Abstract
A 13–year-old neutered male lion was presented with a primary neoplasm arising from the left mandibular salivary gland associated with metastases to regional lymph nodes, thoracic viscera (lungs, heart, esophagus, and diaphragm), and kidney. Histologic and immunohistochemical investigations led to a diagnosis of a high-grade mucoepidermoid carcinoma of the mandibular salivary gland. In this case report, we point out the importance of the immunohistochemical characterization for differential diagnosis between various types of carcinomas of the salivary gland.
Cancer is an uncommon cause of disease and death in captive wild felids, and few neoplasms have been described in lions. Among the tumors reported in captive lions, the most commonly affected sites are the skin and subcutaneous tissue (including a cutaneous mast cell tumor, 20 a basal cell epithelioma, 23 a ductular mammary carcinoma, 10 and a subcutaneous fibrosarcoma 15 ), as well as the alimentary system (including a gastric carcinoma, 8 a malignant lymphoma of the biliary tract, 6 and two cases of gallbladder adenocarcinomas 16 ). In this case report, we describe a malignant tumor of the mandibular salivary gland in a captive lion, with the neoplasm displaying histologic and immunohistochemical features typical of a high-grade mucoepidermoid carcinoma.
A 13-year-old neutered male lion from a zoologic park in France was referred to the Veterinary School of Nantes for clinical signs of anorexia and ptyalism with blood-stained sialorrhea. Physical examination of the oral cavity under anesthesia with a mixture of xylazine and ketamine (2.5 mg/kg and 2 mg/kg, respectively, for an approximate weight of 200 kg) revealed the presence of a voluminous sublingual mass causing a right deviation of the tongue. The caudal part of the mass was palpable in the pharyngeal area under the mandibular bone. An aspirate sample of blood-stained fluid was cytologically noninformative. After a 2-day treatment with a nonsteroidal anti-inflammatory drug (meloxicam, 0.1 mg/kg intramuscular injection and then by oral route), the lion developed a sudden and dramatic deterioration of condition with dyspnea. The lion was euthanized for humane reasons, and a complete necropsy was performed shortly after death.
Postmortem examination revealed the presence of a firm, white mass 15 × 30 cm located in a posterior left sublingual position. Transverse sections of the mass revealed a multicavitary neoformation with extensive multifocal necrotic and hemorrhagic foci (Fig. 1). Lesions that were similar but smaller in size were observed in other tissues and organs, including retropharyngeal and submandibular lymph nodes, lungs, myocardium, endocardium, muscular part of the esophagus, diaphragm, and kidney. Tissue samples were collected, fixed in neutral-buffered 10% formalin, embedded in paraffin, and routinely processed for histologic examination. Three-micrometer–thick sections of the tumor were submitted for staining with hematoxylin-eosin-saffron (HES), periodic