Abstract
Xanthoparmelia chlorochroa, commonly called tumbleweed lichen, is found throughout the Rocky Mountain region. This particular species of lichen was incriminated in the poisoning of cattle and sheep in Wyoming during the 1930s. More than 70 years elapsed before another case was reported. There is virtually no information in the veterinary literature regarding toxicity of this lichen. This report describes X. chlorochroa poisoning in domestic sheep fed lichen collected from different locales and at different times of the year. Affected animals voided red urine and displayed incoordination. A transient spike in serum creatine kinase activity occurred in all ewes during the course of the feeding trial. Histologically, necrosis of a few individual appendicular skeletal myocytes was observed in 1 ewe, but grossly discernible myonecrosis was absent. The severity of clinical signs varied depending on the location and/or time of year the lichen was collected, indicating that toxicity of the lichen may be influenced by environmental conditions. Results demonstrate that domestic sheep are a useful model for further investigation of X. chlorochroa intoxication. The current study should act as a starting point for elucidating the pathogenesis of X. chlorochroa poisoning and aid in the development of a diagnostic assay to confirm lichen poisoning.
Introduction
Xanthoparmelia chlorochroa inhabits the high plains paralleling the Rocky Mountains. 3 It is a folióse, vagrant lichen cited as an indicator of good pronghorn antelope habitat 3 as well as a source of water or forage during harsh winters (Thomas A, Rosentreter R: 1992, Utilization of lichens by pronghorn antelope in three valleys in east-central Idaho. Idaho Bureau of Land Management Technical Bulletin 92–93). 11 Reports of poisoning following ingestion of any lichen species are extremely rare, excluding cases in which the accumulation of metals or radionuclides was a contributing factor. Anecdotal accounts of X. chlorochroa poisoning in Wyoming cattle and sheep date to the late 1930s (Beath OA: 1937, Livestock poisoning from the common “lichen.” Wyoming Agricultural Experiment Station Bulletin 500; Beath OA: 1939, Poisonous plants and livestock poisoning. Wyoming Agricultural Experiment Station Bulletin 231:49–53). These reports identify the lichen as Parmelia molliuscula, now classified as X. chlorochroa. 10 No further incidents of X. chlorochroa poisoning have been documented until a recent elk (Cervus canadensis) mortality, which occurred on the Red Rim—Daley Wildlife Habitat Management Area (RRWHMA) in February and March of 2004. 4 , 5 Clinical signs in the affected elk were similar to those previously reported in cattle and sheep and included ataxia and muscular weakness that rapidly progressed to recumbency. Affected elk voided red urine, absent of myoglobin and hemoglobin, 4 which was an observation not previously made in domestic livestock (Beath OA: 1939, Poisonous plants and livestock poisoning). Although postmortem examination was not described, no distinctive lesions were recorded in lichen-intoxicated cattle or sheep (Beath OA: 1939, Poisonous plants and livestock poisoning). Myodegeneration was observed in elk recumbent for several days, but could have been related to exertional rhabdomyolysis rather than a primary toxic myopathy. 4 Interestingly, cattle and sheep mortality was low (Beath OA: 1937, Livestock poisoning from the common “lichen.”; Beath OA: 1939, Poisonous plants and livestock poisoning) compared to an estimated 60% mortality in the elk herd. 5
Currently, confirmation of X. chlorochroa poisoning is circumstantial. The pathogenesis of the syndrome is undetermined. The responsible toxin(s) within the lichen and their mechanism(s) of action have yet to be identified. Usnic acid, a lichen secondary metabolite, was postulated to be the toxic compound within the lichen responsible for poisoning (Beath OA: 1939, Poisonous plants and livestock poisoning). In a recent experimental study, however, administration of usnic acid orally did not duplicate the syndrome in domestic sheep. 6

Xanthoparmelia chlorochroa along with other vegetation in the Red Rim—Daley Wildlife Habitat Management Area.
Given the lack of information regarding X. chlorochroa toxicity, diagnosis is difficult, and treatment options are nonexistent. A combination of intravenous fluids; vitamins A, B, D, and E; selenium; calcium gluconate; dextrose phosphorus; magnesium; potassium; and anti-inflammatory and corticosteroid drugs had no beneficial effect in 4 symptomatic elk. 5 To date, recommended control or preventive measures are to remove animals from the area and/or to provide a different or supplemental feed source. The objective of the current study was to describe lichen poisoning in a domestic sheep model, which was initiated by feeding domestic sheep X. chlorochroa (University of Nebraska at Omaha Lichen Herbarium No. 15631) collected from 3 Wyoming locations during different times of the year.
