Abstract
Mucoepidermoid carcinoma is one of the most common malignant tumors in salivary glands and is usually associated with mastermind-like transcriptional coactivator 2 (MAML2) rearrangement. Primary nasopharyngeal mucoepidermoid carcinoma is extremely rare, and MAML2 status was reported in only two studies. Herein, we present a 70-year-old male patient with incidentally found nasopharyngeal mucoepidermoid carcinoma. MAML2 translocation was detected by fluorescence in situ hybridization test. Additionally, we conducted a comprehensive literature review and summarized the clinicopathological features of this rare condition. Nasopharyngeal mucoepidermoid carcinoma shows a similar mean age at diagnosis and gender ratio to those of mucoepidermoid carcinoma in salivary glands. More than half of the patients exhibit high histologic grade at the time of diagnosis. As MAML2 status is unreported in almost all published cases, further studies are needed to explore the incidence and prognostic value of MAML2 rearrangement in nasopharyngeal mucoepidermoid carcinoma.
Introduction
Mucoepidermoid carcinoma (MEC) was first described by Stewart et al. in 1945. 1 Modern molecular studies demonstrate that most MECs harbor mastermind-like transcriptional coactivator 2 (MAML2) rearrangement. Approximately half of MECs occur in major salivary glands, predominantly in parotid glands. Other frequent sites include palate and buccal mucosa. 2 Primary nasopharyngeal mucoepidermoid carcinoma (NPMEC) is extremely rare. In a population-based analysis in the United States of America, the overall incidence of nasopharyngeal salivary gland-type cancers was measured at 0.019 per 100,000 people, with MEC being the third most common cancer, accounting for 13.3% of all cases. 3
In our search, we found a total of 115 reported cases of NPMEC in 20 articles. Of the reviewed literature, MAML2 analysis was performed in only two studies, which identified three cases of NPMEC with MAML2 rearrangement.4,5 Herein, we present a case of incidentally found NPMEC with MAML2 translocation that was initially diagnosed in a small biopsy and confirmed by a subsequent resection specimen. Additionally, we conducted a comprehensive review literature regarding the epidemiology, clinicopathological characteristics, treatment, and prognosis of NPMEC.
Case presentation
The patient was a 70-year-old Chinese gentleman with hypertension and diabetes on medications. He claimed to be a social drinker but denied smoking. He did not report any special family history. He was brought to the emergency room of our hospital in January 2023 due to a traffic accident. Brain computerized tomography (CT) scan revealed right frontal lobe subarachnoid hemorrhage and right medial orbital wall fracture. Incidentally, an isodense mass was found in the right lateral wall of nasopharynx with obliteration of right lateral pharyngeal recess in the CT scan. He denied nasal obstruction, epistaxis, tinnitus, hearing loss, headache, or any other specific symptoms.
He was then referred to the department of otorhinolaryngology after his head trauma stabilized. Further magnetic resonance imaging (MRI) examination revealed a 3.0 cm × 1.6 cm × 2.5 cm contrast enhancing mass in the right wall of nasopharynx and extended to right lateral pharyngeal recess of the nasopharynx with right para-pharyngeal space compression (Figure 1(a) and (b)). No detectable abnormal signal image was found in the skull basal region and the visible brain. No enlarged lymph node was found at bilateral carotid spaces. Serum Epstein–Barr virus (EBV)-DNA was not detected. Endoscopic examination found an irregularly surfaced fragile mass in the right lateral wall of nasopharynx (Figure 1(c) and (d)). Tissue from the mass was taken for biopsy.

Magnetic resonance imaging revealed a 3 cm contrast enhancing mass (red arrow) in the right wall of nasopharynx and extended to the right lateral pharyngeal recess of the nasopharynx ((a) Axial view. (b) Coronal view). Endoscopic examination found an irregularly surfaced fragile mass in the right lateral wall of nasopharynx (c). Narrow band imaging revealed multiple dilated and irregular caliber of intra-epithelial papillary capillary loops on the surface (d).