acid–Schiff (PAS), Alcian blue, and mucicarmin. In addition, several immunohistochemical stains were performed based on a standard avidin-biotin-peroxidase complex–based method, including reagents for cytokeratin (panCK, dilution 1:500, clone MNF116; Dako, Carpinteria, CA), alpha-smooth muscle actin (α-SMA, dilution 1:1000, clone 1A4; Dako), S100 protein (PS100, dilution 1:4000; Dako), vimentin (dilution 1:1000, clone V9; Dako), and p63 protein (dilution 1:100, clone 4A4; Dako). This latter protein is a recently characterized p53 homolog representing a nuclear transcription factor and has different functions in the cell cycle, ranging from apoptosis to cell differentiation. In human pathology, p63 is a recognized marker of the epithelial basal cell layer in normal and neoplastic epithelium. 2
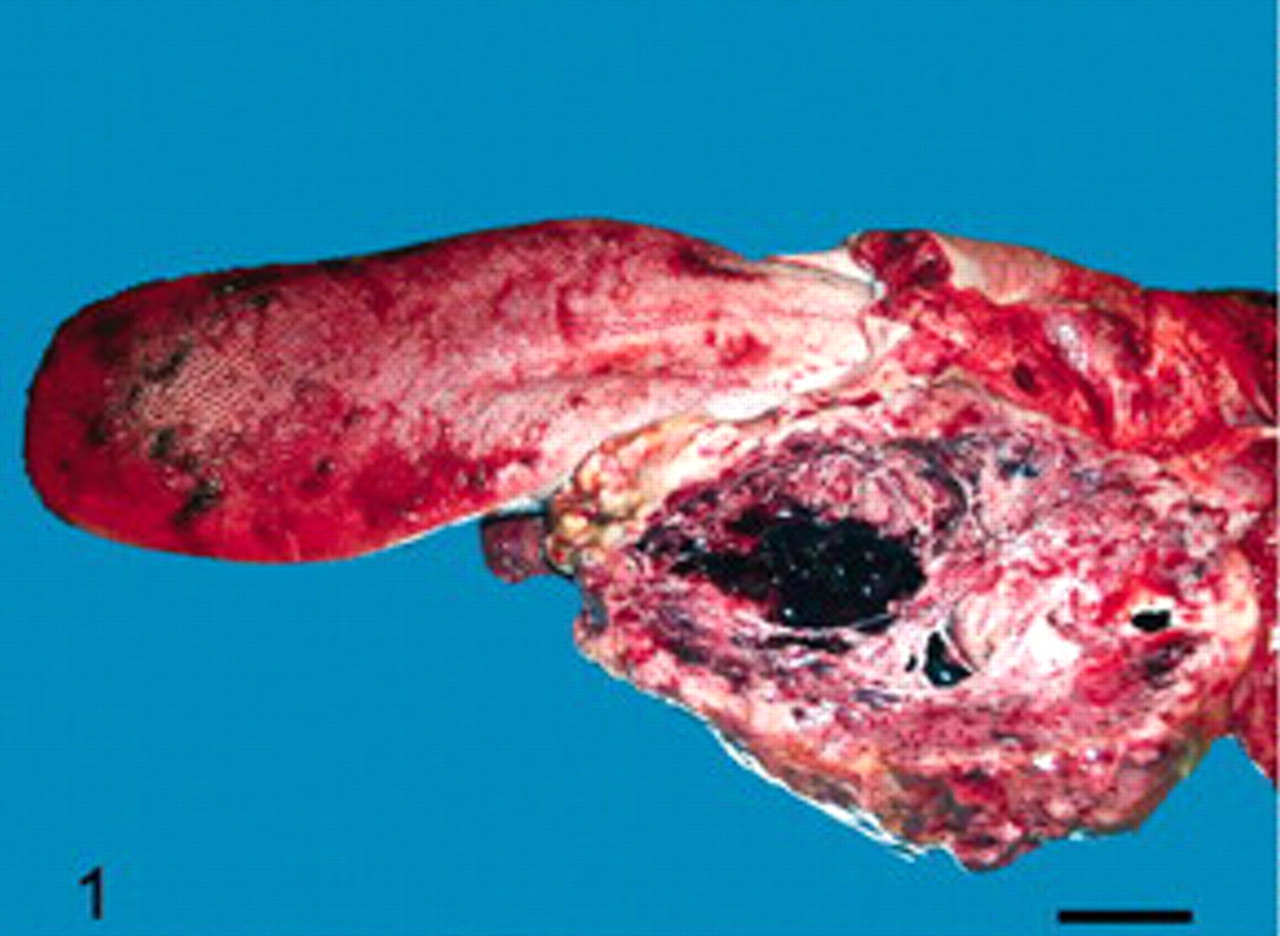
Tongue and pharyngeal area; lion. Macrophotograph of a cut surface of the primary tumor of the mandibular salivary gland: posterior left sublingual and mandibular mass with necrotic and hemorrhagic remodeling. Bar = 6.5 cm.
On microscopic examinations, the tumor was unencapsulated, infiltrative, densely cellular, and poorly delineated. Tumor cells were arranged in anastomosing and irregular solid islets surrounded by a moderately abundant stroma (Fig. 2). Some scarce and small cystic spaces in tumor islets were observed that were occasionally filled with neutrophils, cell debris, and apoptotic cells, as well as a small amount of bluish mucinous fluid, which stained positive with PAS, Alcian blue, and mucicarmin (Fig. 3). Some of these cystic spaces are lined by small, 15-μm diameter tumor cells with hyperchromatic nuclei forming strata several cells thick. Their morphology is consistent with a nonterminally differentiated “intermediate” cell type. Most of the tumor islets, however, were composed of tumor cells that were 20–30 μm in diameter and polygonal in shape, with distinct cell borders and intercellular bridges. These epidermoid-type cells contained an abundant, pale to eosinophilic, homogenous cytoplasm, as well as a round, central nucleus with coarse chromatin, and included a well-marked eosinophilic nucleolus at its center. No keratin pearls were observed. They were admixed in some areas with densely packed smaller cells of the previously described intermediate type. Anisocytosis and anisokaryosis were prominent. Mitotic activity averaged three per high-power field. There were numerous foci of coagulative necrosis and hemorrhage. Neoplastic emboli were present within lymphatic and venular blood vessels. Microscopic evidence of metastases was found in all of the organs with macroscopic lesions (lungs, heart, esophagus, diaphragm, and kidney). Based on the histologic features, a presumptive diagnosis of malignant mucus-secreting trabecular carcinoma of the mandibular salivary gland was made. Due to the presence of squamous and intermediate cell type components and mucous secretion, a mucoepidermoid carcinoma of the salivary gland was suspected.
