to obtain 4 μm thick, hematoxylin and eosin-stained sections.
Clinical signs were similar in all 3 animals of the TG. The first clinical signs, observed 12 hours post-inoculation, were anorexia, apathy, and depression. In addition, the animals had droopy heads and ears, eyes almost entirely closed, bruxism, and ruminal hypomotility. At 24 hours post-inoculation, reluctance to move and bloody diarrhea was observed. At 36 hours post-inoculation, the heart rate was 120 bpm and the pulse was weak. Two sheep from the TG died spontaneously 38 and 72 hours after inoculation, respectively, and the remaining TG animal was euthanized in extremis 74 hours after inoculation. No clinical abnormalities were observed in either CG animal.
Grossly, in all 3 animals of the TG, the content of the small and large intestine was liquid and hemorrhagic, and the mucosa was diffusely red and had multifocal to coalescing, small 0.2 to 0.8 cm dark red ulcers covered by fibrin (Fig. 2a). The ruminal mucosa of the 3 animals of the TG was diffusely hyperemic. Parts of the beetles were observed in the ruminal and intestinal contents of the 3 TG animals; occasionally these fragments were admixed with fibrin in the small intestines (Fig. 2b). There were multifocal to coalescing hemorrhages in the mucosa of the omasal folds of case 1 (Fig. 2c). In the abomasal mucosa of this animal, there were multifocal round or linear erosions (Fig. 2d). Ascites with a few strands of fibrin was observed in case 3. No significant gross abnormalities were observed in either sheep of the CG.
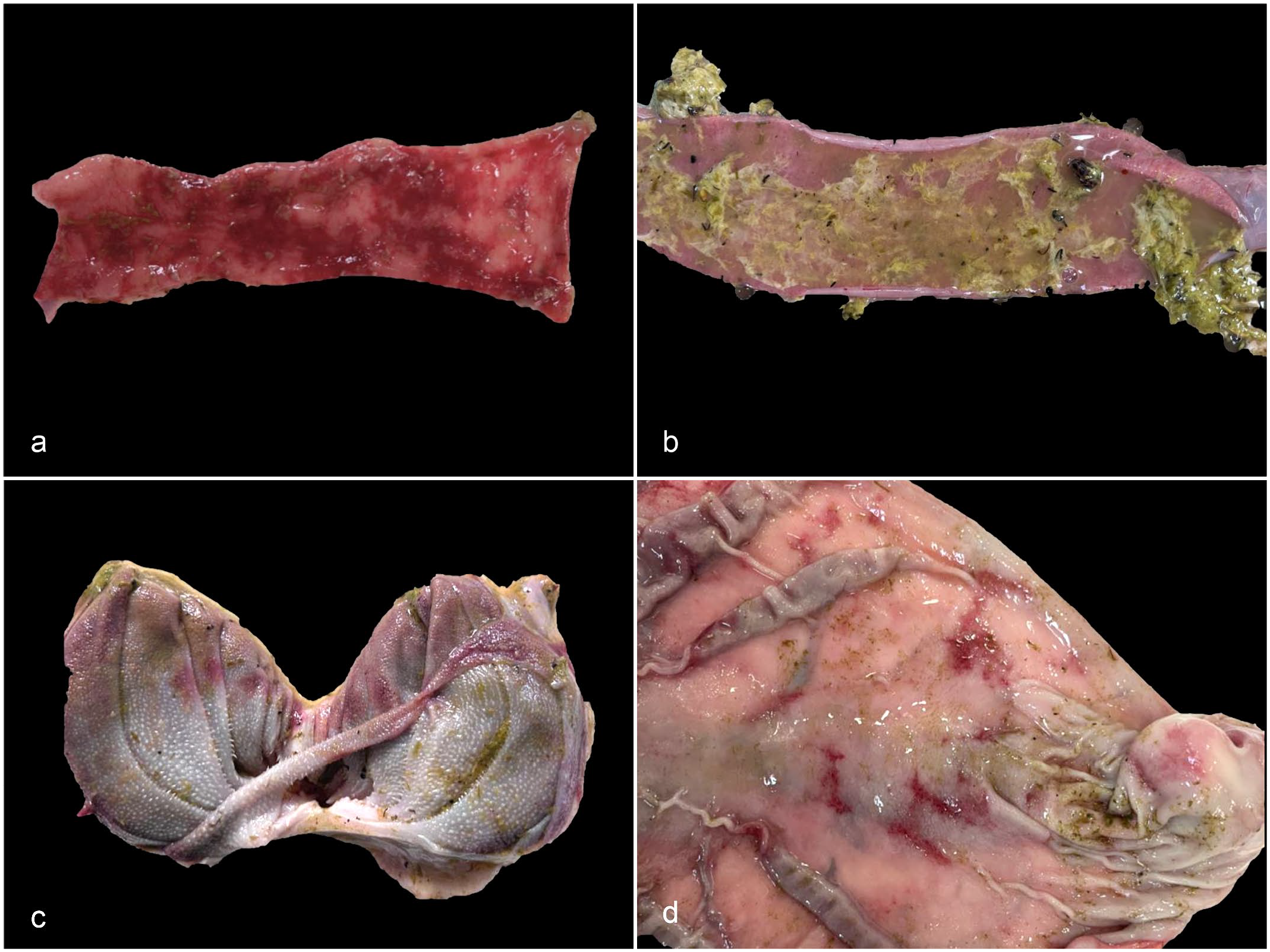
Gross findings in sheep experimentally intoxicated with Astylus atromaculatus. (a) Jejunum. Multifocal to coalescing hemorrhages throughout the eroded jejunal mucosa. (b) Jejunum. The mucosa is covered by a yellowish fibrinous pseudomembrane admixed with dark brown to black beetle fragments. (c) Omasum. Multifocal to coalescing hemorrhages in the mucosa of the omasal folds. (d) Abomasum. Multifocal, random, irregular round or linear erosions in the abomasal mucosa.
