Abstract
Salivary gland neoplasia is uncommon in veterinary species and has rarely been reported in cattle. Adenocarcinoma, squamous cell carcinoma, and pleomorphic carcinoma have been described in the parotid gland of dairy cows. Here we describe a case of high-grade mucoepidermoid carcinoma in a bull. The bull had a chronic history of caudal mandibular soft tissue swelling. Postmortem examination revealed a 30 × 30-cm mass with a caseonecrotic center, hemorrhage and necrosis of the ipsilateral cervical musculature, osteolysis of the right paracondylar process of the skull and right horn base, pulmonary nodules, and enlarged tracheobronchial lymph nodes. Histology of the mass, lungs, and lymph nodes revealed an invasive neoplasm composed of epithelial cells arranged in nests and ductular structures supported by a spindle cell stroma, with frequent central necrosis. Immunohistochemistry revealed that epithelial cells were diffusely positive for pancytokeratin and p63 with multifocal vimentin positivity; stromal cells were diffusely positive for vimentin and α-SMA. A salivary gland mucoepidermoid carcinoma was diagnosed based on these findings. Although rare, salivary gland neoplasia should be considered a differential diagnosis for mandibular masses in cattle.
Salivary gland neoplasms are uncommon in all veterinary species, occurring most often in dogs and cats,11,13 rarely in cattle,2,16,17 and sporadically in nontraditional species5–7; they remain unreported in swine. 13 A retrospective study including almost 600 tumors in cattle over a 45-y period identified no salivary neoplasms. 12 Documented reports of parotid salivary gland neoplasia in cattle include pleomorphic carcinoma (2 cases), 2 adenocarcinoma (2 cases),16,17 and squamous cell carcinoma (1 case), 2 all occurring in dairy cows. The tumors were described as firm nonpainful masses on the side of the face and under the eye.
Mucoepidermoid carcinoma (MEC) has not been reported in cattle but has been reported in a lion, 6 mice, dogs, 11 and an Amazon parrot, 14 although the neoplastic tissue of origin in the parrot was not definitive. MEC accounts for ~30% of malignant salivary tumors in humans,1,15 and was the second most common malignant salivary neoplasm of dogs in a retrospective study. 11
A 6-y-old mixed beef-breed bucking bull was presented to Mississippi State University Animal Health Center (MSU-AHC; USA) for evaluation of a mandibular mass. A limited physical exam was performed given the bull’s aggressive nature. Examination revealed a fluctuant 30 × 30-cm mass and mild soft tissue swelling. Ultrasound of the mass showed a large, central area of hypoechoic fluid surrounded by a thick capsule. Fluid was aspirated from the mass and described as purulent and foul-smelling. The clinical diagnosis was of a soft tissue abscess. The mass was lanced and drained via a stab incision in the ventral-most aspect. The defect was lavaged with copious amounts of a dilute betadine solution and packed with betadine-soaked gauze. This treatment process was repeated 3 additional times during hospitalization. At the time of discharge, the mass had decreased dramatically in size to 15 × 10 cm.
One month after discharge from the hospital, the bull was returned to MSU-AHC because of progressive enlargement of the mass and associated soft tissue swelling. The mass had increased to 38 × 40 cm and extended up the side of the face to the right eye and base of the right ear with marked ventral subcutaneous edema. Given the bull’s aggressive behavior, exploration and treatment under general anesthesia were elected. Shortly after anesthesia was induced, the bull’s respiration ceased. Immediate intubation was attempted but was unsuccessful because of laryngeal edema. The bull was then submitted for autopsy.
Postmortem examination revealed a 30 × 30-cm, irregularly round, fluctuant soft tissue swelling extending from the base of the right ear to the caudoventral right mandible with a central draining tract exuding abundant red-to-yellow caseous material (Fig. 1A). On cut surface, the mass had extensive fibrosis with a caseonecrotic center (Fig. 1B). Given the extensive tissue destruction and/or invasion, the tissue of origin could not be identified but was likely the submandibular salivary gland given the location of the largest mass. There were 2 similar 5-cm masses caudal and ventral to the primary mass. The cervical skeletal muscle from the base of the right horn to the thoracic inlet was dry with extensive dark-red-to-purple discolored streaks. The right paracondylar process of the skull was largely absent, and there were numerous small bony fragments at the point of attachment of the mass to the skull and at the base of the right horn.