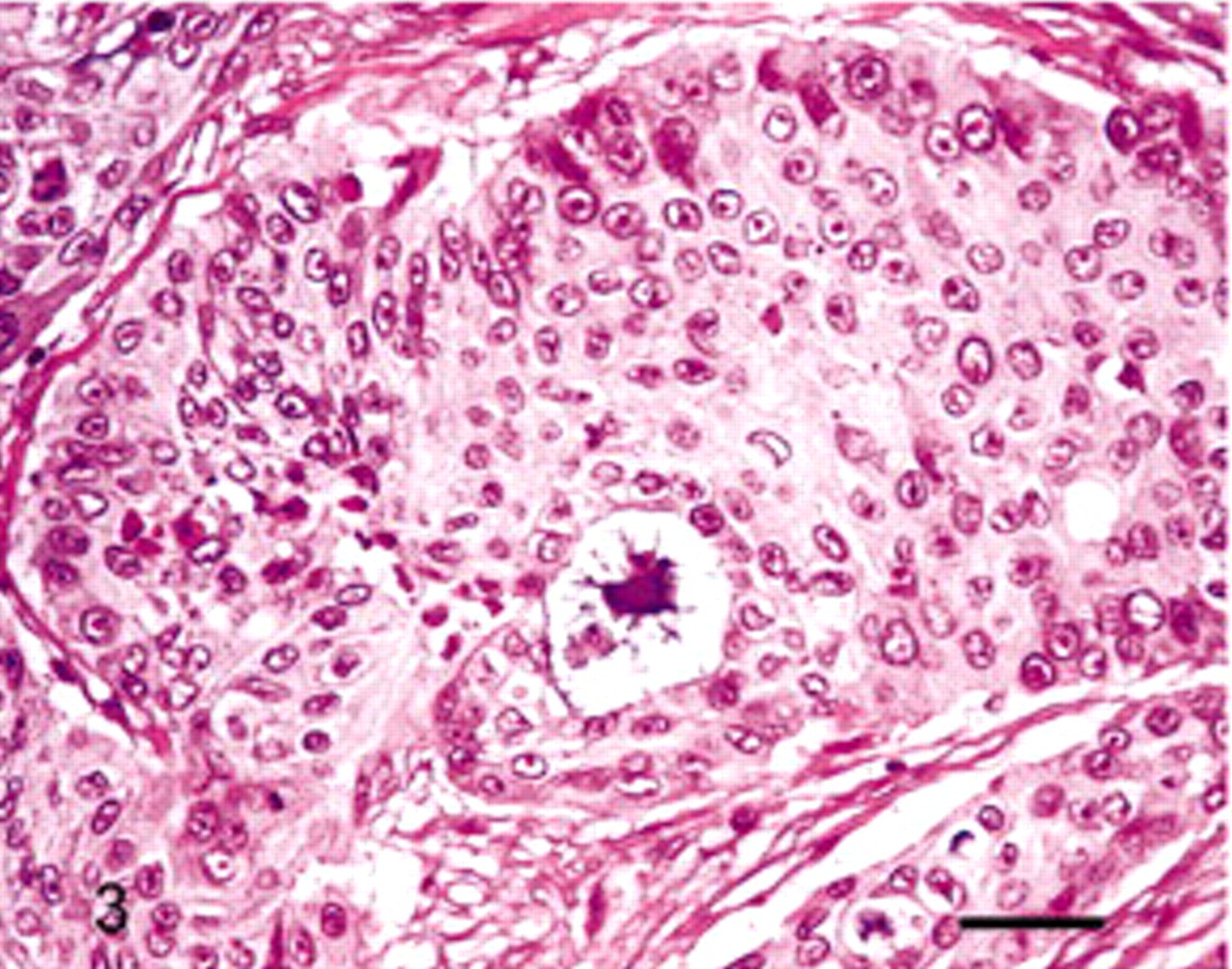
Salivary gland; lion. Sparse and small cystic spaces containing low amounts of bluish fluid were observed in neoplastic islands. HES. Bar = 50 μm.