Materials and methods
Lichen collection
Xanthoparmelia chlorochroa was collected from the RRWHMA immediately following the 2004 elk mortality and again 19 months later (Fig. 1). Xanthoparmelia chlorochroa was also collected from the McCulloch Peaks grazing allotment in northwestern Wyoming and from the Monolith Hunter Management Area in southeastern Wyoming (Table 1). Lichen was collected by hand and allowed to air-dry on an outdoor concrete pad. Once dry, lichen was placed in appropriately labeled, sealed, 30-gallon containers and stored at ambient temperature.
Housing and observation protocol
Three weight-matched, adult Rambouillet ewes were utilized per lichen group. Ewes were housed either on wood chips or on dirt. All ewes were offered municipal water, plain salt, and sufficient ground alfalfa hay to fulfill a 2% body weight basal diet for a 2-week acclimation period. Ewes were observed several times throughout the day and evening for the duration of the trial. Each ewe underwent several neurological exams while on trial; these exams included a visual exam for gait abnormalities as well as conscious proprioception. Neurological exams were performed both prior to observing clinical signs and after clinical signs were evident. Sheep from an adjacent pen, which were fed hay only, were utilized as controls for observation.
Sample collection
Body weight and control blood were collected prior to feeding lichen. Additional blood samples were collected at the first sign of illness, or every 3 days if no clinical signs developed. Body weights were collected throughout the trial and at the time of necropsy. Serum chemistries and a complete blood cell count were conducted on all blood samples, as previously described. 6 Urine was collected from each ewe at least once while the animal was on a 100% lichen diet via metabolism cage. Upon collection, urine samples were immediately filtered a and subsequently refrigerated until urinalysis (typically within 6–24 hr). The presence of hemoglobin was tested via dipstick, b and myoglobin was considered absent if urine was negative for protein and if no erythrocytes were observed in the sediment.
Feeding protocol
The first lichen ration (day 1) was a mixture containing 10% X. chlorochroa and 90% ground alfalfa hay. The percentage of lichen in the diet was increased 10% per day until the entire diet was lichen (day 10). Once the basal body weight diet consisted entirely of lichen, ewes remained on this ration for the remainder of the trial (day 21).
Xanthoparmelia chlorochroa collection dates and locations.

Site of the 2004 elk mortality. 6,7 WHMA = Wildlife Habitat Management Area.
Site of a probable lichen-poisoning event involving domestic cattle (B. McKenzie, personal communication).
Site selected as earlier studies utilized X. chlorochroa from the Laramie plains. 2
Pathology
A complete postmortem examination was performed on all ewes after they were determined to be moribund or at the end of the study (day 21). Tissues obtained for histological evaluation included brain, spinal cord, autonomic ganglia, peripheral nerve, eye, heart, trachea, lung, esophagus, forestomachs, abomasum, small and large intestine, salivary gland, liver, pancreas, spleen, lymph nodes, bone marrow, kidney, urinary bladder, thyroid, and adrenal and pituitary glands. Tissues were immersed in 10% neutral buffered formalin or Davidson fixative (eyes only). Brain, liver, heart, and kidneys were weighed prior to fixation. Skeletal muscles obtained included semitendinosus, semimembranosus, vastus lateralis, rectus femoris, peroneus, longissimus dorsi, psoas major, diaphragm, and tongue. To minimize artifact, skeletal muscle and peripheral nerve samples were extended and stapled to an appropriately labeled wooden tongue depressor. Skeletal muscles were allowed to rest at room temperature for approximately 1 hr prior to being immersed in fixative. Following fixation, the tissues were trimmed, processed for paraffin embedding, sectioned, and stained according to accepted histological technique. Cross sections as well as longitudinal sections were trimmed for skeletal muscles and nerves. All sections were stained with hematoxylin and eosin for light microscopic examination. The University of Wyoming Animal Care and Use Committee reviewed and approved all protocols.