The histological section showed an infiltrative tumor composed of squamoid, mucin-producing and intermediate-type cells arranged in solid or cystic pattern, setting in fibrous to hyaline stroma. The tumor cells exhibited mildly to moderately pleomorphic nuclei with prominent nucleoli. Mitosis was inconspicuous. No lymphovascular permeation or perineural invasion was identified. No necrosis was apparent. On immunohistochemical study, the tumor cells were diffusely positive for keratin 7, partially positive for p63, and negative for SOX10 and thyroid transcription factor 1 (TTF1). In situ hybridization for Epstein–Barr virus-encoded small RNAs (EBER) was negative (Ventana® INFORM EBER probe by Roche, Basel, Switzerland) (Figure 2). Fluorescence in situ hybridization (FISH) test using ZytoLight® SPEC MAML2 dual color break apart probe (ZytoVision, Bremerhaven, Germany) was performed. Split signal of MAML2 was observed in 119 (78.8%) out of 151 informative tumor cell nuclei, exceeding the cutoff of 15% and was interpreted as a positive result (Figure 3). A diagnosis of low-grade MEC was rendered based on the small biopsy even though the anatomical site was unusual.
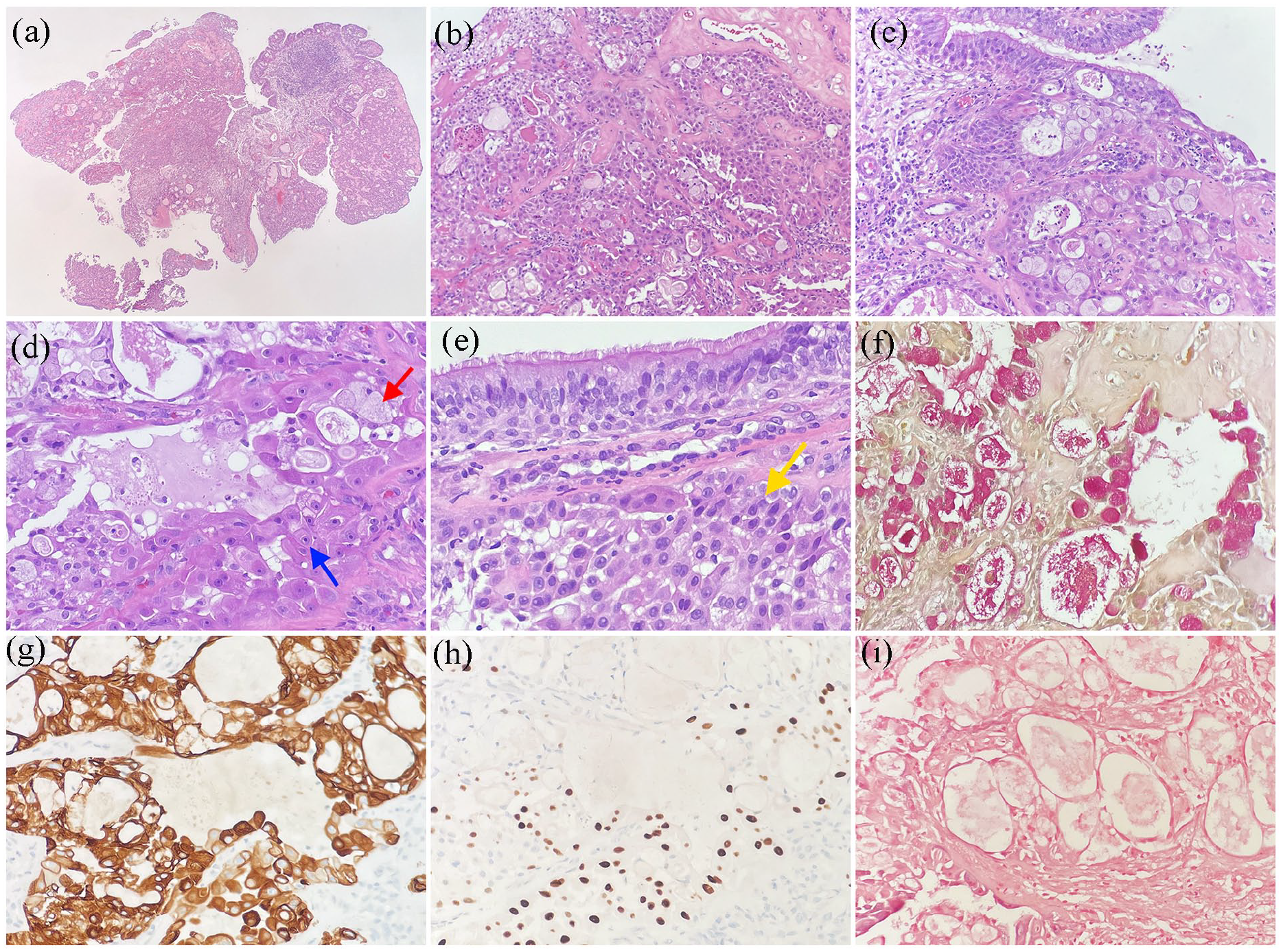
Histological findings of the nasopharyngeal biopsy. Sections showed nasopharyngeal tissue infiltrated by a tumor composed of mucous cells (red arrow), intermediate cells (yellow arrow), and squamoid cells (blue arrow), arranging in solid clusters or cystic pattern. ((a)–(e)) (h&e staining). Mucicarmine stain demonstrated intracytoplasmic mucin and mucinous material in the cystic spaces (f). On immunohistochemical study, the tumor cells were diffusely positive for keratin 7 (g). The squamoid cells and intermediate cells were also positive for p63 (h). In situ hybridization for EBV-encoded small RNA was negative (i). (Original magnification: (a) ×20, (b) ×100, (c) to (i) ×200).