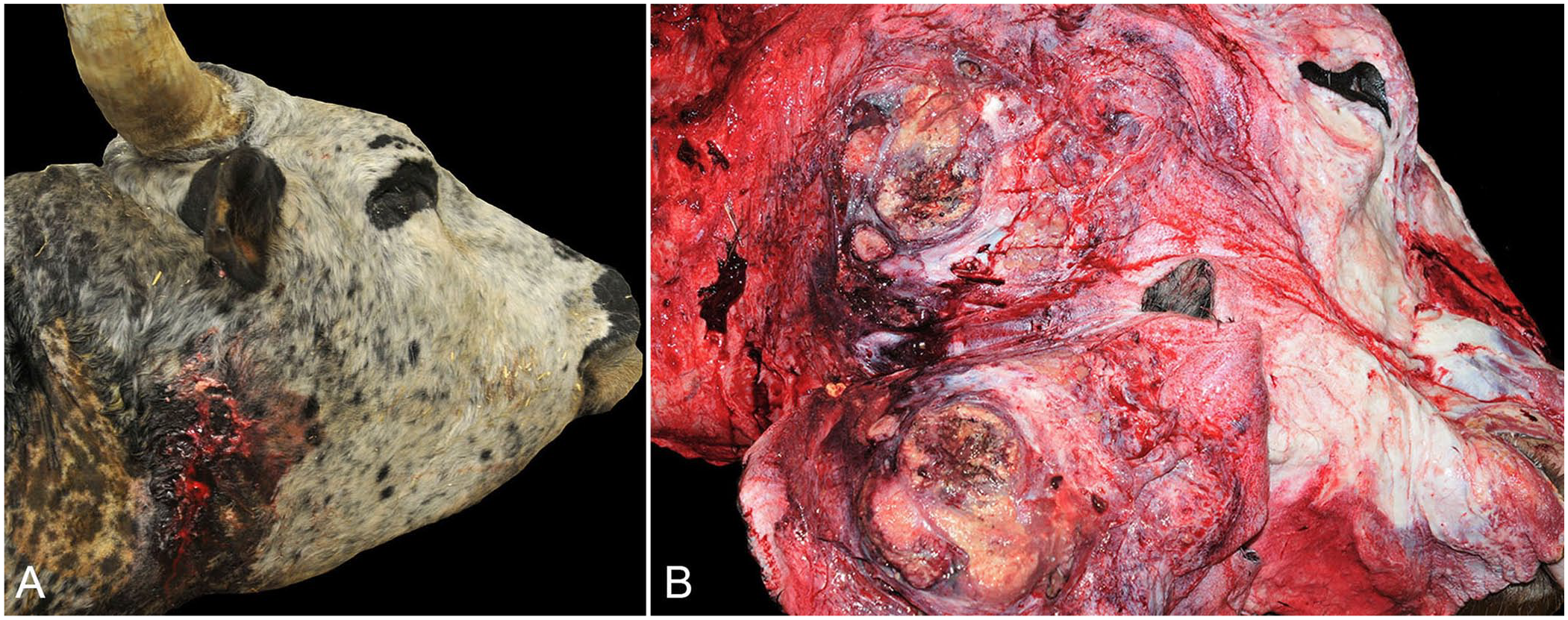
Mucoepidermoid carcinoma of the salivary gland in a bull.
Approximately 70% of the lungs were mottled dark-red-to-purple with numerous, firm, white-to-yellow nodules. Yellow caseous material exuded from the cut surface of the nodules. The tracheobronchial lymph nodes were markedly enlarged; the largest node was 15 cm long. The cut surfaces of the lymph nodes were yellow, firm, and bulged slightly. Other regional lymph nodes had no grossly apparent abnormalities.
A complete set of tissue samples, including the mandibular mass, lungs, and tracheobronchial lymph nodes, were collected and fixed in 10% neutral-buffered formalin. Samples were processed routinely, paraffin-embedded, sectioned at 5 μm, and stained with H&E. Following examination with H&E, periodic acid–Schiff (PAS) and Alcian blue stains were performed. Immunohistochemistry (IHC) for cytokeratin, vimentin, alpha–smooth muscle actin (α-SMA), and tumor protein 63 (p63) was performed on additional sections of the tumor (Table 1). The p63 IHC was performed at the Purdue Animal Disease Diagnostic Laboratory (West Lafayette, IN, USA; Table 1).