Microscopically, the main lesion present in all 3 TG animals was multifocal extensive necrosis and hemorrhage in the duodenum, jejunum (Fig. 3a), and ileum. The lesions were most severe in the jejunum. The necrotic regions were characterized by villus blunting, crypt necrosis and dilation, eosinophilic areas with fragmented nuclei intermingled with fibrillar eosinophilic material, and low numbers of diffusely infiltrating viable and degenerate neutrophils. Overlying the necrotic mucosa, there was a pseudomembrane composed of fibrin, necrotic debris, bacterial colonies, and hemorrhage. Occasionally, multiple ulcers covered by fibrin were observed (Fig. 3a). In 2 animals (cases 1 and 2), chitinous structures were seen on the necrotic surface and rarely embedded in the superficial intestinal mucosa surrounded by hemorrhage and necrosis. In the rumen, multiple foci of epithelial necrosis reaching the basal layer, admixed with few neutrophils and occasionally covered by fibrin, necrotic debris, and entrapped bacterial colonies were present in 2 animals (cases 1 and 2) (Fig. 3b). Lymphocytes and neutrophils multifocally infiltrated the lamina propria. Multifocal necrosis of the omasal mucosa characterized by fragmented nuclei and karyorrhexis of keratinocytes mixed with neutrophils was present in 1 animal (case 1). In the abomasum of 2 animals (cases 2 and 3), neutrophils and fewer lymphocytes were noted diffusely in the lamina propria and submucosa. Multifocal areas of marked congestion and hemorrhage in the superficial mucosa with mild necrosis were observed. In the liver of the 3 TG animals, there was diffuse, mild to moderate microvesicular hepatocellular degeneration.