Mastermind-like transcriptional coactivator 2 (MAML2) break apart fluorescence in situ hybridization of the nasopharyngeal biopsy. Tumor cells with translocation affecting the 11q21 locus as indicated by one separate orange and one separate green signal (white arrow). The 5′ end of MAML2 was labeled with green probe, and the 3′ end of MAML2 was labeled with orange probe.
After a multidisciplinary team discussion, the patient was referred to a tertiary hospital in Hong Kong for endoscopic nasopharyngectomy with right selective neck dissection in August 2023. Pathological examination of the resection specimen confirmed low-grade MEC with clear resection margin. The pTNM staging according to American Joint Committee on Cancer (AJCC) Cancer Staging Manual 8th edition was pT2N0M0. 6 Multidisciplinary team discussion after operation decided for observation, and no further radiotherapy nor chemotherapy was administered. The patient was regularly followed up in our hospital after discharge and showed a good recovery. His follow-up 8 months after operation showed no evidence of tumor recurrence or complication by endoscopy and MRI examinations.
Methods
We performed a literature search following the Preferred Reporting Items of Systematic reviews and Meta-Analyses (PRISMA) guideline 2020. 7 The electronic search was performed up until March 31, 2024, in PubMed, Web of Science and Scopus databases with keywords “(nasopharynx OR nasopharyngeal) AND mucoepidermoid carcinoma” and “(nasopharynx OR nasopharyngeal) AND salivary gland.” No restrictions were placed on the language of the articles or the date of publication. The cited references in the reviewed articles related to the topic were assessed to widen the search for further relevant papers. The following inclusion criteria were used for the selection of articles: (1) human studies mentioning NPMEC. The exclusion criteria were as follows: (1) studies that did not report on NPMEC; (2) the anatomical site of MEC was not clearly specified in nasopharynx; (3) unavailable full-text articles; (4) studies with overlapping data. Two authors (CFC and LFK) independently screened, agreed on the selection of eligible articles and achieved consensus on which studies to include. Since most eligible articles were case reports and case series, we used the tool proposed by Murad et al. to assess the quality of eligible articles. 8 The following data were extracted from each selected study: authors; period of cohort; region; age and gender; tumor grade; TNM stage; MAML2 status; treatment; follow-up. These data were classified and tabulated (Tables 1 and 2).
Nasopharyngeal mucoepidermoid carcinomas reported in the literature.

±: With or without; AWD: Alive with disease; ChT: Chemotherapy; DOD: Dead of disease; F/U: follow-up; F: Female; FISH: Fluorescence in situ hybridization; M: Male; NA: Not available; NED: Alive with no evidence of disease; RT: Radiotherapy; S: Surgery.
Clinicopathological characteristics of 116 nasopharyngeal mucoepidermoid carcinomas.

ChT: Chemotherapy; RT: Radiotherapy; ±: With or without.
Results
The initial search of the databases identified 3,445 articles. One additional record was identified through a manual search in the article references. Abstracts were subsequently analyzed. A total of 31 articles were selected for full-text analysis, which included long-term cohorts and sporadic case reports. Four of these articles were excluded because the full-text was not available. Nine articles from two China-based long-term cohorts contained overlapping data. We selected one article with most complete information for statistical analysis from each of these two cohorts, and the remaining seven articles were excluded. Finally, a total of 115 NPMEC patients reported in 16 English-language studies, three Japanese-language studies and one Chinese-language study were included for review (Figure 4).