Antibodies used to characterize a bovine mucoepidermoid carcinoma, with source and manufacturer, clone, retrieval information, dilution, and detection method.

In all cases, the chromogen used was diaminobenzidine (DAB); the autostainer was IntelliPATH (Biocare).
Heat retrieval, 110°C for 15 min (RD, Reveal Decloaker; Biocare RV1000).
Stain performed at Purdue Animal Disease Diagnostic Laboratory, West Lafayette, IN, USA.
Heat retrieval for 15 min (BORG; Biocare BD1000).
Histologically, the mandibular mass was a densely cellular, highly invasive neoplasm. Neoplastic cells were arranged in nests and ductular structures with frequent central necrosis supported by a robust population of spindle cells (Fig. 2A). Neoplastic cells were polygonal with occasionally prominent intercellular bridging, abundant foamy eosinophilic cytoplasm, and a large round nucleus with coarsely stippled chromatin and variably prominent eosinophilic nucleoli. Rare cells contained one-to-few clear cytoplasmic vacuoles that occasionally displaced the nucleus. Multifocally, cells displayed squamous differentiation and contained keratohyalin granules (Fig. 2B). No keratin pearl formation was noted. Anisocytosis and anisokaryosis were marked with significant cellular pleomorphism. Mitotic figures averaged 20 per 10 hpf, 40× objective, 10× ocular field number (FN) 22 mm (2.37 mm2). Lymphatic and vascular invasion were frequent. Stromal cells were elongate with moderate amounts of eosinophilic cytoplasm and a central oval nucleus with finely stippled chromatin. There was marked multifocal lymphoplasmacytic inflammation and moderate multifocal hemorrhage. Neoplastic infiltration of the cervical musculature with metastasis to the lungs and tracheobronchial lymph nodes was present (Fig. 2C).
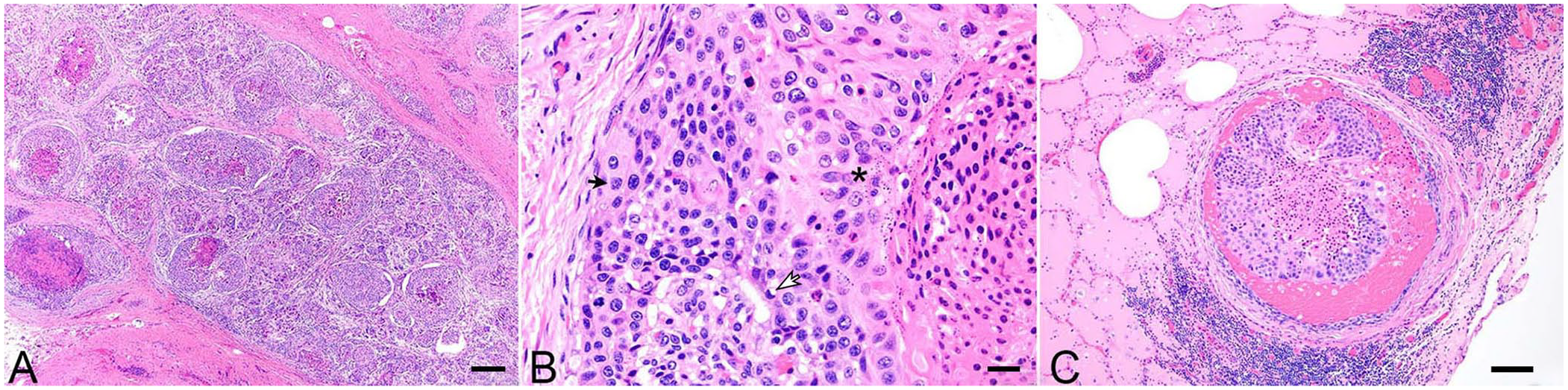
Mucoepidermoid carcinoma of the salivary gland in a bull.
