Abstract
A ventral abdominal subcutaneous mass was removed from each of 2 young adult spayed female ferrets. In both cases, the neoplasms were composed of islands of polygonal cells separated by interlacing streams of spindloid cells reminiscent of ferret adrenocortical tumors with smooth muscle proliferation. Immunohistochemically, the polygonal cells demonstrated strong cytoplasmic reactivity for inhibin and weak cytoplasmic reactivity for pancytokeratin and S-100 protein. Spindloid cells demonstrated strong cytoplasmic reactivity for alpha smooth muscle actin, muscle-specific actin, desmin, and glial fibrillary acidic protein. Ultrastructurally, the polygonal cells contained numerous intracytoplasmic clear vacuoles, mitochondria, scant rough endoplasmic reticulum, and few intermediate filaments. In one tumor, vesicular tubular mitochondria were found in polygonal cells. The spindloid cells contained numerous aggregates of parallel intermediate filaments. The histologic, immunohistochemical, and ultrastructural findings are suggestive of adrenocortical tumors with smooth muscle proliferation, but cannot be differentiated from an ovarian gonadal stromal tumor. Neither ferret had a clinically detected primary adrenal gland tumor or clinical signs of adrenal-associated endocrinopathy.
Domestic ferrets are susceptible to a variety of neoplasms. The most common are islet cell tumors of the pancreas and adrenal cortical tumors. 9 A few sex cord–stromal (gonadostromal) tumors, including thecomas, ovarian stromal tumors, and granulosa cell tumors, have also been reported in this species. 9 This report documents the clinical presentation and histologic, immunohistochemical, and ultrastructural findings in 2 ferrets with ventral abdominal subcutaneous neoplasms that resemble adrenocortical tumors with smooth muscle proliferation.
Ferret No. 1 was an 11-month-old spayed female that presented with a subcutaneous mass of the ventral abdomen near the surgical site of a previous ovariohysterectomy. Clinical signs of hyperestrogenism, that is, endocrine alopecia and swollen vulva, were not evident. Grossly, the mass was firm, lobulated, and tan on cut surface.
Ferret No. 2 was a 1-year-old spayed female that presented with an intestinal foreign body. A subcutaneous mass located near the linea alba of the abdomen was noticed and removed. No clinical signs of hyperestrogenism were noted. Grossly, the mass was firm, tan, spheroid, and 0.7 cm in diameter.
Tissue specimens from both masses were processed by conventional methods, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin.
Immunohistochemical staining was performed on deparaffinized sections using the avidin-biotin-peroxidase complex technique for cytokeratin (Keratin AE1/3, DAKO), inhibin, S-100 protein, glial fibrillary acidic protein (GFAP), neuron-specific enolase (NSE), alpha smooth muscle actin (SMA), muscle specific actin (actin), myoglobin (Ventana Basic DAB kit, Tucson, Arizona), and desmin (prediluted product, Ventana Basic DAB kit, Tucson, AA). Sections were also stained under identical conditions with normal rabbit or normal mouse serum to serve as negative controls. These sections were counterstained with Mayer's or Harris hematoxylin (Richard-Allan Scientific, Kalamazoo, MI).
For transmission electron microscopy samples of formalin-fixed tissue were post fixed in 1% osmium tetroxide, dehydrated, and embedded in effepoxy plastic (Ernest F. Fullam, Latham, NY), sectioned at 800 angstrom, and stained with 4% uranyl acetate and Reynolds lead citrate for ultrastructure examination. Sections were examined and photographed on a Zeiss EM 10 electron microscope (LED Electron Microscopy Inc, Thornwood, NY).
Clinical information was obtained from accompanying biopsy submission forms and follow-up telephone conversations with the attending clinician and/or veterinary technician. The owners did not return to the submitting clinics for further treatment and could not be reached by telephone and/or email.
Histologically, the neoplasm in Ferret No. 1 was composed of islands of 2 to 20 polygonal cells separated by thick interlacing streams of spindloid cells (Fig. 1). The polygonal cells had variably distinct cell borders and moderate amounts of eosinophilic granular to microvacuolated cytoplasm. The nuclei were round and centrally located and had finely stippled chromatin and 1 distinct nucleolus. The mitotic index was low (no mitotic figures were seen). The spindloid cells had indistinct cell borders and small amounts of eosinophilic fibrillar cytoplasm. Their nuclei were oval to fusiform and had finely stippled chromatin and small nucleoli. The mitotic index was approximately 3 per 10 high-power fields. In ferret No. 2, the histologic findings were similar, but the islands of polygonal cells were larger, the islands were separated by thinner streams of spindloid cells, and the mitotic index of the spindloid cells was low (no mitotic figures were seen) (Fig. 2).