Flow diagram of literature search according to Preferred Reporting Items of Systematic reviews and Meta-Analyses (PRISMA) 2020 guideline.
Based on the available data from the reviewed articles and our reported case, the age at diagnosis of NPMEC ranges from 7 to 78 years, with a mean age of 45 years, a median age of 48 years, and a slight female predilection of 1.03:1. The mean age at diagnosis and gender ratio of NPMEC are similar to those in mucoepidermoid carcinoma of salivary glands (SGMEC) reported by the World Health Organization (mean age of 45 years and female predilection of 1.1–1.5:1) (p value >0.05. See Supplemental material Statistical Analysis). 2 The most common histological grade at diagnosis is high-grade (55.8%), followed by low-grade (32.7%), and intermediate grade (11.5%). The TNM staging in the reviewed literature was according to AJCC Cancer Staging Manual spanning from the 5th to the 8th edition. Due to insufficient information provided in the literature, it was difficult to restage all the patients. Thus, we simply summarized the cancer stage from the original papers. Stages I and II account for 53.3% of patients at the time of diagnosis, whereas stages III and IV account for 46.7% of them. Among the patients with available data, 66.1% underwent primary surgery with or without adjuvant radiotherapy, while 33.9% received primary radiotherapy with or without chemotherapy or surgery. Regarding the follow-up survival outcomes, 53.3% are alive with no evidence of disease, 16.7% are alive with disease, and 30.0% are dead of disease. MAML2 rearrangement is detected in four out of six patients.
Discussion
Primary NPMEC is extremely rare, and the incidence is estimated to be 0.0025 per 100,000 people. 3 Our search found a total of 115 reported cases of NPMEC in 20 articles. In this article, we present a new documented case of NPMEC, adding to the 115 previously reported cases.
In human beings, there are about 600 to 1000 minor salivary glands spreading throughout the aerodigestive tract submucosa, with relatively fewer glands located in nasopharynx. 26 In 2020, Valstar et al. described a pair of previously unnoticed macroscopic salivary glands in human nasopharynx, namely tubarial glands. 27 Their subsequent study suggests that tubarial glands closely resemble palatal salivary glands. 28 Salivary gland-type nasopharyngeal tumors are hypothesized to raise from these salivary gland tissues in nasopharynx.
The morphological and immunohistochemical features of NPMEC are similar to those of SGMEC.10,15 Histologically, it is typically composed of a mixture of mucous cells, intermediate cells, and squamoid cells. Significant keratinization is exceptional, though a case of psammomatous NPMEC was reported. 10 According to the WHO Classification of Tumors for Head and Neck, 2 primary nasopharyngeal malignancies include nasopharyngeal carcinoma (NPC) and low-grade nasopharyngeal papillary adenocarcinoma. Exclusion of NPC is essential in the regions with a high incidence rate, such as southern China and Southeast Asia. Since NPC is highly chemo- and radiosensitive, the preferred treatment is radiotherapy with or without concurrent systemic therapy, 29 whereas surgical excision may be the treatment of choice for other histological types of nasopharyngeal malignancies. 2 The typical morphological features of MEC are usually distinguishable from NPC and low-grade nasopharyngeal papillary adenocarcinoma.
Immunohistochemically, MEC expresses p63 and is often positive for keratin 7. 30 In contrast, conventional NPC is negative for keratin 7 and positive for in situ hybridization for EBER. Low-grade nasopharyngeal papillary adenocarcinoma is characteristically positive for TTF1. Furthermore, p63 or p40 expression in absence of S100 protein/SOX10 staining may help to differentiate MEC from other salivary tumors. 2
MEC is specifically associated with a unique t(11;19) translocation, and the resulting CRTC1::MAML2 fusion is a major oncogenic driver for MEC initiation and maintenance. 31 CRTC1::MAML2 fusion is identified in most low- and intermediate-grade SGMEC patients and in some high-grade cases. Rare SGMEC patients harbor CRTC3::MAML2 fusion or EWSR1::POU5F1 fusion. 2 MAML2 analysis is useful in diagnostic workups and may aid in the diagnosis of MEC with unusual features, such as being devoid of squamoid cells by immunohistochemistry. 4
A study by Kuo et al. based on a limited number of patients from Taiwan, China—one of the regions with high incidence of conventional NPC—suggested that the oncogenesis of NPMEC might be related to EBV. 10 However, such a correlation has not been reported by other studies. Our reported case is also from the region with high incidence of NPC but in situ hybridization for EBER is negative.