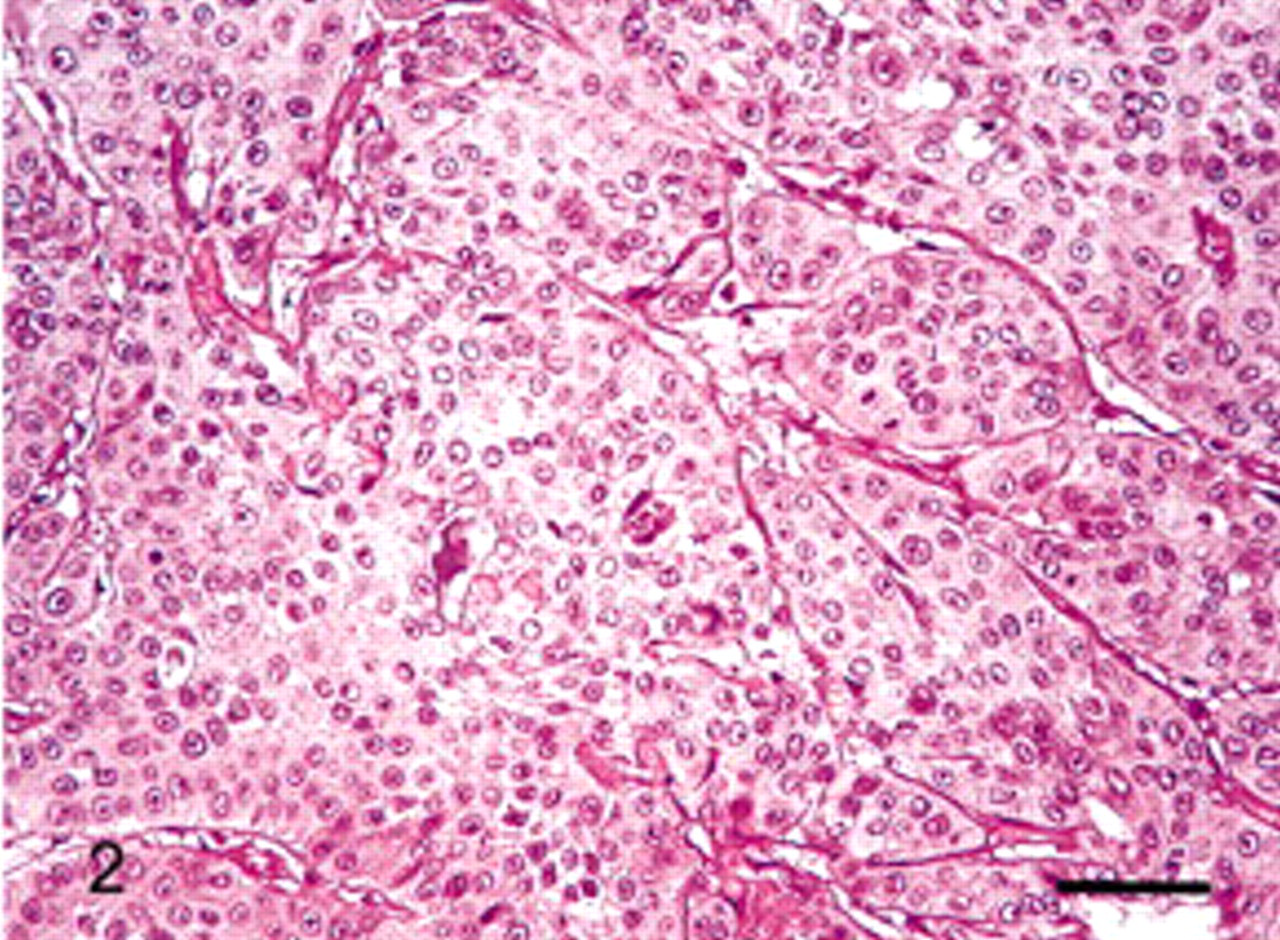
Salivary gland; lion. Tumor cells were organized in a trabecular pattern, with anastomosing, irregular islands surrounded by a moderatly abundant stroma. HES. Bar = 80 μm.
Immunohistochemical stains showed that tumor cells were positive for panCK and negative for S100 protein. Tumor cells were negative for α-SMA, but myofibroblastic stroma was