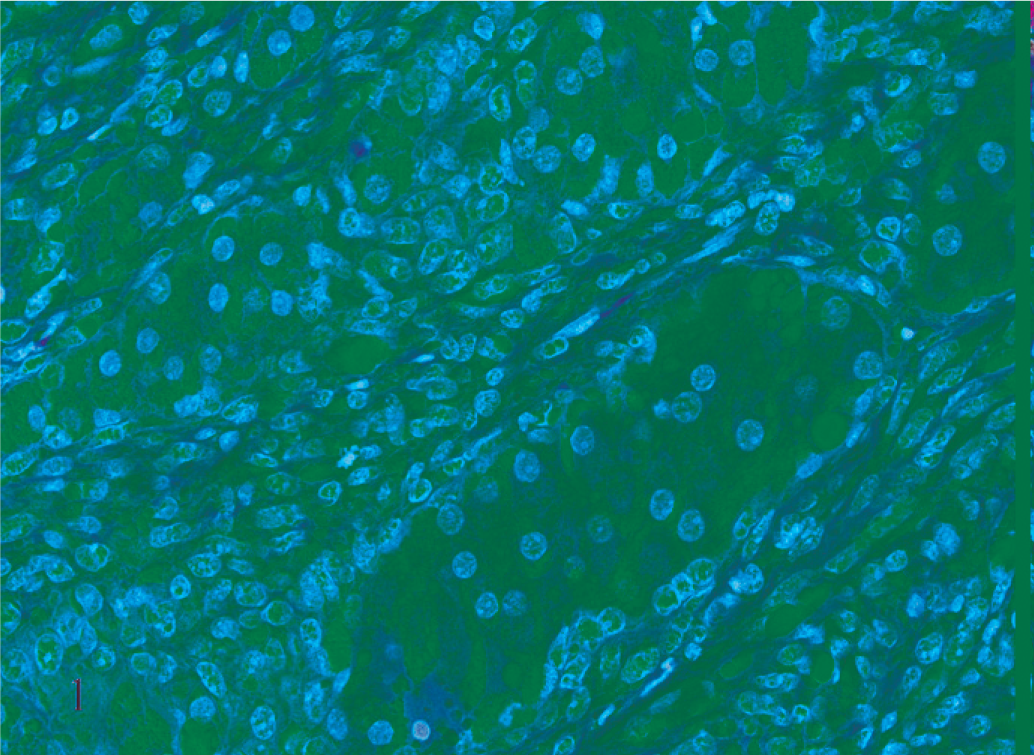
Subcutaneous neoplasm, ferret No. 1. Islands of polygonal cells are separated by thick interlacing streams of spindle cells. HE.

Subcutaneous neoplasm, ferret No. 2. Islands of polygonal cells are larger and separated by thinner streams of spindle cells than ferret 1. HE.
The immunohistochemical findings are summarized in Table 1. The polygonal cells demonstrated strong cytoplasmic reactivity for inhibin (Fig. 3) and weak cytoplasmic reactivity for keratin (Fig. 4) and S-100 protein. Spindloid cells demonstrated strong cytoplasmic reactivity for SMA (Fig. 5), actin, desmin, and GFAP (Fig. 6) and demonstrated weak positivity for NSE.