Currently, there is no consensus on the treatment for NPMEC. Nasopharyngectomy is suggested for resectable cases.11,22,25,32 Schramm et al. reported that occult neck disease was as high as 47% in patients with nasopharyngeal salivary gland malignancy. 11 They recommended elective neck dissection as part of the surgical treatment. Although NPMEC appears to be relatively radioresistant, 22 postoperative radiotherapy is recommended for advanced stages, high-grade tumors, residual tumors, and positive surgical margins.11,16,32,33
A population-based analysis in the United States of America demonstrated the 1-, 5-, and 10-year disease-free survival rates (DFS) of NPMEC were 84.4%, 65.3%, and 52.0%, respectively. 3 Two China-based cohorts reported variable outcomes. Liu et al. reported that the 5-year overall survival (OS) and DFS of 12 NPMEC patients were 50.4% and 41.4%, respectively.33 Sun et al. analyzed the outcomes of 17 patients with NPMEC. The 5-year overall OS, loco-regional failure free survival rate (LRFFS) and distant failure free survival rate were 69.7%, 64.7%, and 86.9%, respectively.32
The studies regarding primary salivary gland-type nasopharyngeal carcinoma (SNPC) found that younger age, earlier stage, and Asian ethnicities were positive prognostic factors, whereas cranial nerve invasion and lymph node metastasis were negative prognostic factors.3,32,33 Additionally, Sun et al. found significant association between the radiotherapy techniques and survival.32 Patients received intensity modulated radiation therapy had better 5-year OS and LRFFS than those underwent two-dimensional radiotherapy. However, their analyses were based on all histological subtypes of SNPC, and they did not further analyze the prognostic factors specific for NPMEC.
CRTC1::MAML2 fusion has been regarded as an additional marker of favorable prognosis in SGMEC. 2 As MAML2 status is unreported in almost all published cases of NPMEC, the incidence and prognostic value of MAML2 rearrangement in NPMEC remain uncertain.
Conclusion
In this article, we report a case of primary NPMEC harboring MAML2 translocation. NPMEC is an extremely rare condition. Our search found a total of 115 NPMEC patients reported in 20 studies. According to our review of literature, the mean age at diagnosis of NPMEC is 45 years old with a slight female predilection, which is similar to those of MEC in salivary glands. More than half of patients exhibit high histologic grade at the time of diagnosis. Nasopharyngectomy is preferred treatment for resectable cases. Postoperative radiotherapy is recommended for patients with advanced stages, high-grade tumors, residual tumors, and positive surgical margins. The prognosis of NPMEC may be influenced by tumor stage, age, race, cranial nerve invasion, lymph node metastases, and radiotherapy technique. As MAML2 status is unreported in almost all published cases, the incidence and prognostic value of MAML2 rearrangement in NPMEC remain uncertain, and further studies are needed to explore this aspect.
Supplemental Material
sj-docx-1-sco-10.1177_2050313X241281323 – Supplemental material for Nasopharyngeal mucoepidermoid carcinoma with mastermind-like transcriptional coactivator 2 translocation: A case report and comprehensive literature review of a rare entity
Supplemental material, sj-docx-1-sco-10.1177_2050313X241281323 for Nasopharyngeal mucoepidermoid carcinoma with mastermind-like transcriptional coactivator 2 translocation: A case report and comprehensive literature review of a rare entity by Chan-Fong Chio, Wan-Pang Sam, Sio-In Wong, Cheong-Un Sio, Lai-Fong Kok, Ki-Man Pai and Thazin Hlaing in SAGE Open Medical Case Reports
Footnotes
Acknowledgements
None.
Author contributions
C.-F.C. Conceptualization, Investigation, Formal analysis, Methodology, Writing—original draft; W.-P.S. Conceptualization, Investigation, Writing—review & editing; S.-I.W. Resources, Writing—review & editing; C.-U.S. Investigation, Writing—review & editing; L.-F.K. Conceptualization, Methodology, Writing—review & editing; K.-M.P. Supervision, Writing—review & editing; T.H. Supervision, Writing—review & editing.
Data availability
The radiology images, endoscopy images, and pathology sections used to support the findings of this case report are included within the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval to report this case was obtained from the Medical Ethic Committee at Conde de São Januário Hospital, Macao SAR, China (0029/MEC/N/2024).
Informed consent
Written informed consent was obtained from the patient for the publication of anonymized information and images included in this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