strongly positive (Fig. 4). Peripheral cells of tumor islets were positive for vimentin and p63, whereas luminal tumor cells were not (Figs. 5, 6). Taken together, the histologic and immunohistochemical results are consistent with the features of mucoepidermoid carcinoma described in humans. 1,4,14,17
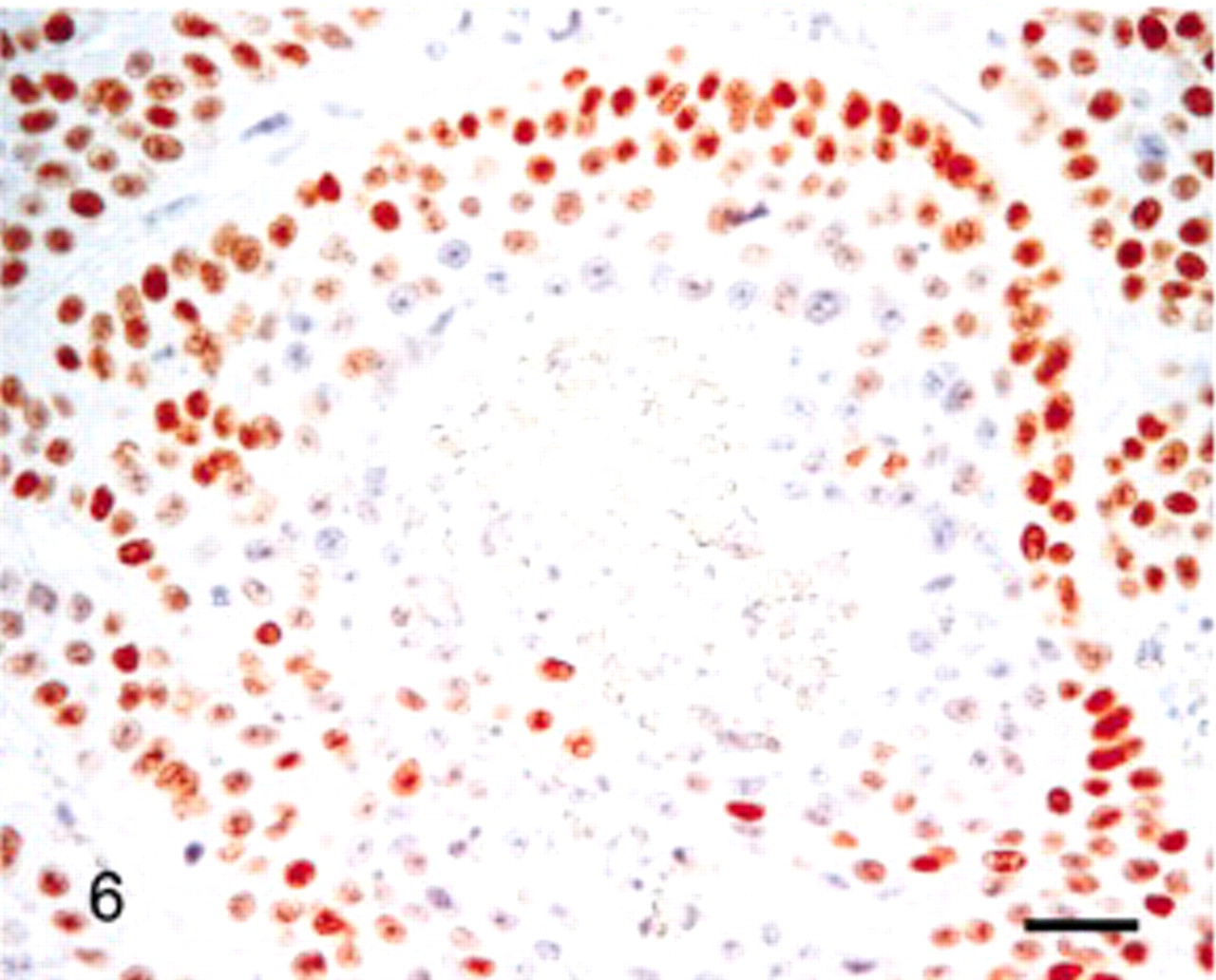