Summary of immunohistochemical results from 2 ferrets with subcutaneous tumors(ferrets No. 1 and 2) and 2 ferrets with adrenocortical tumors with smooth muscle proliferation (ferrets No. 3 and 4).∗
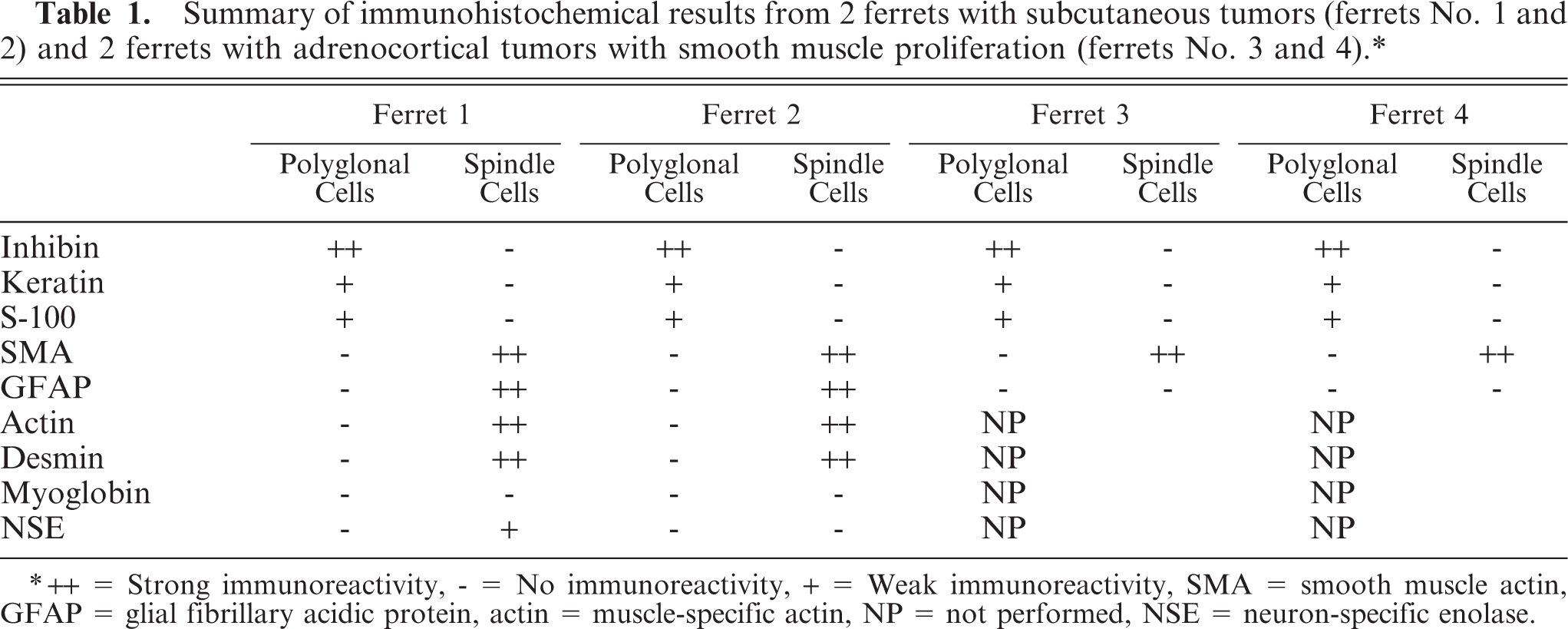
∗++ = Strong immunoreactivity, - = No immunoreactivity, + = Weak immunoreactivity, SMA = smooth muscle actin, GFAP = glial fibrillary acidic protein, actin = muscle-specific actin, NP = not performed, NSE = neuron-specific enolase.