Results
The severity of clinical signs varied appreciably among lichen groups (A, B, C, and D) and between individuals in the same group. Ewes had to remain on a 100% lichen diet for at least 2 days, and in most cases longer, to reproduce the clinical syndrome. The ewes found the lichen palatable and consumed 2% of their body weight daily. Red urine was the only consistent clinical sign in all individuals, and it occurred only after consuming a 100% lichen diet.
Group A
Group A displayed the most progressive and severe clinical signs. These ewes were on a 100% lichen diet for 2 days (day 11) before the onset of clinical signs, the first being voiding of red urine. Locomotor abnormalities, very subtle on the morning of day 12, progressed rapidly to include a stiff-legged, bunny-hopping gait, crossing of the front or hind legs, and leaning on fence panels in ewes A1 and A2. Wool chewing was observed in ewes A1 and A2 after day 13. The 2 ewes were humanely euthanized on day 14. The remaining ewe (A3) displayed transient mild stiffness in both the front and hind limbs on day 16 but was clinically normal on day 17. Ewe A3 was euthanized on day 21.
Group B
Group B was the least affected of all the lichen groups, and all 3 ewes were euthanized on day 21. Ewe B1 remained asymptomatic throughout the entire course of the study. Ewe B2 displayed very subtle stiffness on day 12, but stiffness did not progress and seemed to subside the following day. Ewe B3 displayed mild hind limb incoordination on day 15. Again, clinical signs did not progress, and ewe B3 seemed to recover in subsequent days.
Group C
All 3 ewes in group C displayed mild spasticity in the hind legs by day 16. Spasticity in ewes C1 and C2 did not progress. Ewe C3, however, did progress from spasticity to incoordination and from leaning on fence panels to periodic recumbency. After resting for a period of minutes, she would be able to get back up, but she was unsteady. Ewe C3 was euthanized on day 19, while ewes C1 and C2 were euthanized on day 21.
Group D
All 3 ewes in group D displayed clinical signs that varied in severity and were euthanized on day 21. Clinical signs were first observed on day 15 in ewe D1. Signs consisted of mild stiffness in the hind legs accompanied by bunny hopping when attempting to run. The following day, when attempting to run, ewe D1's front legs would buckle, but she was always able to get back up. Clinical signs thereafter remained static. The remaining 2 ewes (D2 and D3) exhibited spasticity mainly in the front legs beginning on day 16, but clinical signs did not progress.
Clinical pathology
Each ewe exhibited a single transient spike in serum creatine kinase (CK) activity, excluding ewes B3, C1, and D2, whose CK activities did not exceed the average control value (278 ± 123 μ/l) The increase in CK activity did not occur simultaneously among ewes in the same lichen group or across lichen groups. The spike in CK activity also did not invariably coincide with the onset or progression of clinical signs. The largest increase in serum CK activity occurred in group A (3,350 ± 1,577 μ/l) The average values for groups B, C, and D were 818.5 ± 235.5, 1,257.5 ± 695.1, and 1,646.5 ± 1,467.2 μ/l, respectively. No definitive abnormalities were observed in the other parameters measured.
Urine collected directly from the bladder during necropsy, if present, or indirectly via metabolism cage from all ewes was red. All urine samples were negative for myoglobin and hemoglobin. Urine specific gravity ranged from 1.005 to 1.020, and pH ranged from 6 to 8.5. Other parameters were unremarkable.