Salivary gland; lion. Peripheral tumor cells expressed p63, whereas central tumor cells did not. Bar = 40 μm.
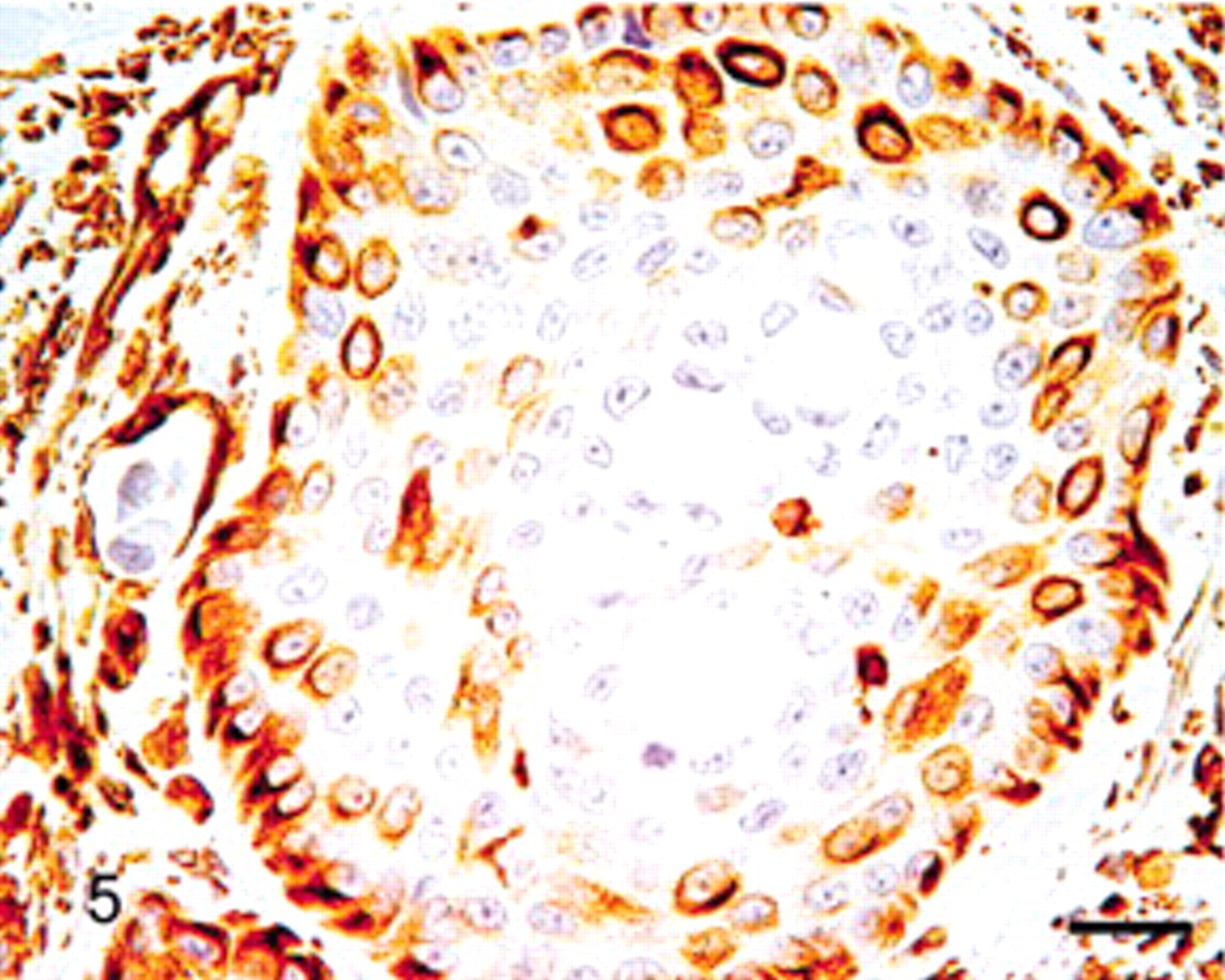
Salivary gland; lion. Peripheral tumor cells stained positive for vimentin, whereas central tumor cells did not. Bar = 40 μm.
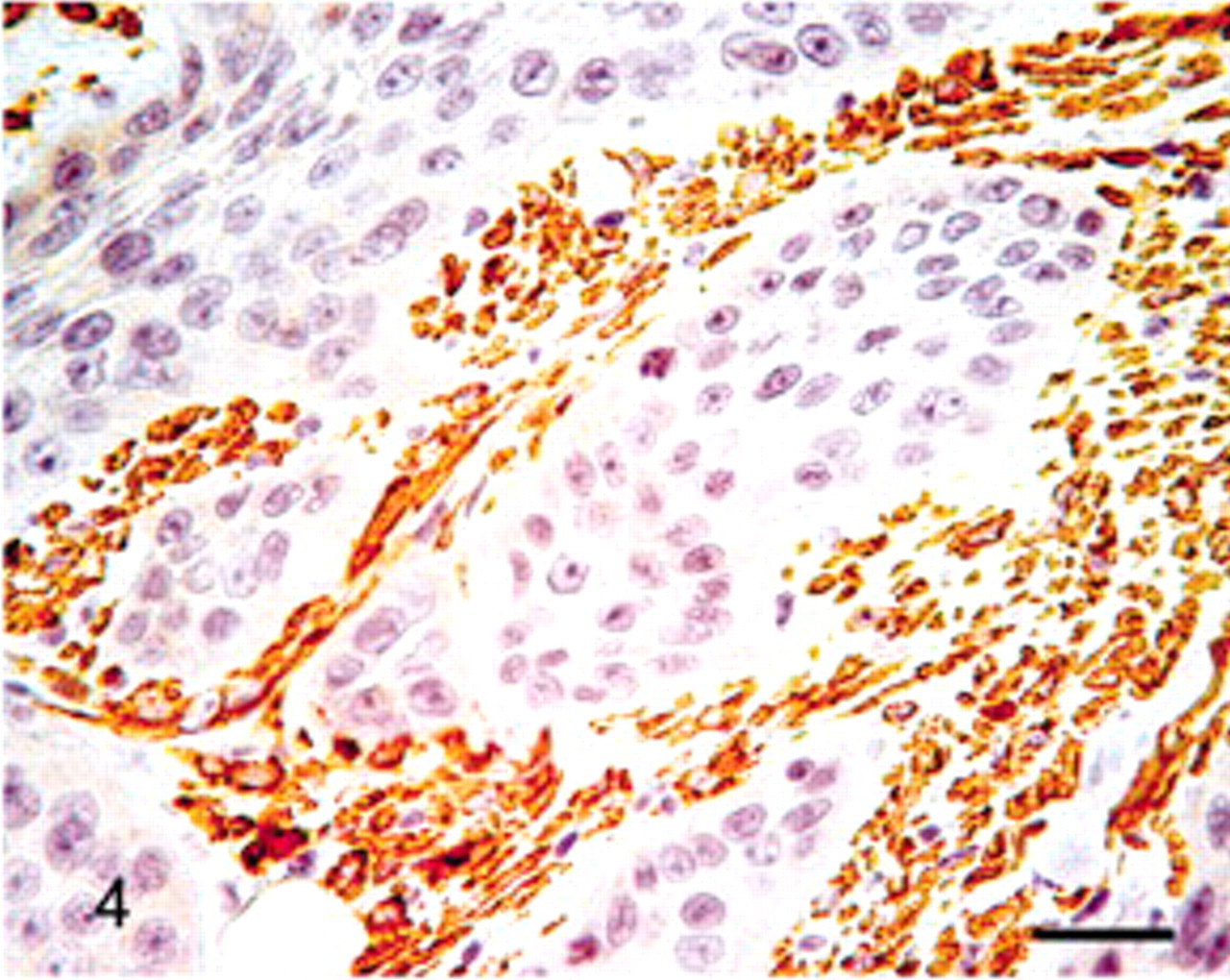
Salivary gland; lion. Tumor cells were negative for α-SMA, but myofibroblastic stroma was strongly positive. Bar = 40 μm.