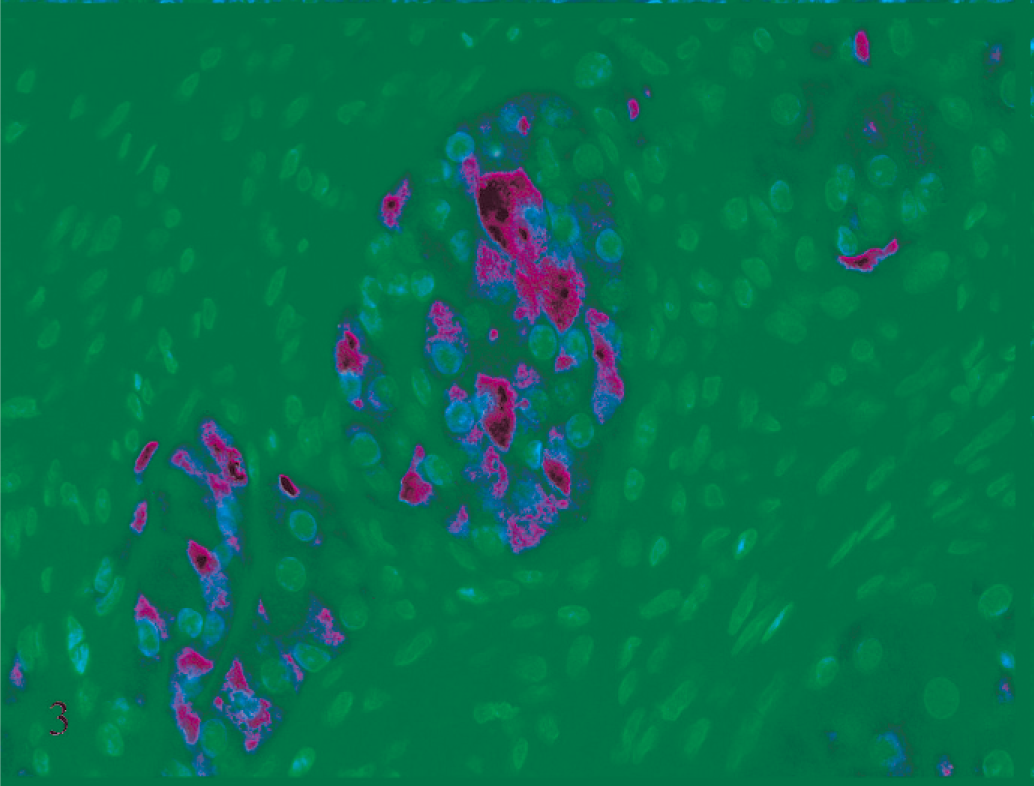
Subcutaneous neoplasm, ferret No. 1. Polygonal cells are strongly immunoreactive with alpha-inhibin. Immunostaining for alpha-inhibin with hematoxylin counterstain.

Subcutaneous neoplasm, ferret No. 1. Polygonal cells are weakly immunoreactive with pancytokeratin. Immunostaining for pancytokeratin with hematoxylin counterstain.
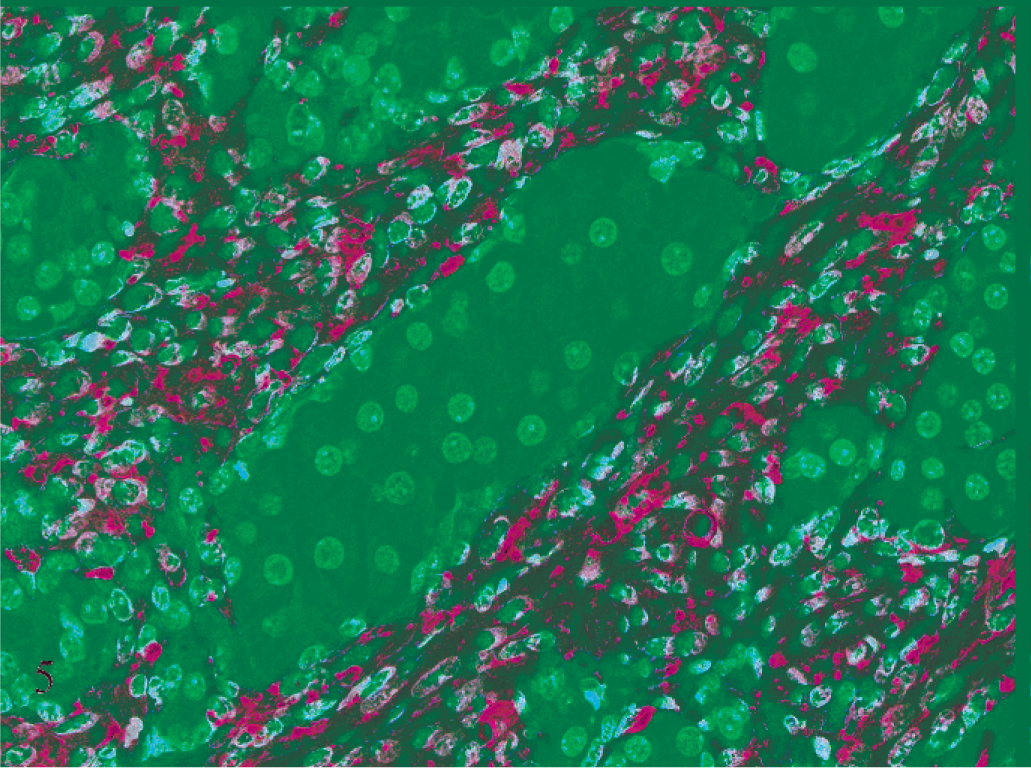
Subcutaneous neoplasm, ferret No. 1. Spindle cells are strongly immunoreactive with SMA. Immunostaining for SMA with hematoxylin counterstain.