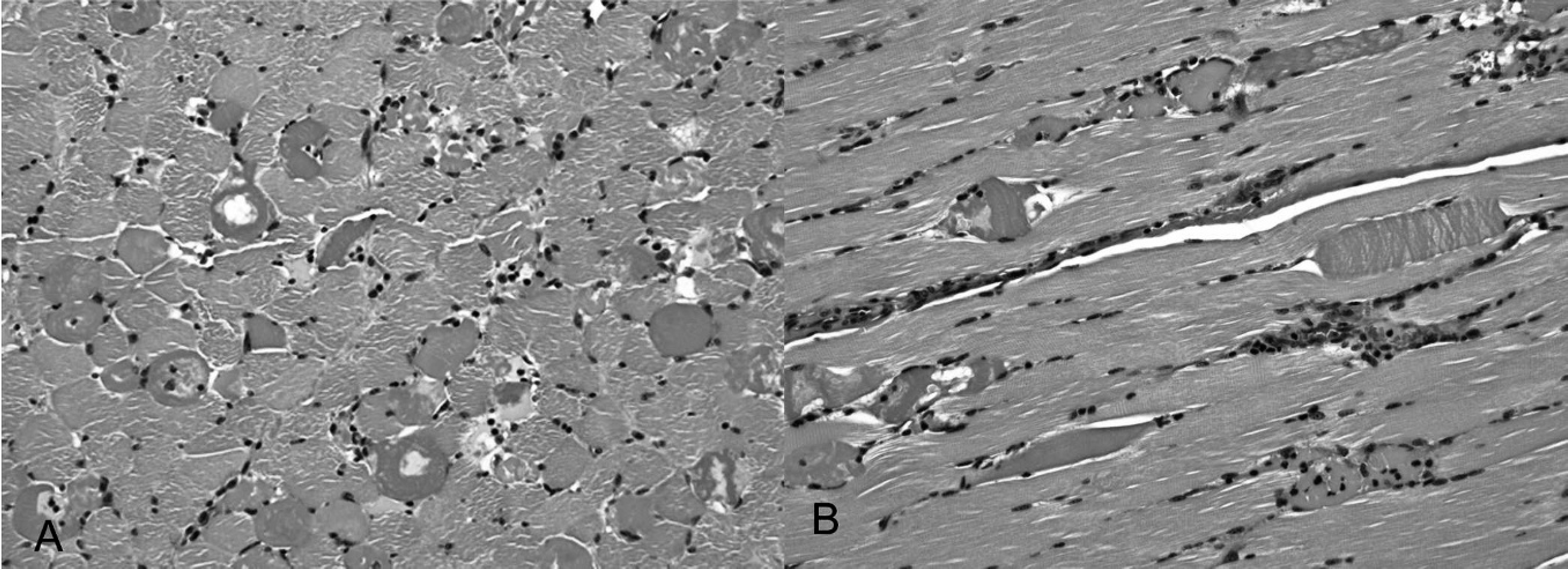
Peroneus muscle from ewe D1.
Histology
Gross lesions consisted of slight pallor of questionable significance in the semimembranosus and semitendinosus muscles from ewes A1 and A2. Histological changes were limited to skeletal muscles, including diaphragm, psoas, rectus femoris, semimembranosus, tongue, vastus lateralis, semitendinosus, and peroneus. Scattered myocytes in these muscles were hypercontracted and hypereosinophilic, changes that are commonly considered artifactual. Irrefutable microscopic lesions were observed only in the semimembranosus and peroneal muscles from ewe D1. In the peroneal muscle, numerous myocytes were hypereosinophilic and hypercontracted. In scattered individual myocytes, these changes progressed to flocculation and fragmentation, with a mild acute influx of neutrophils and macrophages, and myophagocytosis. There was no mineralization or regeneration associated with the myocellular degeneration and necrosis (Fig. 2A, Fig. 2B). Similar but minimal lesions were observed in the semimembranosus muscle. No significant lesions were observed in the other tissues examined.
Discussion
The objective of the current study was to describe lichen poisoning in a domestic sheep model. This model may be useful in defining the pathogenesis of X. chlorochroa poisoning and may aid in the development of a diagnostic assay to confirm lichen poisoning. It is difficult at this time to speculate regarding pathogenesis. Based upon clinical and neurological examinations, incoordination was thought to be of muscular rather than neurological origin. The mild and inconstant skeletal muscle lesions in lichen-poisoned animals do not correlate with the consistent, often dramatic clinical signs observed in the ewes in the present study, however.