In humans, mucoepidermoid carcinoma of the salivary gland is classified as low, intermediate, or high grade according to several histopathologic characteristics, including the number of cystic spaces, number of mucinous cells, number of mitoses, pleomorphism, and metastases. 1 In the present case, the mucoepidermoid carcinoma displayed a small number of cystic spaces and mucous cells, along with a high mitotic index and metastases. So, consistent with the above criteria, the tumor was classified as a high-grade mucoepidermoid carcinoma.
In human pathology, salivary gland neoplasms represent less than 0.5% of all neoplasms and most often involve the major salivary glands (85–90%), particularly the parotid gland (64–84%). 5,21 Only 30% of all salivary gland tumors are malignant in humans. 5 Among these tumor types, mucoepidermoid carcinoma is the most common malignant salivary gland tumor and comprises 5–12% of all salivary gland tumors. 7
To the authors' knowledge, primary tumors of the salivary gland have not been reported in wild felids, but cases have been described in domestic species (dog, cat, horse, cow, sheep, and goat) and laboratory animals (rat, guinea pig, and baboon). 3 Neoplasms of the salivary gland are uncommon in dogs and cats, with a reported incidence of 0.17% and the major salivary glands (mandibular and parotid) being most often affected (75–80% of cases). The mandibular salivary gland is the most common primary site in cats (59%), whereas in dogs, the most common site is the parotid gland (50%). 19 According to Hammer et al., 12 simple adenocarcinoma is the most prevalent tumor type in cats and dogs. Other types in order of decreasing frequency are squamous cell carcinoma, mucoepidermoid carcinoma, anaplastic carcinoma, and complex carcinoma. 12 In the recently revised “WHO International Histologic Classification of Tumors of the Alimentary System of Domestic Animals,” primary and malignant epithelial neoplasms are still the most frequent tumors of the salivary gland. 13 The two most common tumor types are represented by acinic cell carcinoma and adenocarcinoma. 3,12 Acinic cell carcinomas are characterized by neoplastic, round, or polyhedral serous cells that have a basophilic granular cytoplasm and are arranged in sheets or acini. Acinic cell carcinoma resembles the normal parotid gland, apart from the absence of striated ducts and the absence of subdivision into lobules. The tumor cells may produce secretion, which can accumulate and produce spaces containing clear fluid. However, mucicarmin staining is negative for this fluid. 22 Adenocarcinoma is composed of a single cell type, which is arranged to form neoplastic ducts and tubules. The mucous type of adenocarcinoma must be distinguished from mucoepidermoid carcinoma by the presence of at least scanty or poorly formed epidermoid constituents in the latter.
Tumors are reported to be 1.6 times more prevalent in the salivary gland of cats than in dogs. 19 Cats also seem to be affected by more aggressive disease, with more than half of the animals having nodal involvement and/or distant metastases (lung, liver, pancreas, heart, adrenal gland, diaphragm, body wall, and bone) at the time of initial diagnosis. 12 As in domestic cats, the tumor observed in this lion involved the mandibular salivary gland and displayed malignant behavior, with local aggressiveness and distant metastases to several visceral organs.