Neoplastic epithelial cells were diffusely and strongly positive for pancytokeratin (Fig. 3A) with marked, strong nuclear positivity for p63 (Fig. 3B), and multifocal positivity for vimentin (Fig. 3C). Neoplastic epithelial cells were negative for α-SMA. The neoplastic stroma was diffusely and strongly positive for both vimentin and α-SMA (Fig. 3C, 3D) and negative for pancytokeratin and p63. Rare cytoplasmic vacuoles within neoplastic epithelia were positive for PAS and Alcian blue with scant PAS- and Alcian blue–positive extracellular material (Fig. 3E, 3F).
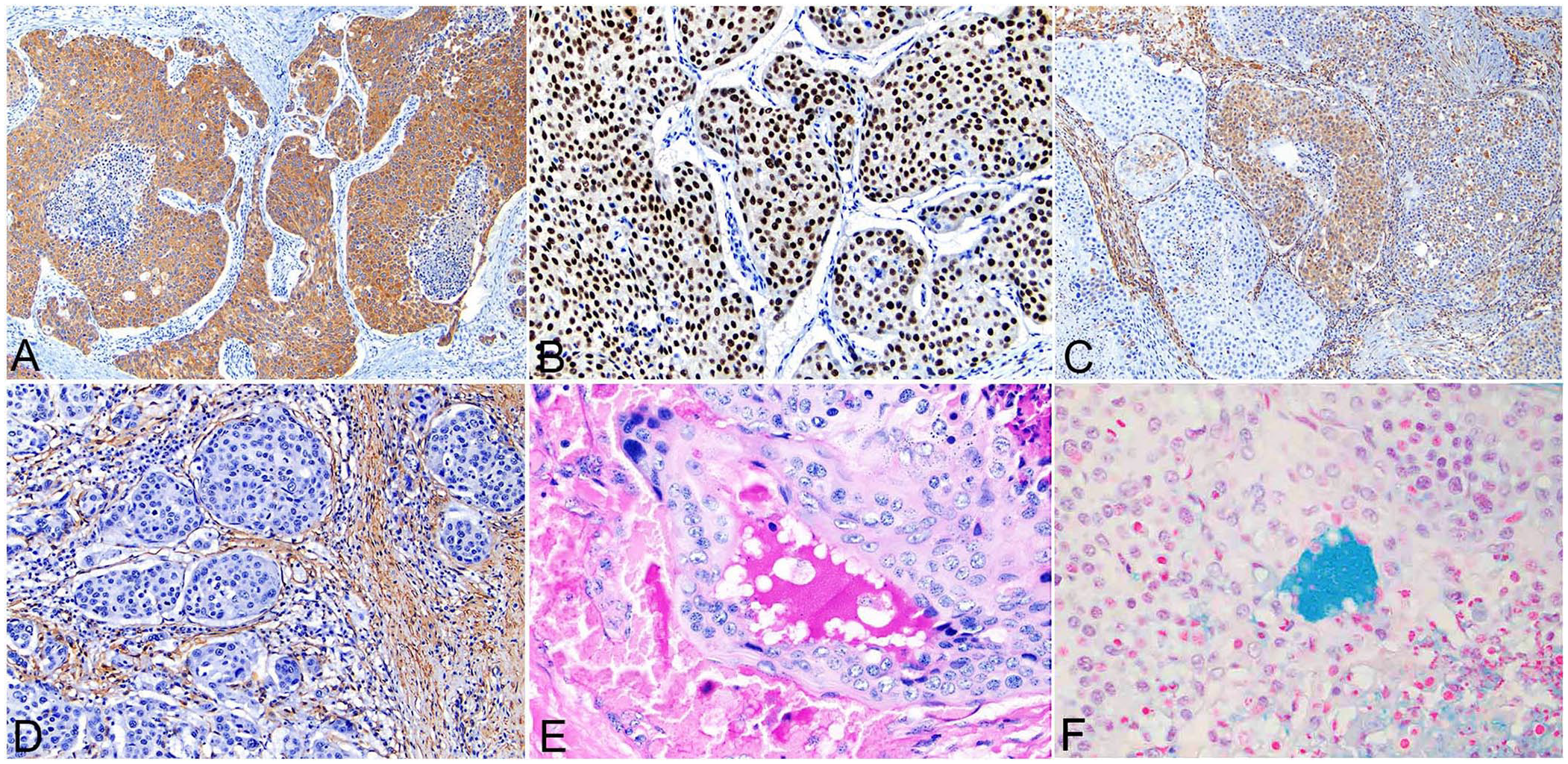
Immunohistochemistry and special stains of a mucoepidermoid carcinoma of the salivary gland in a bull.