Swollen, hypereosinophilic myocytes are generally considered artifactual. Despite efforts to minimize this effect, the change was observed with regularity. This “artifact” occurs with increased frequency, however, in myopathic disorders, including malignant hyperthermia and Duchenne's muscular dystrophy. 6,8,12 The transient increase in serum CK activities may indicate that this is a real change. Overt myocellular degeneration and necrosis were limited to only 1 ewe (D1) in the present study. The limited distribution would indicate that this was not an exertional rhabdomyolysis. Since these lesions occurred in only 1 ewe, however, direct relevance to lichen toxicity could not be determined with certainty. There are many examples of myopathic syndromes and neurotoxicants that alter or disrupt normal functions without producing morphologic cellular damage (e.g., channelopathies and clostridial neurotoxins, respectively). 12,16 There are several secondary metabolites, commonly termed lichen substances, within X. chlorochroa the biological effects of which are largely unknown. Oral administration of (+)-usnic acid, although shown to be toxic in a previous study, 6 did not reproduce the exact clinical syndrome observed in lichen-poisoned animals, indicating that other secondary metabolites within the lichen may be the active toxin(s). 6 These compounds have several proposed functions, such as protection from herbivores, insects, pathogens, and ultraviolet (UV) light; they also aid in rock mineralization. 1 , 3 , 9 , 15
It is interesting to note that while all lichen-fed ewes displayed red urine and varying degrees of locomotor signs, the severity of clinical signs differed dramatically between lichen groups. This is best illustrated by comparing ewes from groups A and B fed lichen from the same location but collected 19 months apart. Group A was the most severely affected of all the lichen-fed groups, while ewes in group B were the least affected. All recent, naturally occurring lichen poisonings, including diagnostic cases submitted to the Wyoming State Veterinary Laboratory, have occurred between November and March. A previous observation also noted that lichen intoxication was primarily a problem in winter months (Beath OA: 1939, Poisonous plants and livestock poisoning). This may simply reflect higher consumption during winter months as the availability of other forages decreases. Conversely, lichen secondary metabolite concentrations fluctuate in response to environmental conditions, 18 which may ultimately affect toxicity. A previous study examined usnic acid concentrations in Flavocetraria nivalis from 4 different locations and found a correlation between time of season (solar radiation and temperature) and usnic acid concentration. 2 Another study reported that when Cladonia mitis, Cladonia rangiferina, Usnea dasypoga, and Bryoria trichodes were exposed to varying levels of UV light, the concentration of usnic acid and atranorin varied. Usnic acid decreased when exposed to visible UVA and UVB light, while atranorin increased under similar conditions (Mitchell JL, Helleur RJ: 2002, Analysis of lichen phenolics as environmental stress indicators by LC-MS. Enviro-Analysis, Proceedings of the Biennial International Conference on Monitoring and Measurement of the Environment, pp. 283–286). While the amount of lichen consumed plays a crucial role in toxicity, the significance of secondary metabolite concentration fluctuations remains to be determined.
Because the production of red urine is not a result of myoglobin or hemoglobin, the red color is probably due to lichen secondary metabolites. Many species of lichen are used to produce red, orange, yellow, or purple dyes. 7 , 14 , 17 Native Americans have utilized X. chlorochroa, which creates a reddish-brown color, to dye wool for blankets. Secondary metabolites thought to be responsible for the dying properties of certain lichens include stictic, norstictic, salazinic, vulpinic, and pinastric acids. 3 Of these, norstictic and salazinic acids have been identified in X. chlorochroa. 3 , 10 Since red urine occurs prior to any gait abnormalities, it may be useful as a diagnostic indicator of potentially toxic exposure.
The lack of information regarding toxicity of X. chlorochroa has made diagnosis and treatment of affected animals extremely difficult. While field cases of tumbleweed lichen poisoning have been reported in both domestic and wild ruminants, to the author's knowledge, the current study is the first experimental reproduction of lichen intoxication in domestic sheep. Utilizing this model, urine has been identified as a promising diagnostic sample. Further investigation of this lichen species in a ruminant model may aid in defining the pathogenesis of X. chlorochroa poisoning and provide insight into the biological effects of lichen secondary metabolites.
Acknowledgements
The authors would like to thank Dr. Robert Egan of the University of Nebraska at Omaha for his assistance with lichen identification and Katie Bardsley of the Wyoming State Veterinary Laboratory for all the clinical pathology work. The authors thank Brent Larson for supplying and transporting study animals and Marce Vasquez, Amanda Fluegel, Dave Edmunds, Jeremy Brown, Dr. Ken Mills, Sarah Riker, Dr. Cynthia Tate, Dr. Todd Cornish, Bryan McKenzie, and Patricia Hatle for lichen collection. Finally, the authors thank Amanda Fluegel and Dr. Walt Cook for comments on the manuscript. This work was supported by a grant from the Wyoming Wildlife Partnership.
Footnotes
a.
Whatman no. 4 filter paper, Whatman International Ltd., Maidstone, UK.
b.
Multistix 10 SG, Bayer Inc., Pittsburg, PA.