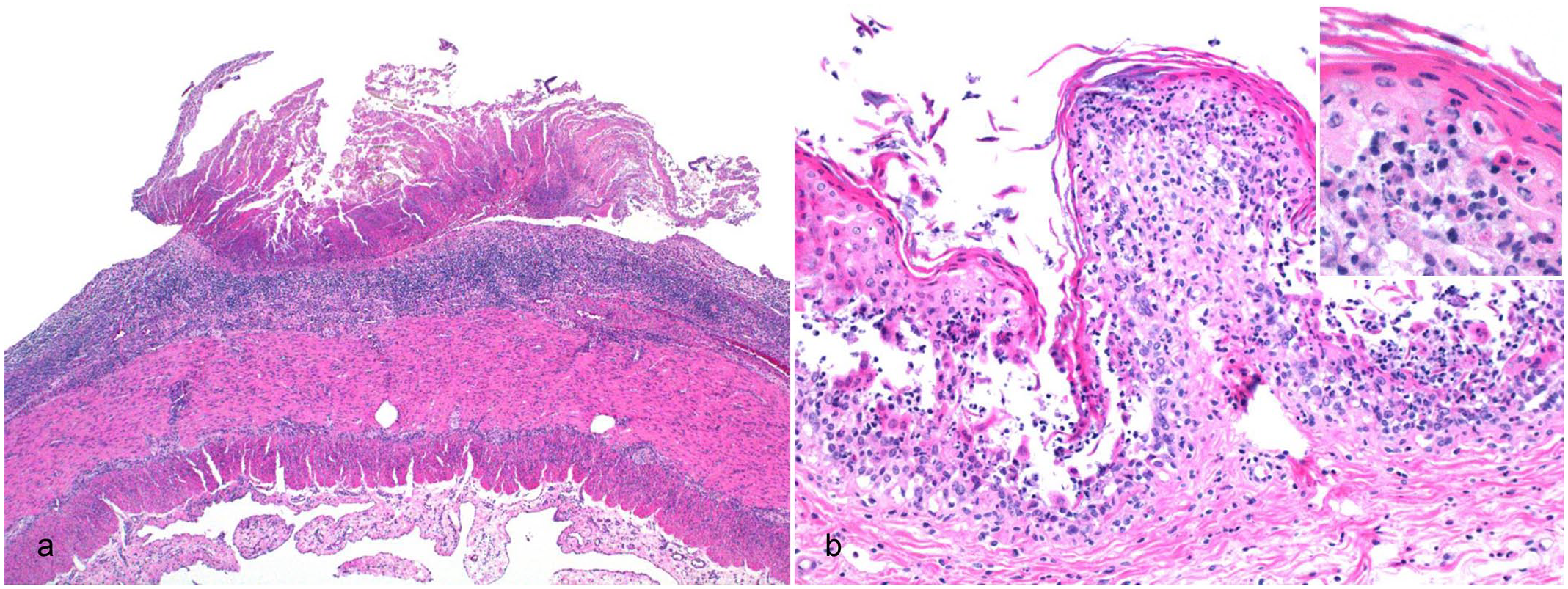
Microscopic findings in sheep experimentally intoxicated with Astylus atromaculatus. Hematoxylin and eosin. (a) Jejunum. Extensive ulceration characterized by complete necrosis of the mucosal layer with overlying fibrinous exudation and karyorrhectic debris. (b) Rumen. Full-thickness necrosis of the mucosa and neutrophilic infiltration. Inset: higher magnification of necrotic keratinocytes in the spinosum layer with infiltrating neutrophils.
No differences in the type or severity of lesions were observed between animals inoculated with 4 or 5 g of beetles per kg BW. No significant microscopic lesions were observed in either sheep of the CG.
We reproduced acute, lethal, gastrointestinal disease in sheep by oral administration of A. atromaculatus. The disease was similar to that described in spontaneous cases of A. atromaculatus in sheep and cattle.4,8 This further supports previous experimental studies conducted in sheep 6 and cattle 5 demonstrating that ingestion of theses beetles causes this condition. Our results, coupled with a recent experimental assay in calves, 5 add to the existing body of scientific literature pointing at a causative role of A. atromaculatus in gastroenteric disease of ruminants.
Given the limited number of animals available for this study, the minimum lethal dose was not determined. Both oral doses tested (ie, 4 and 5 g/kg BW) were lethal for sheep, which is consistent with previous results. 6 In calves, a single dose of 2.5 g/kg BW was lethal and given that this was the lowest dose tested, it seems possible that the lethal dose could be even lower. 5
The clinical signs and postmortem findings in our experiment showed slight differences with those described in spontaneous cases. The only difference in the clinical signs was that in spontaneous cases bloating was observed; this was not seen in the sheep of our study. 8 Although a limited number of animals were available for this study, gross and microscopic lesions in the forestomachs were frequent, unlike what was observed in spontaneous cases and previous experimental challenge in sheep.6,8 Also, insect structures embedded in the superficial intestinal mucosa were not reported in spontaneous cases. 8 This may be associated with multiple variables, including the length of time in which animals were consuming beetles and the amount of insect consumed.4,8 In experimental challenges like the one presented here, the animals are exposed to a high concentration of grounded A. atromaculatus, unlike spontaneous outbreaks, when insects are likely ingested in lower doses over longer periods. Also, in natural intoxication cases, it is likely that the insects are not as thoroughly grounded as they were in this study when they were blended. This could account for less small insect parts being immersed in gastrointestinal tissues in spontaneous cases and could also explain why forestomach lesions were observed in experimentally dosed animals and not in animals spontaneously intoxicated.