MEC is the malignant salivary tumor reported most frequently in humans,1,15 and was the second most common malignant salivary neoplasm of dogs in a 2020 retrospective study. 11 MEC occurs most commonly in the parotid salivary gland of humans4,15 and veterinary species,11,13 but may affect other major or minor salivary glands and even originate from within bone. 4 MEC is a neoplasm composed of variable populations of intermediate cells, epidermoid cells, and mucous cells arranged in cystic and/or solid histologic patterns.4,15,20 Intermediate cells are often the predominant cell type, particularly in high-grade MECs.1,3,10,15 Intermediate cells are pluripotent cells of salivary gland excretory ducts that differentiate into epidermoid and mucous cells,1,4 versus myoepithelial cells, which surround the luminal epithelial cells of individual acini. 8 Epidermoid cells may contain keratohyalin granules and can display keratin pearl formation histologically, resembling squamous cell carcinoma. 9
Accurate grading of MECs is clinically important given that the disease-free interval and therapeutic strategy are vastly different for low-grade versus high-grade MECs. In humans, low-grade MECs have reported 5-y survival rates of 92–100% after surgical removal, whereas the median 5-y survival rate for high-grade MECs drops to 0–43%. 19 Lower clinical grade is also associated with a longer median survival time in dogs with malignant salivary tumors, compared to those with higher clinical grade tumors. 11 Low-grade MECs are characterized by a predominance of mucous cells with mucus-filled cystic spaces, minimal cellular atypia, rare mitoses, and lack of neural invasion.15,19 High-grade MECs are typically more solid with few-to-rare mucous cells and may be composed of up to 95% epidermoid cells with nuclear atypia, high mitotic count, necrosis, peripheral perineural invasion, vascular invasion, and bony invasion.1,4,19 The solid growth pattern, frequent necrosis, marked cellular and nuclear pleomorphism, vascular invasion, multi-organ metastasis, and bony destruction classify the tumor in our case as a high-grade MEC.
IHC has been utilized in the differentiation of MECs. Epithelial cells display diffuse positivity for cytokeratin not only in MECs but in adenocarcinomas, 6 pleomorphic adenomas, 7 basal cell adenocarcinomas, 5 and squamous cell carcinomas. 13 Peripheral cells in MECs can stain positive for vimentin, 6 and 20–25% of epithelial cells in MECs co-express vimentin and glial fibrillary acidic protein. 4 In the veterinary literature, a salivary gland epithelial-myoepithelial carcinoma in a dog 8 and a high-grade MEC in a lion 6 were both described as having peripheral vimentin and p63 staining. In the human literature, diffuse positivity is described for p63, differentiating high-grade MEC from acinic cell carcinoma 18 and salivary duct carcinoma.3,6 Furthermore, the level of p63 staining increased as the proportion of epidermoid cells and tumor grade increased; thus, p63 alone may be a more specific IHC marker for high-grade MEC versus cytokeratin or p63 in combination with cytokeratin. 3
The tumor in our case displayed diffuse cytokeratin and p63 positivity in the epithelial component, consistent with IHC staining patterns in human high-grade MECs.3,18 Vimentin positivity in the epithelial population was multifocal with no discernible pattern, most likely as a result of the loss of stratified epithelial differentiation with increasing malignancy. Alternatively, our case could represent a subset of MECs that co-express cytokeratin and vimentin, 4 as described in the human literature. The tumor stroma in our case was positive for vimentin and α-SMA and negative for cytokeratin, indicating myofibroblastic origin. In the human literature, a myofibroblastic stromal reaction was briefly mentioned following rupture of a tumor 4 ; antemortem manipulation of the tumor could perhaps be associated with the abundant myofibroblastic stroma seen in our case.
The lesion in the bull in our case had been diagnosed clinically as a chronic abscess, which is a common malady in cattle. Although rare, salivary gland neoplasia should be included in the differential diagnosis of chronic, non-resolving masses on and around the head of cattle.
Footnotes
Acknowledgements
We thank the Purdue Animal Disease Diagnostic Laboratory (West Lafayette, IN, USA) for performing the p63 IHC stain.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