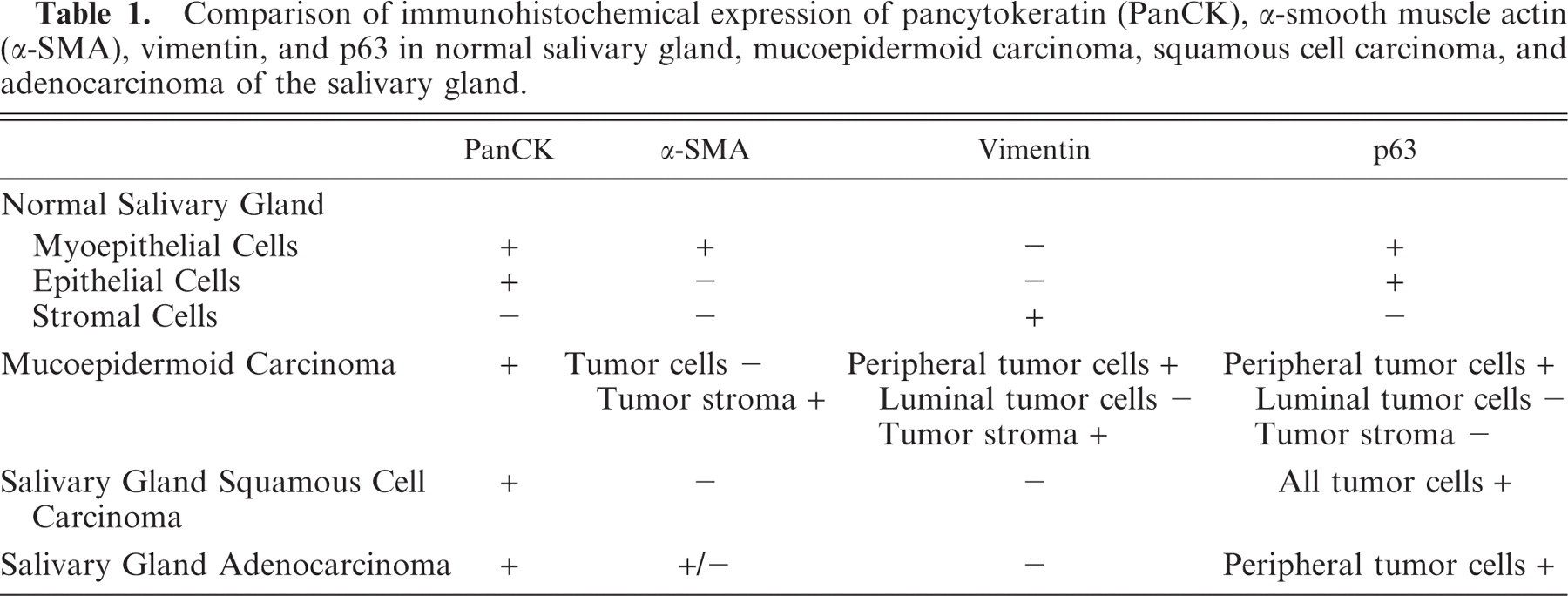
Differential diagnosis of malignant epithelial tumors of the salivary gland in animals includes squamous cell carcinoma, poorly differentiated adenocarcinoma, and mucoepidermoid carcinoma. 17 The histologic diagnosis of a mucoepidermoid carcinoma requires the identification of the three characteristic cell types (i.e., mucous, epidermoid, and intermediate cells), which are present in varying proportions. However, these characteristic features may be difficult to recognize, particularly in high-grade tumors. 13 Thus, in human pathology, the immunohistochemical profile of tumor cells is used to discriminate between these different tumor types. In squamous cell carcinoma, tumor cells are positive for panCK, negative for S100 protein and vimentin, and always negative for α-SMA, whereas all of the tumor cells are positive for p63. In adenocarcinoma, the tumor cells are positive for panCK, negative for S100 protein and vimentin, partially positive for α-SMA, and partially positive for p63 (but only in cells located at the periphery). 9 In mucoepidermoid carcinoma, tumor cells are positive for panCK, negative for S100 protein, always negative for α-SMA, and partially positive for p63 (basal, intermediate, and squamous cells express p63, in contrast to luminal cells). 2 Peripheral tumor cells in mucoepidermoid carcinoma and tumor stroma are positive for vimentin. 14,18 Consequently, the expression of α-SMA by tumor cells discriminates adenocarcinoma (partially positive) versus mucoepidermoid carcinoma and squamous cell carcinoma (negative). Moreover, vimentin expression distinguishes mucoepidermoid carcinoma (expression by peripheral tumor cells) from squamous cell carcinoma and adenocarcinoma (no expression by tumor cells). Finally, p63 differentiates squamous cell carcinoma (positive results for all tumor cells) from mucoepidermoid carcinoma and adenocarcinoma (positive results restricted to peripheral tumor cells). As in human pathology, the combination of α-SMA, vimentin, and p63 immunohistochemistry allowed us in this case to establish that the salivary gland tumor in this lion was a mucoepidermoid carcinoma rather than a squamous cell carcinoma or a poorly differentiated adenocarcinoma (Table 1).
Comparison of immunohistochemical expression of pancytokeratin(PanCK), α-smooth muscle actin (α-SMA), vimentin, and p63 in normal salivary gland, mucoepidermoid carcinoma, squamous cell carcinoma, and adenocarcinoma of the salivary gland.
An accurate diagnosis for high-grade mucoepidermoid carcinoma is important for appropriate prognostic and therapeutic considerations. 11 Indeed, in humans, high-grade mucoepidermoid carcinomas are reported for their highly malignant behavior and metastatic potential when compared to a squamous cell carcinoma, as well as for exhibiting radioresistence relative to an adenocarcinoma. Further studies are needed to verify and extend the utility of this immunohistochemical protocol for dogs, cats, and other species, thus assisting in the differential diagnosis of malignant neoplasms of the salivary gland.
Footnotes
Acknowledgements
We would like to thank Mrs. Catherine Guéreaud and Mr. Bernard Fernandez of the Pathology Department of Nantes Veterinary School for technical assistance.