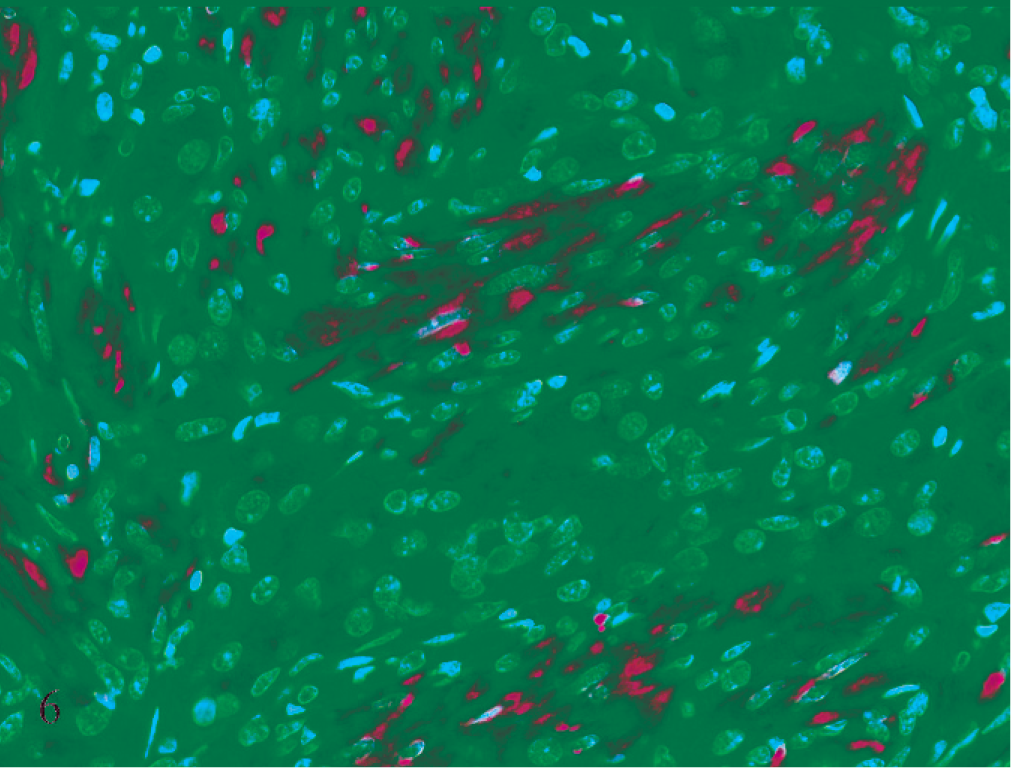
Subcutaneous neoplasm, ferret No. 1. Spindle cells are strongly immunoreactive with GFAP. Immunostaining for GFAP with hematoxylin counterstain.
For comparison, adrenal cortical tumors with smooth muscle proliferation from a 1-year-old female ferret (ferret No. 3) and a 4-year-old female ferret (ferret No. 4) were evaluated by immunohistochemistry. Neoplastic cortical cells demonstrated strong cytoplasmic reactivity for inhibin, and multifocally mild reactivity for keratin and S-100 protein. Non-neoplastic cortical cells demonstrated weak cytoplasmic reactivity for keratin and S-100 protein and were negative for inhibin. The spindloid cells demonstrated diffuse strong cytoplasmic reactivity with SMA and were negative for inhibin, keratin, GFAP, and S-100 protein.
Ultrastructurally, the polygonal cells contained numerous intracytoplasmic clear vacuoles, mitochondria, scant rough endoplasmic reticulum, and few intermediate filaments. The spindle cells contained numerous aggregates of parallel intermediate filaments. In ferret No. 2, vesicular tubular mitochondria were found in a few polygonal cells.