The clinical and postmortem findings of this study are compatible with previous reports of spontaneous A. atromaculatus intoxication in livestock.4,8 Although experimental intoxication in sheep was conducted nearly 50 years ago in South Africa, 6 the study reported limited information on clinical and pathologic findings. Our study showed a faster onset of clinical signs and death compared to the experiment in sheep in South Africa where clinical signs appeared after 48 hours, and death occurred between 6 and 9 days post-inoculation. 6 This could be due to a difference in toxicity between insects from different locations, though the nature of the toxic principle is unknown. In the study previously referred to, 6 the lowest lethal dose was 5 g/kg BW, while we determined that 4 g/kg were lethal for sheep. With an average weight of 27.3 mg/beetle, 5 this would mean for a 20 kg sheep a difference of 733 beetles (2930 beetles in 4 g/kg BW dose and 3663 in a 5 g/kg BW dose). The latter is important considering that the ingestion of beetles in spontaneous outbreaks could occur at lower doses over a more prolonged period.
In spontaneous outbreaks in sheep, watery diarrhea, depression, anorexia, and ruminal atony were observed; 8 all these clinical signs were also observed in our study. Necrotic enteritis and occasional lesions in forestomachs and abomasum have been reported in spontaneous outbreaks and experimentally in calves.4,5 Although a physical damage to the intestine as a pathogenic mechanism was suggested for cattle, 5 in our study, fragments of A. atromaculatus were rarely observed embedded in damaged intestinal mucosa, probably as common feature of entrapment as occurs with other materials such as vegetal parts. Further studies are necessary to elucidate the gastrointestinal mechanism of action of A. atromaculatus. An important postmortem finding was the presence of lesions in the abomasum and forestomachs, which was not reported by Kellerman et al, 6 but was observed in spontaneous outbreaks in sheep 8 and calves. 4
Ingestion of A. atromaculatus should be considered for a differential diagnosis of cantharidin poisoning due to ingestion of beetles from the Meloidae family, known as blister beetles, with similar gastrointestinal lesions.2,9,12 Other diseases, with similar lesions, frequently observed in the region where spontaneous cases of intoxication by A. atromaculatus occurred are intoxication by Baccharis corifidolia, 11 Ricinus communis, 7 and lead arsenate; 10 gastrointestinal helminthiasis; coccidiosis; and infectious agents such as Salmonella enterica, bovine coronavirus, and bovine viral diarrhea virus. 14
The mechanism of action of A. atromaculatus remains unknown. Extensive toxicology testing was carried out with no toxin yet detected.4,5 Consequently, other pathogenic mechanisms may be considered, including bacteria or viruses from the beetle microbiota acting as pathogens in the alimentary tract of ruminants, direct physical damage, and/or immune-mediated damage caused by insect fragments. The possibility of variance of insect toxicity between different geographic areas reinforces the need to carry out experimental studies and further toxicologic and pathogenesis investigations. Particularly, inoculation of beetles stored at 4°C for 5 weeks in 4 sheep (not shown) resulted in only ruminal stasis in 1 animal with recovery, 6 reducing potential toxicity. Further studies are necessary to identify possible toxin or other mechanisms of action, as well as the minimum lethal dose to know the potential risk of the beetle.
Footnotes
Acknowledgements
We thank to Dr Carla Faliveni who provided the facilities and experimental animals, Valeria Scioli and Leticia Ferreira for histopathological processing, Bruno Canneva (Facultad de Veterinaria, Universidad de la República, Uruguay) for beetle identification, and the veterinarian students Joaquín Heredia and Mariana Perdomo for assistance with experimental animals.
Author Contributions
J.A.G., C.G.S., and F.A.U. conceived the study; J.A.G., C.G.S., and F.D. designed the experiments and J.M.L., A.C.C., A.C., and J.E. performed the experiments and collected the insects. J.M.L. and J.E. collected clinical data and J.M.L., J.A.G., C.G.S., A.C.C., A.C., and J.E. performed the necropsies; J.A.G., F.A.U., and F.D. performed the histopathologic examinations and obtained microphotographs; F.A.U., G.C., C.M., and F.G. helped with the interpretation of the pathological findings and provided valuable insights based on their experience with natural or experimental cases of this disease; C.G.S. and J.A.G. wrote the first draft of the manuscript. All authors reviewed, edited, and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