There have been no previous reports of similar neoplasms in the subcutis of ferrets. The origin of the tumors is not known. Histologically, the presence of islands of microvacuolated polygonal cells that are inhibin positive and separated by streams of spindloid cells is reminiscent of ferret adrenocortical tumors with smooth muscle proliferation. 4 Recently, 2 cases of neoplastic transformation of presumed ovarian remnants that had histologic features similar to tumors of the ferret adrenal cortex were reported. 14 An ovarian origin was favored over an adrenal cortical one based on location of the tumors in the ovarian pedicles. In humans, tumors that stain positively for inhibin have been reported in the liver, 17 pancreas, 7 digestive tract, 7 pituitary gland, 16 and breast 10 and include endometrioid tumors of the uterus 18 and granular cell tumors; 8 however, these tumor types with histomorphology similar to adrenal cortical tumors with smooth muscle proliferation have not been described in ferrets. Therefore, adrenal cortical tumor and sex cord–stromal tumor are considered most likely in the differential diagnosis for these subcutaneous neoplasms.
Steroid-producing cells of the adrenal cortex and gonads arise from common progenitors in the urogenital ridge of the embryo. 1 Tumors of these 2 cell types have similar histologic and immunohistochemical features. Neoplasms of the adrenal cortex and gonads have been reported to be immunoreactive with inhibin and GATA-4 in ferrets and with inhibin, melan A, and keratin in humans. 1, 2, 19, 21 In addition, MIS immunoreactivity has been reported in adrenocortical tumors and in normal adrenal cortical cells and granulosa cells of ferrets. 14 Therefore, the immunohistochemical results do not help to differentiate adrenocortical tumor from sex cord-stromal tumor.
The SMA, actin, and desmin positivity of the spindloid cells are compatible with smooth muscle proliferation and are consistent with what has been reported in some adrenocortical tumors of ferrets, but the GFAP positivity of these cells was not expected. GFAP positivity can be seen occasionally in non-neural tumors, that is, 40% of human fetal rhabdomyomas and occasional myoepithelial cells in human breast tumors are positive for GFAP. 2, 6
Ultrastructurally, endocrine cells have abundant smooth endoplasmic reticulum containing lipid and vesicular tubular mitochondria. 20 In these cases, the ultrastructural detail was obscured by suboptimal tissue processing. Smooth endoplasmic reticulum was not apparent; however, the numerous clear vacuoles, which were likely lipid vacuoles, and occasional cells with vesicular tubular mitochondria in ferret No. 2 are supportive of cells of endocrine origin. The aggregates of parallel intermediate filaments in the spindle cells are consistent with muscle cells.
Both of these tumors occurred in young spayed female ferrets along the ventral abdominal midline. Metastasis from a primary adrenal tumor seems unlikely as no primary adrenal tumor was identified by the clinicians, and clinical signs of hyperestrogenism that are typically associated with adrenocortical tumors in ferrets were not present. Similarly as these ferrets were both young spayed females, metastatic sex cord–stromal tumor is unlikely. Ectopic adrenal tissue has been reported in ferrets, but only close to the adrenal glands or in the cranial perirenal adipose tissue. 5, 12 Therefore, transformation of subcutaneous ectopic adrenal tissue is not likely.
Although metastatic neoplasia cannot be ruled out because these animals were lost to follow up, we speculate that adrenocortical or ovarian cells may have been seeded to the subcutis during ovariohysterectomy and later transformed. The concept of surgically transplanting tumors cells to the subcutis or other locations that survive and grow is well established in human and veterinary literature. 3, 13 While, to the best of our knowledge, the concept of seeding non-neoplastic cells to the subcutis that later transform has not been reported, there are reports of experimental transplantation of adrenocortical and ovarian tissue into the subcutis of mice and rats in which the transplanted cells survive and are functional. 11, 15 This seems the most likely explanation for transformed adrenocortical or sex cord–stromal cells within the subcutis near the sites of the spay incisions. Hopefully, this report will result in increased recognition of this tumor in ferrets and lead to better understanding of its histogenesis and biological behavior.
Footnotes
Acknowledgements
We thank Dr. Bruce Williams for his consultation and Mr. John Jenkins, HT, HTL, ASCP, for electron microscope service. Mark A. Smith is a major in the U.S. Army. The opinions or assertions herein are those of the authors and do not necessarily reflect the view of the Department of the Army or the Department of Defense.
